question_answer 1) A heavy uniform chain lies on a horizontal top of a table. If the coefficient of friction between the chain and the table is 0.25, then the maximum percentage over the length of the chain that can hang over one edge of the table is
A)
20%
done
clear
B)
25%
done
clear
C)
35%
done
clear
D)
15%
done
clear
View Answer play_arrow
question_answer 2) A weight w is suspended from the midpoint of a rope, whose ends are at the same level. In order to make the rope perfectly horizontal, the force applied to each of its ends must be
A)
less than w
done
clear
B)
equal to w
done
clear
C)
equal to 2 w
done
clear
D)
infinitely large
done
clear
View Answer play_arrow
question_answer 3) A disc of mass 100 g is kept floating horizontally in air by firing bullets, each of mass 5 g with the same velocity at the same rate of 10 bullets per second. The bullets rebound with the same speed in opposite direction, the velocity of each bullet at the time of impact is
A)
196cm/s
done
clear
B)
9.8 cm/s
done
clear
C)
98 cm/s
done
clear
D)
980 cm/s
done
clear
View Answer play_arrow
question_answer 4) The velocity of a particle at an instant is 10 m/s. After 3s its velocity will become 16 m/s. The velocity at 2 s, before the given instant will be
A)
6 m/s
done
clear
B)
4 m/s
done
clear
C)
2 m/s
done
clear
D)
1 m/s
done
clear
View Answer play_arrow
question_answer 5) A heavy stone hanging from a massless string of length 15 m is projected horizontally with speed 147 m/s. The speed of the particle at the point where the tension in the string equals the weight of the particle is
A)
10 m/s
done
clear
B)
7 m/s
done
clear
C)
12 m/s
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 6) When a body moves with constant speed in a circular path then
A)
work done will be zero
done
clear
B)
acceleration will be zero
done
clear
C)
no force acts on the body
done
clear
D)
its velocity remains constant
done
clear
View Answer play_arrow
question_answer 7) Two stones are projected with same velocity v at an angle 6 and \[\left( \text{9}0{}^\circ \text{ }-\theta \right).\] If H and \[{{H}_{1}}\]are the greatest height in the two paths, what is the relation between R, H and\[{{H}_{1}}\]?
A)
\[R=4\sqrt{H{{H}_{1}}}\]
done
clear
B)
\[R=\sqrt{H{{H}_{1}}}\]
done
clear
C)
\[R=H{{H}_{1}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 8) A body initially at rest is moving with uniform acceleration a. Its velocity after n sec is v. The displacement of the body in last 2 s is
A)
\[\frac{2v(n-1)}{n}\]
done
clear
B)
\[\frac{v(n-1)}{n}\]
done
clear
C)
\[\frac{v(n+1)}{n}\]
done
clear
D)
\[\frac{2v(n+1)}{n}\]
done
clear
View Answer play_arrow
question_answer 9) The magnetic force on a point charge is \[\overset{\to }{\mathop{F}}\,=q(\overset{\to }{\mathop{V}}\,\times \overset{\to }{\mathop{B}}\,)\] Here, q = electric charge \[\overset{\to }{\mathop{V}}\,\]= velocity of point charge \[\overset{\to }{\mathop{B}}\,\]= magnetic field The dimension of \[\overset{\to }{\mathop{B}}\,\]is
A)
\[\left[ \text{ML}{{\text{T}}^{-1}}\text{A} \right]\]
done
clear
B)
\[\left[ {{\text{M}}^{2}}\text{L}{{\text{T}}^{-2}}{{\text{T}}^{-1}} \right]\]
done
clear
C)
\[\left[ \text{M}{{\text{T}}^{-2}}{{A}^{-1}} \right]\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 10) The first diffraction minimum due to single slit diffraction is 9, for a light of wavelength \[5000\overset{\text{o}}{\mathop{\text{A}}}\,\]. It the width of the slit is \[\text{1}\times \text{1}{{0}^{-\text{4}}}\text{ cm},\] then the value of 9 is
A)
\[30{}^\circ \]
done
clear
B)
\[45{}^\circ \]
done
clear
C)
\[60{}^\circ \]
done
clear
D)
\[15{}^\circ \]
done
clear
View Answer play_arrow
question_answer 11) A body of mass 2 kg is placed on rough horizontal plane. The coefficient of friction between body and plane is 0.2. Then
A)
body will move in forward direction if F = 5 N
done
clear
B)
body will move in backward direction with acceleration 0.5 m/s2, if force F = 3 N
done
clear
C)
If F = 3 N then body will be in rest condition
done
clear
D)
Both (a) and (b) are correct
done
clear
View Answer play_arrow
question_answer 12) A particle moves with a velocity \[\left( \text{5}\overset{\hat{\ }}{\mathop{\text{i}}}\,-\text{3}\overset{\hat{\ }}{\mathop{\text{j}}}\,\text{ }+\text{ 6}\overset{\hat{\ }}{\mathop{\text{k}}}\, \right)\text{ m}/\text{s}\]under the influence of a constant force \[\overset{\to }{\mathop{\text{F}}}\,=\text{1}0\overset{\hat{\ }}{\mathop{\text{i}}}\,+\text{1}0\overset{\hat{\ }}{\mathop{\text{j}}}\,+\text{2}0\overset{\hat{\ }}{\mathop{\text{k}}}\,\text{ N}.\] The instantaneous power applied to the particle is
A)
200 J/s
done
clear
B)
40 J/s
done
clear
C)
140 J/s
done
clear
D)
170 J/s
done
clear
View Answer play_arrow
question_answer 13) The potential energy of a particle of mass 5 kg moving in the x-y plane is given by \[\text{U}=\left( -\text{7x}+\text{24y} \right)\text{ J},\text{ x}\]and y being in metre. If the particle starts from rest from origin, then speed of particle at t = 2 s is
A)
5 m/s
done
clear
B)
14 m/s
done
clear
C)
17.5 m/s
done
clear
D)
10 m/s
done
clear
View Answer play_arrow
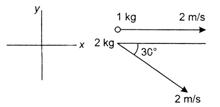
question_answer 14)
Find the velocity of centre of mass of the system shown in the figure?
A)
\[\left( \frac{2+2\sqrt{3}}{3} \right)\hat{i}-\frac{2}{3}\hat{j}\]
done
clear
B)
\[4\hat{i}\]
done
clear
C)
\[\left( \frac{2-2\sqrt{3}}{3} \right)\,\overset{\hat{\ }}{\mathop{i}}\,-\frac{2}{3}\overset{\hat{\ }}{\mathop{j}}\,\]
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 15) The ratio of radii of gyration of a circular disc and a circular ring of the same radii and same mass about a tangential axis in the plane is
A)
1 : 2
done
clear
B)
\[\sqrt{5}:\sqrt{6}\]
done
clear
C)
2 : 3
done
clear
D)
2 : 1
done
clear
View Answer play_arrow
question_answer 16) A particle performs uniform circular motion with an angular momentum L. If the frequency of particle motion is doubled and its KE is halved the angular momentum becomes
A)
2 L
done
clear
B)
4 L
done
clear
C)
L / 2
done
clear
D)
L / 4
done
clear
View Answer play_arrow
question_answer 17) A body of mass m rises to height h = R/S from earths surface, where R is earths radius. Ifg is acceleration due to gravity at earths surface, the increase in potential energy is
A)
\[mgh\]
done
clear
B)
\[\frac{4}{5}mgh\]
done
clear
C)
\[\frac{5}{6}mgh\]
done
clear
D)
\[\frac{6}{7}mgh\]
done
clear
View Answer play_arrow
question_answer 18) A satellite is moving on a circular path of radius r around the earth has a time period T. If its radius slightly increases by \[\Delta \]r, the change in it; time period is
A)
\[\frac{3}{2}\left( \frac{T}{r} \right)\Delta r\]
done
clear
B)
\[\left( \frac{T}{r} \right)\Delta r\]
done
clear
C)
\[\frac{3}{2}\left( \frac{{{T}^{2}}}{{{r}^{2}}} \right)\Delta r\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 19) A particle executes SHM, its time period is 16 s If its passes through the centre of oscillation then its velocity is 2 m/s at times 2 s. The amplitude will be
A)
7.2 m
done
clear
B)
4 cm
done
clear
C)
6 cm
done
clear
D)
0.72 m
done
clear
View Answer play_arrow
question_answer 20) A particle executes simple harmonic motior. with a frequency f. The frequency with which the potential energy oscillates is
A)
\[f\]
done
clear
B)
\[f\]/2
done
clear
C)
2\[f\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 21) An astronaut is approaching the moon. He sends out a radio signal of frequency 5000 MHz and the frequency of echo is different from that of the original frequency b) 100 kHz. His velocity of approach with respec- to the moon is
A)
2km/s
done
clear
B)
3 km/s
done
clear
C)
4 km/s
done
clear
D)
5 km/s
done
clear
View Answer play_arrow
question_answer 22) When a sphere is taken to bottom of sea 1 krr: deep, it contracts by 0.01%. The bulk modulus of elasticity of the material of sphere is \[\left( \text{Given }:\text{ Density of water }=\text{ 1 g}/\text{c}{{\text{m}}^{\text{3}}} \right)\]
A)
\[\text{9}.\text{8}\times \text{1}{{0}^{\text{1}0}}\text{ N}/{{\text{m}}^{\text{2}}}\]
done
clear
B)
\[\text{1}0.\text{2}\times \text{l}{{0}^{\text{1}0}}\text{ N}/{{\text{m}}^{\text{2}}}\]
done
clear
C)
\[0.\text{98}\times \text{1}{{0}^{\text{1}0}}\text{ N}/{{\text{m}}^{\text{2}}}\]
done
clear
D)
\[\text{8}.\text{4}\times \text{l}{{0}^{\text{1}0}}\text{ N}/{{\text{m}}^{\text{2}}}\]
done
clear
View Answer play_arrow
question_answer 23) The temperature of hydrogen at which the rms velocity of its molecules is seven times the rms velocity the molecules of nitrogen at 300 K is
A)
2100 K
done
clear
B)
1700 K
done
clear
C)
1350 K
done
clear
D)
1050 K
done
clear
View Answer play_arrow
question_answer 24) The inside and outside temperatures of a refrigerator are 273 K and 303 K respectively. Assuming that refrigerator cycle is reversible, for every joule of work done the heat delivered to the surrounding will be
A)
10 J
done
clear
B)
20 J
done
clear
C)
30 J
done
clear
D)
50 J
done
clear
View Answer play_arrow
question_answer 25) In an energy recycling process, X g of steam at \[100{}^\circ C\] becomes water at \[100{}^\circ C\] which converts Y g of ice at \[0{}^\circ C\] into water at \[100{}^\circ C\]. The ratio of X and Y will be
A)
\[\frac{1}{3}\]
done
clear
B)
\[\frac{2}{3}\]
done
clear
C)
3
done
clear
D)
2
done
clear
View Answer play_arrow
question_answer 26) The surface temperature of the sun is T K and the solar constant for a plate is S. The sun subtends an angle 6 at the planet. Then
A)
\[\text{S}\propto {{\text{T}}^{\text{4}}}\]
done
clear
B)
\[\text{S}\propto {{\text{T}}^{2}}\]
done
clear
C)
\[\text{S}\propto {{\theta }^{2}}\]
done
clear
D)
\[\text{S}\propto \theta \]
done
clear
View Answer play_arrow
question_answer 27) A body at a temperature of \[727{}^\circ C\] and has surface area 5 cm2, radiates 300 J of energy each minute. The emissivity is (Given : Boltzmann constant \[=5.67\times {{10}^{-8}}W{{m}^{2}}{{K}^{4}})\]
A)
\[e\]=0.18
done
clear
B)
\[e\]=0.02
done
clear
C)
\[e\]=0.2
done
clear
D)
\[e\]=0.15
done
clear
View Answer play_arrow
question_answer 28) If at NTP velocity of sound in a gas is 1150 m/s, then the rms velocity of gas molecules at NTP is \[\left( \text{Given}:\text{R}=\text{ 8}.\text{3J}/\text{mol K},{{\text{C}}_{\text{p}}}\text{ }=\text{ 4}.\text{8 cal}/\text{mol K} \right)\]
A)
1600 m/s
done
clear
B)
1532.19 m/s
done
clear
C)
160 m/s
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 29) If\[\sigma \]surface charge density, \[\varepsilon \]= electric permittivity the dimensions of \[\frac{\sigma }{\varepsilon }\] are same as
A)
electric force
done
clear
B)
electric field intensity
done
clear
C)
pressure
done
clear
D)
electric charge
done
clear
View Answer play_arrow
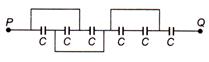
question_answer 30)
For circuit the equivalent capacitance between P and Q is
A)
6 C
done
clear
B)
4 C
done
clear
C)
\[\frac{3C}{2}\]
done
clear
D)
\[\frac{6C}{11}\]
done
clear
View Answer play_arrow
question_answer 31) A wire has resistance 12\[\Omega \]. It is bent in the form of a circle. The effective resistance between the two points on any diameter of the circle is
A)
12\[\Omega \]
done
clear
B)
24\[\Omega \]
done
clear
C)
6\[\Omega \]
done
clear
D)
3\[\Omega \]
done
clear
View Answer play_arrow
question_answer 32) If two identical heaters each rated as (1000 W, 220 V) are connected in parallel to 220 V, then the total power consumed is
A)
200 W
done
clear
B)
2500 W
done
clear
C)
250 W
done
clear
D)
2000 W
done
clear
View Answer play_arrow
question_answer 33) A conducting circular loop of radius r carries a constant current . It is placed in a uniform magnetic field bq such that By is perpendicular to the plane of the loop. The magnetic force acting on the loop is
A)
\[\text{Ir}{{\text{B}}_{\text{o}}}\]
done
clear
B)
\[\text{2}\pi I\text{r}{{\text{B}}_{\text{o}}}\]
done
clear
C)
\[\pi I\text{r}{{\text{B}}_{\text{o}}}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 34) In the given figure, the loop is fixed but straight wire can move. The straight wire will
A)
remain stationary
done
clear
B)
move towards the loop
done
clear
C)
move away from the loop
done
clear
D)
rotates about the axis
done
clear
View Answer play_arrow
question_answer 35) At a point on the right bisector of a magnetic dipole, the magnetic
A)
potential varies as \[\frac{1}{{{r}^{2}}}\].
done
clear
B)
potential is zero at all point on the right bisector
done
clear
C)
field varies as \[{{r}^{3}}\]
done
clear
D)
field is perpendicular to the axis of dipole
done
clear
View Answer play_arrow
question_answer 36) The couple acting on a magnet of length10 cm and pole strength 15 Am, kept in a field of \[\text{B}=\text{2}\times \text{1}{{0}^{-\text{5}}}\text{ T},\]at an angle of 30° is
A)
\[\text{1}.\text{5}\times \text{l}{{\text{0}}^{-5}}\text{N}-\text{m}\]
done
clear
B)
\[\text{1}.\text{5}\times \text{l}{{\text{0}}^{-3}}\text{N}-\text{m}\]
done
clear
C)
\[\text{1}.\text{5}\times \text{l}{{\text{0}}^{-2}}\text{N}-\text{m}\]
done
clear
D)
\[\text{1}.\text{5}\times \text{l}{{\text{0}}^{-6}}\text{N}-\text{m}\]
done
clear
View Answer play_arrow
question_answer 37) If a circuit made up of a resistance 1 n and inductance 0.01 H, an alternating emf 200 V at 50 Hz is connected, then the phase difference between the current and the emf in the circuit is
A)
\[\text{ta}{{\text{n}}^{-\text{1}}}\text{ }\left( \pi \right)\]
done
clear
B)
\[\text{ta}{{\text{n}}^{-\text{1}}}\text{ }\left( \frac{\pi }{2} \right)\]
done
clear
C)
\[\text{ta}{{\text{n}}^{-\text{1}}}\text{ }\left( \frac{\pi }{4} \right)\]
done
clear
D)
\[\text{ta}{{\text{n}}^{-\text{1}}}\text{ }\left( \frac{\pi }{3} \right)\]
done
clear
View Answer play_arrow
question_answer 38) An AC is represented by e = 220 sin (100 \[\pi \]) t V and is applied over a resistance of 110 n. The heat produced in 7 min is
A)
\[\text{11}\times \text{l}{{0}^{\text{3}}}\text{ cal}\]
done
clear
B)
\[\text{22}\times \text{1}{{0}^{\text{3}}}\text{ cal}\]
done
clear
C)
\[33\times \text{1}{{0}^{\text{3}}}\text{ cal}\]
done
clear
D)
\[25\times \text{1}{{0}^{\text{3}}}\text{ cal}\]
done
clear
View Answer play_arrow
question_answer 39) The wavelength of a radiowave of frequency of 1 MHz is
A)
400 m
done
clear
B)
300 m
done
clear
C)
350 m
done
clear
D)
200 m
done
clear
View Answer play_arrow
question_answer 40) The correct option, if speed of gamma rays. X-rays and microwaves are \[{{v}_{g}},{{v}_{X}}\]and \[{{v}_{m}}\]respectively will be
A)
\[{{v}_{g}}>{{v}_{X}}>{{v}_{m}}\]
done
clear
B)
\[{{v}_{g}}<{{v}_{X}}<{{v}_{m}}\]
done
clear
C)
\[{{v}_{g}}<{{v}_{X}}>{{v}_{m}}\]
done
clear
D)
\[{{v}_{g}}={{v}_{X}}={{v}_{m}}\]
done
clear
View Answer play_arrow
question_answer 41) The electric field E and magnetic field B in electromagentic wave are
A)
parallel to each other
done
clear
B)
inclined at an angle of \[45{}^\circ \]
done
clear
C)
perpendicular to each other
done
clear
D)
opposite to each other
done
clear
View Answer play_arrow
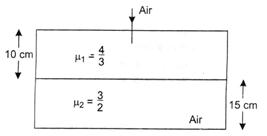
question_answer 42)
Considering normal incidence of ray, the equivalent refractive index of combination of two slabs shown in figure is
A)
1.8
done
clear
B)
1.43
done
clear
C)
2
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 43) To obtain a good photographic print an exposure of 2 s at a distance of 1 m from a 75 cd bulb is done. To obtain an equally satisfactory result, what should be the distance, if time of exposure is 12 s from a 50 cd bulb?
A)
1 m
done
clear
B)
2 m
done
clear
C)
3 m
done
clear
D)
4 m
done
clear
View Answer play_arrow
question_answer 44) In a Youngs experiment, one of the slit is covered with a transparent sheet of thickness \[\text{3}.\text{6}\times \text{1}{{\text{0}}^{-3}}\text{ cm}\]due to which position of central fringe shifts to a position originally occupied by 30th bright fringe. The refractive index of the sheet, if K = 6000 Å is
A)
1.5
done
clear
B)
1.2
done
clear
C)
1.3
done
clear
D)
1.7
done
clear
View Answer play_arrow
question_answer 45) Polarisation of light proves
A)
corpuscular nature of light
done
clear
B)
quantum nature of light
done
clear
C)
transverse wave nature of light
done
clear
D)
longitudinal wave nature of light
done
clear
View Answer play_arrow
question_answer 46) In terms of Rydberg constant R, the wave number of the first Balmer line is
A)
\[R\]
done
clear
B)
\[3R\]
done
clear
C)
\[\frac{5R}{36}\]
done
clear
D)
\[\frac{8R}{9}\]
done
clear
View Answer play_arrow
question_answer 47) In nuclear reaction \[_{2}H{{e}^{4}}{{+}_{Z}}{{X}^{A}}{{\to }_{Z+2}}{{Y}^{A+3}}{{+}_{Z}}{{M}^{A}}\] where M denotes
A)
electron
done
clear
B)
positron
done
clear
C)
proton
done
clear
D)
neutron
done
clear
View Answer play_arrow
question_answer 48) If the de-Broglie wavelength of a proton is \[\text{1}{{0}^{-\text{13}}}\text{ m},\]the electric potential through which it must have been accelerated is
A)
\[\text{4}.0\text{7}\times \text{1}{{0}^{\text{4}}}\text{ V}\]
done
clear
B)
\[\text{8}.\text{2}\times \text{1}{{0}^{\text{4}}}\text{ V}\]
done
clear
C)
\[\text{8}.\text{2}\times \text{1}{{0}^{\text{3}}}\text{ V}\]
done
clear
D)
\[\text{4}.0\text{7}\times \text{1}{{0}^{\text{5}}}\text{ V}\]
done
clear
View Answer play_arrow
question_answer 49) \[n\]alpha particles per second are emitted from \[N\]atoms of a radioactive element. The half-life of radioactive element is (in sec)
A)
\[\frac{n}{N}\]
done
clear
B)
\[\frac{N}{n}\]
done
clear
C)
\[\frac{0.693N}{n}\]
done
clear
D)
\[\frac{0.693\,n}{N}\]
done
clear
View Answer play_arrow
question_answer 50) In a semiconductor diode, the barrier potential offers opposition to only
A)
free electron in n-region
done
clear
B)
holes in p-region
done
clear
C)
free electrons in n-region and holes in p-region
done
clear
D)
free electrons in p-region and holes in n-region
done
clear
View Answer play_arrow
question_answer 51) Maximum entropy will be in which of the following?
A)
Ice
done
clear
B)
Liquid water
done
clear
C)
Snow
done
clear
D)
Water vapours
done
clear
View Answer play_arrow
question_answer 52) What is obtained when chlorine is passed in boiling toluene and product is hydrolysed?
A)
o-cresol
done
clear
B)
p-cresol
done
clear
C)
2, 4-dihydroxytoluene
done
clear
D)
Benzyl alcohol
done
clear
View Answer play_arrow
question_answer 53) Which of the following has covalent bond?
A)
\[N{{a}_{2}}S\]
done
clear
B)
\[AlC{{l}_{3}}\]
done
clear
C)
NaH
done
clear
D)
\[MgC{{l}_{2}}\]
done
clear
View Answer play_arrow
question_answer 54) Which of the following acts as an oxidising as well as reducing agent?
A)
\[N{{a}_{2}}S\]
done
clear
B)
\[N{{a}_{2}}{{O}_{2}}\]
done
clear
C)
\[NaN{{O}_{3}}\]
done
clear
D)
\[NaN{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 55) What is the oxidation state of P in Ba\[{{({{H}_{2}}P{{O}_{2}})}_{2}}\]?
A)
+1
done
clear
B)
+2
done
clear
C)
+3
done
clear
D)
-1
done
clear
View Answer play_arrow
question_answer 56) Which of the following molecules has pyramidal shape?
A)
\[PC{{l}_{3}}\]
done
clear
B)
\[S{{O}_{3}}\]
done
clear
C)
\[CO_{3}^{2-}\]
done
clear
D)
\[NO_{3}^{-}\]
done
clear
View Answer play_arrow
question_answer 57) Maximum number of hydrogen bonds in \[{{H}_{2}}O\] is
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 58) Number of isomers possible for \[{{C}_{4}}{{H}_{8}}O\] is
A)
3
done
clear
B)
4
done
clear
C)
5
done
clear
D)
6
done
clear
View Answer play_arrow
question_answer 59) \[{{C}_{6}}{{H}_{5}}CH=CHCHO\xrightarrow{X}{{C}_{6}}{{H}_{5}}CH\]\[=CHC{{H}_{2}}OH\] In the above sequence X can be
A)
\[{{H}_{2}}/Ni\]
done
clear
B)
\[NaE{{H}_{4}}\]
done
clear
C)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}/{{H}^{+}}\]
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 60) For a reaction \[{{H}_{2}}+{{I}_{2}}2HI\] at 721 K, the value of equilibrium constant is 50. If 0.5 moles each of \[{{H}_{2}}\] and \[{{I}_{2}}\] is added to the system the value of equilibrium constant will be
A)
40
done
clear
B)
60
done
clear
C)
50
done
clear
D)
30
done
clear
View Answer play_arrow
question_answer 61) Schottky defect generally appears in
A)
NaCI
done
clear
B)
KCl
done
clear
C)
CsCI
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 62) The ability of a given substance to assume two or more crystalline structure is called
A)
amorphism
done
clear
B)
isomorphism
done
clear
C)
polymorphism
done
clear
D)
isomerism
done
clear
View Answer play_arrow
question_answer 63) A cricket ball of 0.5 kg is moving with a velocity of 100 m/s. The wavelength associated with its motion is
A)
1/100 cm
done
clear
B)
\[6.6\times {{10}^{-34}}m\]
done
clear
C)
\[1.32\times {{10}^{-35}}m\]
done
clear
D)
\[6.6\times {{10}^{-28}}m\]
done
clear
View Answer play_arrow
question_answer 64) Which among the following species have the same number of electrons in its outermost as well as penultimate shell?
A)
\[M{{g}^{2+}}\]
done
clear
B)
\[{{O}^{2-}}\]
done
clear
C)
\[{{F}^{-}}\]
done
clear
D)
\[C{{a}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 65) \[\Delta {{E}^{o}}\] of combustion of isobutylene is\[-X\,kJ\,mo{{l}^{-1}}\]. The value of \[\Delta {{H}^{o}}\] is
A)
\[=\Delta {{E}^{o}}\]
done
clear
B)
\[>\Delta {{E}^{o}}\]
done
clear
C)
= 0
done
clear
D)
\[<\Delta {{E}^{o}}\]
done
clear
View Answer play_arrow
question_answer 66) At \[{{90}^{o}}C\], pure water has \[{{H}_{3}}{{O}^{+}}\] ion concentration \[{{10}^{-6}}mol\,{{L}^{-1}}\]. The \[{{K}_{w}}\] at \[{{90}^{o}}C\] is
A)
\[{{10}^{-6}}\]
done
clear
B)
\[{{10}^{-14}}\]
done
clear
C)
\[{{10}^{-12}}\]
done
clear
D)
\[{{10}^{-8}}\]
done
clear
View Answer play_arrow
question_answer 67) A gas is found to have a formula \[{{[CO]}_{x}}\]. If its vapour density is 70, the value of x is
A)
2.5
done
clear
B)
3
done
clear
C)
5
done
clear
D)
6
done
clear
View Answer play_arrow
question_answer 68) Which of the following represents soap?
A)
\[{{C}_{17}}{{H}_{35}}COOK\]
done
clear
B)
\[{{C}_{17}}{{H}_{35}}COOH\]
done
clear
C)
\[{{C}_{15}}{{H}_{31}}COOH\]
done
clear
D)
\[{{({{C}_{17}}{{H}_{35}}COO)}_{2}}Ca\]
done
clear
View Answer play_arrow
question_answer 69) Aspirin is chemically
A)
methyl benzoate
done
clear
B)
ethyl salicylate
done
clear
C)
acetyl salicylic acid
done
clear
D)
o-hydroxy benzoic acid
done
clear
View Answer play_arrow
question_answer 70) Vitamin \[{{B}_{6}}\] is known as
A)
pyndoxine
done
clear
B)
thiamine
done
clear
C)
tocopherol
done
clear
D)
riboflavin
done
clear
View Answer play_arrow
question_answer 71) Which of the following compounds is found abundantly in nature?
A)
Fructose
done
clear
B)
Starch
done
clear
C)
Glucose
done
clear
D)
Cellulose
done
clear
View Answer play_arrow
question_answer 72) Synthetic polymer which resembles natural rubber is
A)
neoprene
done
clear
B)
chloroprene
done
clear
C)
glyptal
done
clear
D)
nylon
done
clear
View Answer play_arrow
question_answer 73) Which of the following is not a nitro derivative?
A)
\[{{C}_{6}}{{H}_{5}}N{{O}_{2}}\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}ONO\]
done
clear
C)
done
clear
D)
\[{{C}_{6}}{{H}_{4}}(OH)N{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 74) The reduction of which of the following compound would yield secondary amine?
A)
Alkylnitrile
done
clear
B)
Carbylamine
done
clear
C)
Primary amine
done
clear
D)
Secondary nitro compound
done
clear
View Answer play_arrow
question_answer 75) Which of the aldehyde is most reactive?
A)
\[{{C}_{6}}{{H}_{5}}-CHO\]
done
clear
B)
\[C{{H}_{3}}CHO\]
done
clear
C)
HCHO
done
clear
D)
All the equally reactive
done
clear
View Answer play_arrow
question_answer 76) Which of the following does not contain ?COOH group?
A)
Aspirin
done
clear
B)
Benzoic acid
done
clear
C)
Picric acid
done
clear
D)
All have?COOH group
done
clear
View Answer play_arrow
question_answer 77) Which of the following is dihydric alcohol?
A)
Glycerol
done
clear
B)
Ethylene glycol
done
clear
C)
Catechol
done
clear
D)
Resorcinol
done
clear
View Answer play_arrow
question_answer 78) Ethyl alcohol is heated with cone. H2S04. The product formed is
A)
\[C{{H}_{3}}-\overset{\begin{smallmatrix} O \\ || \end{smallmatrix}}{\mathop{C}}\,-O{{C}_{2}}{{H}_{5}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{4}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
View Answer play_arrow
question_answer 79) In the first order reaction, the concentration of the reactants is reduced to 25% in one hour. The half-life period of the reaction is
A)
2h
done
clear
B)
4h
done
clear
C)
1/2h
done
clear
D)
1/4 h
done
clear
View Answer play_arrow
question_answer 80) For a reaction, X(g) \[\xrightarrow{{}}\] Y(g) + Z(g) the half-life period is 10 min. In what period of time would the concentration of X be reduced to 10% of original concentration?
A)
20 min
done
clear
B)
33 min
done
clear
C)
15 mm
done
clear
D)
25 min
done
clear
View Answer play_arrow
question_answer 81) The molar freezing point constant for water is\[-{{1.86}^{o}}C\]. If 342 g of cane sugar \[({{C}_{12}}{{H}_{22}}{{O}_{11}})\] is dissolved in 1000 g of water, the solution will freeze at
A)
\[-{{1.86}^{o}}C\]
done
clear
B)
\[{{1.86}^{o}}C\]
done
clear
C)
\[{{3.92}^{o}}C\]
done
clear
D)
\[{{2.42}^{o}}C\]
done
clear
View Answer play_arrow
question_answer 82) The movement of solvent molecules through a semipermeable membrane is called
A)
electrolysis
done
clear
B)
electrophoresis
done
clear
C)
osmosis
done
clear
D)
cataphoresis
done
clear
View Answer play_arrow
question_answer 83) Which of the following is a primary halide?
A)
150-propyl iodide
done
clear
B)
Secondary butyl iodide
done
clear
C)
Tertiary butyl bromide
done
clear
D)
neo-hexyl chloride
done
clear
View Answer play_arrow
question_answer 84) A gas is found to have the formula \[{{(CO)}_{n}}\]. If its vapour density is 56, the value of n will be
A)
7
done
clear
B)
5
done
clear
C)
4
done
clear
D)
3
done
clear
View Answer play_arrow
question_answer 85) Aromatisation of n-heptane by passing over \[(A{{l}_{2}}{{O}_{3}}+C{{r}_{2}}{{O}_{3}})\] catalyst at 773 K gives
A)
benzene
done
clear
B)
toluene
done
clear
C)
mixture of Both (a) and (b)
done
clear
D)
heptylene
done
clear
View Answer play_arrow
question_answer 86) \[{{C}_{6}}{{H}_{5}}C{{H}_{3}}\xrightarrow{Cr{{O}_{2}}C{{l}_{2}}}Z\]In the given sequence Z is
A)
benzaldehyde
done
clear
B)
toluic acid
done
clear
C)
phenyl acetic acid
done
clear
D)
benzoic acid
done
clear
View Answer play_arrow
question_answer 87) Nitroethane can exhibit which one of the following kinds of isomerism?
A)
Metamerism
done
clear
B)
Optical activity
done
clear
C)
Tautomerism
done
clear
D)
Position isomerism
done
clear
View Answer play_arrow
question_answer 88) A compound has 3 chiral carbon atoms. The number of possible optical isomers it can have,
A)
3
done
clear
B)
3
done
clear
C)
8
done
clear
D)
4
done
clear
View Answer play_arrow
question_answer 89) \[4{{K}_{2}}C{{r}_{2}}{{O}_{7}}\xrightarrow{Heat}4{{K}_{2}}Cr{{O}_{4}}+3{{O}_{2}}+X\]. In the above reaction X is
A)
\[Cr{{O}_{3}}\]
done
clear
B)
\[Cr{{ }_{2}}{{O}_{7}}\]
done
clear
C)
\[C{{r}_{2}}{{O}_{3}}\]
done
clear
D)
\[Cr{{O}_{5}}\]
done
clear
View Answer play_arrow
question_answer 90) The coordination number and oxidation number of X in the following compound \[[\,X(S{{O}_{4}}){{(N{{H}_{3}})}_{5}}]Cl\] will be
A)
10 and 3
done
clear
B)
2 and 6
done
clear
C)
6 and 3
done
clear
D)
6 and 4
done
clear
View Answer play_arrow
question_answer 91) In the electrolysis of water, one faraday of electrical energy would evolve
A)
one mole of oxygen
done
clear
B)
one g atom of oxygen
done
clear
C)
8 g of oxygen
done
clear
D)
22.4 L of oxygen
done
clear
View Answer play_arrow
question_answer 92) In which of these processes platinum is used as a catalyst?
A)
Oxidation of ammonia to form \[HN{{O}_{3}}\]
done
clear
B)
Hardening of oils
done
clear
C)
Production of synthetic rubber
done
clear
D)
Synthesis of methanol
done
clear
View Answer play_arrow
question_answer 93) If the half-life of an isotope X is 10 yr, its decay constant is
A)
6.932 \[y{{r}^{-1}}\]
done
clear
B)
0.6932 \[y{{r}^{-1}}\]
done
clear
C)
0.06932 \[y{{r}^{-1}}\]
done
clear
D)
0.006932 \[y{{r}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 94) On strong heating sodium bicarbonate changes into
A)
sodium monoxide
done
clear
B)
sodium hydroxide
done
clear
C)
sodium carbonate
done
clear
D)
sodium peroxide
done
clear
View Answer play_arrow
question_answer 95) Which of the following is not an ore of magnesium?
A)
Camallite
done
clear
B)
Magnesite
done
clear
C)
Dolomite
done
clear
D)
Gypsum
done
clear
View Answer play_arrow
question_answer 96) Aluminium reacts with caustic soda to form
A)
aluminium hydroxide
done
clear
B)
aluminium oxide
done
clear
C)
sodium metaaluminate
done
clear
D)
sodium tetraaluminate
done
clear
View Answer play_arrow
question_answer 97) In laboratory burners, we use
A)
producer gas
done
clear
B)
oil gas
done
clear
C)
gobargas
done
clear
D)
coal gas
done
clear
View Answer play_arrow
question_answer 98) Iron is dropped in dil. \[HN{{O}_{3}}\], it gives
A)
ferric nitrate
done
clear
B)
ferric nitrate and \[N{{O}_{2}}\]
done
clear
C)
ferrous nitrate and ammonium nitrate
done
clear
D)
ferrous nitrate and nitric oxide
done
clear
View Answer play_arrow
question_answer 99) When tin is treated with concentrated nitric acid
A)
it converts into stannous nitrate
done
clear
B)
it converts into stannic nitrate
done
clear
C)
it converts into metastannic acid
done
clear
D)
it becomes passive
done
clear
View Answer play_arrow
question_answer 100) The chief impurity present in red bauxite is
A)
\[Si{{O}_{3}}\]
done
clear
B)
\[F{{e}_{2}}{{O}_{3}}\]
done
clear
C)
\[{{K}_{2}}S{{O}_{4}}\]
done
clear
D)
NaF
done
clear
View Answer play_arrow
question_answer 101) Cladistics can be best defined as
A)
the relationship between sub-species and species
done
clear
B)
relationship between two most forward races
done
clear
C)
relationship between endangered and surviving species
done
clear
D)
method of classification that attempt to interfere phylogenetic relationship
done
clear
View Answer play_arrow
question_answer 102) Saltatory conduction occurs in
A)
myelinated nerve fibres
done
clear
B)
non- myelinated nerve fibres
done
clear
C)
liver cells
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 103) Which one is related to urine concentration in mammals?
A)
Testosterone hormone
done
clear
B)
Antidiuretic hormone
done
clear
C)
Oxytocin hormone
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 104) The point by which Annelida advanced over Nematoda
A)
true coelom
done
clear
B)
metameric segmentation
done
clear
C)
closed circulation
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 105) Adjacent epithelial cells are held together by, means of
A)
microsomes
done
clear
B)
liposomes
done
clear
C)
desmosomes
done
clear
D)
glyoxysomes
done
clear
View Answer play_arrow
question_answer 106) An enzyme that uses viral RNA as template for the synthesis of DNA is
A)
RNA polymerase
done
clear
B)
reverse transcriptase
done
clear
C)
viral nuclease
done
clear
D)
RNA replicase
done
clear
View Answer play_arrow
question_answer 107) Photochemical smog mostly contains
A)
\[{{O}_{3}}\]
done
clear
B)
\[CO\]
done
clear
C)
\[P{{O}_{4}}\]
done
clear
D)
\[C{{H}_{4}}\]
done
clear
View Answer play_arrow
question_answer 108) The carrier which transfers the electrons in electron transport system
A)
phytochrome
done
clear
B)
cytochrome
done
clear
C)
quantasomes
done
clear
D)
fucoxanthin
done
clear
View Answer play_arrow
question_answer 109) The source of agar- agar is
A)
Chlamydornonas
done
clear
B)
Chlorella
done
clear
C)
Gelidium
done
clear
D)
Spirogyra
done
clear
View Answer play_arrow
question_answer 110) Aggregate fruit is found in
A)
Ananas sativus
done
clear
B)
Annona squamosal
done
clear
C)
Artocarpus integrifolia
done
clear
D)
Pyrus malus
done
clear
View Answer play_arrow
question_answer 111) A term helotism is used for the symbiosis of
A)
algae and fungi
done
clear
B)
algae and Cycas
done
clear
C)
algae and bacteria
done
clear
D)
Pinus and fungi
done
clear
View Answer play_arrow
question_answer 112) Kupffer cells are found in
A)
liver
done
clear
B)
heart
done
clear
C)
blood
done
clear
D)
kidney
done
clear
View Answer play_arrow
question_answer 113) Which one of these is associated with multiple phenotypes?
A)
Mutation
done
clear
B)
Pleiotropy
done
clear
C)
Epistasis
done
clear
D)
Polygenic inheritance
done
clear
View Answer play_arrow
question_answer 114) First \[C{{O}_{2}}\]acceptor in \[{{C}_{4}}\]plants is
A)
PEP
done
clear
B)
PGA
done
clear
C)
RuBP
done
clear
D)
Pyruvic acid
done
clear
View Answer play_arrow
question_answer 115) The blood group known as universal acceptor is
A)
A
done
clear
B)
B
done
clear
C)
AB
done
clear
D)
O
done
clear
View Answer play_arrow
question_answer 116) The common point of attachment of all the arms of polytene chromosome is known as
A)
cencromere
done
clear
B)
chromomere
done
clear
C)
chromocentre
done
clear
D)
centrosome
done
clear
View Answer play_arrow
question_answer 117) In the members of family- Malvaceae anthers are described as
A)
diadelphous and dithecous
done
clear
B)
diadelphous and monothecous
done
clear
C)
monoadelphous and monothecous
done
clear
D)
monoadelphous and dithecous
done
clear
View Answer play_arrow
question_answer 118)
Column- I lists the components of body defense and column-II lists the corresponding descriptions. Match the two columns. Choose the correct option from those given Column- I Column- II A. Active natural B. First line of defense C. Passive natural immunity D. Second line of defense 1. Injection of gamma globulins 2. Complement proteins and interferons 3. Direct contact with the pathogens that have entered inside 4. Surface barriers 5. Antibodies transferred through the placenta
A)
A B C D 4 3 5 2
done
clear
B)
3 4 2 5
done
clear
C)
3 4 5 2
done
clear
D)
5 3 2 1
done
clear
View Answer play_arrow
question_answer 119) Haploid structure of Funaria is
A)
calyptras
done
clear
B)
protonema
done
clear
C)
apophysis
done
clear
D)
operculum
done
clear
View Answer play_arrow
question_answer 120) Sal and teak are dominant in
A)
tropical rain forest
done
clear
B)
temperate broad leaf forest
done
clear
C)
temperate needle leaf forest
done
clear
D)
tropical deciduous forest
done
clear
View Answer play_arrow
question_answer 121) In the angiosperm ovule, central cell of the embryo sac, prior to the entry of pollen tube, contains
A)
a single haploid nucleus
done
clear
B)
one diploid and one haploid nuclei
done
clear
C)
two haploid polar nuclei
done
clear
D)
one diploid secondary nucleus
done
clear
View Answer play_arrow
question_answer 122) Which of the following represents a condition where the motility of the sperms is highly reduced?
A)
Oligospermia
done
clear
B)
Athenospermia
done
clear
C)
Azoospermia
done
clear
D)
Polyspermy
done
clear
View Answer play_arrow
question_answer 123) Which of the following is not an influence auxins?
A)
Apical dominance
done
clear
B)
Parthenocarpy
done
clear
C)
Tropic movements
done
clear
D)
Bolting
done
clear
View Answer play_arrow
question_answer 124) Which type of cells is absent in sponges?
A)
Trophocytes
done
clear
B)
Myocytes
done
clear
C)
Archaeocytes
done
clear
D)
Cnidocytes
done
clear
View Answer play_arrow
question_answer 125) Which one of the following pairs of the kind of cells and their secretion is correctly matched?
A)
Oxyntic cells?a secretion with pH between 2.0 and 3.0
done
clear
B)
Alpha cells of islets of Langerhans secretion that decreases blood sugar level
done
clear
C)
Kupffer cells?a digestive enzyme that hydrolyses nucleic acids
done
clear
D)
Sebaceous glands?a secretion that evaporates for cooling
done
clear
View Answer play_arrow
question_answer 126) Which plant hormone promotes seed, bud dormancy and causes stomatal closure?
A)
I AA
done
clear
B)
Abscisic acid
done
clear
C)
\[G{{A}_{1}}\]
done
clear
D)
Cytokinin
done
clear
View Answer play_arrow
question_answer 127) Green-house effect is due to the increased concentration of
A)
\[C{{O}_{2}}\]
done
clear
B)
\[Ne\]
done
clear
C)
\[S{{O}_{2}}\]
done
clear
D)
\[N{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 128) Sickle cell anaemia has not been eliminated from the African population because
A)
it is controlled by recessive genes
done
clear
B)
it is not a fatal disease
done
clear
C)
it provides immunity against malaria
done
clear
D)
it is controlled by dominant genes
done
clear
View Answer play_arrow
question_answer 129) Mycorrhiza helps in
A)
nutrition up taking
done
clear
B)
food manufacturing
done
clear
C)
disease resistance
done
clear
D)
disease prevention
done
clear
View Answer play_arrow
question_answer 130) Fever in malaria is due to
A)
release of merozoites from RBCs
done
clear
B)
entry of sporozoites into blood capillaries
done
clear
C)
entry of cryptomerozoites into RBCs
done
clear
D)
entry of merozoites into liver cells
done
clear
View Answer play_arrow
question_answer 131) Protein in silk thread is
A)
keratin
done
clear
B)
globulin
done
clear
C)
fibroin
done
clear
D)
albumin
done
clear
View Answer play_arrow
question_answer 132) Which of the following is purely motor cranial nerve?
A)
Optic
done
clear
B)
Olfactory
done
clear
C)
Vagus
done
clear
D)
Abducens
done
clear
View Answer play_arrow
question_answer 133) Which one of the following is correctly matched?
A)
Body louse - Typhoid
done
clear
B)
House fly - Yellow fever
done
clear
C)
Anopheles - Malaria
done
clear
D)
Aedes - Plague
done
clear
View Answer play_arrow
question_answer 134) Which one of the following is not correctly matched?
A)
Helminthosporium - Bengal famine
done
clear
B)
Melanospora - Red rust of tea
done
clear
C)
Aspergillus - Aflatoxins
done
clear
D)
Cercospora - Tikka disease of groundnut
done
clear
View Answer play_arrow
question_answer 135) In earthworm self fertilization cannot occur due to
A)
protogyny
done
clear
B)
protoandry
done
clear
C)
epigyny
done
clear
D)
hypogyny
done
clear
View Answer play_arrow
question_answer 136) Which one of the following parasite shows alternation of generation and alternation of host in its life cycle?
A)
Fasdola
done
clear
B)
Ascaris
done
clear
C)
Wuchereria
done
clear
D)
Taenia
done
clear
View Answer play_arrow
question_answer 137) Which of the following is an opiate narcotic?
A)
Morphine
done
clear
B)
LSD
done
clear
C)
Amphetamines
done
clear
D)
Barbiturates
done
clear
View Answer play_arrow
question_answer 138) Amphids are cuticular elevations on ventrolateral lips of Ascar is. These are
A)
tangoreceptors
done
clear
B)
taetoreceptors
done
clear
C)
olfactoreceptors
done
clear
D)
chemoreceptors
done
clear
View Answer play_arrow
question_answer 139) The chemicals which are produced by host plants due to infection as a defence reaction to pathogen are called
A)
toxin
done
clear
B)
phytotoxin
done
clear
C)
phytotron
done
clear
D)
phytoalexins
done
clear
View Answer play_arrow
question_answer 140) Meiosis in Dryopteris takes place during
A)
gamete formation
done
clear
B)
spore germination
done
clear
C)
zygote formation
done
clear
D)
spore formation
done
clear
View Answer play_arrow
question_answer 141) Which malaria parasite has longest incubation period?
A)
Plasmodium vivax
done
clear
B)
P. falciparum
done
clear
C)
P. malariae
done
clear
D)
P. ovate
done
clear
View Answer play_arrow
question_answer 142) Scurvy is caused due to deficiency of vitamin
A)
B
done
clear
B)
A
done
clear
C)
E
done
clear
D)
C
done
clear
View Answer play_arrow
question_answer 143) Pollination by snail and slug is known as
A)
entomophily
done
clear
B)
ornithophily
done
clear
C)
anemophily
done
clear
D)
malacophily
done
clear
View Answer play_arrow
question_answer 144) In which group of the following would you place the plants having vascular tissue and lacking seeds?
A)
Algae
done
clear
B)
Fungi
done
clear
C)
Bryophytes
done
clear
D)
Pteridophytes
done
clear
View Answer play_arrow
question_answer 145) The long bones are hollow and connected by air passage. They are characteristic of
A)
Mammalia
done
clear
B)
Aves
done
clear
C)
Reptilia
done
clear
D)
Sponges
done
clear
View Answer play_arrow
question_answer 146) Inflorescence of Ficus is
A)
raceme
done
clear
B)
spike
done
clear
C)
hypanthodium
done
clear
D)
verticillaster
done
clear
View Answer play_arrow
question_answer 147) The plant hormone controlling fruit ripening is
A)
IAA
done
clear
B)
GA
done
clear
C)
Ethylene
done
clear
D)
ABA
done
clear
View Answer play_arrow
question_answer 148) Which is the example of conditioned reflex?
A)
Your kneeing took up a stone than dog run away
done
clear
B)
Eye closed when anything enter into it
done
clear
C)
Hand took up when piercing with needle
done
clear
D)
Digestive food goes forward in alimentary canal
done
clear
View Answer play_arrow
question_answer 149) HIV has a protein coat and genetic material
A)
ssRNA
done
clear
B)
dsRNA
done
clear
C)
ssDNA
done
clear
D)
dsDNA
done
clear
View Answer play_arrow
question_answer 150) Colchicine prevents the mitosis of cells at which of the following stage?
A)
Anaphase
done
clear
B)
Metaphase
done
clear
C)
Prophase
done
clear
D)
Interphase
done
clear
View Answer play_arrow
question_answer 151) The poisonous fluid present in nematocyst of Hydra is
A)
venom
done
clear
B)
haematin
done
clear
C)
toxin
done
clear
D)
hypnotoxin
done
clear
View Answer play_arrow
question_answer 152) XXY chromosome constitution is represented by
A)
Downs syndrome
done
clear
B)
Turners syndrome
done
clear
C)
Klinefelters syndrome
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 153) The fruit which develops from inflorescence is called
A)
achene
done
clear
B)
berry
done
clear
C)
etaerio
done
clear
D)
composite fruit
done
clear
View Answer play_arrow
question_answer 154) The enzyme responsible for the reduction of molecular nitrogen to the level of ammonia in the leguminous root nodule is
A)
amminase
done
clear
B)
nitrogenase
done
clear
C)
nitrate reductase
done
clear
D)
nitrite reductase
done
clear
View Answer play_arrow
question_answer 155) Hydrolytic enzymes are found in
A)
lysosomes
done
clear
B)
peroxisomes
done
clear
C)
lomasomes
done
clear
D)
ribosomes
done
clear
View Answer play_arrow
question_answer 156) In Cycas diploxylic vascular bundles are found in
A)
stem
done
clear
B)
root
done
clear
C)
leaflet
done
clear
D)
rachis and leaflet
done
clear
View Answer play_arrow
question_answer 157) The binomial names were accepted by all after the publication of the work by
A)
Linnaeus
done
clear
B)
Hooker
done
clear
C)
Bentham
done
clear
D)
Darwin
done
clear
View Answer play_arrow
question_answer 158) Which of the following is used in genetic engineering?
A)
Plastid
done
clear
B)
Plasmid
done
clear
C)
Mitochondria
done
clear
D)
ER
done
clear
View Answer play_arrow
question_answer 159) If frogs brain is crushed, even than its leg moves on pinpointing. It is called as
A)
conditional reflex
done
clear
B)
simple reflex
done
clear
C)
neurotransmitter function
done
clear
D)
auconomic nerve conditions
done
clear
View Answer play_arrow
question_answer 160) When centromere is present at one end, the chromosome is
A)
metacentric
done
clear
B)
excentric
done
clear
C)
telocentric
done
clear
D)
apocentric
done
clear
View Answer play_arrow
question_answer 161) Xenia refers to
A)
effect of pollen on stem
done
clear
B)
effect of pollen on taste of fruit
done
clear
C)
effect of pollen on vascular tissue
done
clear
D)
effect of pollen on endosperm
done
clear
View Answer play_arrow
question_answer 162) Curdling of milk in small intestine takes place due to
A)
trypsin
done
clear
B)
rennin
done
clear
C)
ptyalin
done
clear
D)
chymotrypsin
done
clear
View Answer play_arrow
question_answer 163) The pair of disease caused by virus is
A)
typhoid, tetanus
done
clear
B)
rabies, mumps
done
clear
C)
cholera, tuberculosis
done
clear
D)
AIDS, syphilis
done
clear
View Answer play_arrow
question_answer 164) Pasteurization is a process which means heating of drinks at
A)
120°C for 60 minutes
done
clear
B)
80°C for 30 minutes
done
clear
C)
60°-70°C for 30 minutes
done
clear
D)
70°C for 60 minutes
done
clear
View Answer play_arrow
question_answer 165) When an animal has both the characters of male and female, it is called
A)
intersex
done
clear
B)
super female
done
clear
C)
super male
done
clear
D)
gynandromorphy
done
clear
View Answer play_arrow
question_answer 166) An angiospermic leaf carries 16 chromosomes. The number of chromosomes in its endosperm will be
A)
16
done
clear
B)
24
done
clear
C)
12
done
clear
D)
8
done
clear
View Answer play_arrow
question_answer 167) Identify the correctly matched pair
A)
Gir forest ? Rhino
done
clear
B)
Kaziranga ? Elephant
done
clear
C)
Corbettpark ? Aves
done
clear
D)
Runn of Kutch ? Wild ass
done
clear
View Answer play_arrow
question_answer 168) Elater mechanism or spore dispersal is exhibited by
A)
Riccia
done
clear
B)
Funaria
done
clear
C)
Liverworts
done
clear
D)
Marchantia
done
clear
View Answer play_arrow
question_answer 169) Which of the following is living fossil?
A)
Gnetum
done
clear
B)
Cycas
done
clear
C)
Ginkgo
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
question_answer 170) The most common indicator organism which represents polluted water is
A)
Salmonella typhi
done
clear
B)
E. coli
done
clear
C)
Entamoeba histolydca
done
clear
D)
Vibrio cholera
done
clear
View Answer play_arrow
question_answer 171) The progenies are found to be male sterile after crossing two plants. This is due to some genes which are present in
A)
mitochondria
done
clear
B)
cytoplasm
done
clear
C)
nucleus
done
clear
D)
chloroplast
done
clear
View Answer play_arrow
question_answer 172) Gobar gas contains mainly
A)
\[C{{H}_{4}}+C{{O}_{2}}\]
done
clear
B)
\[C{{H}_{4}}+{{O}_{3}}\]
done
clear
C)
\[C{{O}_{2}}+{{H}_{2}}\]
done
clear
D)
\[C{{O}_{2}}+S{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 173) A fertilizing is produced from
A)
polar bodies
done
clear
B)
middle piece of sperm
done
clear
C)
acrosome
done
clear
D)
mature eggs
done
clear
View Answer play_arrow
question_answer 174) Heterospory and seed habit is exhibited by a plant having
A)
petiole
done
clear
B)
spa the
done
clear
C)
prothallus
done
clear
D)
ligule
done
clear
View Answer play_arrow
question_answer 175) Which of the following best shown the common origin of man and chimpanzee?
A)
Chromosome banding
done
clear
B)
Binocular vision
done
clear
C)
Cranial capacity
done
clear
D)
Dental formula
done
clear
View Answer play_arrow
question_answer 176) The vitamin which can be synthesized by bacteria inside
A)
B
done
clear
B)
D
done
clear
C)
K
done
clear
D)
E
done
clear
View Answer play_arrow
question_answer 177) Free living aerobic non-photosynthetic nitrogen fixing bacterium is
A)
Azotobacter
done
clear
B)
B. coli
done
clear
C)
Nostoc
done
clear
D)
Salmonella
done
clear
View Answer play_arrow
question_answer 178) In an ecosystem, keystone are those which
A)
are most frequent
done
clear
B)
are present in maximum number
done
clear
C)
attain a large biomass
done
clear
D)
contribute to ecosystem properties
done
clear
View Answer play_arrow
question_answer 179) Which of the following hormone stimulates the stomach to secrete gastric juice?
A)
Gastrin
done
clear
B)
Enterokinase
done
clear
C)
Rennin
done
clear
D)
Zymase
done
clear
View Answer play_arrow
question_answer 180) Which one of the following contributes to social forestry?
A)
Leucaena leucocephala
done
clear
B)
Mangifera indica
done
clear
C)
Jatropha
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 181) The meristem responsible for extra stellar secondary growth in dicot stem is
A)
inter fascicular cambium
done
clear
B)
intra fascicular cambium
done
clear
C)
inter calary meristem
done
clear
D)
phellogen
done
clear
View Answer play_arrow
question_answer 182) The smallest known agents of infectious disease are
A)
viruses
done
clear
B)
mycoplasma
done
clear
C)
viroids
done
clear
D)
Actinomycetes
done
clear
View Answer play_arrow
question_answer 183) The plant most commonly used as green manure is
A)
Delbergia sissoo
done
clear
B)
Polyalthea
done
clear
C)
Sesbania aculeate
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 184) Minamata disease prevalent in industrial area due to pollution is due to
A)
lead
done
clear
B)
cadmium
done
clear
C)
mercury
done
clear
D)
zinc
done
clear
View Answer play_arrow
question_answer 185) As a fungus completes its life cycle on two hosts, it is termed as
A)
heteroecious
done
clear
B)
autoecious
done
clear
C)
heterothallic
done
clear
D)
monothallic
done
clear
View Answer play_arrow
question_answer 186) Beer and butter milk are products of fermentation by
A)
Rhizopus stolonifer
done
clear
B)
Caedobacter taeniospiralls
done
clear
C)
Bacillus subtilis
done
clear
D)
Saccharomyces cerevisae
done
clear
View Answer play_arrow
question_answer 187) The Indian wild ass is in the category of........... by wild life protection act of government of India.
A)
rare species
done
clear
B)
endangered species
done
clear
C)
endemic species
done
clear
D)
vulnerable species
done
clear
View Answer play_arrow
question_answer 188) Which of the following is commonly called emergency gland of body?
A)
Thymus
done
clear
B)
Testis
done
clear
C)
Adrenal
done
clear
D)
Pituitary
done
clear
View Answer play_arrow
question_answer 189) The set of ions necessary for muscle contraction is
A)
\[C{{a}^{2+}}\]and\[M{{g}^{2+}}\]
done
clear
B)
\[N{{a}^{+}}\]and \[M{{g}^{2+}}\]
done
clear
C)
\[N{{a}^{+}}\]and \[{{K}^{+}}\]
done
clear
D)
\[N{{a}^{+}}\] and \[C{{a}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 190) A haemophilic man marries a carrier woman, their daughter progenies are
A)
all haemophilic
done
clear
B)
half haemophilic
done
clear
C)
half haemophilic and half carrier
done
clear
D)
all normal
done
clear
View Answer play_arrow
question_answer 191) Insectivorous plants grow in deficient soil
A)
Na
done
clear
B)
N
done
clear
C)
C
done
clear
D)
\[{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 192) In an ovule the wall curvature is more pronounced and embryo sac becomes horse shoe shaped. The ovule is
A)
campylotropous
done
clear
B)
anatropous
done
clear
C)
amphitropous
done
clear
D)
orthotropous
done
clear
View Answer play_arrow
question_answer 193) Genetically engineered human insulin humulin was launched by American drug company on
A)
5th July 1998
done
clear
B)
5th July 1993
done
clear
C)
5th July 1973
done
clear
D)
5th July 1983
done
clear
View Answer play_arrow
question_answer 194) Uterine endometrium, epithelial glands and connective tissue are broken in menstrual phase. This is due to
A)
over secretion of FSH
done
clear
B)
lack of estrogen
done
clear
C)
lack of progesterone
done
clear
D)
over production of progesterone
done
clear
View Answer play_arrow
question_answer 195) The structure in Amoeba functionally similar to human kidney is
A)
nucleus
done
clear
B)
plasmodesmata
done
clear
C)
plasma membrane
done
clear
D)
contractile vacuole
done
clear
View Answer play_arrow
question_answer 196) Phenomenon of industrial melanism demonstrates
A)
induced mutation
done
clear
B)
natural selection
done
clear
C)
reproductive isolation
done
clear
D)
geographical isolation
done
clear
View Answer play_arrow
question_answer 197) Organs which differ in origin but performing similar function are
A)
analogous
done
clear
B)
homologous
done
clear
C)
vestigial
done
clear
D)
atavism
done
clear
View Answer play_arrow
question_answer 198) In Mollusca the osphraidium has function of
A)
reproduction
done
clear
B)
respiration
done
clear
C)
testing physical and chemical qualities of food
done
clear
D)
excretion
done
clear
View Answer play_arrow
question_answer 199) The stomata in CAM plants open during
A)
day
done
clear
B)
night
done
clear
C)
Both (a) and (b)
done
clear
D)
always closed
done
clear
View Answer play_arrow
question_answer 200) The organelles which take part in photo respiration are
A)
chloroplast, mitochondria, nucleus
done
clear
B)
chloroplast, mitochondria, lysosome
done
clear
C)
mitochondria, chloroplast, peroxisome
done
clear
D)
mitochondria, lysosomes, peroxisome
done
clear
View Answer play_arrow
 \[\left( \frac{2+2\sqrt{3}}{3} \right)\,\overset{\hat{\ }}{\mathop{i}}\,-\frac{2}{3}\overset{\hat{\ }}{\mathop{j}}\,\] \[4\overset{\hat{\ }}{\mathop{i}}\,\]
\[\left( \frac{2+2\sqrt{3}}{3} \right)\,\overset{\hat{\ }}{\mathop{i}}\,-\frac{2}{3}\overset{\hat{\ }}{\mathop{j}}\,\] \[4\overset{\hat{\ }}{\mathop{i}}\,\]