question_answer 1) The quantities RC and \[\left( \frac{L}{R} \right)\] ( where R, L and C stand for resistance, inductance and capacitance respectively) have the dimensions of
A)
force
done
clear
B)
linear momentum
done
clear
C)
linear velocity
done
clear
D)
time
done
clear
View Answer play_arrow
question_answer 2) The correct vector relation between linear velocity \[\vec{v}\] and angular velocity\[\vec{\omega }\]in rigid body dynamics is ( where \[\vec{r}\] is the postion vector)
A)
\[\vec{\omega }=\vec{v}\times \vec{r}\]
done
clear
B)
\[\vec{v}=\vec{r}/\vec{\omega }\]
done
clear
C)
\[\vec{v}=\vec{\omega }\times \vec{r}\]
done
clear
D)
\[\vec{r}=\vec{v}\times \vec{\omega }\]
done
clear
View Answer play_arrow
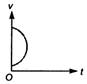
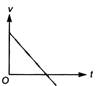
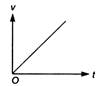
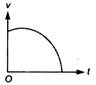
question_answer 3) Which of the following cannot be speed-time graph?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 4) Magnitudes of four pairs of displacement vectors are given. Which pair of displacement vectors, under vector addition, fails to give a resultant vector of magnitude 3 cm?
A)
2 cm, 7 cm
done
clear
B)
1 cm, 4 cm
done
clear
C)
2 cm, 3 cm
done
clear
D)
2 cm, 4 cm
done
clear
View Answer play_arrow
question_answer 5) The maximum range of a projectile is 100 m. The maximum height reached by it is
A)
100 m
done
clear
B)
25 m
done
clear
C)
200 m
done
clear
D)
75 m
done
clear
View Answer play_arrow
question_answer 6) The working principle of rocket propulsion is conservation of
A)
angular momentum
done
clear
B)
mass
done
clear
C)
linear momentum
done
clear
D)
kinetic energy
done
clear
View Answer play_arrow
question_answer 7) In inelastic collision
A)
momentum, kinetic energy and total energy are conserved
done
clear
B)
momentum, kinetic energy and total energy are not conserved
done
clear
C)
momentum, and kinetic energy are conserved but total energy is not conserved
done
clear
D)
total energy and momentum are conserved but kinetic energy is not conserved
done
clear
View Answer play_arrow
question_answer 8) A cricket ball of mass 0.5 kg strikes a cricket bat normally with a velocity of 20 ms 1 and rebounds with velocity of 10 ms-1. The impulse of the force exerted by the ball on the bat is
A)
15 Ns
done
clear
B)
25 Ns
done
clear
C)
30 Ns
done
clear
D)
10 Ns
done
clear
View Answer play_arrow
question_answer 9) Assuming earth to be an inertial frame, an example for inertial frame observer is
A)
a driver in a train which is slowing down to stop
done
clear
B)
a person in a car moving with uniform velocity
done
clear
C)
a girl revolving in a merry-go-round
done
clear
D)
a passenger in an aircraft which is taking off
done
clear
View Answer play_arrow
question_answer 10) If the force acting on a body is inversely proportional to its speed, then its kinetic energy is
A)
linearly related to time
done
clear
B)
inversely proportional to time
done
clear
C)
inversely proportional to the square of time
done
clear
D)
a constant
done
clear
View Answer play_arrow
question_answer 11) A body, possessing kineric energy T, moving on a rough horizontal surface, is stopped in a distance y. The frictional force exerted on the body is
A)
Ty
done
clear
B)
\[\frac{\sqrt{T}}{y}\]
done
clear
C)
\[\frac{T}{y}\]
done
clear
D)
\[\frac{T}{\sqrt{y}}\]
done
clear
View Answer play_arrow
question_answer 12) A particle is describing uniform circular motion. Its acceleration is
A)
along the radius of the circular path pointing towards the centre
done
clear
B)
along the tangent to the circular path
done
clear
C)
along the radius of the circular path pointing away from the centre
done
clear
D)
zero
done
clear
View Answer play_arrow
question_answer 13) \[{{I}_{1}},\]and \[{{I}_{2}}\]are the moments of inertia of two circular discs about their central axes perpendicular to their surfaces. Their angular frequencies of rotation are \[{{\omega }_{1}}\]and \[{{\omega }_{2}}\] respectively. If they are brought into contact face to face with their axes of rotation coinciding with each other, the angular frequency of the composite disc will be
A)
\[\frac{{{I}_{1}}+{{I}_{2}}}{{{\omega }_{1}}+{{\omega }_{2}}}\]
done
clear
B)
\[\frac{{{I}_{2}}{{\omega }_{1}}-{{I}_{1}}{{\omega }_{2}}}{{{I}_{1}}-{{I}_{2}}}\]
done
clear
C)
\[\frac{{{I}_{2}}{{\omega }_{1}}+{{I}_{1}}{{\omega }_{2}}}{{{I}_{1}}+{{I}_{2}}}\]
done
clear
D)
\[\frac{{{I}_{1}}{{\omega }_{1}}+{{I}_{2}}{{\omega }_{2}}}{{{I}_{1}}+{{I}_{2}}}\]
done
clear
View Answer play_arrow
question_answer 14) A child stands at one end of a boat moving with a speed v in still water. If the child starts running towards the other end of the boat with a speed u, the centre of mass of the system (boat and child) will move with a speed
A)
v - u
done
clear
B)
v
done
clear
C)
u
done
clear
D)
v +u
done
clear
View Answer play_arrow
question_answer 15) In planetary motion, the quantity that remains unchanged is
A)
radius of the orbit
done
clear
B)
speed along the orbit
done
clear
C)
total angular momentum
done
clear
D)
angular speed
done
clear
View Answer play_arrow
question_answer 16) Work done in taking a mass from one point to another in a gravitational field depends on
A)
the end points only
done
clear
B)
the path followed
done
clear
C)
the velocity of the mass
done
clear
D)
both the length of the path and the end points
done
clear
View Answer play_arrow
question_answer 17) A body is projected up from the surface of the earth with a velocity equal to \[\frac{3}{4}\]of its escape velocity. If R be the radius of earth, the height it reaches is
A)
\[\frac{3R}{10}\]
done
clear
B)
\[\frac{9R}{7}\]
done
clear
C)
\[\frac{8R}{5}\]
done
clear
D)
\[\frac{9R}{5}\]
done
clear
View Answer play_arrow
question_answer 18) A satellite moving round the earth in circular orbit of radius r and speed v suddenly loses some of its energy. Then,
A)
r will increase and v will decrease
done
clear
B)
both r and v will decrease
done
clear
C)
both r and v will increase
done
clear
D)
r will decrease and v will increase
done
clear
View Answer play_arrow
question_answer 19) The stress required to double the length of wire of Youngs modulus Y is
A)
\[\frac{y}{2}\]
done
clear
B)
2y
done
clear
C)
Y
done
clear
D)
4Y
done
clear
View Answer play_arrow
question_answer 20) Water flows through a pipe of varying cross-section. Then the ratio of the speeds of water at two points 1 and 2, where the radii of the pipe are \[{{r}_{1}}\]and \[{{r}_{2}}\]is
A)
\[\frac{r_{2}^{2}}{r_{1}^{2}}\]
done
clear
B)
\[\frac{{{r}_{2}}}{{{r}_{1}}}\]
done
clear
C)
\[\frac{r_{1}^{2}}{r_{2}^{2}}\]
done
clear
D)
\[\frac{{{r}_{1}}}{{{r}_{2}}}\]
done
clear
View Answer play_arrow
question_answer 21) A piece of ice, with a stone embedded inside it, is floating in water contained in a vessel. When the ice melts completely, the level of water in vessel
A)
remains unchanged
done
clear
B)
rises
done
clear
C)
falls
done
clear
D)
falls in the beginning and rises to the same level later
done
clear
View Answer play_arrow
question_answer 22) The excess pressure inside a cylindrical drop of liquid or a cylindrical bubble of radius R in a liquid of surface tension T is
A)
\[\frac{T}{4R}\]
done
clear
B)
\[\frac{T}{R}\]
done
clear
C)
\[\frac{2T}{R}\]
done
clear
D)
\[\frac{4T}{R}\]
done
clear
View Answer play_arrow
question_answer 23) Two small spheres of radii r and 4r fall through a viscous liquid with the same terminal velocity. The ratio between the viscous forces acting on them is
A)
1 : 2
done
clear
B)
4 : 1
done
clear
C)
1:16
done
clear
D)
1 : 4
done
clear
View Answer play_arrow
question_answer 24) A certain quantity of heat energy is given to a diatomic ideal gas which expands at constant pressure. The fraction of the heat energy that is converted into work is
A)
2/3
done
clear
B)
2/7
done
clear
C)
1/5
done
clear
D)
1/7
done
clear
View Answer play_arrow
question_answer 25) When the temperature of a gas is increased
A)
its molecular kinetic energy increases
done
clear
B)
molecular potential energy decreases and molecular kinetic energy also decreases, total energy remaining constant
done
clear
C)
molecular potential energy increases and molecular kinetic energy decreases; total energy remaining constant
done
clear
D)
its molecular potential energy increases
done
clear
View Answer play_arrow
question_answer 26) Two monoatomic ideal gases A and B occupying the same volume V, are at the same temperature T and pressure p. If they are mixed, the resultant mixture has volume V and temperature T. The pressure of the mixture is
A)
p
done
clear
B)
\[\frac{p}{2}\]
done
clear
C)
4p
done
clear
D)
2p
done
clear
View Answer play_arrow
question_answer 27) The requirement for heat conduction to take place in a solid is
A)
density gradient
done
clear
B)
uniform density
done
clear
C)
temperature gradient
done
clear
D)
uniform temperature
done
clear
View Answer play_arrow
question_answer 28) A Carnots engine working between \[27{}^\circ C\] and \[127{}^\circ C\] has a work output of 200 J/cycle. The energy supplied to the engine from the source in each cycle is
A)
400 J
done
clear
B)
800 J
done
clear
C)
600 J
done
clear
D)
500 J
done
clear
View Answer play_arrow
question_answer 29) A particle is executing linear simple harmonic motion. The fraction of the total energy that is potential, when its displacement is \[\frac{1}{4}\] of the amplitude is
A)
1/16
done
clear
B)
1/8
done
clear
C)
1/2
done
clear
D)
¼
done
clear
View Answer play_arrow
question_answer 30) The equation \[y\text{ }=\text{ }A\text{ }sin\text{ }k(vt-x)\]represents\[\left( k=\frac{2\pi }{\lambda } \right)\]
A)
a plane progressive wave travelling along negative x-direction
done
clear
B)
a plane progressive wave travelling along positive X-direction
done
clear
C)
a stationary wave
done
clear
D)
a plane progressive wave travelling along positive K-direction
done
clear
View Answer play_arrow
question_answer 31) The third overtone of an open organ pipe is in resonance with the second overtone of a closed organ pipe. If the length of the open pipe is 8 cm, then the length of closed pipe is
A)
10 cm
done
clear
B)
8 cm
done
clear
C)
12 cm
done
clear
D)
5 cm
done
clear
View Answer play_arrow
question_answer 32) When a wave undergoes refraction
A)
its frequency changes
done
clear
B)
its amplitude changes
done
clear
C)
its velocity changes
done
clear
D)
both amplitude and frequency change
done
clear
View Answer play_arrow
question_answer 33) A sound wave with frequency 256 Hz falls normally on a perfectly reflecting wall. The shortest distance from the wall at which the air particles will have maximum amplitude of vibrations is nearly ( velocity of sound in air is\[336\text{ }m{{s}^{-1}}\])
A)
32.8 cm
done
clear
B)
50 cm
done
clear
C)
65.8 cm
done
clear
D)
25 cm
done
clear
View Answer play_arrow
question_answer 34) When air medium in which two charges kept apart at a distance r is replaced by a dielectric medium of dielectric constant K, the force between the charges
A)
remains unchanged
done
clear
B)
decreases K times
done
clear
C)
increases K times
done
clear
D)
increases K2 times
done
clear
View Answer play_arrow
question_answer 35) The magnitude of electric field E required to balance an oil drop of mass m, carrying charge q is (g = acceleration due to gravity)
A)
\[\frac{q}{m}\]
done
clear
B)
\[\frac{mg}{{{q}^{2}}}\]
done
clear
C)
mgq
done
clear
D)
\[\frac{mg}{q}\]
done
clear
View Answer play_arrow
question_answer 36)
Three charges -q, + Q and -q are placed in a straight line as shown.
A)
2
done
clear
B)
5.5
done
clear
C)
4
done
clear
D)
1.5
done
clear
View Answer play_arrow
question_answer 37) Electric flux emanating through a surface element \[d\vec{s}=5\hat{i}\] placed in an electric field \[\vec{E}=4\hat{i}+4\hat{j}+4\hat{k}\]is
A)
10 units
done
clear
B)
20 units
done
clear
C)
4 units
done
clear
D)
16 units
done
clear
View Answer play_arrow
question_answer 38) A parallel plate capacitor is charged to a potential of V volt. The battery is then disconnected and the distance between the plates of the capacitor is increased using an insulating handle. The potential difference between the plates of the capacitor will
A)
increase
done
clear
B)
decrease
done
clear
C)
not change
done
clear
D)
become zero
done
clear
View Answer play_arrow
question_answer 39) Energy stored per unit volume of a parallel plate capacitor having plate area A and plate separation d when charged to a potential of V volts is (air space in between the plates)
A)
\[\frac{1}{2}{{C}^{2}}{{V}^{2}}\]
done
clear
B)
\[\frac{{{q}^{2}}}{4C}\]
done
clear
C)
\[\frac{1}{2}{{\varepsilon }_{0}}\left( \frac{v}{d} \right)\]
done
clear
D)
\[\frac{1}{2}{{\varepsilon }_{0}}\left( \frac{{{v}^{2}}}{{{d}^{2}}} \right)\]
done
clear
View Answer play_arrow
question_answer 40) The mutual electrostatic potential energy between two protons which are at a distance of \[9\times {{10}^{-15}}m,\,in{{\,}_{19}}{{\mathsf{U}}^{235}}\]nucleus is
A)
\[1.56\times {{10}^{-14}}J\]
done
clear
B)
\[5.5\times {{10}^{-14}}J\]
done
clear
C)
\[2.56\times {{10}^{-14}}J\]
done
clear
D)
\[4.56\times {{10}^{-14}}J\]
done
clear
View Answer play_arrow
question_answer 41) The SI units of electron mobility are
A)
\[{{m}^{2}}{{s}^{-1}}{{v}^{-1}}\]
done
clear
B)
\[ms\,\,{{V}^{-1}}\]
done
clear
C)
\[m{{s}^{-1}}V\]
done
clear
D)
\[{{m}^{2}}{{s}^{-2}}{{V}^{-2}}\]
done
clear
View Answer play_arrow
question_answer 42) A copper wire and a silicon wire are heated from room temperature to \[60{}^\circ C\]. Then
A)
resistance of both the wires decreases
done
clear
B)
resistance of both the wires increases
done
clear
C)
resistance of copper wire decreases and that of silicon wire increases
done
clear
D)
resistance of copper wire increases and that of silicon wire decreases
done
clear
View Answer play_arrow
question_answer 43) The tungsten filaments of two electric bulbs are of the same length. If one of them gives 25 W power and the other 60 W power, then
A)
both the filaments are of same thickness
done
clear
B)
25 W bulb has thicker filament
done
clear
C)
60 W bulb has thicker filament
done
clear
D)
both the filaments have same cross-section area
done
clear
View Answer play_arrow
question_answer 44) Heater coil A takes t1 second to boil certain quantity of water. Heater coil B takes \[{{t}_{2}}\]second to boil same quantity of water. If A and B are connected in series, the time taken to boil the same quantity of water by the combination is
A)
\[\sqrt{{{t}_{1}}{{t}_{2}}}\]
done
clear
B)
\[{{t}_{1}}+{{t}_{2}}\]
done
clear
C)
\[\frac{1}{2}({{t}_{1}}+{{t}_{2}})\]
done
clear
D)
\[\frac{{{t}_{1}}{{t}_{2}}}{{{t}_{1}}+{{t}_{2}}}\]
done
clear
View Answer play_arrow
question_answer 45) When the cold and hot junctions of a thermocouple are interchanged, the thermo emf
A)
changes sign
done
clear
B)
remains the same
done
clear
C)
becomes zero
done
clear
D)
is doubled
done
clear
View Answer play_arrow
question_answer 46) A charge q coulomb makes n revolutions in one second in a circular orbit of radius r. The magnetic field at the centre of the orbit in \[N{{A}^{-1}}{{m}^{-1}}\]is
A)
\[\frac{2\pi rn}{q}\times {{10}^{-7}}\]
done
clear
B)
\[\left( \frac{2\pi q}{r} \right)\times {{10}^{-7}}\]
done
clear
C)
\[\left( \frac{2\pi q}{nr} \right)\times {{10}^{-7}}\]
done
clear
D)
\[\left( \frac{2\pi nq}{r} \right)\times {{10}^{-7}}\]
done
clear
View Answer play_arrow
question_answer 47) In a moving coil galvanometer, to make the field radial
A)
coil is wound on wooden frame
done
clear
B)
magnetic poles are cylindrically cut
done
clear
C)
a horse-shoe magnet is used
done
clear
D)
the number of windings in the coil is decreased
done
clear
View Answer play_arrow
question_answer 48) An electron travelling with velocity\[\vec{v}\], enters a region of space in which electric and magnetic fields exist. Then the electron goes undeflected for all values of fields
A)
if both electric and magnetic fields are normal to\[\vec{v}\]
done
clear
B)
if the magnetic field alone in normal to v
done
clear
C)
if both electric and magnetic fields are parallel to \[\vec{v}\]
done
clear
D)
if the electric field alone is normal to \[\vec{v}\]
done
clear
View Answer play_arrow
question_answer 49) Magnetic field at the centre of a coil in the form of a square of side 2 cm carrying a current of 1.414 A is
A)
\[8\times {{10}^{-5}}T\]
done
clear
B)
\[8\times {{10}^{-5}}T\]
done
clear
C)
\[1.5\times {{10}^{-5}}T\]
done
clear
D)
\[6\times {{10}^{-5}}T\]
done
clear
View Answer play_arrow
question_answer 50) The resistance of an ideal voltmeter is
A)
zero
done
clear
B)
low
done
clear
C)
high
done
clear
D)
infinity
done
clear
View Answer play_arrow
question_answer 51) The relation connecting magnetic susceptibility \[{{X}_{m}}\]and relative permeability \[\mu \] is
A)
\[{{X}_{m}}={{\mu }_{r}}+1\]
done
clear
B)
\[{{X}_{m}}={{\mu }_{r}}-1\]
done
clear
C)
\[{{X}_{m}}=\frac{1}{{{\mu }_{r}}}\]
done
clear
D)
\[{{X}_{m}}=3(1+{{\mu }_{r}})\]
done
clear
View Answer play_arrow
question_answer 52) Whenever there is a relative motion between a coil and a magnet, the magnitude of induced emf set up in the coil does not depend upon the
A)
relative speed between the coil and magnet
done
clear
B)
magnetic moment of the coil
done
clear
C)
resistance of the coil
done
clear
D)
number of turns in the coil
done
clear
View Answer play_arrow
question_answer 53) A uniformly wound coil of self-inductance \[1.2\times {{10}^{-4}}H\] and resistance 3\[\Omega \] is broken up into two identical coils. These coils are then connected parallely across a 6V battery of negligible resistance. The time constant for the current in the circuit is (neglect mutual inductance)
A)
\[0.4\times {{10}^{-\text{ }4}}s\]
done
clear
B)
\[0.2\times {{10}^{-\text{ }4}}s\]
done
clear
C)
\[0.5\times {{10}^{-4}}s\]
done
clear
D)
\[0.1\times {{10}^{-4}}s\]
done
clear
View Answer play_arrow
question_answer 54) In AC circuit Ohms law is applicable for
A)
instantaneous values of current and voltage only
done
clear
B)
rms values of current and voltage only
done
clear
C)
peak values of current and voltage only
done
clear
D)
all values of current and voltage
done
clear
View Answer play_arrow
question_answer 55) The average power dissipated in a pure capacitance AC circuit is
A)
CV
done
clear
B)
zero
done
clear
C)
\[\frac{1}{C{{V}^{2}}}\]
done
clear
D)
\[\frac{1}{4}C{{V}^{2}}\]
done
clear
View Answer play_arrow
question_answer 56) The instantaneous values of current and voltage in an AC circuit are given by \[I=6\sin \left( 100\pi t+\frac{\pi }{4} \right),\]\[V=5\sin \left( 100\pi t+\frac{\pi }{4} \right),then\]
A)
current leads the voltage by \[45{}^\circ \]
done
clear
B)
voltage leads the current by \[90{}^\circ \]
done
clear
C)
current leads the voltage by \[90{}^\circ \]
done
clear
D)
voltage leads the current by \[45{}^\circ \]
done
clear
View Answer play_arrow
question_answer 57) The velocity of an electromagnetic wave in vacuum can be changed by changing
A)
frequency
done
clear
B)
amplitude
done
clear
C)
wavelength
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 58) The relationship between phase difference \[\Delta \phi \] and the path difference \[\Delta x\]between two interfering waves is given by (\[\lambda \] = wavelength)
A)
\[\Delta x=\left( \frac{\lambda }{2\pi } \right)\Delta \phi \]
done
clear
B)
\[\Delta x=\left( \frac{2\pi }{\lambda } \right)\Delta \phi \]
done
clear
C)
\[\Delta \phi =\left( \frac{\lambda }{\pi } \right)\Delta x\]
done
clear
D)
\[\Delta \phi =(2\pi )\Delta x\]
done
clear
View Answer play_arrow
question_answer 59) In Youngs double slit experiment, the fringe width with light of wavelength 6000 \[_{A}^{0}\] is 3 mm. The fringe width, when the wavelength of light is changed to 4000 \[_{A}^{0}\]is
A)
3 mm
done
clear
B)
1 mm
done
clear
C)
2 mm
done
clear
D)
4 mm
done
clear
View Answer play_arrow
question_answer 60) If the width of the slit in single slit diffraction experiment is doubled, then the central maximum of diffraction pattern becomes
A)
broader and brighter
done
clear
B)
sharper and brighter
done
clear
C)
sharper and fainter
done
clear
D)
broader and fainter
done
clear
View Answer play_arrow
question_answer 61) Transverse nature of light was confirmed by the phenomenon of
A)
refraction of light
done
clear
B)
diffraction of light
done
clear
C)
dispersion of light
done
clear
D)
polarization of light
done
clear
View Answer play_arrow
question_answer 62) If a transparent parallel plate of uniform thickness t and refractive index \[\mu \] is interposed perpendicularly in the path of a light beam, the optical path is
A)
increased by\[(~\mu -\text{1})t\]
done
clear
B)
decreased by \[\mu \,t\]
done
clear
C)
decreased by \[(~\mu -1)t\]
done
clear
D)
increased by \[~\mu \,t\]
done
clear
View Answer play_arrow
question_answer 63) If the photoelectric work function for a metallic surface is 4.125 eV, the cut-off wavelength for photoelectric phenomenon for the surface is
A)
4500\[\overset{0}{\mathop{A}}\,\]
done
clear
B)
1700 \[\overset{0}{\mathop{A}}\,\]
done
clear
C)
2800 \[\overset{0}{\mathop{A}}\,\]
done
clear
D)
3000 \[\overset{0}{\mathop{A}}\,\]
done
clear
View Answer play_arrow
question_answer 64) The masses of two particles having same kinetic energies are in the ratio 1 : 2. Then their de-Broglie wavelengths are in the ratio
A)
2 : 1
done
clear
B)
1 : 2
done
clear
C)
\[\sqrt{2}:1\]
done
clear
D)
\[\sqrt{3}:1\]
done
clear
View Answer play_arrow
question_answer 65) Balmer series of hydrogen atom lies in
A)
microwave region
done
clear
B)
visible region
done
clear
C)
ultraviolet region
done
clear
D)
infrared region
done
clear
View Answer play_arrow
question_answer 66) The shortest wavelength of X-rays coming from an X-ray tube depends on the
A)
voltage applied to the tube
done
clear
B)
current in the tube
done
clear
C)
atomic number of target element
done
clear
D)
nature of gas in the tube
done
clear
View Answer play_arrow
question_answer 67) The nuclear force
A)
is purely an electrostatic force
done
clear
B)
obeys inverse square law of distance
done
clear
C)
is equal in strength to gravitational force
done
clear
D)
is short range force
done
clear
View Answer play_arrow
question_answer 68) A radioactive isotope A with a half-life of \[1.25\times {{10}^{10}}yr\]decays into B which is stable. A sample of rock from a planet is found to contain both A and B present in the ratio 1:15. The age of the rock is (in years)
A)
\[9.6\times {{10}^{10}}\]
done
clear
B)
\[4.2\times {{10}^{10}}\]
done
clear
C)
\[5\times {{10}^{10}}\]
done
clear
D)
\[~1.95\times {{10}^{10}}\]
done
clear
View Answer play_arrow
question_answer 69) Enriched uranium is used in nuclear reactors because, it contains greater proportion of
A)
\[{{U}^{238}}\]
done
clear
B)
\[{{U}^{235}}\]
done
clear
C)
\[{{U}^{239}}\]
done
clear
D)
\[{{U}^{233}}\]
done
clear
View Answer play_arrow
question_answer 70) The weakest bond in solids is
A)
van der Waals
done
clear
B)
metallic
done
clear
C)
covalent
done
clear
D)
ionic
done
clear
View Answer play_arrow
question_answer 71) In a semiconducting material l/5th of the total current is carried by the holes and the remaining is carried by the electrons. The drift speed of electrons is twice that of holes at this temperature, the ratio between the number densities of electrons and holes is
A)
21/6
done
clear
B)
5
done
clear
C)
3/8
done
clear
D)
2
done
clear
View Answer play_arrow
question_answer 72) In a transistor, the base is
A)
a conductor with high conductivity
done
clear
B)
an insulator
done
clear
C)
an extrinsic semiconductor
done
clear
D)
an intrinsic semiconductor
done
clear
View Answer play_arrow
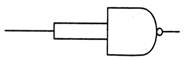
question_answer 73)
The following figure represents
A)
OR gate
done
clear
B)
NOT gate
done
clear
C)
NOR gate
done
clear
D)
AND gate
done
clear
View Answer play_arrow
question_answer 74) In amplitude modulation, the bandwidth is
A)
twice the audio signal frequency
done
clear
B)
thrice the audio signal frequency
done
clear
C)
thrice the carrier wave frequency
done
clear
D)
twice the carrier wave frequency
done
clear
View Answer play_arrow
question_answer 75) In a typical optical fibre, the difference between the refractive indices of core material and cladding material is of the order of
A)
\[{{10}^{-5}}\]
done
clear
B)
\[{{10}^{-6}}\]
done
clear
C)
\[{{10}^{-1}}\]
done
clear
D)
\[{{10}^{-3}}\]
done
clear
View Answer play_arrow
question_answer 76) The equivalent mass of potassium permanganate in alkaline medium is its molar
A)
\[\frac{molar\,mass}{5}\]
done
clear
B)
\[\frac{molar\,mass}{3}\]
done
clear
C)
\[\frac{molar\,mass}{2}\]
done
clear
D)
molar mass itself
done
clear
View Answer play_arrow
question_answer 77) The number of molecules in 18 mg of water in terms of Avogadro number N is
A)
\[{{10}^{-3}}N\]
done
clear
B)
\[{{10}^{-2}}N\]
done
clear
C)
\[{{10}^{-1}}N\]
done
clear
D)
\[10N\]
done
clear
View Answer play_arrow
question_answer 78) If the de-Broglie wavelength of a particle of mass m is 100 times its velocity then its value in terms of its mass (m) and Plancks constant (h) is
A)
\[\frac{1}{10}\sqrt{m/h}\]
done
clear
B)
\[10\sqrt{h/m}\]
done
clear
C)
\[\frac{1}{10}\sqrt{h/m}\]
done
clear
D)
\[10\sqrt{m/h}\]
done
clear
View Answer play_arrow
question_answer 79) How much volume of oxygen at STP, in litre is required to burn 4 g of methane gas completely?
A)
11.2
done
clear
B)
5.6
done
clear
C)
2.8
done
clear
D)
8
done
clear
View Answer play_arrow
question_answer 80) The set of quantum numbers\[n=4,l=0\text{ }m=0\] and\[s=+\frac{1}{2}\]correspond to the most loosely bound, ground state electron of which one of the following atoms?
A)
Na
done
clear
B)
\[Cl\]
done
clear
C)
Cr
done
clear
D)
\[Rb\]
done
clear
View Answer play_arrow
question_answer 81) The\[\beta \]decay of a radioactive element results in the formation of its
A)
isotope
done
clear
B)
isobar
done
clear
C)
isodiaphere
done
clear
D)
nuclear isomer
done
clear
View Answer play_arrow
question_answer 82) In the radioactive, decay, \[_{y}{{X}^{z}}{{\xrightarrow[{}]{(-8\text{ }\alpha \text{ }and\text{ }6\beta )}}_{82}}P{{b}^{2.6}},X,y\]and z are
A)
U, 92 and 235
done
clear
B)
Th, 90 and 232
done
clear
C)
Pu, 94 and 238
done
clear
D)
U, 92 and 238
done
clear
View Answer play_arrow
question_answer 83) In which one of the following equilibria, the increase of pressure over the equilibrium will favour the backward reaction?
A)
Decomposition equilibrium of\[HI\]
done
clear
B)
Formation equilibrium of\[S{{O}_{3}}\]
done
clear
C)
Decomposition equilibrium of\[N{{H}_{3}}\]
done
clear
D)
Formation equilibrium of\[PC{{l}_{5}}\]
done
clear
View Answer play_arrow
question_answer 84) Which one of the following is the correct quadratic form of the Ostwalds dilution law equation?
A)
\[{{\alpha }^{2}}C+\alpha K-K=0\]
done
clear
B)
\[{{\alpha }^{2}}C-\alpha K-K=0\]
done
clear
C)
\[{{\alpha }^{2}}C-\alpha K+K=0\]
done
clear
D)
\[{{\alpha }^{2}}C+\alpha K+K=0\]
done
clear
View Answer play_arrow
question_answer 85) Which one of the following aqueous solutions of salts has the lowest, pH value?
A)
\[C{{H}_{3}}COONa\]
done
clear
B)
\[NaCl\]
done
clear
C)
\[N{{H}_{4}}OOCC{{H}_{3}}\]
done
clear
D)
\[N{{H}_{4}}Cl\]
done
clear
View Answer play_arrow
question_answer 86) The solubility product of a sparingly soluble metal hydroxide\[M{{(OH)}^{2}}\]at 298 K is\[5\times {{10}^{-16}}\] \[mo{{l}^{3}}d{{m}^{-9}}\]. The pH value of its aqueous and saturated solution is
A)
5
done
clear
B)
9
done
clear
C)
11.5
done
clear
D)
2.5
done
clear
View Answer play_arrow
question_answer 87) In the synthesis of ammonia from nitrogen and hydrogen gases, if\[6\times {{10}^{-2}}\]moles of hydrogen disappears in 10 min, the number of moles of ammonia formed in 3 min is
A)
\[1.8\times {{10}^{-2}}\]
done
clear
B)
\[1.2\times {{10}^{-2}}\]
done
clear
C)
\[4.0\times {{10}^{-2}}\]
done
clear
D)
\[3.6\times {{10}^{-2}}\]
done
clear
View Answer play_arrow
question_answer 88) In a reversible reaction, the enthalpy change and the activation energy in the forward direction are respectively\[-x\text{ }kJmo{{l}^{-1}}\] and y \[kJmo{{l}^{-1}}\]. Therefore, the energy of activation in the backward direction, in\[kJ\,mo{{l}^{-1}}\], is
A)
\[(y-x)\]
done
clear
B)
\[(x+y)\]
done
clear
C)
\[(x-y)\]
done
clear
D)
\[-(x+y)\]
done
clear
View Answer play_arrow
question_answer 89) The rate constant for a first order reaction is\[6.909\text{ }mi{{n}^{-1}}\]. Therefore, the time required, in minute, for the participation of 75% of the initial reactant is
A)
2/3 log 2
done
clear
B)
2/3 log 4
done
clear
C)
3/2 log 2
done
clear
D)
3/2 log 4
done
clear
View Answer play_arrow
question_answer 90) A solution with negative deviation among the following is
A)
ethanol-acetone
done
clear
B)
chlorobenzene-bromobenzene
done
clear
C)
chloroform-acetone
done
clear
D)
benzene-toluene
done
clear
View Answer play_arrow
question_answer 91) At 300 K, two pure liquids A and B have vapour pressures respectively 150 mm Hg and 100 mm Hg. In an equimolar liquid mixture of A and B, the mole fraction of B in the vapour mixture at this temperature is
A)
0.6
done
clear
B)
0.5
done
clear
C)
0.8
done
clear
D)
0.4
done
clear
View Answer play_arrow
question_answer 92) The molar mass of the solute sodium hydroxide obtained from the measurement of the osmotic pressure of its aqueous solution at\[{{27}^{o}}C\]is\[25g\,mo{{l}^{-1}}\]. Therefore, its ionization percentage in this solution is
A)
75
done
clear
B)
60
done
clear
C)
80
done
clear
D)
70
done
clear
View Answer play_arrow
question_answer 93) 25 g of a solute of molar mass\[250\text{ }g\text{ }mo{{l}^{-1}}\] is dissolved in 10 mL of water to obtain a solution whose density is\[1.25\text{ }g{{(mL)}^{-1}}\]. The molarity and molality of the solution are respectively
A)
0.75 M and 1 m
done
clear
B)
0.8 M and 1 m
done
clear
C)
1M and 0.8 m
done
clear
D)
1 M and 0.75 m (e) None of the above
done
clear
View Answer play_arrow
question_answer 94) The standard enthalpies of formation of\[A(N{{H}_{3}}),B(C{{O}_{2}}),C(HI)\]and\[D(S{{O}_{2}})\]are respectively\[-46.19,-393.4,+24.94\] and\[-296.9\]\[kJmo{{l}^{-1}}\]. The increasing order of their stability is
A)
\[B<D<A<C\]
done
clear
B)
\[C<A<D<B\]
done
clear
C)
\[D<B<C<A\]
done
clear
D)
\[A<C<D<B\]
done
clear
View Answer play_arrow
question_answer 95) When 400 mL of 0.2 N solution of a weak acid is neutralised by a dilute aqueous solution of sodium hydroxide under standard conditions, 4.4 kJ amount of heat is liberated. Therefore, the standard enthalpy of neutralisation of this weak acid in,\[kJ\text{ }equi{{v}^{-1}},\]is
A)
\[-\,11\]
done
clear
B)
\[-\,44\]
done
clear
C)
\[-\,55\]
done
clear
D)
\[-\,22\]
done
clear
View Answer play_arrow
question_answer 96) The incorrect statement among the following is
A)
The entropy of the universe remains constant
done
clear
B)
Heat can be completely converted into work only under specified conditions
done
clear
C)
The absolute entropy of a perfect crystalline solid at absolute zero temperature is zero
done
clear
D)
The total energy of an isolated system remains constant
done
clear
View Answer play_arrow
question_answer 97) Which one of the following is always not negative?
A)
Enthalpy of combustion
done
clear
B)
Enthalpy of formation
done
clear
C)
Enthalpy of neutralization
done
clear
D)
Lattice enthalpy
done
clear
View Answer play_arrow
question_answer 98) The oxidation numbers of the sulphur atoms in peroxomonosulphuric acid\[({{H}_{2}}S{{O}_{5}})\]and peroxodisulphuric acid\[({{H}_{2}}{{S}_{2}}{{O}_{8}})\]are respectively
A)
+8 and +7
done
clear
B)
+3 and +3
done
clear
C)
+6 and+6
done
clear
D)
+4 and+6
done
clear
View Answer play_arrow
question_answer 99) In the electrolysis of aqueous solution of\[CuS{{O}_{4}}\]using copper electrodes, the process taking place at the anode is
A)
\[SO_{4}^{2-}\xrightarrow[{}]{{}}S{{O}_{4}}+2{{e}^{-}}\]
done
clear
B)
\[Cu\xrightarrow{{}}C{{u}^{+}}+{{e}^{-}}\]
done
clear
C)
\[2O{{H}^{-}}\xrightarrow{{}}{{H}_{2}}O+\frac{1}{2}{{O}_{2}}+2{{e}^{-}}\]
done
clear
D)
\[Cu\xrightarrow{{}}C{{u}^{2+}}+2{{e}^{-}}\]
done
clear
View Answer play_arrow
question_answer 100) The correct expression in SI system relating the equivalent conductance\[({{\Lambda }_{c}}),\]specific conductance\[(k)\]and equivalent concentration is (where C is the number of gram-equivalents of the electrolyte in one litre of the solution)
A)
\[{{\Lambda }_{c}}=\frac{k}{C}\]
done
clear
B)
\[{{\Lambda }_{c}}=k\times \frac{1000}{c}\]
done
clear
C)
\[{{\Lambda }_{c}}=k\times \frac{{{10}^{-3}}}{c}\]
done
clear
D)
\[{{\Lambda }_{c}}=k\times \frac{{{10}^{6}}}{c}\]
done
clear
View Answer play_arrow
question_answer 101) The standard reduction electrode potentials of the three electrodes P, Q and R are respectively\[-1.76\text{ }V,\text{ }0.34\text{ }V\]and 0.8 V, then
A)
metal Q will displace the cation of P from its aqueous solution and deposit the metal P
done
clear
B)
both metals Q and R will displace the cation of P from its aqueous solution and deposit the metal P
done
clear
C)
metal R will displace the cation of P from its aqueous solution and deposit the metal P
done
clear
D)
metal P will displace the cation of R from its aqueous solution and deposit the metal R
done
clear
View Answer play_arrow
question_answer 102) If the ratio of the rates of diffusion of two gases A and B is 4:1, the ratio of their densities in the same order is
A)
16 : 1
done
clear
B)
1 : 4
done
clear
C)
4 : 1
done
clear
D)
1 : 16
done
clear
View Answer play_arrow
question_answer 103) The van der Waals constants for four gases P, Q, R and S are 4.17, 3.59, 6.71 and 3.8 atm\[{{L}^{2}}.mo{{l}^{-2}}\]. Therefore, the ascending order of their liquefaction is
A)
\[R<P<S<Q\]
done
clear
B)
\[Q<S<R<P\]
done
clear
C)
\[Q<S<P<R\]
done
clear
D)
\[R<P<Q<S\]
done
clear
View Answer play_arrow
question_answer 104) The unit cell of a binary alloy composed of A and B metals, has a ccp structure with A atoms? occupying the comers and B atoms occupying centres of each face of the cube. If during the crystallisation of this alloy, in the unit cell two A atoms are missed, the overall composition per unit cell is
A)
\[A{{B}_{6}}\]
done
clear
B)
\[A{{B}_{4}}\]
done
clear
C)
\[A{{B}_{8}}\]
done
clear
D)
\[{{A}_{5}}{{B}_{24}}\]
done
clear
View Answer play_arrow
question_answer 105) When an excess and a very dilute aqueous solution of\[KI\]is added to a very dilute aqueous solution of silver nitrate, the colloidal particles of silver iodide are associated with the Helmholtz double layer
A)
\[AgI:\text{ }A{{g}^{+}}:NO_{3}^{-}\]
done
clear
B)
\[AgI:{{K}^{+}}:NO_{3}^{-}\]
done
clear
C)
\[AgI:\text{ }NO_{3}^{-}:A{{g}^{+}}\]
done
clear
D)
\[AgI:{{I}^{-}}:{{K}^{+}}\]
done
clear
View Answer play_arrow
question_answer 106) In the Freundlichs adsorption isotherm equation \[\log \frac{x}{m}=\log k+\left( \frac{1}{n} \right)\log p,\]the value of n is
A)
any value from 0 to 1
done
clear
B)
a negative integer
done
clear
C)
a positive integer
done
clear
D)
a positive or a negative fractional number
done
clear
View Answer play_arrow
question_answer 107) The polymer used in the manufacture of orlon is
A)
PTFE
done
clear
B)
PAN
done
clear
C)
PMMA
done
clear
D)
PVC
done
clear
View Answer play_arrow
question_answer 108) Which one of the following is d-block element?
A)
Gd
done
clear
B)
Hs
done
clear
C)
Es
done
clear
D)
Cs
done
clear
View Answer play_arrow
question_answer 109) The atom of smallest atomic radius among the following is
A)
Na
done
clear
B)
K
done
clear
C)
Br
done
clear
D)
Li
done
clear
View Answer play_arrow
question_answer 110) The d orbital involved in the hybridisation in the\[PC{{l}_{5}}\]molecule is
A)
\[3{{d}_{{{x}^{2}}-{{y}^{2}}}}\]
done
clear
B)
\[3{{d}_{{{z}^{2}}}}\]
done
clear
C)
\[3{{d}_{xy}}\]
done
clear
D)
\[4{{d}_{{{x}^{2}}-{{y}^{2}}}}\]
done
clear
View Answer play_arrow
question_answer 111) The shape of\[XeO{{F}_{2}}\]on the basis of VSEPR theory is
A)
sea saw
done
clear
B)
V-shaped
done
clear
C)
trigonal planar
done
clear
D)
T-shaped
done
clear
View Answer play_arrow
question_answer 112) Among the following the molecule possessing highest dipole moment is
A)
\[C{{O}_{2}}\]
done
clear
B)
\[B{{F}_{3}}\]
done
clear
C)
\[S{{O}_{2}}\]
done
clear
D)
Trans 2-butene
done
clear
View Answer play_arrow
question_answer 113) Which one of the following molecules is paramagnetic?
A)
\[{{F}_{2}}\]
done
clear
B)
\[{{B}_{2}}\]
done
clear
C)
\[L{{i}_{2}}\]
done
clear
D)
\[{{N}_{2}}\]
done
clear
View Answer play_arrow
question_answer 114) Among the following the least thermally stable is
A)
\[{{K}_{2}}C{{O}_{3}}\]
done
clear
B)
\[N{{a}_{2}}C{{O}_{3}}\]
done
clear
C)
\[BaC{{O}_{3}}\]
done
clear
D)
\[L{{i}_{2}}C{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 115) Which one of the following has the highest Lewis acid strength?
A)
\[B{{I}_{3}}\]
done
clear
B)
\[BB{{r}_{3}}\]
done
clear
C)
\[B{{F}_{3}}\]
done
clear
D)
\[BC{{l}_{3}}\]
done
clear
View Answer play_arrow
question_answer 116) The most powerful oxidising agent of the following is
A)
\[{{I}_{2}}\]
done
clear
B)
\[{{F}_{2}}\]
done
clear
C)
\[B{{r}_{2}}\]
done
clear
D)
\[C{{l}_{2}}\]
done
clear
View Answer play_arrow
question_answer 117) Which one of the following is non-reducing?
A)
\[{{H}_{2}}S\]
done
clear
B)
\[{{H}_{2}}Te\]
done
clear
C)
\[{{H}_{2}}Se\]
done
clear
D)
\[{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 118) The atom of which one of the following elements has the highest number of unpaired electrons?
A)
\[_{25}Mn\]
done
clear
B)
\[_{24}Cr\]
done
clear
C)
\[_{96}Cm\]
done
clear
D)
\[_{26}Fe\]
done
clear
View Answer play_arrow
question_answer 119) The ion of least magnetic moment among the following is
A)
\[T{{i}^{3+}}\]
done
clear
B)
\[N{{i}^{2+}}\]
done
clear
C)
\[C{{o}^{2+}}\]
done
clear
D)
\[M{{n}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 120) The composition of Bell metal is
A)
Cu (80%), Zn (20%)
done
clear
B)
Cu (60%), Ni (40%)
done
clear
C)
Cu (90%), Sn (10%)
done
clear
D)
Cu (80%), Sn (20%)
done
clear
View Answer play_arrow
question_answer 121) The amphoteric oxide among the following is
A)
\[C{{r}_{2}}{{O}_{3}}\]
done
clear
B)
\[M{{n}_{2}}{{O}_{7}}\]
done
clear
C)
\[{{V}_{2}}{{O}_{3}}\]
done
clear
D)
\[CrO\]
done
clear
View Answer play_arrow
question_answer 122) The coordination compound of which one of the following compositions will produce two equivalents of\[AgCl\]on reaction with aqueous silver nitrate solution?
A)
\[CoC{{l}_{3}}.3N{{H}_{3}}\]
done
clear
B)
\[CoC{{l}_{3}}.6N{{H}_{3}}\]
done
clear
C)
\[CoC{{l}_{3}}.4N{{H}_{3}}\]
done
clear
D)
\[CoC{{l}_{3}}.5N{{H}_{3}}\]
done
clear
View Answer play_arrow
question_answer 123) The optically active coordination complex ion among the following is
A)
\[trans{{[Co{{(en)}_{2}}C{{l}_{2}}]}^{+}}\]
done
clear
B)
\[cis{{[Co(en){{(N{{H}_{3}})}_{6}}]}^{2+}}\]
done
clear
C)
\[{{[Co{{(N{{H}_{3}})}_{6}}]}^{3+}}\]
done
clear
D)
\[{{[Fe{{(CN)}_{6}}]}^{3-}}\]
done
clear
View Answer play_arrow
question_answer 124) Which one of the following complex ions has the highest magnetic moment?
A)
\[{{[Cr{{(N{{H}_{3}})}_{6}}]}^{3+}}\]
done
clear
B)
\[{{[Fe(C{{N}_{6}})]}^{3-}}\]
done
clear
C)
\[{{[Fe(C{{N}_{6}})]}^{4-}}\]
done
clear
D)
\[{{[Zn{{(N{{H}_{3}})}_{6}}]}^{2+}}\]
done
clear
View Answer play_arrow
question_answer 125) The non-existant metal carbonyl among the following is
A)
\[Cr{{(CO)}_{6}}\]
done
clear
B)
\[Mn{{(CO)}_{5}}\]
done
clear
C)
\[Ni{{(CO)}_{4}}\]
done
clear
D)
\[Fe{{(CO)}_{5}}\]
done
clear
View Answer play_arrow
question_answer 126) The auto reduction process is not used in the metallurgy of
A)
\[Hg\]
done
clear
B)
\[Cu\]
done
clear
C)
\[Pb\]
done
clear
D)
\[Fe\]
done
clear
View Answer play_arrow
question_answer 127) The incorrect statement among the following is
A)
hydrogen is used to reduce\[NiO\]
done
clear
B)
zirconium is refined by van-Arkel method
done
clear
C)
the sulphide ore galena is concentrated by hydraulic washing process
done
clear
D)
in the metallurgy of iron, the flux used is \[Si{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 128) The percentage of carbon in cast iron is
A)
5-10
done
clear
B)
0.25-2.5
done
clear
C)
2.5-4.5
done
clear
D)
0.12-0.2
done
clear
View Answer play_arrow
question_answer 129) The IUPAC name of the molecule \[C{{H}_{3}}-\overset{\begin{smallmatrix} O \\ || \end{smallmatrix}}{\mathop{C}}\,-\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\mathop{C}}\,=\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\mathop{C}}\,-\overset{\begin{smallmatrix} O \\ || \end{smallmatrix}}{\mathop{C}}\,-OH\]
A)
4-oxo-2, 3-dimethyl pent-2-en-l-oic acid
done
clear
B)
2-carboxy-3 methyl pent-2-en-3-one
done
clear
C)
4-carboxy-3 methyl pent-3-en-2-one
done
clear
D)
2, 3-dimethyM-oxo pent-2-en-l-oic-acid
done
clear
View Answer play_arrow
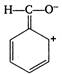
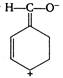
question_answer 130) The ascending order of stability of the carbanion\[\overline{C}{{H}_{3}}(P),{{C}_{6}}{{H}_{5}}\overline{C}{{H}_{2}}(Q),\] \[{{(C{{H}_{3}})}_{2}}\overline{C}H(R)\]and\[{{H}_{2}}\overline{C}-CH=C{{H}_{2}}(S)\] is
A)
\[P<R<S<Q\]
done
clear
B)
\[R<P<S<Q\]
done
clear
C)
\[R<P<Q<S\]
done
clear
D)
\[P<R<Q<S\]
done
clear
View Answer play_arrow
question_answer 131) The descending order of stability of the carbonium ions \[{{C}_{6}}{{H}_{5}}\overset{+}{\mathop{C{{H}_{2}}}}\,(I),p(C{{H}_{3}}O){{C}_{6}}{{H}_{4}}\overset{+}{\mathop{C{{H}_{2}}}}\,(II),p(N{{O}_{2}})\] \[{{C}_{6}}{{H}_{4}}\overset{+}{\mathop{C{{H}_{2}}}}\,(III)\,and\,p(C{{H}_{3}}){{C}_{6}}{{H}_{4}}\overset{+}{\mathop{C{{H}_{2}}}}\,(IV)\]is
A)
\[IV>II>I>III\]
done
clear
B)
\[II>IV>III>I\]
done
clear
C)
\[II>IV>I>III\]
done
clear
D)
\[IV>II>III>I\]
done
clear
View Answer play_arrow
question_answer 132) Which one of the following is aromatic?
A)
Cyclopentadienyl cation
done
clear
B)
Cyclooctatetraene
done
clear
C)
Cycloheptatriene
done
clear
D)
Cycloheptatrienylcation
done
clear
View Answer play_arrow
question_answer 133) The total number of acyclic structural and optical isomers possible for a hydrocarbon of molecular formula\[{{C}_{7}}{{H}_{16}}\]is
A)
12
done
clear
B)
8
done
clear
C)
10
done
clear
D)
6
done
clear
View Answer play_arrow
question_answer 134) The optical rotaion of an optically active compound is
A)
directly proportional to the length of the polanmeter tube only
done
clear
B)
directly proportional to the molar concentration of the compound
done
clear
C)
independent of the length of the polanmeter tube and concentration of the compound
done
clear
D)
directly proportional to both the length of the polarimeter tube and the molar concentration of the compound
done
clear
View Answer play_arrow
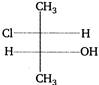
question_answer 135)
The absolute configurations of the\[{{C}_{2}}\]and\[{{C}_{3}}\] atoms in the molecule with the structure is
A)
25, 35
done
clear
B)
2R, 3S
done
clear
C)
2S, 3R
done
clear
D)
2R, 3R
done
clear
View Answer play_arrow
question_answer 136) Hydration of which one of the following yields a ketone?
A)
Propyne
done
clear
B)
Ethene
done
clear
C)
Propene
done
clear
D)
Ethyne
done
clear
View Answer play_arrow
question_answer 137) The most easily hydrolysed molecule under \[{{S}_{N}}1\]conditions is
A)
allyl chloride
done
clear
B)
ethyl chloride
done
clear
C)
isopropyi chloride
done
clear
D)
benzyl chloride
done
clear
View Answer play_arrow
question_answer 138) The most acidic among the following is
A)
p-cresol
done
clear
B)
o-cresol
done
clear
C)
p-nitrophenol
done
clear
D)
p-chlorophenol
done
clear
View Answer play_arrow
question_answer 139) Which one of the following does not undergo iodoform reaction?
A)
Secondary butyl alcohol
done
clear
B)
Iso-propyi alcohol
done
clear
C)
Diethyl ketone
done
clear
D)
Ethyl alcohol
done
clear
View Answer play_arrow
question_answer 140) Glycerol on oxidation with bismuth nitrate forms
A)
mesooxalic acid
done
clear
B)
glyceraldehyde
done
clear
C)
dihydroxy acetone
done
clear
D)
tartronic acid
done
clear
View Answer play_arrow
question_answer 141) Among the following, the alkene on ozonolysis giving rise to only one aldehyde as the product
A)
1-butene
done
clear
B)
propene
done
clear
C)
2-butene
done
clear
D)
2-methyl-prop-1-ene
done
clear
View Answer play_arrow
question_answer 142) Which one of the following does not form sodium bisulphite addition product with sodium bisulphite solution?
A)
\[C{{H}_{2}}O\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}COC{{H}_{3}}\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}CHO\]
done
clear
D)
\[C{{H}_{3}}CHO\]
done
clear
View Answer play_arrow
question_answer 143) The carboxylic acid of least strength among the following is
A)
p-nitrobenzoic acid
done
clear
B)
p-methylbenzoic acid
done
clear
C)
p-chlorobenzoic acid
done
clear
D)
p-methoxybenzoic acid
done
clear
View Answer play_arrow
question_answer 144) The most reactive of the following is
A)
acetone
done
clear
B)
benzophenone
done
clear
C)
benzaldehyde
done
clear
D)
acetaldehyde
done
clear
View Answer play_arrow
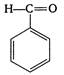
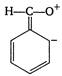
question_answer 145) Which of the following is not a resonating form of benzaldehyde?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
question_answer 146) The compound that does not undergo Hell-Volhard Zeiinsky reaction is
A)
ethanoic acid
done
clear
B)
propionic acid
done
clear
C)
Lso-butyric acid
done
clear
D)
trichloroacetic acid
done
clear
View Answer play_arrow
question_answer 147) Nitrobenzene can be converted into hydrazobenzene by reduction with
A)
Zn and alcoholic\[NaOH\]
done
clear
B)
Zn and aqueous\[NaOH\]
done
clear
C)
\[N{{H}_{2}}N{{H}_{2}}\]and alcoholic\[KOH\]
done
clear
D)
\[Zn\]and\[HCl\]
done
clear
View Answer play_arrow
question_answer 148) Which of the following compounds gives carbylamine test?
A)
N-methyl-o-methyl aniline
done
clear
B)
N, N-dimethyl aniline
done
clear
C)
2, 4-diethyl aniline
done
clear
D)
p-methyl-N-methyl benzylamine
done
clear
View Answer play_arrow
question_answer 149) By which of the following reagents, both the aldehyde and the primary alcoholic group of glucose are oxidised?
A)
Toilers reagent
done
clear
B)
\[B{{r}_{2}}+{{H}_{2}}O\]
done
clear
C)
\[HN{{O}_{3}}\]
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 150) Lactoflavin is
A)
vitamin\[{{B}_{1}}\]
done
clear
B)
vitamin C
done
clear
C)
vitamin\[{{B}_{3}}\]
done
clear
D)
vitamin\[{{B}_{2}}\]
done
clear
View Answer play_arrow
question_answer 151) Which one is the correct heirarchial order in Taxonomy?
A)
Genus < species < class < order
done
clear
B)
Genus < class < order < family
done
clear
C)
Species < order < class < phylum
done
clear
D)
Genus < class < division < order
done
clear
View Answer play_arrow
question_answer 152) Systematic botany means
A)
system analysis
done
clear
B)
systematic arrangement of organs of plants
done
clear
C)
systematic study of organelles and tissues
done
clear
D)
methodical study of plants, dealing with identification, naming and classification
done
clear
View Answer play_arrow
question_answer 153) The kingdom of prokaryotes is
A)
Protista
done
clear
B)
Monera
done
clear
C)
Fungi
done
clear
D)
Plantae
done
clear
View Answer play_arrow
question_answer 154) The non-living characteristic of viruses is
A)
ability to multiply only inside the host
done
clear
B)
ability to cause diseases in the host
done
clear
C)
ability to undergo mutation
done
clear
D)
ability to be crystallized
done
clear
View Answer play_arrow
question_answer 155) The term bacteria was coined by
A)
Leeuwenhoek
done
clear
B)
Louis Pasteur
done
clear
C)
Robert Koch
done
clear
D)
Ehrenberg
done
clear
View Answer play_arrow
question_answer 156) The Prayer sanctuary is located in
A)
Kerala
done
clear
B)
Tamil Nadu
done
clear
C)
Karnataka
done
clear
D)
Andhra Pradesh
done
clear
View Answer play_arrow
question_answer 157) The photosynthetic or assimilatory roots are observed in
A)
Banyan
done
clear
B)
Vanda
done
clear
C)
Cuscuta
done
clear
D)
Tinospora
done
clear
View Answer play_arrow
question_answer 158) The bladder serving as floats and for trapping insects is found in
A)
Zizyphus
done
clear
B)
Utriculari
done
clear
C)
Nephenthes
done
clear
D)
Acacia
done
clear
View Answer play_arrow
question_answer 159) The order of opening of flower parts from the periphery towards the centre is called
A)
acropetal
done
clear
B)
centripetal
done
clear
C)
centrifugal
done
clear
D)
basipetal
done
clear
View Answer play_arrow
question_answer 160) Tobacco and Petunia belong to the family
A)
Poaceae
done
clear
B)
Fabaceae
done
clear
C)
Solanaceae
done
clear
D)
Brassicaceae
done
clear
View Answer play_arrow
question_answer 161) Simple, cluster of radial leaves, stipulate and parallel venation leaves and cyme or umbel inflorescence are the characteristics of
A)
Poaceae
done
clear
B)
Liliaceae
done
clear
C)
Asteraceae
done
clear
D)
Fabaceae
done
clear
View Answer play_arrow
question_answer 162) The gene, which controls many characters is called
A)
codominant gene
done
clear
B)
poly gene
done
clear
C)
pleiotropic gene
done
clear
D)
multiple gene
done
clear
View Answer play_arrow
question_answer 163) Which of the following movements in plants is related to the changes in the auxin level?
A)
Movement of shoot towards the source of light
done
clear
B)
Nyctinasty
done
clear
C)
Movement of sunflower towards sun
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 164) Embryo developed from the somatic cells are called
A)
cybrids
done
clear
B)
embryoid
done
clear
C)
cellus
done
clear
D)
hybrids
done
clear
View Answer play_arrow
question_answer 165) Vegetative fertilization is also called
A)
triple fusion
done
clear
B)
true fertilization
done
clear
C)
syngamy
done
clear
D)
generative fertilization
done
clear
View Answer play_arrow
question_answer 166) Vivipary is observed in
A)
Banyan
done
clear
B)
Bryophyllum
done
clear
C)
Ipomoea
done
clear
D)
Rhizophora
done
clear
View Answer play_arrow
question_answer 167) The scientist, who performed some experiments with oat coleoptiles for the presence of a substance which could diffuse into agar blocks is
A)
Ganong
done
clear
B)
Went
done
clear
C)
Boysen-Jensen
done
clear
D)
Fujikura
done
clear
View Answer play_arrow
question_answer 168) Micropyle is useful for the entry of
A)
pollen grain
done
clear
B)
pollen tube
done
clear
C)
water
done
clear
D)
male gamete
done
clear
View Answer play_arrow
question_answer 169) Abcisic acid is primarily synthesized in
A)
lysosomes
done
clear
B)
Golgi complex
done
clear
C)
chloroplast
done
clear
D)
ribosomes
done
clear
View Answer play_arrow
question_answer 170) Which one of the following inhibit seed germination for a particular period?
A)
Light
done
clear
B)
Water
done
clear
C)
Carbon dioxide
done
clear
D)
Dormancy
done
clear
View Answer play_arrow
question_answer 171) Flocculation or coagulation of protoplasm is the
A)
interchangeability between sol and gel states
done
clear
B)
ability to scatter the beam of light
done
clear
C)
erratic zig-zag movement of protoplasmic particles
done
clear
D)
ability of protoplasm to increase in size when they lose charges
done
clear
View Answer play_arrow
question_answer 172) Plasmolysis is the result of
A)
exosmosis
done
clear
B)
endosmosis
done
clear
C)
reverse osmosis
done
clear
D)
diffusion
done
clear
View Answer play_arrow
question_answer 173) Which one is incorrect statement?
A)
Movement of water is expressed in terms of free energy
done
clear
B)
Free energy determines the direction by which physical and chemical changes should occur
done
clear
C)
Water potential is the sum of free energy of water molecules in pure water and in any other system
done
clear
D)
Water potential of pure water is zero
done
clear
View Answer play_arrow
question_answer 174) Ascent of sap in plants was demonstrated by
A)
Girdling experiment
done
clear
B)
Ganongs experiment
done
clear
C)
Went experiment
done
clear
D)
Lever auxanometer
done
clear
View Answer play_arrow
question_answer 175) Cohesion and adhesion theory is otherwise called
A)
relay pump theory
done
clear
B)
pulsation theory
done
clear
C)
root pressure theory
done
clear
D)
transpiration pull theory
done
clear
View Answer play_arrow
question_answer 176) Molybdenum is the essential constituent of
A)
nitrogenase
done
clear
B)
respiratory chain
done
clear
C)
growth regulators
done
clear
D)
chlorophyll
done
clear
View Answer play_arrow
question_answer 177) Which one is true about guttation?
A)
It occurs through specialized pores called hydathodes
done
clear
B)
It occurs in herbaceous plants when root pressure is low and transpiration is high
done
clear
C)
It only occur during the day time
done
clear
D)
It occurs in plants growing under conditions of low soil moisture and high humidity
done
clear
View Answer play_arrow
question_answer 178) Which one does not occur in cyclic photophosphorylation?
A)
Oxygen is not given off
done
clear
B)
Water is not consumed
done
clear
C)
Only photosystem-I is involved
done
clear
D)
\[NADP{{H}_{2}}\] formation
done
clear
View Answer play_arrow
question_answer 179) Chlorophyll-a and b differ in having
A)
Chlorophyll-a has an methyl group and chlorophyll-b has aldehyde group in position X
done
clear
B)
Chlorophyll-a has an aldehyde group and chlorophyll-b has a methyl group in position X
done
clear
C)
Chlorophyll-a has a carboxyl group and chlorophyll-b has an aldehyde group in position X
done
clear
D)
Chlorophyll-a has an ethyl group and chlorophyll-b has an aldehyde group in position X
done
clear
View Answer play_arrow
question_answer 180) The assemblage of all the populations of different species that function as an integrated unit through coevolved metabolic transformation in a specific area is called
A)
biome
done
clear
B)
biotic community
done
clear
C)
population
done
clear
D)
ecosystem
done
clear
View Answer play_arrow
question_answer 181) Genetically adapted population to a particular habitat is called
A)
ecotone
done
clear
B)
ecotype
done
clear
C)
biome
done
clear
D)
niche
done
clear
View Answer play_arrow
question_answer 182) Which one is the edaphic factor in biosphere?
A)
Light
done
clear
B)
Temperature
done
clear
C)
Water
done
clear
D)
Soil
done
clear
View Answer play_arrow
question_answer 183) \[C{{O}_{2}},C{{H}_{4}},{{N}_{2}}O\] and CFCs are called green-house gases because they absorb and emit
A)
UV rays
done
clear
B)
heat rays
done
clear
C)
X rays
done
clear
D)
gamma rays
done
clear
View Answer play_arrow
question_answer 184) In plant succession, when climax community is reached, the net productivity
A)
continues to increase
done
clear
B)
becomes zero
done
clear
C)
becomes reduced
done
clear
D)
becomes stable
done
clear
View Answer play_arrow
question_answer 185) The species of plants that play a vital role in controlling the relative abundance of other species in a community are called
A)
edge species
done
clear
B)
key stone species
done
clear
C)
pioneer species
done
clear
D)
serial species
done
clear
View Answer play_arrow
question_answer 186) Ratio between mortality and natality is called
A)
population ratio
done
clear
B)
vital index
done
clear
C)
density coefficient
done
clear
D)
census ratio
done
clear
View Answer play_arrow
question_answer 187) Which one is not the renewable energy of natural resources?
A)
Tidal energy
done
clear
B)
Wind energy
done
clear
C)
Fossil fuel
done
clear
D)
Solar energy
done
clear
View Answer play_arrow
question_answer 188) Choanocytes form the lining of paragastral cavity in
A)
jelly fish
done
clear
B)
sponges
done
clear
C)
helminthes
done
clear
D)
echinoderms
done
clear
View Answer play_arrow
question_answer 189) Siphonophora is the order in
A)
Protozoa
done
clear
B)
Cnidaria
done
clear
C)
Porifera
done
clear
D)
Annelida
done
clear
View Answer play_arrow
question_answer 190) The second largest number of species containing phylum in the animal kingdom is
A)
Annelida
done
clear
B)
Arthropoda
done
clear
C)
Mollusca
done
clear
D)
Chordata
done
clear
View Answer play_arrow
question_answer 191) Limbless amphibians are called
A)
paddle worms
done
clear
B)
glow worms
done
clear
C)
caecilian worms
done
clear
D)
pin worms
done
clear
View Answer play_arrow
question_answer 192) The second largest aquatic vertebrate is
A)
blue whale
done
clear
B)
whale shark
done
clear
C)
sea elephant
done
clear
D)
dugongs
done
clear
View Answer play_arrow
question_answer 193) The snake eating snake is
A)
black cobra
done
clear
B)
king cobra
done
clear
C)
rattle snake
done
clear
D)
anaconda
done
clear
View Answer play_arrow
question_answer 194) The layer of cells forming tissue that appears to be multilayered but actually some of the cells extend from the basement membrane to the surface is
A)
simple columnar epithelium
done
clear
B)
pseudo stratified epithelium
done
clear
C)
stratified columnar epithelium
done
clear
D)
stratified cuboidal epithelium
done
clear
View Answer play_arrow
question_answer 195) Haversian lamellae are the structures found in
A)
hyaline cartilage
done
clear
B)
fibrous cartilage
done
clear
C)
bone marrow
done
clear
D)
myelin sheath
done
clear
View Answer play_arrow
question_answer 196) The malnutrition disease in man is
A)
Cri du chat syndrome
done
clear
B)
Klinefelter syndrome
done
clear
C)
Potbelly syndrome
done
clear
D)
Edward syndrome
done
clear
View Answer play_arrow
question_answer 197) Which one is not true about vitamins?
A)
Vitamins are organic catalysts
done
clear
B)
Vitamins are indispensable for life
done
clear
C)
Vitamins act as a source of energy
done
clear
D)
Tocopherol b anti sterility vitamin
done
clear
View Answer play_arrow
question_answer 198) The process of resynthesis of food materials from simpler food molecules is called
A)
biosynthesis
done
clear
B)
catabolism
done
clear
C)
absorption
done
clear
D)
assimilation
done
clear
View Answer play_arrow
question_answer 199) The conversion of dangerous nitrogenous waste into less toxic excretory matter is carried out in man in the
A)
blood
done
clear
B)
liver
done
clear
C)
kidney
done
clear
D)
skin
done
clear
View Answer play_arrow
question_answer 200) The total number of lobes and alveoli present in both the lungs of man are
A)
17 and 30 millions, respectively
done
clear
B)
5 and 300 millions, respectively
done
clear
C)
19 and 300 millions, respectively
done
clear
D)
18 and 300 lakh, respectively
done
clear
View Answer play_arrow
question_answer 201) The hormones oxytocin and vasopressin are secreted by
A)
neurohypophysis
done
clear
B)
adenohypophysis
done
clear
C)
hypothalamus
done
clear
D)
adrenal medulla
done
clear
View Answer play_arrow
question_answer 202) When the stimulus reaches the end of one neuron, it is conducted to the adjacent neuron through the secretions of
A)
acetaldehyde
done
clear
B)
acetylcholine
done
clear
C)
acetylcholine esterase
done
clear
D)
acetyl Co-A
done
clear
View Answer play_arrow
question_answer 203) The fusion of male and female pronuclear of the gametes is called
A)
fertilization
done
clear
B)
conjugation
done
clear
C)
amphimixis
done
clear
D)
panmixis
done
clear
View Answer play_arrow
question_answer 204) Long flattened, usually unbranched units arranged in parallel stacks in endoplasmic reticulum are called
A)
cisternae
done
clear
B)
cristae
done
clear
C)
vesicles
done
clear
D)
tubules
done
clear
View Answer play_arrow
question_answer 205) TCA cycle enzymes are present in
A)
cytoplasm
done
clear
B)
inter membrane space of mitochondria
done
clear
C)
mitochondrial matrix
done
clear
D)
inner membrane of mitochondria
done
clear
View Answer play_arrow
question_answer 206) In cellular respiration the final acceptor molecule of electron is
A)
NAD
done
clear
B)
FAD
done
clear
C)
NADP
done
clear
D)
Oxygen
done
clear
View Answer play_arrow
question_answer 207) Which one is not the nonsense codon?
A)
UAA
done
clear
B)
UGA
done
clear
C)
UCA
done
clear
D)
UAG
done
clear
View Answer play_arrow
question_answer 208) Which one is referred to as soluble RNA?
A)
mRNA
done
clear
B)
tRNA
done
clear
C)
rRNA
done
clear
D)
ssRNA
done
clear
View Answer play_arrow
question_answer 209) The autonomously independent self replicating extra nuclear DNA imparting certain factors to some bacterium is called
A)
plastid
done
clear
B)
plasmid
done
clear
C)
phagemid
done
clear
D)
cosmid
done
clear
View Answer play_arrow
question_answer 210) In microbial genetics, which one is referred to as Griffith effect?
A)
Conjugation
done
clear
B)
Transduction
done
clear
C)
Transformation
done
clear
D)
Sexduction
done
clear
View Answer play_arrow
question_answer 211) The phenotypic ratio in the \[{{F}_{2}}\]generation of dihybrid cross is
A)
9 : 3 : 3 :1
done
clear
B)
1 : 2 : 2 : 4 : 1 : 2 : 1 : 2 : 1
done
clear
C)
7 : 1 : 1 : 7
done
clear
D)
12 : 8 : 4
done
clear
View Answer play_arrow
question_answer 212) Which one is regarded as a molecular scissor in biotechnology?
A)
Reverse transcriptase
done
clear
B)
Restriction endonuclease
done
clear
C)
Taq polymerase
done
clear
D)
Topoisomerase
done
clear
View Answer play_arrow
question_answer 213) Which one does not conform to the theory of biogenesis?
A)
Francisco red is experiment
done
clear
B)
Spallanzanis experiment
done
clear
C)
Louis Pasteurs experiment
done
clear
D)
Von Helmonts experiment
done
clear
View Answer play_arrow
question_answer 214) Urey-Millers experiment mixture had the following except
A)
methane
done
clear
B)
\[C{{O}_{2}}\]
done
clear
C)
hydrogen
done
clear
D)
water vapour
done
clear
View Answer play_arrow
question_answer 215) Among the following which one is the mutagenic agent?
A)
Visible light
done
clear
B)
Penicillin
done
clear
C)
Formalin
done
clear
D)
Water vapour
done
clear
View Answer play_arrow
question_answer 216) Ontogeny Recapitulates Phylogeny is narrated in which of the evidences for organic evolution?
A)
Palaeontological evidence
done
clear
B)
Physiological evidence
done
clear
C)
Embryological evidence
done
clear
D)
Anatomical evidence
done
clear
View Answer play_arrow
question_answer 217) Which one of the following was not explained by the Darwinism?
A)
Natural selection
done
clear
B)
Struggle for existence
done
clear
C)
Arrival of the fittest
done
clear
D)
Origin of species
done
clear
View Answer play_arrow
question_answer 218) The chronological order of human evolution from early to the recent is
A)
Ramapithecus-Australo pithecus-Homo habilis-Homo erectus
done
clear
B)
Australopithecus-Ramapithecus-Homo habilis -Homo erectus
done
clear
C)
Pithecanthropus pekinensis-Homo habilis- Homo erectus
done
clear
D)
Australopithecus-Ramapithecus-Pithecant hropus pekinensis-Homo erectus
done
clear
View Answer play_arrow
question_answer 219) Phenomenal and rapid increase of population in a short period is called
A)
natural increase
done
clear
B)
population growth
done
clear
C)
population explosion
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 220) Amphetamines are the drugs of
A)
narcotics
done
clear
B)
sedatives
done
clear
C)
stimulants
done
clear
D)
hallucinogens
done
clear
View Answer play_arrow
question_answer 221) Cyclosporin and endosporins are the drugs that are used as
A)
anti retro viral drugs
done
clear
B)
immuno-suppressants
done
clear
C)
immuno-modulators
done
clear
D)
immuno-vaccines
done
clear
View Answer play_arrow
question_answer 222) The tests that are used in the diagnosis of AIDS are
A)
ELISA and immunoblot
done
clear
B)
Northern blot and ELISA
done
clear
C)
ELISA and southern blot
done
clear
D)
Western blot and ELISA
done
clear
View Answer play_arrow
question_answer 223) Which of the following causes prostate cancer?
A)
Chromium
done
clear
B)
Cadmium oxide
done
clear
C)
Vinyl chloride
done
clear
D)
Aflatoxins
done
clear
View Answer play_arrow
question_answer 224) Magic bullets are the
A)
recombinant vaccines
done
clear
B)
monoclonal antibodies
done
clear
C)
chemotherapy drugs for cancer
done
clear
D)
anabolic steroids
done
clear
View Answer play_arrow
question_answer 225) The interferons are
A)
antibacterial drugs
done
clear
B)
antiviral drugs
done
clear
C)
antibiotic drugs
done
clear
D)
immunosuppressive drugs
done
clear
View Answer play_arrow