-
Write the Lewis dot structure of
CO molecule.
View Answer play_arrow
-
Write the Lewis structure of the
nitrite ion, \[NO_{2}^{-}\].
View Answer play_arrow
-
Explain the structure of \[CO_{3}^{2-}\]
ion in terms of resonance.
View Answer play_arrow
-
Explain the structure of \[C{{O}_{2}}\]
molecule.
View Answer play_arrow
-
Explain the formation of a
chemical bond.
View Answer play_arrow
-
Write Lewis dot symbols for atoms of the following elements
and predict their valencies.
Mg, Na, B, O, Br
View Answer play_arrow
-
"Write
the Lewis dot symbols for the following atoms and ions:
(i) S and \[{{S}^{2-}}\] (it)
Al and \[A{{l}^{3+}}\] (iii H and \[{{H}^{-}}\]
View Answer play_arrow
-
Write
the Lewis structures for the following molecules/ions:
\[{{H}_{2}}S,\,\,SiC{{l}_{4}},\,\,Be{{F}_{2}},\,\,ClO_{3}^{-},\,\,HCOOH\]
View Answer play_arrow
-
Define octet rule. What is its
significance?
View Answer play_arrow
-
Write the favourable factors for the
formation of an ionic bond.
View Answer play_arrow
-
Discuss
the shapes of the following molecules using VSEPR model:
\[BeC{{l}_{2}},BC{{l}_{3}},SiC{{l}_{4}},As{{F}_{3}},{{H}_{2}}S,P{{H}_{3}}\]
View Answer play_arrow
-
Although geometries of \[N{{H}_{3}}\]
and \[{{H}_{2}}O\]molecules are distorted tetrahedral, bond angle in water is
less than that of ammonia. Discuss.
View Answer play_arrow
-
How do you express the bond length
in terms of bond order?
View Answer play_arrow
-
Define the bond length.
View Answer play_arrow
-
Explain the important aspects of
resonance with reference to \[CO_{3}^{2-}\] ion.
View Answer play_arrow
-
\[{{H}_{3}}P{{O}_{3}}\] can
be represented by the structures I and II shown below. Can these structures be taken
as the canonical forms of the resonance hybrid of \[{{H}_{3}}P{{O}_{3}}\]? If not,
give reason for the same.

-
Write the resonance structures for
\[S{{O}_{3}},N{{O}_{2}}\] and \[NO_{3}^{-}\].
View Answer play_arrow
-
Use
Lewis symbols to show electron transfer between the following atoms to form
cations and onions :
(a) K and S (b) Ca and 0 (c) Al and N
View Answer play_arrow
-
Although
both \[C{{O}_{2}}\] and \[{{H}_{2}}O\] are triatomic molecules, the shape of \[{{H}_{2}}O\]
molecule is bent while that of \[C{{O}_{2}}\] is linear.
Explain this on the basis of dipole
moment.
View Answer play_arrow
-
Write the significance of dipole
moment.
View Answer play_arrow
-
Define electronegativity. How does
it differ from electron gain enthalpy?
View Answer play_arrow
-
Explain with the help of a
suitable example polar covalent bond.
View Answer play_arrow
-
Arrange
the bonds in order of increasing ionic character in the molecules.
\[LiF,\,{{K}_{2}}O,\,{{N}_{2}},\,S{{O}_{2}},\,Cl{{F}_{3}}\]
View Answer play_arrow
-
The skeletal structure of \[C{{H}_{3}}COOH\]
as shown below is correct, but some of the bonds are shown incorrectly. Write the
correct Lewis structure for acetic acid.
View Answer play_arrow
-
Apart from tetrahedral geometry,
another possible geometry for \[C{{H}_{4}}\] is square planar with four H-atoms
at the comers of the square and C-atom at its centre. Explain why \[C{{H}_{4}}\]
is not square planar?
View Answer play_arrow
-
Explain why \[Be{{H}_{2}}\]
molecule has a zero dipole moment although BeH
bonds are polar?
View Answer play_arrow
-
Which out of \[N{{H}_{3}}\] and \[N{{F}_{3}}\]
has higher dipole moment and why?
View Answer play_arrow
-
What
is meant by hybridization of atomic orbitals?
Describe the shapes of \[sp,\,\,s{{p}^{2}},\,\,s{{p}^{3}}\]
hybrid orbitals.
View Answer play_arrow
-
Describe
the change in hybridization (if any) of the Al-atom in the following reaction.
\[AlC{{l}_{3}}+C{{l}^{-}}\to
AlCl_{4}^{-}\]
View Answer play_arrow
-
Is
there any change in the hybridization of B and N-atoms as a result of the
following reaction?
\[B{{F}_{3}}+N{{H}_{3}}\to
{{F}_{3}}BN{{H}_{3}}\]
View Answer play_arrow
-
Draw the diagrams showing the
formation of a double bond and a triple bond between carbon atoms in \[{{C}_{2}}{{H}_{4}}\]
and \[{{C}_{2}}{{H}_{2}}\] molecules.
View Answer play_arrow
-
What
is the total number of sigma and pi bonds in the following molecules?
(a) \[{{C}_{2}}{{H}_{2}}\] (b)
\[{{C}_{2}}{{H}_{4}}\]
View Answer play_arrow
-
Considering
X-axis as the intemuclear axis, which out of following will not form a sigma
bond and why?
(a) 1s and 1s (b) 1s and \[2{{p}_{x}}\]
(c) \[2{{p}_{y}}\] and \[2{{p}_{y}}\]
(d) 1s and 1s
View Answer play_arrow
-
Which hybrid orbitals are used by carbon atoms in the following
molecules?
(a) \[C{{H}_{3}}-C{{H}_{3}}\] (b) \[C{{H}_{3}}-CH=C{{H}_{2}}\]
(c) \[C{{H}_{3}}-C{{H}_{2}}-OH\] (d) \[C{{H}_{3}}-CHO\]
(e) \[C{{H}_{3}}-COOH\]
View Answer play_arrow
-
What do you understand by bond
pairs and lone pairs of electrons?
View Answer play_arrow
-
Distinguish between a sigma bond
and a pi bond
View Answer play_arrow
-
Explain the formation of H^
molecule on the basis of valence bond theory.
View Answer play_arrow
-
Write the important conditions
required for the linear combination of atomic orbitals to form molecular
orbitals.
View Answer play_arrow
-
Use molecular orbital theory to
explain why the \[B{{e}_{2}}\] molecule does not exist?
View Answer play_arrow
-
Compare
the relative stabilities of the following species and indicate their magnetic
properties
\[{{O}_{2}},O_{2}^{+},O_{2}^{-}\]
and \[O_{2}^{2-}\]
View Answer play_arrow
-
What is the significance of a plus
and a minus sign shown in representing the
orbitals?
View Answer play_arrow
-
Describe the hybridization in case
of\[PC{{l}_{5}}\]. Why are the axial bonds longer as compared to equatorial
bonds?
View Answer play_arrow
-
Define hydrogen bond. Is it weaker
or stronger than the van der Waals' forces?
View Answer play_arrow
-
What is meant by the term bond
order? Calculate the bond order of \[{{N}_{2}},{{O}_{2}},O_{2}^{+}\] and \[O_{2}^{-}\].
View Answer play_arrow
-
question_answer45)
In the following questions only one
option is correct.
Isostructural species are those
which have the same shapeand hybridisation. Among the given species identify
the isostructural pairs :
(a) \[[N{{F}_{3}}and\,B{{F}_{3}}]\] (b)
\[[BF_{4}^{-}and\,NH_{4}^{+}]\] (c)\[[BC{{l}_{3}}\,and\,BeC{{l}_{3}}]\] (d)
{\[N{{H}_{3}}and\,NO_{3}^{-}\]]
View Answer play_arrow
-
question_answer46)
Polarity in a molecule and hence
the dipole momentdepends primarily on electronegativity of the constituentatoms
and shape of molecule. Which of the following has the highest dipole moment?
(a) \[C{{O}_{2}}\] (b)
\[HI\] (c)\[{{H}_{2}}O\] (d)
\[S{{O}_{2}}\]
View Answer play_arrow
-
question_answer47)
The types of hybrid orbitals of
nitrogen in \[NO_{2}^{+},NO_{3}^{-}\]and \[NH_{4}^{+}\] respectively are
expected to be :
(a) \[sp,\text{ }s{{p}^{3}}\]and\[s{{p}^{2}}\]
(b) \[sp,\text{ }s{{p}^{2}}\]and \[s{{p}^{3}}\] (c)\[~s{{p}^{2}},\text{
}sp\]and\[s{{p}^{3}}\] (d) \[s{{p}^{2}},\text{
}s{{p}^{3}}\]and \[sp\]
View Answer play_arrow
-
question_answer48)
Hydrogen bonds are formed in
many compounds e.g.,\[{{H}_{2}}O,HF,N{{H}_{3}}\]. The boiling point of such
compoundsdepends to a large extent on the strength of hydrogen bondand the
number of hydrogen bonds. The correct decreasingorder of boiling points of
above compounds is:
(a) \[HF>{{H}_{2}}O>N{{H}_{3}}\] (b)
\[{{H}_{2}}O>HF>N{{H}_{3}}\]
(c) \[N{{H}_{3}}>HF>{{H}_{2}}O\] (d)
\[N{{H}_{3}}>{{H}_{2}}O>HF\]
View Answer play_arrow
-
question_answer49)
In \[PO_{4}^{3-}\] ion, the
formal charge on the oxygen atom of P- O bond is :
(a) +1 (b)
-1 (c) -0.75 (d)
+0.75
View Answer play_arrow
-
question_answer50)
In \[NO_{3}^{-}\] ion, the
number of bond pairs and lone pairs of electrons on nitrogen atom are :
(a) 2, 2 (b) 3,
1 (c) 1, 3 (d)
4, 0
View Answer play_arrow
-
question_answer51)
Which of the following species
has tetrahedral geometry?
(a) \[BH_{4}^{-}\] (b)\[NH_{2}^{-}\] (c)
\[CO_{3}^{2-}\] (d) \[{{H}_{3}}\overset{+}{\mathop{O}}\,\]
View Answer play_arrow
-
question_answer52)
Number of \[\pi \]-bonds and \[\sigma
\]-bonds in the following structure is :
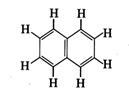 (a) 6, 19 (b)
4, 20 (c) 5, 19 (d)
5, 20
(a) 6, 19 (b)
4, 20 (c) 5, 19 (d)
5, 20
View Answer play_arrow
-
question_answer53)
Which molecule or ion out of the
following does not contain unpaired electrons?
(a) \[N_{2}^{+}\] (b)
\[{{O}_{2}}\] (c)\[O_{2}^{2-}\] (d)
\[{{B}_{2}}\]
View Answer play_arrow
-
question_answer54)
In which of the following
molecules/ions all the bonds arenot equal?
(a)\[Xe{{F}_{4}}\] (b)
\[BF_{4}^{-}\] (c) \[{{C}_{2}}{{H}_{4}}\] (d)
\[Si{{F}_{4}}\]
View Answer play_arrow
-
question_answer55)
In which of the following
substances will hydrogen bond be strongest?
(a)\[HCl\] (b)\[{{H}_{2}}O\] (c)\[HI\] (d)
\[{{H}_{2}}S\]
View Answer play_arrow
-
question_answer56)
If the electronic configuration
of an element is
\[1{{s}^{2}}\text{
}2{{s}^{2}}\text{ }2{{p}^{6}}\text{ }3{{s}^{2}}\text{ }3{{p}^{6}}\text{
}3{{d}^{2}}\text{ }4{{s}^{2}}\], the four electrons involved inchemical bond
formation will be :
(a)\[3{{p}^{6}}\] (b)
\[3{{p}^{6}},4{{s}^{2}}\] (c)\[3{{p}^{6}},3{{d}^{2}}\] (d)
\[3{{d}^{2}},4{{s}^{2}}\]
View Answer play_arrow
-
question_answer57)
Which of the following angle
corresponds to \[s{{p}^{2}}\]hybridisation?
(a)\[90{}^\circ \] (b)
\[120{}^\circ \] (c)\[180{}^\circ \] (d)
\[109{}^\circ \]
View Answer play_arrow
-
question_answer58)
The electronic configurations
of three elements A, and C are given below. Answer the questions 14 to 17 on
the basis of these configurations.
A \[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}\]
B \[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{3}}\]
C \[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{5}}\]
Stable form of A may be
represented by the formula :
(a) \[A\] (b)\[{{A}_{2}}\] (c)\[{{A}_{3}}\] (d)\[{{A}_{4}}\]
View Answer play_arrow
-
question_answer59)
Stable form of C may be
represented by the formula :
(a) \[C\] (b)
\[{{C}_{2}}\] (c)\[{{C}_{3}}\] (d)
\[{{C}_{4}}\]
View Answer play_arrow
-
question_answer60)
The molecular formula of the
compound formed between B and C will be :
(a)\[BC\] (b)
\[{{B}_{2}}C\] (c)\[B{{C}_{2}}\] (d)\[B{{C}_{3}}\]
View Answer play_arrow
-
question_answer61)
The bond between B and C will be
:
(a) ionic (b)
covalent (c) hydrogen (d) coordinate
View Answer play_arrow
-
question_answer62)
Which of the following order of
energies of molecular orbitals of \[{{N}_{2}}\] is correct?
(a)\[(\pi 2{{p}_{y}})<(\sigma
2{{p}_{z}})<({{\pi }^{*}}2{{p}_{x}})\approx ({{\pi }^{*}}2{{p}_{y}})\] (b)\[(\pi
2{{p}_{y}})>(\sigma 2{{p}_{z}})>({{\pi }^{*}}2{{p}_{x}})\approx ({{\pi
}^{*}}2{{p}_{y}})\]
(c)\[(\pi 2{{p}_{y}})<(\sigma
2{{p}_{z}})>({{\pi }^{*}}2{{p}_{x}})\approx ({{\pi }^{*}}2{{p}_{y}})\] (d)\[(\pi
2{{p}_{y}})>(\sigma 2{{p}_{z}})<({{\pi }^{*}}2{{p}_{x}})\approx ({{\pi }^{*}}2{{p}_{y}})\]
View Answer play_arrow
-
question_answer63)
Which of the following
statements is not correct from theview point of molecular theory?
(a) \[B{{e}_{2}}\]is not a
stable molecule
(b) \[H{{e}_{2}}\]is not stable
but \[He_{2}^{+}\] is expected to exist
(c) Bond strength of \[{{N}_{2}}\]
is maximum among homonuclear diatomic molecules belonging to second period
(d) The order of energies of
molecular orbitals in \[{{N}_{2}}\]molecule is \[\sigma 2s<{{\sigma
}^{*}}2s<\sigma 2{{p}_{z}}<(\pi 2{{p}_{x}}=\pi 2{{p}_{y}})<\]\[({{\pi
}^{*}}2{{p}_{x}}={{\pi }^{*}}2{{p}_{y}})<{{\sigma }^{*}}2{{p}_{z}}\]
View Answer play_arrow
-
question_answer64)
Which of the following options
represents the correct bond order?
(a) \[O_{2}^{-}>{{O}_{2}}>O_{2}^{+}\] (b)
\[O_{2}^{-}<{{O}_{2}}<O_{2}^{+}\] (c) \[O_{2}^{-}>{{O}_{2}}<O_{2}^{+}\] (d)
\[O_{2}^{-}<{{O}_{2}}>O_{2}^{+}\]
View Answer play_arrow
-
question_answer65)
The electronic configuration of
the outermost shell of most electronegative element is :
(a)\[2{{s}^{2}}2{{p}^{5}}\] (b)
\[3{{s}^{2}}3{{p}^{5}}\] (c) \[4{{s}^{2}}4{{p}^{5}}\] (d)
\[5{{s}^{2}}5{{p}^{5}}\]
View Answer play_arrow
-
question_answer66)
Amongst the following elements
whose electronic configurations are given below, the one having the highest
ionisation enthalpy is
(a) \[[Ne]3{{s}^{2}}3{{p}^{1}}\] (b)
\[[Ne]3{{s}^{2}}3{{p}^{3}}\] (c) \[[Ne]3{{s}^{2}}3{{p}^{2}}\] (d)
\[[Ar]3{{d}^{10}}4{{s}^{2}}4{{p}^{3}}\]
View Answer play_arrow
-
question_answer67)
In the following questions two
or more options may be correct.
Which of the following have
identical bond order?
(a) \[C{{N}^{-}}\] (b)
\[N{{O}^{+}}\] (c) \[O_{2}^{-}\] (d)
\[O_{2}^{2-}\]
View Answer play_arrow
-
question_answer68)
Which of the following attain
the linear structure?
(a) \[BeC{{l}_{2}}\] (b)
\[NC{{O}^{+}}\] (c) \[N{{O}_{2}}\] (d)\[C{{S}_{2}}\]
View Answer play_arrow
-
question_answer69)
CO is isoelectronic with :
(a) \[N{{O}^{+}}\] (b)
\[{{N}_{2}}\] (c) \[SnC{{l}_{2}}\] (d)
\[N{{O}_{2}}\]
View Answer play_arrow
-
question_answer70)
Which of the following species
have the same shape?
(a) \[C{{O}_{2}}\] (b)
\[CC{{l}_{4}}\] (c)\[{{O}_{3}}\] (d)
\[NO_{2}^{-}\]
View Answer play_arrow
-
question_answer71)
Which of the following
statements are correct about \[CO_{3}^{2-}\]?
(a) The hybridisation of central
atom is \[s{{p}^{3}}\]
(b) Its resonance structure has
one C-O single bond and two C = O double bonds
(c) The average formal charge on
each oxygen atom is -0.67 units
(d) All C-O bond lengths are
equal
View Answer play_arrow
-
question_answer72)
Diamagnetic species are those
which contain no unpaired electrons. Which among the following are diamagnetic?
(a) \[{{N}_{2}}\] (b)
\[N_{2}^{-}\] (c) \[{{O}_{2}}\] (d)
\[O_{2}^{2-}\]
View Answer play_arrow
-
question_answer73)
Species having same bond order
are :
(a) \[{{N}_{2}}\] (b)
\[N_{2}^{-}\] (c)\[{{O}_{2}}\] (d)
\[O_{2}^{-}\]
View Answer play_arrow
-
question_answer74)
Which of the following
statements are not correct?
(a) \[NaCl\]being an ionic
compound is a good conductor ofelectricity in the solid state
(b) In canonical structures
there is difference in the arrangement of atoms
(c) Hybrid orbitals form
stronger bonds than pure orbitals
(d) VSEPR theory can explain the
square planar geometry of\[Xe{{F}_{4}}\]
View Answer play_arrow
-
question_answer75)
Explain the non-linear shape of \[{{H}_{2}}S\]
and non-planar shape of \[PC{{l}_{3}}\] using valence shell electron pair
repulsion theory.
View Answer play_arrow
-
question_answer76)
Using molecular orbital theory,
compare the bond energy and magnetic character of \[O_{2}^{+}\] and \[O_{2}^{-}\]
species.
View Answer play_arrow
-
question_answer77)
Explain the shape of \[Br{{F}_{5}}\]
View Answer play_arrow
-
question_answer78)
Structures of molecules of two
compounds are given below :
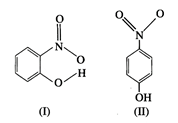 (a) Which of the two compounds
will have intermolecular hydrogen bonding and which compound is expected to show
intramolecular hydrogen bonding?
(b) The melting point of a
compound depends on, among other things, the extent of hydrogen bonding. On
this basis explain which of the above two compounds will show higher melting
point?
(c) Solubility of compounds in
water depends on power toform hydrogen bonds with water. Which of the
abovecompounds will form hydrogen bond with water easilyand be more soluble in
it?
(a) Which of the two compounds
will have intermolecular hydrogen bonding and which compound is expected to show
intramolecular hydrogen bonding?
(b) The melting point of a
compound depends on, among other things, the extent of hydrogen bonding. On
this basis explain which of the above two compounds will show higher melting
point?
(c) Solubility of compounds in
water depends on power toform hydrogen bonds with water. Which of the
abovecompounds will form hydrogen bond with water easilyand be more soluble in
it?
View Answer play_arrow
-
question_answer79)
Why does type of overlap given
in the following figure not result in bond formation?
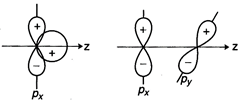
View Answer play_arrow
-
question_answer80)
Explain why \[PC{{l}_{5}}\] is
trigonal bipyramidal whereas \[I{{F}_{5}}\] is square pyramidal?
View Answer play_arrow
-
question_answer81)
In both water and dimethyl ether
\[(C{{H}_{3}}-\underset{\centerdot \centerdot }{\overset{\centerdot \centerdot
}{\mathop{O}}}\,-C{{H}_{3}})\]oxygen atom is central atom and has the same hybridisation,
yet they have different bond angles. Which one has greater bond angle? Give
reason.
View Answer play_arrow
-
question_answer82)
Write Lewis structure of the
following compounds and show formal charges on each of them.
\[HN{{O}_{3}},N{{O}_{2}},{{H}_{2}}S{{O}_{4}}\]
View Answer play_arrow
-
question_answer83)
The energy of\[\sigma 2{{p}_{z}}\]
molecular orbital is greater than \[\pi 2{{p}_{x}}\] and \[\pi 2{{p}_{y}}\]
molecular orbitals in nitrogen molecule. Write complete sequence of energy
levels in the increasing order of energy in the molecule. Compare the relative
stability and magnetic behaviour of the following species :
\[{{N}_{2}},N_{2}^{+},N_{2}^{-},N_{2}^{2+}\]
View Answer play_arrow
-
question_answer84)
What is the effect of the following
processes on the bond order in \[{{N}_{2}}\]and\[{{O}_{2}}\]?
(i) \[{{N}_{2}}\to
N_{2}^{+}+{{e}^{-}}\]
(ii) \[{{O}_{2}}\to
O_{2}^{+}+{{e}^{-}}\]
View Answer play_arrow
-
question_answer85)
Give reasons for the following :
(a) Covalent bonds are
directional bonds while ionic bonds are non-directional.
(b) Water molecule has bent
structure whereas carbondioxide molecule is linear.
(c) Ethyne molecule is linear.
View Answer play_arrow
-
question_answer86)
What is an ionic bond? With two suitable
examples, explain the difference between an ionic and a covalent bond.
View Answer play_arrow
-
question_answer87)
Arrange the following bonds in
order of increasing ionic character giving reason.
\[N-H,F-H,C-H\]and\[O-H\]
View Answer play_arrow
-
question_answer88)
Explain why \[CO_{3}^{2-}\] ion cannot
be represented by a single Lewis structure? How can it be best represented?
View Answer play_arrow
-
question_answer89)
Predict the hybridisation of
each carbon atom in the molecule of organic compound given below. Also indicate
the total number of \[\sigma \]- and \[\pi \]-bonds in this molecule.
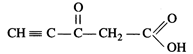
View Answer play_arrow
-
question_answer90)
Group the following as linear
and non-linear molecules.
\[{{H}_{2}}O,HOCl,BeC{{l}_{2}},C{{l}_{2}}O\]
View Answer play_arrow
-
question_answer91)
Elements X, Y and Z have 4, 5 and 7
valence electrons respectively.
(i) Write the molecular formula
of the compound formed by these elements individually with hydrogen.
(ii) Which of these compound
will have the highest dipole moments?
View Answer play_arrow
-
question_answer92)
Draw the resonating structures
of :
(i) Ozone molecule
(ii) Nitrate ion
View Answer play_arrow
-
question_answer93)
Predict the shapes of the following
molecules on the basis of hybridisation
\[BC{{l}_{3}},C{{H}_{4}},C{{O}_{2}},NH3\]
View Answer play_arrow
-
question_answer94)
All the C- O bonds in carbonate ion \[(CO_{3}^{2-})\]are
equal in length Explain.
View Answer play_arrow
-
question_answer95)
What is meant by average bond
enthalpy? Why is there difference in bond enthalpy of O - H bond in ethanol and
water?
View Answer play_arrow
-
question_answer96)
Matching the species in column I
with the type of hybrid orbitals in column ll.
|
Column I
|
Column II
|
|
(i)\[S{{F}_{4}}\]
|
(a) \[s{{p}^{3}}{{d}^{2}}\]
|
|
(ii) \[I{{F}_{5}}\]
|
(b) \[{{d}^{2}}s{{p}^{3}}\]
|
|
(iii)\[NO_{2}^{+}\]
|
(c) \[s{{p}^{3}}d\]
|
|
(iv) \[NH_{4}^{+}\]
|
(d) \[s{{p}^{3}}\]
|
|
|
(e) \[sp\]
|
View Answer play_arrow
-
question_answer97)
Match the species in column I with
geometry/ shape in column II.
|
Column I
|
Column II
|
|
(i) \[{{H}_{2}}{{O}^{+}}\]
|
(a) Linear
|
|
(ii) \[HC\equiv CH\]
|
(b) Angular
|
|
(iii)\[ClO_{2}^{-}\]
|
(c) Tetrahedral
|
|
(iv) \[NH_{4}^{+}\]
|
(d) Trigonal
bipyramidal
|
|
|
(e) Pyramidal
|
View Answer play_arrow
-
question_answer98)
Match the species in column I
with bond order in column II.
|
Column I
|
Column II
|
|
(i)\[NO\]
|
(a) 1.5
|
|
(ii)\[CO\]
|
(b) 2.0
|
|
(iii)\[O_{2}^{-}\]
|
(c) 2.5
|
|
(iv) \[{{O}_{2}}\]
|
(d) 3.0
|
View Answer play_arrow
-
question_answer99)
Match the items in column I with
examples gives in column II.
|
Column I
|
Column II
|
|
(i) Hydrogen bond
|
(a) C
|
|
(ii) Resonance
|
(b) \[LiF\]
|
|
(iii) Ionic solid
|
(c) \[{{H}_{2}}\]
|
|
(iv) Covalent solid
|
(d) \[HF\]
|
|
|
(e)\[{{O}_{3}}\]
|
View Answer play_arrow
-
question_answer100)
Match the shape of the molecules
in column I with the type of hybridisation in column II.
|
Column I
|
Column II
|
|
(i) Tetrahedral
|
(a) \[s{{p}^{2}}\]
|
|
(ii) Trigonal
|
(b)\[sp\]
|
|
(iii) Linear
|
(c)\[s{{p}^{3}}\]
|
View Answer play_arrow
-
question_answer101)
In the following questions a
statement of Assertion (A) followed by a statement of Reason (R) is given.
Choose the correct option out of the choices given below :
(a) A and R both are true and R
is the correct explanation of A.
(b) A and R both are true but R
is not the correct explanation of A.
(c) A is true but R is false.
(d) A and R both are false.
Assertion (A) : Sodium
chloride formed by the action of chlorine gas on sodium metal is a stable
compound.
Reason (R) : This is
because sodium and chloride ions acquire octet in sodium chloride formation.
View Answer play_arrow
-
question_answer102)
Assertion (A) : Though
the central atom of both \[N{{H}_{3}}\] and \[{{H}_{2}}O\]molecules are \[s{{p}^{3}}\]
-hybridised. Yet H-N-H bond angle is greater than that of H-O-H.
Reason (R) : This is
because nitrogen atom has one lone pair and oxygen atom has two lone pairs.
View Answer play_arrow
-
question_answer103)
Assertion (A) : Among the
two O-H bonds in water molecule, the energy required to break the first O - H
bond and the other O-H bond is the same.
Reason (R) : This is
because the electronic environment around oxygen is the same even after
breakage of one O-H bond.
View Answer play_arrow
-
question_answer104)
(i) Discuss the
significance/applications of dipole moment.
(ii) Represent diagrammatically
the bond moments and the resultant dipole moment in \[C{{O}_{2}},N{{F}_{3}}\]
and \[CHC{{l}_{3}}\]
View Answer play_arrow
-
question_answer105)
Use the molecular orbital energy
level diagram to show that \[{{N}_{2}}\] would be expected to have a triple
bond, \[{{F}_{2}}\], a single bond and \[N{{e}_{2}}\], no bond.
View Answer play_arrow
-
question_answer106)
Briefly describe the valence
bond theory of covalent bond formation by taking an example of hydrogen. How
can you interpret energy changes taking place in the formation of di- hydrogen?
View Answer play_arrow
-
question_answer107)
Describe hybridisation in case
of \[PC{{l}_{5}}\] and \[S{{F}_{6}}\]. The axial bonds are longer as compared
to equatorial bonds in \[PC{{l}_{5}}\] whereas in \[S{{F}_{6}}\]both axial
bonds and equatorial bonds have same length. Explain.
View Answer play_arrow
-
question_answer108)
(i) Discuss the concept of
hybridisation. What are its different types in carbon atom?
(ii) What is the type of hybridisation
of carbon atoms marked with star?
(a)\[\overset{*}{\mathop{C}}\,{{H}_{2}}=CH-\overset{\overset{\text{O}}{\mathop{||}}\,}{\mathop{{}^{*}C}}\,-O-H\]
(b)\[C{{H}_{3}}-\overset{*}{\mathop{C}}\,{{H}_{2}}-OH\]
(c)\[C{{H}_{3}}-C{{H}_{2}}-\overset{\overset{\text{O}}{\mathop{||}}\,}{\mathop{{}_{*}C}}\,-H\]
(d)\[\overset{*}{\mathop{C}}\,{{H}_{3}}-CH=CH-C{{H}_{3}}\]
(e) \[C{{H}_{3}}-\overset{*}{\mathop{C}}\,\equiv
CH\]
View Answer play_arrow
-
question_answer109)
Comprehension given below is followed
by some multiple choice questions. Each question has one correct option. Choose
the correct option.
Molecular orbitals are formed by
overlap of atomic orbitals. Two atomic orbitals combine to form two molecular
orbitals called bonding molecular orbital (BMO) and antibonding molecular
orbital (ABMO). Energy of antibonding orbital is raised above the parent atomic
orbitals that have combined and the energy of bonding orbital is lowered than
the parent atomic orbitals. Energies of various molecular orbitals for elements
hydrogen to nitrogen increase in the order \[\sigma 1s<{{\sigma
}^{*}}1s<\sigma 2s<{{\sigma }^{*}}2s<\]\[(\pi 2{{p}_{x}}\approx \pi
2{{p}_{y}})\]\[<\sigma 2{{p}_{z}}<({{\pi }^{*}}2{{p}_{x}}\approx {{\pi
}^{*}}2{{p}_{y}})<{{\sigma }^{*}}2{{p}_{z}}\] and for oxygen and fluorine order of energy of molecular
orbitals is given below :
\[\sigma 1s<{{\sigma
}^{*}}1s<\sigma 2s<{{\sigma }^{*}}2s<\sigma 2{{p}_{z}}<\]
\[(\pi 2{{p}_{x}}\approx \pi
2{{p}_{y}})<({{\pi }^{*}}2{{p}_{x}}\approx {{\pi
}^{*}}2{{p}_{y}})<{{\sigma }^{*}}2{{p}_{z}}.\]
Different atomic orbitals of one
atom combine with those atomic orbitals of the second atom which have
comparable energies and proper orientation. Further, if the overlapping is head
on, the molecular orbital is called 'sigma' \[(\sigma )\] and if the overlap is
lateral, the molecular orbital is called 'pi', (\[\pi \]). The molecular
orbitals are filled with electrons according to the same rules as followed for
filling of atomic orbitals. However, the order for filling is not the same for
all molecules or their ions. Bond order is one of the most important parameters
to compare the strength of the bonds.
Which of the following
statements is correct?
(a) In the formation of dioxygen
from oxygen atoms 10 molecular orbitals will be formed.
(b) All the molecular orbitals
in the dioxygen will be completely filled.
(c) Total number of bonding
molecular orbitals will not be same as total number of antibonding orbitals in dioxygen.
(d) Number of filled bonding
orbitals will be same as number of filled antibonding orbitals.
View Answer play_arrow
-
question_answer110)
Which of the following molecular
orbitals has maximum number of nodal planes?
(a) \[{{\sigma }^{*}}1s\]
(b) \[{{\sigma }^{*}}2{{p}_{x}}\] (c)
\[\pi 2{{p}_{x}}\] (d) \[{{\pi
}^{*}}2{{p}_{y}}\]
View Answer play_arrow
-
question_answer111)
Which of the following pairs is
expected to have the same bond order?
(a) \[{{O}_{2}},{{N}_{2}}\]
(b) \[O_{2}^{+},N_{2}^{-}\] (c) \[O_{2}^{-},N_{2}^{+}\]
(d) \[O_{2}^{-},N_{2}^{-}\]
View Answer play_arrow
-
question_answer112)
In which of the following
molecules \[\sigma 2{{p}_{z}}\]molecular orbital is filled after \[\pi
2{{p}_{x}}\] and \[\pi 2{{p}_{y}}\] molecular
orbitals?
(a) \[{{O}_{2}}\] (b)
\[{{F}_{2}}\] (c) \[{{N}_{2}}\]
(d) None of these
View Answer play_arrow

