Answer:
(i)
Applications of dipole moment.
(a)
To decide polarity of the molecule
(b)
To determine percentage of ionic character
(c)
To determine geometry of molecules
(d)
To determine orientation in benzene ring
(e)
To distinguish cis and trans forms of geometrical forms
(f) To
determine bond moment
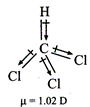
(ii)
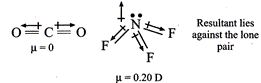
You need to login to perform this action.
You will be redirected in
3 sec
