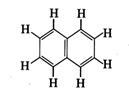
 (a) 6, 19 (b)
4, 20 (c) 5, 19 (d)
5, 20
(a) 6, 19 (b)
4, 20 (c) 5, 19 (d)
5, 20
Answer:
(c)
Every single bond has one sigma bond and every double bond has one sigma and
one \[\pi \]-bond. The given structure has five double bonds and fourteen
single bonds.
\[\therefore \] \[\pi \]-bonds = 5
\[\sigma \]-bonds = 14 + 5 = 19
You need to login to perform this action.
You will be redirected in
3 sec
