Answer:
(c)\[{{C}_{2}}{{H}_{4}}\]has
double and single bonds in its structure.
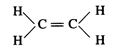 The
bond length of double bond is shorter than C-H single bonds.
The
bond length of double bond is shorter than C-H single bonds.
You need to login to perform this action.
You will be redirected in
3 sec
