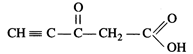
Answer:
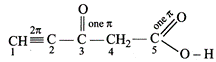
Total
number of sigma bonds = 11, \[\pi \]-bonds = 4
Carbon No.
Hybridisation
1
\[sp\]
2
\[sp\]
3
\[s{{p}^{2}}\]
4
\[s{{p}^{3}}\]
5
\[s{{p}^{2}}\]
You need to login to perform this action.
You will be redirected in
3 sec
