question_answer 1) A simple pendulum is made of a body which is a hollow sphere containing mercury suspended by means of a wire. If a little mercury is drained off, the period of pendulum will:
A)
remain unchanged
done
clear
B)
increase
done
clear
C)
decrease
done
clear
D)
become erratic
done
clear
View Answer play_arrow
question_answer 2) Two balloons are filled, one with pure He gas and the other by air, respectively. If the pressure and temperature of these balloons are same then the number of molecules per unit volume is:
A)
more in the He filled balloon
done
clear
B)
same in both balloons
done
clear
C)
more in air filled balloon
done
clear
D)
in the ratio of 1 : 4
done
clear
View Answer play_arrow
question_answer 3) For inelastic collision between two spherical rigid bodies:
A)
the total kinetic energy is conserved
done
clear
B)
the total mechanical energy is not conserved
done
clear
C)
the linear momentum is not conserved
done
clear
D)
the linear momentum is conserved
done
clear
View Answer play_arrow
question_answer 4) We consider a thermodynamic system. If \[\Delta U\] represents the increase in its internal energy and IV the work done by the system, which of the following statements is true?
A)
\[\Delta U\] = - W in an adiabatic process
done
clear
B)
\[\Delta U\] = W in an isothermal process
done
clear
C)
\[\Delta U\] = - W in an isothermal process
done
clear
D)
\[\Delta U\] = W in an adiabaitc process
done
clear
View Answer play_arrow
question_answer 5) Aerofils are so designed that the speed of air:
A)
on top side is more than on lower side
done
clear
B)
on lop side is less than on lower side
done
clear
C)
is same on both sides
done
clear
D)
is turbulent
done
clear
View Answer play_arrow
question_answer 6) According to Bernoulli's equation \[\frac{P}{\rho g}+h+\frac{1}{2}\frac{{{v}^{2}}}{g}=cons\tan t\] The terms A. B and C are generally called respectively:
A)
gravitational head, pressure head and velocity head
done
clear
B)
gravity, gravitational head and velocity head
done
clear
C)
pressure head. gravitational head and velocity head
done
clear
D)
gravity, pressure and velocity head
done
clear
View Answer play_arrow
question_answer 7) The time of reverberation of a room A is one second. What will be the lime (in seconds) of reverberation of a room, having all the dimensions double of those of room A?
A)
2
done
clear
B)
4
done
clear
C)
\[\frac{1}{2}\]
done
clear
D)
1
done
clear
View Answer play_arrow
question_answer 8) In refraction, light waves are bent on passing from one medium to the second medium, because, in the second medium:
A)
the frequency is different
done
clear
B)
the coefficient of elasticity is different
done
clear
C)
the speed is different
done
clear
D)
the amplitude is smaller
done
clear
View Answer play_arrow
question_answer 9) Light enters at an angle of incidence in a transparent rod of refractive index of the material of the rod the light once entered into it will not leave it through its lateral face whatsoever be the value of angle of incidence?
A)
\[n>\sqrt{2}\]
done
clear
B)
n = 1
done
clear
C)
\[n=1.1\]
done
clear
D)
n = 1.3
done
clear
View Answer play_arrow
question_answer 10) 27 small drops each having charge q and radius r coalesce to from big drop. How many times charge and capacitance will become?
A)
3, 27
done
clear
B)
27, 3
done
clear
C)
27, 27
done
clear
D)
3, 3
done
clear
View Answer play_arrow
question_answer 11) The magnetic moment has dimensions of:
A)
[LA]
done
clear
B)
[L2A]
done
clear
C)
[LT-1A]
done
clear
D)
[L2T-1A]
done
clear
View Answer play_arrow
question_answer 12) Which of the following circular rods, (given radius r and length 1) each made of the same material and whose ends are maintained at the same temperature will conduct most heat?
A)
\[r=2{{r}_{0}};l=2{{l}_{0}}\]
done
clear
B)
\[r=2{{r}_{0}};l={{l}_{0}}\]
done
clear
C)
\[r={{r}_{0}};l={{l}_{0}}\]
done
clear
D)
\[r={{r}_{0}};l=2{{l}_{0}}\]
done
clear
View Answer play_arrow
question_answer 13) For a satellite moving in an orbit around the earth, the ratio of kinetic energy to potential energy is:
A)
2
done
clear
B)
\[\frac{1}{2}\]
done
clear
C)
\[\frac{1}{\sqrt{2}}\]
done
clear
D)
\[\sqrt{2}\]
done
clear
View Answer play_arrow
question_answer 14) 300 J of work is done in sliding a 2 kg block up an inclined plane of height 10 m. Taking \[g=10m/{{s}^{2}},\]work done against friction is:
A)
200 J
done
clear
B)
100 J
done
clear
C)
zero
done
clear
D)
1000 J
done
clear
View Answer play_arrow
question_answer 15) Interference was observed in an interference chamber when air was present. Now, the chamber is evacuated and if the same light is used, a careful observation will show:
A)
no interference
done
clear
B)
interference with bright band
done
clear
C)
interference with dark bands
done
clear
D)
interference in which breadth of the fringe will be slightly increased
done
clear
View Answer play_arrow
question_answer 16) When a charged particle moving with velocity \[\overrightarrow{v}\] is subjected to a magnetic field of induction\[\overrightarrow{B,}\]the force on it is non-zero. This implies that:
A)
angle between \[\overrightarrow{v}\] and \[\overrightarrow{B,}\]is necessarily 90°
done
clear
B)
angle between \[\overrightarrow{v}\] and \[\overrightarrow{B,}\]can have any value other than 90°
done
clear
C)
angle between \[\overrightarrow{v}\] and \[\overrightarrow{B,}\] can have any value other than zero and 180°
done
clear
D)
angle between \[\overrightarrow{v}\] and \[\overrightarrow{B,}\] is either zero or 180°
done
clear
View Answer play_arrow
question_answer 17) A particle moves along a straight line OX. At a time t (in seconds) the distance x (in metres) of the particle from O is given by \[x=40+12t-{{t}^{3}}\] How long would the particle travel before coming to rest?
A)
24 m
done
clear
B)
40 m
done
clear
C)
56 m
done
clear
D)
16 m
done
clear
View Answer play_arrow
question_answer 18) Which one of the following statements is true
A)
Both light and sound waves in air are transverse
done
clear
B)
The sound waves in air are longitudinal while the light waves are transverse
done
clear
C)
Both light and sound waves in air are longitudinal
done
clear
D)
Both light and sound waves can travel in vacuum
done
clear
View Answer play_arrow
question_answer 19) When power is drawn from the secondary coil of the transformer, the dynamic resistance:
A)
increases
done
clear
B)
decreases
done
clear
C)
remains unchanged
done
clear
D)
changes erratically
done
clear
View Answer play_arrow
question_answer 20) A reference frame attached to the earth:
A)
is an inertial frame by definition
done
clear
B)
cannot be an inertial frame because earth is revolving round the sun
done
clear
C)
is an inertial frame because Newton's law are applicable
done
clear
D)
is an inertial frame because the earth is rotating about its own axis
done
clear
View Answer play_arrow
question_answer 21) 9Pressure of an ideal gas is increased by keeping temperature constant. What is the effect on kinetic energy of molecules?
A)
Increase
done
clear
B)
Decrease
done
clear
C)
No change
done
clear
D)
Can't be determined
done
clear
View Answer play_arrow
question_answer 22) What maximum frequency can be reflected from ionosphere?
A)
5 MHz
done
clear
B)
6 GHz
done
clear
C)
5 kHz
done
clear
D)
500 MHz
done
clear
View Answer play_arrow
question_answer 23) A telescope has focal length of objective m eyepiece as 200 cm and 5 cm respectively. What is magnification of telescope?
A)
40
done
clear
B)
80
done
clear
C)
50
done
clear
D)
0.01
done
clear
View Answer play_arrow
question_answer 24) A small disc of radius 2 cm is cut from n disc of radius 6 cm. If the distance between their centres is 3.2 cm, what is the shift in the centre of mass of the disc?
A)
0.4 cm
done
clear
B)
2.4 cm
done
clear
C)
1.8 cm
done
clear
D)
1.2 cm
done
clear
View Answer play_arrow
question_answer 25) Which of the following is not a process involved in fabrication of IC?
A)
Polymerization
done
clear
B)
Diffusion
done
clear
C)
Photolithography
done
clear
D)
Metallisation
done
clear
View Answer play_arrow
question_answer 26) Permanent magnet has properties retentivity and coercivity respectively:
A)
high- high
done
clear
B)
low- low
done
clear
C)
low- high
done
clear
D)
high- low
done
clear
View Answer play_arrow
question_answer 27) Hydrogen bomb is based upon:
A)
fission
done
clear
B)
fusion
done
clear
C)
chemical reaction
done
clear
D)
transmutation
done
clear
View Answer play_arrow
question_answer 28) What is not true for equipotential surface for uniform electric field?
A)
Equipotential surface is flat
done
clear
B)
Equipotential surface is spherical
done
clear
C)
Electric lines are perpendicular to equipotential surface
done
clear
D)
Work done is zero
done
clear
View Answer play_arrow
question_answer 29) A closed organ pipe of length 20 cm is sounded with tuning fork in resonance. What is the frequency of tuning fork? (y = 332 m/s)
A)
300 Hz
done
clear
B)
350 Hz
done
clear
C)
375 Hz
done
clear
D)
415 Hz
done
clear
View Answer play_arrow
question_answer 30) The displacement of particle is given by \[x={{a}_{0}}+\frac{{{a}_{1}}t}{2}-\frac{{{a}_{2}}{{t}^{2}}}{3}\]What is its acceleration?
A)
\[\frac{2{{a}_{2}}}{3}\]
done
clear
B)
\[-\frac{2{{a}_{2}}}{3}\]
done
clear
C)
\[{{a}_{2}}\]
done
clear
D)
Zero
done
clear
View Answer play_arrow
question_answer 31) The area of the acceleration-displacement curve of a body gives:
A)
impulse
done
clear
B)
change in momentum per unit mass
done
clear
C)
change in KE per unit mass
done
clear
D)
total change in energy
done
clear
View Answer play_arrow
question_answer 32) The speed of earth's rotation about its axis is \[\omega \]. Its speed is increased to x times to make the effective acceleration due to gravity equal to zero at the equator. Then x is:
A)
1
done
clear
B)
8.5
done
clear
C)
17
done
clear
D)
34
done
clear
View Answer play_arrow
question_answer 33) Hard X-rays for the study of fractures in bones should have a minimum wavelength of 10-11 m. The accelerating voltage for electrons in X-ray machine should be:
A)
< 124 kV
done
clear
B)
> 124 RV
done
clear
C)
between 60 kV and 70 kV
done
clear
D)
= 100 kV
done
clear
View Answer play_arrow
question_answer 34) In photoelectric effect, the electrons are ejected from metals if the incident light has a certain minimum:
A)
wavelength
done
clear
B)
frequency
done
clear
C)
amplitude
done
clear
D)
angle of incidence
done
clear
View Answer play_arrow
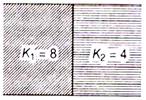
question_answer 35)
A capacitor having capacitance 1\[\mu \]F with air, is filled with two dielectrics as shown. How many times capacitance will increase?
A)
12
done
clear
B)
6
done
clear
C)
8/3
done
clear
D)
3
done
clear
View Answer play_arrow
question_answer 36) A leaf which contains only green pigments, is illuminated by a laser light of wavelength 0.6328\[\mu m\]. It would appear to be:
A)
brown
done
clear
B)
black
done
clear
C)
red
done
clear
D)
green
done
clear
View Answer play_arrow
question_answer 37) The operation of a nuclear reactor is said to be critical, if the multiplication factor (k) has a value:
A)
1
done
clear
B)
1.5
done
clear
C)
2.1
done
clear
D)
2.5
done
clear
View Answer play_arrow
question_answer 38) A galvanometer having a resistance of 8\[\Omega \] is shunted by a wire of resistance 2\[\Omega \]. If the total current is 1 A, the part of it passing through the shunt will be:
A)
0.25 A
done
clear
B)
0.8 A
done
clear
C)
0.2 A
done
clear
D)
0.5 A
done
clear
View Answer play_arrow
question_answer 39) Flash light equipped with a new set of batteries, produces bright white light. As the batteries wear out:
A)
the light intensity gets reduced with no change in its color
done
clear
B)
light color changes first to yellow and then red with no change in intensity
done
clear
C)
it stops working suddenly while giving white light
done
clear
D)
color changes to red and also intensity gets reduced
done
clear
View Answer play_arrow
question_answer 40) If alpha, beta and gamma rays carry same momentum, which has the longest wavelength?
A)
Alpha rays
done
clear
B)
Beta rays
done
clear
C)
Gamma rays
done
clear
D)
None, all have same wavelength
done
clear
View Answer play_arrow
question_answer 41) An amplifier has a voltage gain Ay = 1000. The voltage gain in dB is:
A)
30 dB
done
clear
B)
60 dB
done
clear
C)
3 dB
done
clear
D)
20 dB
done
clear
View Answer play_arrow
question_answer 42) When you make ice cubes, the entropy of water:
A)
does not change
done
clear
B)
increases
done
clear
C)
decreases
done
clear
D)
may either increase or decrease depending on the process used
done
clear
View Answer play_arrow
question_answer 43) Half- lives of two radioactive substances A and 3 are respectively 20 min and 40 min. Initially the samples of A and B have equal number of nuclei. After 80 min the ratio of remaining number of A and B nuclei is:
A)
1 : 16
done
clear
B)
4 : 1
done
clear
C)
1 : 4
done
clear
D)
1 : 1
done
clear
View Answer play_arrow
question_answer 44) Given that y = A sin \[\left[ \left( \frac{2\pi }{\lambda }(ct-x \right) \right],\]where y and x are measured in metres. Which of the following statements is true?
A)
The unit of \[\lambda \] is same as that of x and A
done
clear
B)
The unit of \[\left( ct-x \right)\]is same as that of x but not of A
done
clear
C)
The unit of c is same as that of \[\frac{2\pi }{\lambda }\]
done
clear
D)
The unit of (ct - x) is same as that of \[\frac{2\pi }{\lambda }\]
done
clear
View Answer play_arrow
question_answer 45) A projectile is thrown in the upward direction making an angle of 600 with the horizontal direction with a velocity of 147 ms-1. Then the time after which its inclination with the horizontal is 45°, is:
A)
15 s
done
clear
B)
10.98 s
done
clear
C)
5.49 s
done
clear
D)
2.745 s
done
clear
View Answer play_arrow
question_answer 46) A motor cycle is going on an over bridge of radius R. The driver maintains a constant speed. As the motor cycle is ascending on the over bridge the normal force on it:
A)
In creases
done
clear
B)
decreases
done
clear
C)
remains the same
done
clear
D)
fluctuates erratically
done
clear
View Answer play_arrow
question_answer 47) If we throw a body upwards with velocity of 4 m/s, at what height does its kinetic energy reduce to half of the initial value? (Take g= 10 ms-2)
A)
4 m
done
clear
B)
2 m
done
clear
C)
1 m
done
clear
D)
0.4 m
done
clear
View Answer play_arrow
question_answer 48) Two glass plates are separated by water. If surface tension of water is 75 dynes/cm and 1 area of each plate wetted by water is \[8\text{ }c{{m}^{2}}\]and) the distance between the plates is 0.12 mm, then the force applied to separate the two plates is:
A)
102 dyne
done
clear
B)
104 dyne
done
clear
C)
105 dyne
done
clear
D)
106 dyne
done
clear
View Answer play_arrow
question_answer 49) Which of the following is true for rays coming from infinity?
A)
Two images are formed
done
clear
B)
Continuous image is formed between focal points of upper and lower lens
done
clear
C)
One image is formed
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 50) The north pole of a long horizontal bar magnet is being brought closer to a vertical conducting plane along the perpendicular direction. The direction of the induced current in the conducting plane will be:
A)
horizontal
done
clear
B)
vertical
done
clear
C)
clockwise
done
clear
D)
anticlockwise
done
clear
View Answer play_arrow
question_answer 51) Calcium carbide reacts with\[{{\text{H}}_{\text{2}}}\text{O}\] to produce :
A)
ethane
done
clear
B)
methane
done
clear
C)
ethylene
done
clear
D)
acetylene
done
clear
View Answer play_arrow
question_answer 52) RNA differ from DNA in respect of base :
A)
thyamine
done
clear
B)
adenine
done
clear
C)
cytosine
done
clear
D)
guanine
done
clear
View Answer play_arrow
question_answer 53) The oxidation number of nitrogen in \[\text{NaN}{{\text{O}}_{\text{2}}}\]is:
A)
\[\text{+3}\]
done
clear
B)
+5
done
clear
C)
-3
done
clear
D)
-5
done
clear
View Answer play_arrow
question_answer 54) Bauxite is an ore of:
A)
Al
done
clear
B)
Ca
done
clear
C)
Cu
done
clear
D)
Ni
done
clear
View Answer play_arrow
question_answer 55) The best source of vitamin A is :
A)
beans
done
clear
B)
pulses
done
clear
C)
orange
done
clear
D)
carrot
done
clear
View Answer play_arrow
question_answer 56) Plaster of parts is :
A)
\[CaS{{O}_{4}}.\frac{1}{2}{{H}_{2}}O\]
done
clear
B)
\[CaS{{O}_{4}}.2{{H}_{2}}O\]
done
clear
C)
\[CaS{{O}_{4}}.{{H}_{2}}O\]
done
clear
D)
\[CaS{{O}_{4}}.4{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 57) Penicillin is :
A)
analgesic
done
clear
B)
antipyretic
done
clear
C)
antimalarials
done
clear
D)
antibiotic
done
clear
View Answer play_arrow
question_answer 58) The most stable compound is :
A)
LiF
done
clear
B)
LiCi
done
clear
C)
LiBr
done
clear
D)
Lil
done
clear
View Answer play_arrow
question_answer 59) Heavy water is :
A)
\[CaS{{O}_{4}}\]
done
clear
B)
water contain \[CaS{{O}_{4}},MgS{{O}_{4}}\]
done
clear
C)
\[{{D}_{2}}P\]
done
clear
D)
water contain \[CaC{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 60) When copper reacts with hot and conc. \[{{H}_{2}}S{{O}_{4}},\] gives :
A)
\[{{H}_{2}}\]
done
clear
B)
\[{{N}_{2}}\]
done
clear
C)
\[{{O}_{2}}\]
done
clear
D)
\[S{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 61) \[\text{Ba}{{\text{O}}_{\text{2}}}\]and ozone reacts to produce :
A)
Ba
done
clear
B)
\[B{{a}_{2}}{{O}_{3}}\]
done
clear
C)
BaO
done
clear
D)
\[Ba{{(OH)}_{3}}\]
done
clear
View Answer play_arrow
question_answer 62) Heisenberg uncertainty principle can be explained as :
A)
\[\Delta x\ge \frac{\Delta P\times h}{4\pi }\]
done
clear
B)
\[\Delta x\times \Delta P\ge \frac{h}{4\pi }\]
done
clear
C)
\[\Delta x\times \Delta P\ge \frac{h}{\pi }\]
done
clear
D)
\[\Delta P\ge \frac{\pi h}{\Delta x}\]
done
clear
View Answer play_arrow
question_answer 63) A gas mixture contains \[{{\text{O}}_{\text{2}}}\] and \[{{\text{N}}_{\text{2}}}\]in the ratio of 1: 4 by weight. The ratio of their number of molecules is :
A)
1:8
done
clear
B)
1 : 4
done
clear
C)
3: 16
done
clear
D)
7: 32
done
clear
View Answer play_arrow
question_answer 64) Bleaching powder is obtained by treating \[\text{C}{{\text{l}}_{2}}\]with :
A)
\[Ca{{(OH)}_{2}}\]
done
clear
B)
\[CaO\]
done
clear
C)
\[CaC{{O}_{3}}\]
done
clear
D)
\[CaC{{l}_{2}}\]
done
clear
View Answer play_arrow
question_answer 65) Magnalium contains :
A)
\[Mg+Al\]
done
clear
B)
\[Mg+Cu\]
done
clear
C)
\[Mg+Fe\]
done
clear
D)
\[Mg+Mn\]
done
clear
View Answer play_arrow
question_answer 66) The de-Broglie wavelenght of a particle with mass 1 kg and velocity 100 m/s is :
A)
\[6.6\times {{10}^{-33}}m\]
done
clear
B)
\[6.6\times {{10}^{-36}}m\]
done
clear
C)
\[3.3\times {{10}^{+33}}m\]
done
clear
D)
\[3.3\times {{10}^{-36}}m\]
done
clear
View Answer play_arrow
question_answer 67) The volume of a gas measured at \[{{27}^{o}}C\] and 1 atm pressure is 10 L. To reduce the volume to 2 L at 1 atm. pressure, the temperature required is:
A)
60 K
done
clear
B)
75 K
done
clear
C)
150 K
done
clear
D)
225 K
done
clear
View Answer play_arrow
question_answer 68) Isotonic solutions have :
A)
same vapour pressure
done
clear
B)
same osmotic pressure
done
clear
C)
same boiling point
done
clear
D)
same temperature
done
clear
View Answer play_arrow
question_answer 69) Epsom salt is :
A)
\[BaS{{O}_{4}}.2{{H}_{2}}O\]
done
clear
B)
\[CaS{{O}_{4}}-{{H}_{2}}O\]
done
clear
C)
\[MgS{{O}_{4}}.2{{H}_{2}}O\]
done
clear
D)
\[~MgS{{O}_{4}}.7{{H}_{2}}O\]
done
clear
View Answer play_arrow
question_answer 70) Paracetamol is an :
A)
analgesic
done
clear
B)
antipyretic
done
clear
C)
both (a) and (b)
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 71) The number of moles of oxygen obtained by the electrolytic decomposition of 108 g water is:
A)
2.5
done
clear
B)
3
done
clear
C)
5
done
clear
D)
7.5
done
clear
View Answer play_arrow
question_answer 72) The change in entropy for the fusion, of 1 mole of ice is [mp of ice = 273 K, molar enthalpy of fusion for ice\[=6.0\,kJ\,mo{{l}^{-1}}\]]
A)
\[11.73\text{ }J{{K}^{-1}}\text{ }mo{{l}^{-1}}\]
done
clear
B)
\[18.84\text{ }J{{K}^{-1}}\text{ }mo{{l}^{-1}}\]
done
clear
C)
\[~21.97J{{K}^{-1}}\text{ }mo{{l}^{-1}}\]
done
clear
D)
\[24.47\text{ }J{{K}^{-1}}\text{ }mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
question_answer 73) Which does not give a precipitate with \[\text{AgN}{{\text{O}}_{\text{3}}}\] solution?
A)
\[[Co{{(N{{H}_{3}})}_{6}}]C{{l}_{3}}\]
done
clear
B)
\[[Co{{(N{{H}_{3}})}_{5}}Cl]C{{l}_{2}}\]
done
clear
C)
\[[Co{{(N{{H}_{3}})}_{4}}C{{l}_{2}}]Cl\]
done
clear
D)
\[[Co{{(N{{H}_{3}})}_{3}}C{{l}_{3}}]\]
done
clear
View Answer play_arrow
question_answer 74) Total volume of atoms present in a face centred cubic unit cell of a metal is : (r is atomic radius)
A)
\[\frac{16}{3}\pi {{r}^{3}}\]
done
clear
B)
\[\frac{20}{3}\pi {{r}^{3}}\]
done
clear
C)
\[\frac{24}{3}\pi {{r}^{3}}\]
done
clear
D)
\[\frac{12}{3}\pi {{r}^{3}}\]
done
clear
View Answer play_arrow
question_answer 75) Pure silicon doped with phosphorus is a :
A)
metallic conductor
done
clear
B)
insulator
done
clear
C)
n-type semiconductor
done
clear
D)
p-type semiconductor
done
clear
View Answer play_arrow
question_answer 76) Neutron is discovered by :
A)
Chadwick
done
clear
B)
Rutherford
done
clear
C)
Yukawa
done
clear
D)
Dalton Neutron is discovered by Chadwick.
done
clear
View Answer play_arrow
question_answer 77) What is X in the following nuclear reaction? \[_{7}{{N}^{14}}{{+}_{1}}{{H}^{1}}{{\xrightarrow{{}}}_{8}}{{O}^{15}}+X\]
A)
\[_{0}{{n}^{1}}\]
done
clear
B)
\[_{-1}{{e}^{0}}\]
done
clear
C)
\[_{+1}{{e}^{0}}\]
done
clear
D)
\[\gamma \]
done
clear
View Answer play_arrow
question_answer 78) Solubility product of \[\text{PbC}{{\text{l}}_{\text{2}}}\]at 298 K is \[\text{1}\times {{10}^{-6}}.\]At this temperature solubility of \[\text{PbC}{{\text{l}}_{\text{2}}}\]in mol/L is :
A)
\[{{(1\times {{10}^{-6}})}^{1/2}}\]
done
clear
B)
\[{{(1\times {{10}^{-6}})}^{1/3}}\]
done
clear
C)
\[{{(0.25\times {{10}^{-6}})}^{1/3}}\]
done
clear
D)
\[{{(0.25\times {{10}^{-6}})}^{1/2}}\]
done
clear
View Answer play_arrow
question_answer 79) The pH of a 0.001 M solution of \[HCl\]is :
A)
0
done
clear
B)
3
done
clear
C)
5
done
clear
D)
10
done
clear
View Answer play_arrow
question_answer 80) Gold number is associated with :
A)
amount of gold
done
clear
B)
protective colloids
done
clear
C)
purple of cassius
done
clear
D)
electrophoresis
done
clear
View Answer play_arrow
question_answer 81) Noble gases are used in discharge tubes to give different colours. Reddish-orange glow is due to :
A)
Ar
done
clear
B)
Ne
done
clear
C)
Xe
done
clear
D)
Kr
done
clear
View Answer play_arrow
question_answer 82) The set representing the correct order for first ionisation potential is :
A)
K > Na > Li
done
clear
B)
Be > Mg > Ca
done
clear
C)
B > C > N
done
clear
D)
Ge > Si > C
done
clear
View Answer play_arrow
question_answer 83) Dry ice is :
A)
solid\[C{{O}_{2}}\]
done
clear
B)
solid camphor
done
clear
C)
solid\[S{{O}_{2}}\]
done
clear
D)
solid \[N{{O}_{2}} \]
done
clear
View Answer play_arrow
question_answer 84) Methanol and ethanol are miscible in water due to:
A)
covalent character
done
clear
B)
hydrogen bonding character
done
clear
C)
oxygen bonding character
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 85) Stereoisomers differ in:
A)
configuration
done
clear
B)
conformation
done
clear
C)
they do not differ
done
clear
D)
none of the above
done
clear
View Answer play_arrow
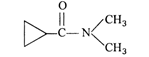
question_answer 86)
IUPAC name of the following compound :
A)
N, N-dimethylcyclopropanecarboxamide
done
clear
B)
N-methylcyclopropanamide
done
clear
C)
cyclopropionamide
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 87) The product of following reaction is : \[2C{{H}_{3}}MgBr+C{{H}_{2}}O\xrightarrow{{}}\]
A)
\[C{{H}_{3}}OH\]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
C)
\[C{{H}_{4}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
View Answer play_arrow
question_answer 88) Freon used as refrigerant is :
A)
\[C{{F}_{2}}=C{{F}_{2}}\]
done
clear
B)
\[C{{H}_{2}}{{F}_{2}}\]
done
clear
C)
\[CC{{l}_{2}}{{F}_{2}}\]
done
clear
D)
\[C{{F}_{4}}\]
done
clear
View Answer play_arrow
question_answer 89) Lucas reagent is:
A)
anhy. \[ZnC{{l}_{2}}\]and \[N{{H}_{3}}\]
done
clear
B)
anhy. \[ZnC{{l}_{2}}\]and \[CaC{{l}_{2}}\]
done
clear
C)
anhy. \[ZnC{{l}_{2}}\]and cone. \[HCl\]
done
clear
D)
anhy. \[ZnC{{l}_{2}}\] and \[HCl\]gas
done
clear
View Answer play_arrow
question_answer 90) The enzyme which can catalyse the conversion of glucose to ethanol is :
A)
zymase
done
clear
B)
invertase
done
clear
C)
maltase
done
clear
D)
diastase
done
clear
View Answer play_arrow
question_answer 91) When dihydroxy acetone reacts with \[\text{HI}{{\text{O}}_{\text{4}}}\text{,}\]the product is/are :
A)
HCHO
done
clear
B)
HCOOH
done
clear
C)
HCHO and HCOOH
done
clear
D)
HCHO and \[C{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 92) Which of the following does not reduce Fehling's solution?
A)
Benzaldehyde
done
clear
B)
Formic acid
done
clear
C)
Glucose
done
clear
D)
Fructose
done
clear
View Answer play_arrow
question_answer 93) Sodium formate on heating gives:
A)
oxalic acid and \[{{\text{H}}_{\text{2}}}\]
done
clear
B)
sodium oxalate and \[{{\text{H}}_{\text{2}}}\]
done
clear
C)
sodium oxalate
done
clear
D)
\[\text{C}{{\text{O}}_{2}}\]and caustic soda
done
clear
View Answer play_arrow
question_answer 94) Reaction of ethyl formate with excess of \[\text{C}{{\text{H}}_{\text{3}}}\text{Mgl}\]followed by hydrolysis gives :
A)
n-propyi alcohol
done
clear
B)
isopropyi alcohol
done
clear
C)
acetaldehyde
done
clear
D)
acetone
done
clear
View Answer play_arrow
question_answer 95) Hydrolysis of phenyl isocyanide forms :
A)
benzoic acid
done
clear
B)
formic acid
done
clear
C)
acetic acid
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 96) Styrene can be purified by :
A)
simple distillation
done
clear
B)
fractional distillation
done
clear
C)
steam distillation
done
clear
D)
vacuum distillation
done
clear
View Answer play_arrow
question_answer 97) Which of the following is not reducing sugar?
A)
Glucose
done
clear
B)
Fructose
done
clear
C)
Lactose
done
clear
D)
Sucrose
done
clear
View Answer play_arrow
question_answer 98) The monomer of teflon is:
A)
\[CHF=C{{H}_{2}}\]
done
clear
B)
\[C{{F}_{2}}=C{{F}_{2}}\]
done
clear
C)
\[CHCl=CHCl\]
done
clear
D)
\[CHF=CHCl\]
done
clear
View Answer play_arrow
question_answer 99) The hybridisation state of carbon in fullerene is :
A)
\[sp\]
done
clear
B)
\[s{{p}^{2}}\]
done
clear
C)
\[s{{p}^{3}}\]
done
clear
D)
\[s{{p}^{3}}d\]
done
clear
View Answer play_arrow
question_answer 100) A fruity smell is produced by the reaction of \[{{C}_{2}}{{H}_{2}}OH\]with:
A)
\[~C{{H}_{3}}COC{{H}_{3}}\]
done
clear
B)
\[C{{H}_{3}}COOH\]
done
clear
C)
\[PC{{l}_{5}}\]
done
clear
D)
\[C{{H}_{3}}CHO\]
done
clear
View Answer play_arrow
question_answer 101) \[N{{a}^{+}}-{{K}^{+}}\]pump is found in membranes of many cells, like nerve cells. It works against electro chemical gradient and involve an integral protein ATPase. For each molecule of ATP used :
A)
3 ions of \[N{{a}^{+}}\]are pumped out and two \[{{K}^{+}}\]are taken in
done
clear
B)
3 ions of \[N{{a}^{+}}\]are taken in and \[2{{K}^{+}}\]are pumped out
done
clear
C)
2 ions of\[N{{a}^{+}}\]are thrown out and \[3{{K}^{+}}\] are absorbed
done
clear
D)
3 ions of \[{{K}^{+}}\] are absorbed and \[3N{{a}^{+}}\] are pumped out
done
clear
View Answer play_arrow
question_answer 102) According to the Neo-Darwinian theory which of the following is responsible for the origin of new species?
A)
Mutations
done
clear
B)
Useful variations
done
clear
C)
Mutations together with natural selection
done
clear
D)
Hybridization
done
clear
View Answer play_arrow
question_answer 103) Enzymes found attached to inner membrane of mitochondria instead of matrix is/are :
A)
succinic dehydrogenase
done
clear
B)
cytochrome oxidase
done
clear
C)
both (a) and (b)
done
clear
D)
malicdehydrogenose
done
clear
View Answer play_arrow
question_answer 104) VAM are :
A)
saprophytic bacteria
done
clear
B)
saprophytic fungi
done
clear
C)
symbiotic fungi
done
clear
D)
symbiatic bacteria
done
clear
View Answer play_arrow
question_answer 105) Part of the plant which is cultured to obtain virus free clones is :
A)
Leaf
done
clear
B)
Root tip
done
clear
C)
shoottip
done
clear
D)
embryo
done
clear
View Answer play_arrow
question_answer 106) If we ignore the effect of crossing over, how many different haploid cells arise by meiosis in a diploid cell having\[2n=12\]?
A)
8
done
clear
B)
16
done
clear
C)
32
done
clear
D)
64
done
clear
View Answer play_arrow
question_answer 107) Double fertilization is fusion of :
A)
two eggs
done
clear
B)
two eggs and polar nuclei
done
clear
C)
one male gamete with egg and other with synergid
done
clear
D)
one male gamete with egg and other with secondary nucleus
done
clear
View Answer play_arrow
question_answer 108) Inheritance of ?ABO? blood group shows :
A)
polyploidy
done
clear
B)
incomplete dominance
done
clear
C)
multipleallelism
done
clear
D)
polygeny
done
clear
View Answer play_arrow
question_answer 109) Photosynthetic enhancement with flashing light was first observed by :
A)
Benson and Calvin
done
clear
B)
Hill and Calvin
done
clear
C)
Hatch and Slack
done
clear
D)
Emerson and Arnold
done
clear
View Answer play_arrow
question_answer 110) Gibberellin was first extracted from :
A)
Gibberellafujikuroi
done
clear
B)
Gelidium
done
clear
C)
Gracelaria
done
clear
D)
Aspergillus
done
clear
View Answer play_arrow
question_answer 111) In E. coli. an operator gene combine with :
A)
inducer gene to switch on structural gene action
done
clear
B)
inducer gene to switch off structural gene action
done
clear
C)
regulator protein (repressor) to switch off structural gene action
done
clear
D)
regulator protein to switch on gene action
done
clear
View Answer play_arrow
question_answer 112) Slime moulds in the division Myxomycota (true slime moulds) have :
A)
pseudoplasmodia
done
clear
B)
sproes that develop into free living amoeboid cells
done
clear
C)
spores that develop into flagellated gametes
done
clear
D)
feeding stages consisting of solitary individual cells
done
clear
View Answer play_arrow
question_answer 113) The stage of Ornithine cycle at which arginase enzyme is used :
A)
\[\text{Ornithine}\to \text{citrulline}\]
done
clear
B)
\[\text{arginine}\to \text{Ornithine}\]
done
clear
C)
\[\text{citrulline}\to \text{arginosuccinic acid}\]
done
clear
D)
\[\text{Ornithine}\to \text{urea}\]
done
clear
View Answer play_arrow
question_answer 114) Immunodeficiency makes a person highly susceptible to infection. It is caused by :
A)
lack of B cells
done
clear
B)
lack of T cells
done
clear
C)
lack of both B and T cells
done
clear
D)
none of the above
done
clear
View Answer play_arrow
question_answer 115) At retrogressive metamorphosis the uro chordate larva :
A)
loss notochord
done
clear
B)
loss tail
done
clear
C)
experience reduction of nervous system to a visceral ganglion
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 116) A species inhabiting different geographical area is known as :
A)
Allopatric
done
clear
B)
Sympatric
done
clear
C)
Biospecies
done
clear
D)
Sibling species
done
clear
View Answer play_arrow
question_answer 117) Pedogamy is the term where :
A)
actual fusion never occur
done
clear
B)
gametes tend to fuse but fusion product never survive
done
clear
C)
the conjugation takes place between parent cell and a bud
done
clear
D)
such term is never known in fungi
done
clear
View Answer play_arrow
question_answer 118) Which type of respiratory organs are present in spiders and scropions ?
A)
book lungs
done
clear
B)
gills
done
clear
C)
gill books
done
clear
D)
lungs
done
clear
View Answer play_arrow
question_answer 119) Energy storage at consumer level is called:
A)
gross primary productivity
done
clear
B)
secondly productivity
done
clear
C)
net primary productivity
done
clear
D)
net productivity
done
clear
View Answer play_arrow
question_answer 120) After examining the blood groups of husband and wife, the doctor advised them not to have more than one child, the blood group of the couple are likely to be :
A)
male\[R{{h}^{-}}\] and female \[\text{R}{{\text{h}}^{+}}\]
done
clear
B)
female\[R{{h}^{-}}\] and male \[\text{R}{{\text{h}}^{+}}\]
done
clear
C)
male\[\text{R}{{\text{h}}^{+}}\] and female \[\text{R}{{\text{h}}^{+}}\]
done
clear
D)
male\[R{{h}^{-}}\] and female \[R{{h}^{-}}\]
done
clear
View Answer play_arrow
question_answer 121) In fishes the lateral line receptors are neuromast organs. These are :
A)
olfactoreceptors
done
clear
B)
gustatoreceptors
done
clear
C)
rheoreceptors
done
clear
D)
chemoreceptors
done
clear
View Answer play_arrow
question_answer 122) How many hearts are found in earth worm?
A)
8 (four pairs)
done
clear
B)
2 (one pair)
done
clear
C)
6 (three pairs)
done
clear
D)
12 (six pairs)
done
clear
View Answer play_arrow
question_answer 123) Schizocoelomates and enterocoelomates are:
A)
acoelomates
done
clear
B)
true coelomates
done
clear
C)
invertebrates
done
clear
D)
echinoderms only
done
clear
View Answer play_arrow
question_answer 124) \[C{{O}_{2}}\] is transported as :
A)
dissolved in blood plasma
done
clear
B)
as carbonic acid
done
clear
C)
incarbaminohaemoglobin
done
clear
D)
ascarbaminohaemoglobin and carbonic acid
done
clear
View Answer play_arrow
question_answer 125) Which of the damaged cells cannot be repaired?
A)
Liver cells
done
clear
B)
Brain cells
done
clear
C)
Bone cells
done
clear
D)
Epidermal cells
done
clear
View Answer play_arrow
question_answer 126) Superficial meroblastic cleavage occurs in :
A)
reptiles
done
clear
B)
birds
done
clear
C)
mammals
done
clear
D)
insects
done
clear
View Answer play_arrow
question_answer 127) Axolotl larva of Ambyostoma normally fails to metamorphose due to:
A)
lack of Ca and Mg ions in water
done
clear
B)
absence of phosphorus in water
done
clear
C)
lack of iodine in water or diet
done
clear
D)
high concentration of iodine in body
done
clear
View Answer play_arrow
question_answer 128) Kreb?s cycle was discovered by Krebs in pigeon muscles in 1940. Which step is called gateway step/link reaction/transition reaction in respiration?
A)
Glycolysis
done
clear
B)
Formation of acetyl-coA
done
clear
C)
citric acid formation
done
clear
D)
ETS terminal oxidation
done
clear
View Answer play_arrow
question_answer 129) When released from ovary, human egg contain :
A)
one Y chromosome
done
clear
B)
two X chromosome
done
clear
C)
one X chromosome
done
clear
D)
XY chromosome
done
clear
View Answer play_arrow
question_answer 130) In the body, the membrane surrounding the bone is known as :
A)
periosteum
done
clear
B)
endosteum
done
clear
C)
perichondrium
done
clear
D)
chondrocytes
done
clear
View Answer play_arrow
question_answer 131) Carbamates pesticides act by combining with acetylcholine esterase enzyme. Which one of the following is a carbamate?
A)
Propoxur (baygon)
done
clear
B)
Aldicarb (temik)
done
clear
C)
Carbofuran (furadan)
done
clear
D)
All of the above
done
clear
View Answer play_arrow
question_answer 132) Some organisms resemble other organisms and thus escape from enemies. This henomenon is known as :
A)
homology
done
clear
B)
variation
done
clear
C)
analogy
done
clear
D)
mimicry
done
clear
View Answer play_arrow
question_answer 133) Recent reports of acid rains in big industrial cities are due to the effect of atmospheric pollution by:
A)
morerelase of \[N{{O}_{2}}\] and \[S{{O}_{2}}\] by burning of fossil fuels
done
clear
B)
morerelase of \[C{{O}_{2}}\] by burning of coal/wood cutting of forests and increasingpopultions
done
clear
C)
excessive release of \[N{{H}_{3}}\] by coal gas/industries
done
clear
D)
excessive release of \[CO\] by incomplete combustion of carbonaceous fuels
done
clear
View Answer play_arrow
question_answer 134) There is more competition for survival between :
A)
different animals of same niche
done
clear
B)
same animals of same niche
done
clear
C)
different animals of different niche
done
clear
D)
same animals of different niche
done
clear
View Answer play_arrow
question_answer 135) Which set includes pathogenic Arthropods ?
A)
Tse-tse fly, mosquito, flea-plague
done
clear
B)
Crab, Culex, spider
done
clear
C)
Anopheles, Culex, cray fish
done
clear
D)
Silver fish, house fly, s and fly
done
clear
View Answer play_arrow
question_answer 136) In polytene chromosomes dark bands are visible. These bands are formed by the aposition of:
A)
protein particles
done
clear
B)
chromomeres on chromonemata
done
clear
C)
nucleosomes
done
clear
D)
none
done
clear
View Answer play_arrow
question_answer 137) In the blind spot where the optic nerves leaves the eyes :
A)
rods and cones are absent
done
clear
B)
only cones are present
done
clear
C)
only rods are present
done
clear
D)
special neurons are present
done
clear
View Answer play_arrow
question_answer 138) Which of the following blood vessels in the circulatory system of frog has moreoxygenated blood ?
A)
Pulmocutaneous artery
done
clear
B)
Pulmocutaneous vein
done
clear
C)
Pulmonary artery
done
clear
D)
Precaval veins
done
clear
View Answer play_arrow
question_answer 139) The enzyme, which helps to cut one strand of DNA duplex to release tension of coiling of two strands is :
A)
DNA ligase
done
clear
B)
DNA polymerase
done
clear
C)
topo-isomerase
done
clear
D)
swielases (helicase or unwindases)
done
clear
View Answer play_arrow
question_answer 140) Blood vascular system in hemichordata is :
A)
open
done
clear
B)
reduced
done
clear
C)
closed
done
clear
D)
absent
done
clear
View Answer play_arrow
question_answer 141) The nitrogeneous metabolic waste in Hydra is mostly :
A)
ammonia and is removed from whole surface of body
done
clear
B)
urea and is removed mainly by tentacles
done
clear
C)
urea and is removed from whole surface of body
done
clear
D)
uric acid and is removed from whole surface of body
done
clear
View Answer play_arrow
question_answer 142) Anaemia is due to deficiency of :
A)
\[Ca\]
done
clear
B)
\[Fe\]
done
clear
C)
\[Mg\]
done
clear
D)
\[P\]
done
clear
View Answer play_arrow
question_answer 143) Mechanical support, enzyme circulation, protein synthesis and detoxification of drugs are function of:
A)
ER
done
clear
B)
ribosomes
done
clear
C)
dictyosome
done
clear
D)
chloroplast
done
clear
View Answer play_arrow
question_answer 144) Characteristic of mammalian liver is :
A)
Kupffer?s cells and leucocytes
done
clear
B)
Leucocytes and canaliculae
done
clear
C)
Glisson?s capsules and Kupffer cells
done
clear
D)
Glisson?s capsules and leucocytes
done
clear
View Answer play_arrow
question_answer 145) Synthesis of amino acids to prove that amino acids were formed in primitive ocean was experimentaly proved by :
A)
Sydeny Fox
done
clear
B)
Oparin
done
clear
C)
Haldane
done
clear
D)
Stanley Miller
done
clear
View Answer play_arrow
question_answer 146) Bacterial flagella do not show ATPase activity and 9 + 2 organization. These are chemically:
A)
flagellin
done
clear
B)
pilin
done
clear
C)
tubulin
done
clear
D)
bacterin
done
clear
View Answer play_arrow
question_answer 147) Phytochromes are :
A)
chemicals regulating flowering
done
clear
B)
chemicals regulating growth from seed to adulthood
done
clear
C)
hormones regulating growth from seed to adult hood
done
clear
D)
regulators synthesized by plants and influencing physiological process
done
clear
View Answer play_arrow
question_answer 148) Certain bacteria living in the soil poor in oxygen convert nitrates into nitrites and then to free nitrogen and such bacteria are termed as:
A)
nitrogen fixing bacteria
done
clear
B)
denitrifying bacteria
done
clear
C)
ammonifying bacteria
done
clear
D)
saprophytic bacteria
done
clear
View Answer play_arrow
question_answer 149) Food chain is a series of population which starts with producers. Its is concerning with :
A)
biotic components only
done
clear
B)
energy flow and transfer of nutrients
done
clear
C)
both (a) and (b)
done
clear
D)
biotic and decomposers
done
clear
View Answer play_arrow
question_answer 150) The leaf less stem of onion which is produced to bear flower is called :
A)
thalamus
done
clear
B)
scape
done
clear
C)
torus
done
clear
D)
Pedicel
done
clear
View Answer play_arrow
question_answer 151) Sudden breaking of rock-layers below the earth, results:
A)
earthquakes
done
clear
B)
valcones
done
clear
C)
floods
done
clear
D)
cyclones
done
clear
View Answer play_arrow
question_answer 152) How may members in RajyaSabha are nominated by President of India?
A)
12
done
clear
B)
13
done
clear
C)
9
done
clear
D)
14
done
clear
View Answer play_arrow
question_answer 153) Which one of the following is the deepest lake in the world?
A)
Dal lake
done
clear
B)
Naini lake
done
clear
C)
Wolan lake
done
clear
D)
Baikal lake
done
clear
View Answer play_arrow
question_answer 154) Which one of the following state has the largest land area?
A)
Madhya Pradesh
done
clear
B)
Uttar Pradesh
done
clear
C)
Maharashtra
done
clear
D)
Rajasthan
done
clear
View Answer play_arrow
question_answer 155) Tides are caused by :
A)
gravitational pull of the sun and moon
done
clear
B)
violent activity under sea
done
clear
C)
high pressure formed on the sea
done
clear
D)
seismic vibration under earth
done
clear
View Answer play_arrow
question_answer 156) At which place did Mahavir get enlightment?
A)
Samath
done
clear
B)
Vaishali
done
clear
C)
Lumbini
done
clear
D)
Gaya
done
clear
View Answer play_arrow
question_answer 157) Duck is associated in which of the following sprots
A)
Cricket
done
clear
B)
Basket ball
done
clear
C)
Golf
done
clear
D)
Hockey
done
clear
View Answer play_arrow
question_answer 158) The battler of HaldiGhati was fought in :
A)
1756 BC
done
clear
B)
1756 AD
done
clear
C)
1576 BC
done
clear
D)
1576 AD
done
clear
View Answer play_arrow
question_answer 159) Who decides the question of disqualification of a member of parliament?
A)
The Election Commission
done
clear
B)
The Supreme Court
done
clear
C)
The President
done
clear
D)
The Speaker
done
clear
View Answer play_arrow
question_answer 160) Lunar space craft is used for :
A)
study of the each from satellite
done
clear
B)
study of energy liberated by sun
done
clear
C)
transmission of radio-signal
done
clear
D)
study of moon
done
clear
View Answer play_arrow
question_answer 161) In which of the following movement Mahatma Gandhi gave the slogan ?Do or die??
A)
Khilafat movement
done
clear
B)
Dandi march
done
clear
C)
Non-cooperation movement
done
clear
D)
Quit-cooperation movement
done
clear
View Answer play_arrow
question_answer 162) Which one of the following is capital of Danmark?
A)
Copen-hagen
done
clear
B)
Tokyo
done
clear
C)
Rome
done
clear
D)
Otawa
done
clear
View Answer play_arrow
question_answer 163) Which of the following country is the largest producer of copper?
A)
Russia
done
clear
B)
India
done
clear
C)
Japan
done
clear
D)
USA
done
clear
View Answer play_arrow
question_answer 164) The abbreviation of ?BARC? stand for :
A)
Bhabha Atomic Research Centre
done
clear
B)
Bharat Atomic Reactor Centre
done
clear
C)
Baroda Artificial Rayon Corporation
done
clear
D)
Bharat Aluminium Rea search Corporation
done
clear
View Answer play_arrow
question_answer 165) The pH value is a measure of:
A)
acidity of aqueous solution
done
clear
B)
concentration of dissolved impurities
done
clear
C)
alkalinity of aqueous solution
done
clear
D)
salinity of aqueous solution
done
clear
View Answer play_arrow
question_answer 166) ?Biwi Ka Maqbara? is the tomb of:
A)
Wife of Aurangzeb
done
clear
B)
Nurjahan
done
clear
C)
Mumtaj Mahal
done
clear
D)
Sister of Hymayun
done
clear
View Answer play_arrow
question_answer 167) What was the name of the town established by Akbar?
A)
Agra
done
clear
B)
Fatehpur Sikari
done
clear
C)
Nizamabad
done
clear
D)
Delhi
done
clear
View Answer play_arrow
question_answer 168) Who was the ruler of India who transferred his capital from Delhi to Daultabad or Devagiri?
A)
Mohd. Tughlak
done
clear
B)
FerozTughlak
done
clear
C)
Aurangjeb
done
clear
D)
AllauddinKhalzi
done
clear
View Answer play_arrow
question_answer 169) India became World Champion in cricket in :
A)
1979
done
clear
B)
1975
done
clear
C)
1984
done
clear
D)
1983
done
clear
View Answer play_arrow
question_answer 170) In which of the following cities is head quarters of the Unit Trust of India located?
A)
Mumbai
done
clear
B)
New Delhi
done
clear
C)
Chennai
done
clear
D)
Ahmedabad
done
clear
View Answer play_arrow
question_answer 171) Fill in the blanks. 2Z5, 7Y7, 14X9, 23W11, 34V13,.........
A)
27U24
done
clear
B)
47U15
done
clear
C)
45U15
done
clear
D)
47V14
done
clear
View Answer play_arrow
question_answer 172) Fill in the blanks. CAT, FDW, IGZ,.........
A)
KTC
done
clear
B)
LJC
done
clear
C)
KJA
done
clear
D)
LHD
done
clear
View Answer play_arrow
question_answer 173) The least number divisible by 12, 15, 20 and is a perfect square, is :
A)
900
done
clear
B)
400
done
clear
C)
36
done
clear
D)
256
done
clear
View Answer play_arrow
question_answer 174) The mean proportional between (2+ % 3) and (8 - v/48)is :
A)
2
done
clear
B)
2 : 3
done
clear
C)
3
done
clear
D)
3 : 1
done
clear
View Answer play_arrow
question_answer 175) How many times will a wheel of diameter105 cm rotate in covering a distance of 330 m?
A)
100 revolutions
done
clear
B)
110 revolutions
done
clear
C)
90 revolutions
done
clear
D)
105 revolutions
done
clear
View Answer play_arrow
question_answer 176) If the two parallel sides of a trapezium are 15 cm and 25 cm respectively and the distance between them is 7 cm, then the area of the trapezium is :
A)
\[105c{{m}^{2}}\]
done
clear
B)
\[125c{{m}^{2}}\]
done
clear
C)
\[140c{{m}^{2}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 177) The multiplicative inverse of \[\left( \frac{1}{2}+\frac{1}{3} \right)-1\]is :
A)
6
done
clear
B)
-6
done
clear
C)
5
done
clear
D)
\[\frac{1}{6}\]
done
clear
View Answer play_arrow
question_answer 178) If \[\text{111}.\text{744}\div \text{28}.\text{8}=\text{ 3}.\text{88},\] then the value of \[\text{1117}.\text{44}+\text{288}\] is:
A)
3.88
done
clear
B)
0.388
done
clear
C)
388.0
done
clear
D)
38.8
done
clear
View Answer play_arrow
question_answer 179) 4 buses runs between Bhopal and Gwalior. If a man goes from Gwalior to Bhopal by a bus and comes back to Gwalior by another bus, then the total possible ways are :
A)
12
done
clear
B)
16
done
clear
C)
4
done
clear
D)
8
done
clear
View Answer play_arrow
question_answer 180) How many words comprising of any three letters of the word UNIVERSAL can be formed :
A)
504
done
clear
B)
405
done
clear
C)
540
done
clear
D)
450
done
clear
View Answer play_arrow
question_answer 181) I found his crude manners abominable.
A)
detestable
done
clear
B)
flattering
done
clear
C)
attractive
done
clear
D)
sloppy
done
clear
View Answer play_arrow
question_answer 182) The rebels returned home under an amnesty.
A)
general pardon
done
clear
B)
financial assistance
done
clear
C)
police security
done
clear
D)
threat
done
clear
View Answer play_arrow
question_answer 183) A posthumous award was given to the poet.
A)
postal
done
clear
B)
after death
done
clear
C)
creditable
done
clear
D)
literary
done
clear
View Answer play_arrow
question_answer 184) I doubt the veracity of your statement.
A)
tenacity
done
clear
B)
wisdom
done
clear
C)
truthfulness
done
clear
D)
reliability
done
clear
View Answer play_arrow
question_answer 185) Would you please rectify the mistake ?
A)
forget
done
clear
B)
clarify
done
clear
C)
condone
done
clear
D)
regularize
done
clear
View Answer play_arrow
question_answer 186) He appears to be a phoney person.
A)
beautiful
done
clear
B)
unread
done
clear
C)
genuine
done
clear
D)
ugly
done
clear
View Answer play_arrow
question_answer 187) He was in a dejected mood.
A)
jubilant
done
clear
B)
rejected
done
clear
C)
irritable
done
clear
D)
romantic
done
clear
View Answer play_arrow
question_answer 188) Philosophers say that the world is an illusion.
A)
a fiction
done
clear
B)
a reality
done
clear
C)
an actuality
done
clear
D)
a truth
done
clear
View Answer play_arrow
question_answer 189) There was a marked deterioration in his condition.
A)
improvement
done
clear
B)
revision
done
clear
C)
reformation
done
clear
D)
amendment
done
clear
View Answer play_arrow
question_answer 190) This offer has come as a great boon to me.
A)
curse
done
clear
B)
blemish
done
clear
C)
tension
done
clear
D)
misfortune
done
clear
View Answer play_arrow
question_answer 191) Some of the following sentences are grammatically incorrect and some are correct. Find out which part of a sentence has an error and mark that part. If there is no error, your answer is (d).
A)
\[\frac{M\text{y friend is}\,\text{equally}}{(a)}\text{,}\]
done
clear
B)
\[\frac{\text{hard working and intelligent}}{(b)}\text{, }\]
done
clear
C)
\[\frac{\text{as your sister}}{(c)}\text{,}\]
done
clear
D)
\[\frac{\text{No error}.}{(d)}\]
done
clear
View Answer play_arrow
question_answer 192) Some of the following sentences are grammatically incorrect and some are correct. Find out which part of a sentence has an error and mark that part. If there is no error, your answer is (d).
A)
\[\frac{\text{I enquired of him}}{(a)}\text{,}\]
done
clear
B)
\[\frac{\text{ why he is so negligent }}{(b)},\]
done
clear
C)
\[\frac{\text{in his studies}.}{(c)}\]
done
clear
D)
\[\frac{\text{No error}.}{(d)}\]
done
clear
View Answer play_arrow
question_answer 193) Some of the following sentences are grammatically incorrect and some are correct. Find out which part of a sentence has an error and mark that part. If there is no error, your answer is (d).
A)
\[\frac{\text{They had been}}{(a)}\]
done
clear
B)
\[\frac{\text{very close friends }}{(b)}\]
done
clear
C)
\[\frac{\text{unless they quarrelled}.\text{ }}{(c)}\]
done
clear
D)
\[\frac{\text{No error}.}{(d)}\]
done
clear
View Answer play_arrow
question_answer 194) Some of the following sentences are grammatically incorrect and some are correct. Find out which part of a sentence has an error and mark that part. If there is no error, your answer is (d).
A)
\[\frac{\text{May I know}}{(a)}\]
done
clear
B)
\[\frac{\text{who you want}}{(b)}\]
done
clear
C)
\[\frac{\text{to see please}.~}{(c)}\]
done
clear
D)
\[\frac{\text{No error}~}{(d)}\]
done
clear
View Answer play_arrow
question_answer 195) Some of the following sentences are grammatically incorrect and some are correct. Find out which part of a sentence has an error and mark that part. If there is no error, your answer is (d).
A)
\[\frac{c\text{hildren often}}{(a)}\]
done
clear
B)
\[\frac{\text{quarrel on}}{(b)}\]
done
clear
C)
\[\frac{\text{petty issues}\text{.}}{(c)}\]
done
clear
D)
\[\frac{\text{No error}\text{.}}{(d)}\]
done
clear
View Answer play_arrow
question_answer 196) The U.S. Soya bean : P : in the developing world Q : and when famine strikes R : is America?s single most lucrative export S : American soya beans are a major source of high protein sustenance
A)
R Q S P
done
clear
B)
P R S Q
done
clear
C)
P S R Q
done
clear
D)
R Q P S
done
clear
View Answer play_arrow
question_answer 197) The ancient people of Mediterranean : P : like a horse with a long horn Q : believed in the existence of the unicorn R : on its forehead S : a mythical animal supposed to look
A)
P R Q S
done
clear
B)
R S Q P
done
clear
C)
Q S P R
done
clear
D)
Q P S R
done
clear
View Answer play_arrow
question_answer 198) The exhibition committee : P : attractive and useful Q : to make the exhibition R : making efforts S : has been
A)
S R P Q
done
clear
B)
Q P S R
done
clear
C)
Q P R S
done
clear
D)
S R Q P
done
clear
View Answer play_arrow
question_answer 199) You must leave a note : P : otherwise she will be terribly worried Q : for your mother R : at the usual hour S : if you are not back home
A)
Q P S R
done
clear
B)
Q P R S
done
clear
C)
S R Q P
done
clear
D)
S R P Q
done
clear
View Answer play_arrow
question_answer 200) Though he was only a child : P: he often worked much harder Q: than was either necessary R: in order to please his parents S: or good for him
A)
P Q S R
done
clear
B)
R P Q S
done
clear
C)
R Q S P
done
clear
D)
P R Q S
done
clear
View Answer play_arrow