A physical quantity P is described by the relation \[P={{a}^{1/2}}{{b}^{2}}{{c}^{3}}{{d}^{-4}}.\] If the relative errors in the measurement of a, b, c and d respectively, are 2%, 1%, 3% and 5%, then the relative error in P will be
A)
8%
done
clear
B)
12%
done
clear
C)
32%
done
clear
D)
25%
done
clear
View Answer play_arrow
When a mass is rotating in a plane about a fixed point, its angular momentum is directed along the
A)
radius of orbit
done
clear
B)
tangent to the orbit
done
clear
C)
line parallel to plane of rotation
done
clear
D)
line perpendicular to plane of rotation
done
clear
View Answer play_arrow
An ideal gas heat engine operates in a Carnot cycle between \[227{}^\circ C\] and \[127{}^\circ C.\] It absorbs 6 kcal at the higher temperature. The amount of heat (in kcal) converted into work is equal to
A)
12
done
clear
B)
4.8
done
clear
C)
3.5
done
clear
D)
1.6
done
clear
View Answer play_arrow
If \[{{\mu }_{r}},\] \[{{\mu }_{k}}\] and \[{{\mu }_{s}}\] represent the coefficients of rolling friction, kinetic and static friction, then
A)
\[{{\mu }_{r}}={{\mu }_{s}}={{\mu }_{k}}\]
done
clear
B)
\[{{\mu }_{r}}>{{\mu }_{k}}>{{\mu }_{s}}\]
done
clear
C)
\[{{\mu }_{r}}<{{\mu }_{s}}<{{\mu }_{k}}\]
done
clear
D)
\[{{\mu }_{r}}<{{\mu }_{k}}<{{\mu }_{s}}\]
done
clear
View Answer play_arrow
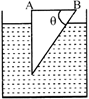
A glass prism of refractive index 1.5 is immersed in water (refractive index 4/3). A light beam incident normally on the face AB is totally reflected to reach on the face BC if
A)
\[\sin \theta \ge \frac{8}{9}\]
done
clear
B)
\[\frac{2}{3}<\sin \theta <\frac{8}{9}\]
done
clear
C)
\[\sin \theta \le \frac{2}{3}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
Kirchhoff's I and II laws are based on conservation of
A)
energy and charge respectively
done
clear
B)
charge and energy respectively
done
clear
C)
mass and charge respectively
done
clear
D)
None of these
done
clear
View Answer play_arrow
If the density of a small planet is the same as that of earth, while the radius of the planet is 0.2 times that of the earth, the gravitational acceleration on the surface of the planet is
A)
0.2 g
done
clear
B)
0.4 g
done
clear
C)
2 g
done
clear
D)
4 g
done
clear
View Answer play_arrow
The work function of a metallic substance is 5 eV. The threshold frequency is approximately
A)
\[1.6\times {{10}^{7}}\,\,Hz\]
done
clear
B)
\[8.68\times {{10}^{15}}\,\,Hz\]
done
clear
C)
\[9.68\times {{10}^{17}}\,\,Hz\]
done
clear
D)
\[1.2\times {{10}^{15}}\,\,Hz\]
done
clear
View Answer play_arrow
A ball is dropped from the top of a tower of height 100 m and at the same time another ball is projected vertically upwards from ground with a velocity \[25\,\,m{{s}^{-1}}.\] Then the distance from the top of the tower, at which the two balls meet is
A)
68.4 m
done
clear
B)
48.4 m
done
clear
C)
18.4 m
done
clear
D)
78.4 m
done
clear
View Answer play_arrow
What is the ratio of the shortest wavelength of the Balmer series to the shortest wavelength of the Lyman series?
A)
4 : 1
done
clear
B)
4 : 3
done
clear
C)
4 : 9
done
clear
D)
5 : 9
done
clear
View Answer play_arrow
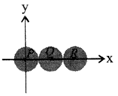
Three identical spheres, each of mass 1 kg are kept as shown in figure, touching each other, with their centres on a straight line. If their centres are marked P, Q, R respectively, the distance of centre of mass of the system from P is
A)
\[\frac{PQ+PR+QR}{3}\]
done
clear
B)
\[\frac{PQ+PR}{3}\]
done
clear
C)
\[\frac{PQ+QR}{3}\]
done
clear
D)
\[\frac{PR+QR}{3}\]
done
clear
View Answer play_arrow
Use of eddy currents is done in the following except
A)
moving coil galvanometer
done
clear
B)
electric brakes
done
clear
C)
induction motor
done
clear
D)
dynamo
done
clear
View Answer play_arrow
A satellite of mass m is put into a circular orbit of height h from the surface of the earth (mass \[={{M}_{e}},\] radius \[={{R}_{e}}\] ). The orbital speed of satellite is
A)
\[\sqrt{\frac{2G{{M}_{e}}h}{{{R}_{e}}({{R}_{e}}+h)}}\]
done
clear
B)
\[\sqrt{\frac{2G{{M}_{e}}}{{{R}_{e}}+h}}\]
done
clear
C)
\[\sqrt{\frac{G{{M}_{e}}}{({{R}_{e}}+{{h}^{3}})}}\]
done
clear
D)
\[\sqrt{\frac{G{{M}_{e}}}{2({{R}_{e}}+h)}}\]
done
clear
View Answer play_arrow
Consider the following statements and select the incorrect statement.
A)
The presence of a large magnetic flux through a coil maintains a current in the coil if the circuit is continuous.
done
clear
B)
A coil of a metal wire kept stationary in a non-uniform magnetic field has an e.m.f induced in it.
done
clear
C)
A charged particle enters a region of uniform magnetic field at an angle of \[85{}^\circ \] to the magnetic lines of force, the path of the particle is a circle.
done
clear
D)
There is no change in the energy of a charged particle moving in a magnetic field although a magnetic force is acting on it.
done
clear
View Answer play_arrow
16 g of oxygen at \[37{}^\circ C\] is mixed with 14 g of nitrogen at \[27{}^\circ C,\] the temperature of the mixture will be
A)
\[30.5{}^\circ C\]
done
clear
B)
\[37{}^\circ C\]
done
clear
C)
\[27{}^\circ C\]
done
clear
D)
\[32{}^\circ C\]
done
clear
View Answer play_arrow
At the centre of a cubical box +Q charge is placed. The value of total flux that is coming out a wall is
A)
\[Q/{{\varepsilon }_{o}}\]
done
clear
B)
\[Q/3{{\varepsilon }_{o}}\]
done
clear
C)
\[Q/4{{\varepsilon }_{o}}\]
done
clear
D)
\[Q/6{{\varepsilon }_{o}}\]
done
clear
View Answer play_arrow
A particle moves under the effect of a force \[F=cx\] from \[x=0\] to \[x={{x}_{1}},\] the work done in the process is
A)
\[cx_{1}^{2}\]
done
clear
B)
\[\frac{1}{2}cx_{1}^{2}\]
done
clear
C)
\[2\,cx_{1}^{2}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
According to the kinetic theory of gases, the pressure exerted by a gas on the walls is measured as
A)
rate of change of momentum imparted to the walls per second per unit area.
done
clear
B)
momentum imparted to the walls per unit area
done
clear
C)
change of momentum imparted to the walls per unit area.
done
clear
D)
change in momentum per unit volume
done
clear
View Answer play_arrow
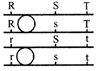
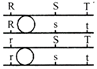
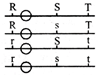
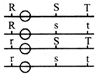
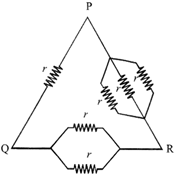
Six equal resistances are connected between points P, Q and R as shown in figure. Then net resistance will be maximum between:
A)
P and R
done
clear
B)
P and Q
done
clear
C)
Q and R
done
clear
D)
Any two points
done
clear
View Answer play_arrow
The fundamental frequency of a closed end organ pipe is n. Its length is doubled and radius is halved. Its frequency will become nearly
A)
n/2
done
clear
B)
n/3
done
clear
C)
n
done
clear
D)
2n
done
clear
View Answer play_arrow
A rain drop of radius 0.3 mm has a terminal velocity in air = 1 m/s. the viscosity of air is \[8\times {{10}^{-5}}\]poise. The viscous force on it is
A)
\[45.2\times {{10}^{-4}}\text{dyne}\]
done
clear
B)
\[101.73\times {{10}^{-5}}\text{dyne}\]
done
clear
C)
\[16.95\times {{10}^{-4}}\text{dyne}\]
done
clear
D)
\[16.95\times {{10}^{-5}}\text{dyne}\]
done
clear
View Answer play_arrow
A particle moves such that its acceleration 'a' is given by a = -bx where x is the displacement from equilibrium position and b is constant. The period of oscillation is
A)
\[2\pi /b\]
done
clear
B)
\[2\pi /\sqrt{b}\]
done
clear
C)
\[\sqrt{2\pi /b}\]
done
clear
D)
0\[2\sqrt{\pi /b}\]
done
clear
View Answer play_arrow
A person who can see things most clearly at a distance of 10 cm, requires spectacles to enable to see clearly things at a distance of 30 cm. What should be the focal length of the spectacles?
A)
15 cm (Concave)
done
clear
B)
15 cm (Convex)
done
clear
C)
10 cm
done
clear
D)
0
done
clear
View Answer play_arrow
The magnetic field of earth at the equator is approximately \[4\times {{10}^{-5}}\,T.\] The radius of earth is \[6.4\times {{10}^{6}}\,m.\] Then the dipole moment of the earth will be nearly of the order of:
A)
\[{{10}^{23}}\,\,\text{A}\,\,{{\text{m}}^{2}}\]
done
clear
B)
\[{{10}^{20}}\,\,\text{A}\,\,{{\text{m}}^{2}}\]
done
clear
C)
\[{{10}^{16}}\,\,\text{A}\,\,{{\text{m}}^{2}}\]
done
clear
D)
\[{{10}^{10}}\,\,\text{A}\,\,{{\text{m}}^{2}}\]
done
clear
View Answer play_arrow
A 2V battery is connected across AB as shown in the figure. The value of the current supplied by the battery when in one case battery's positive terminal is connected to A and in other case when positive terminal of battery is connected to B will respectively be:
A)
0.4 A and 0.2 A
done
clear
B)
0.2 A and 0.4 A
done
clear
C)
0.1 A and 0.2 A
done
clear
D)
0.2A and 0.1 A
done
clear
View Answer play_arrow
A passenger travels along the straight road for half the distance with velocity \[{{\text{v}}_{1}}\] and the remaining half distance with velocity\[{{\text{v}}_{1}}.\] Then average velocity is given by
A)
\[{{\text{v}}_{1}}{{\text{v}}_{2}}\]
done
clear
B)
\[{{\text{v}}_{2}}^{2}/{{\text{v}}_{1}}^{2}\]
done
clear
C)
\[({{\text{v}}_{1}}+{{\text{v}}_{2}})/2\]
done
clear
D)
\[2\,{{\text{v}}_{1}}{{\text{v}}_{2}}/({{\text{v}}_{1}}+{{\text{v}}_{2}})\]
done
clear
View Answer play_arrow
A point initially at rest moves along x-axis. Its acceleration varies with time as a \[=(6t+5)m/{{s}^{2}}.\]If it starts from origin, the distance covered in 2s is
A)
20 m
done
clear
B)
18 m
done
clear
C)
16 m
done
clear
D)
25 m
done
clear
View Answer play_arrow
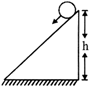
A solid cylinder of mass m & radius R rolls down inclined plane without slipping. The speed of its C.M. when it reaches the bottom is
A)
\[\sqrt{2gh}\]
done
clear
B)
\[\sqrt{4gh/3}\]
done
clear
C)
\[\sqrt{3/4gh}\]
done
clear
D)
\[\sqrt{4gh}\]
done
clear
View Answer play_arrow
A nuclear reaction is given by \[{{Z}^{X}}^{^{A}}{{\to }_{Z+1}}Y+-{{1}^{{{e}^{0}}}}+\overline{v},\] represents
A)
fission
done
clear
B)
\[\beta \]-decay
done
clear
C)
\[\alpha \]-decay
done
clear
D)
fusion
done
clear
View Answer play_arrow
Steam is passed into 22 gm of water at \[20{}^\circ C.\] The mass of water that will be present when the water acquires a temperature of \[90{}^\circ C\] (Latent heat of steam is 540 cal/g) is
A)
24.83 gm
done
clear
B)
24 gm
done
clear
C)
36.6 gm
done
clear
D)
30 gm
done
clear
View Answer play_arrow
Electromotive force is the force which is able to maintain a constant
A)
potential difference
done
clear
B)
power
done
clear
C)
resistance
done
clear
D)
current
done
clear
View Answer play_arrow
A prism of refractive index has a refracting angle of \[60{}^\circ .\] At what angle a ray must be incident on it so that it suffers a minimum deviation?
A)
\[45{}^\circ \]
done
clear
B)
\[60{}^\circ \]
done
clear
C)
\[90{}^\circ \]
done
clear
D)
\[180{}^\circ \]
done
clear
View Answer play_arrow
Select the incorrect statement about Lorentz Force.
A)
In presence of electric field \[\overrightarrow{E}(r)\] and magnetic field \[\overrightarrow{B}(r)\] the force on a moving electric charge is \[\overrightarrow{F}=q[\overrightarrow{E}(r)+v\times \overrightarrow{B}(r)]\]
done
clear
B)
The force, due to magnetic field on a negative charge is opposite to that on a positive charge.
done
clear
C)
The force due to magnetic field are become zero if velocity and magnetic field are parallel or antiparallel.
done
clear
D)
For a static charge the magnetic force is maximum.
done
clear
View Answer play_arrow
In an AC generator, a coil with N turns, all of the same area A and total resistance R, rotates with frequency\[\omega \]in a magnetic field B. The maximum value of emf generated in the coil is
A)
N A B R\[\omega \]
done
clear
B)
N A B
done
clear
C)
N A B R
done
clear
D)
N A B\[\omega \]
done
clear
View Answer play_arrow
A diametrical tunnel is dug across the earth. A ball is dropped into the tunnel from one side. The velocity of the ball when it reaches the centre of the earth is (Given : gravitational potential at the centre of earth\[=-\frac{3}{2}\frac{GM}{R})\]
A)
\[\sqrt{R}\]
done
clear
B)
\[\sqrt{gR}\]
done
clear
C)
\[\sqrt{2.5gR}\]
done
clear
D)
\[\sqrt{7.1gR}\]
done
clear
View Answer play_arrow
A spring of force constant 800 N/m has an extension of 5 cm. The work done in extending it from 5 cm to 15 cm is
A)
16 J
done
clear
B)
8 J
done
clear
C)
32 J
done
clear
D)
24 J
done
clear
View Answer play_arrow
If it takes 5 minutes to fill a 15 litre bucket from a water tap of diameter \[\frac{2}{\sqrt{\pi }}cm\] then the Reynolds number for the flow is close to (density of water \[={{10}^{3}}kg/{{m}^{3}}\] and viscosity of water \[={{10}^{-3}}Pa.s\]):
A)
1100
done
clear
B)
11,000
done
clear
C)
550
done
clear
D)
5500
done
clear
View Answer play_arrow
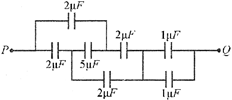
The effective capacitance between the points P and Q of the arrangement shown in the figure is
A)
\[\frac{1}{2}\mu F\]
done
clear
B)
\[1\mu F\]
done
clear
C)
\[2\mu F\]
done
clear
D)
\[1.33\mu F\]
done
clear
View Answer play_arrow
A glass rod 20 cm long is clamped at the middle. It is set into the longitudinal vibration. If the emitted sound frequency is 4000 Hz, the velocity of sound in glass will be
A)
2800 m/s
done
clear
B)
3200 m/s
done
clear
C)
1600 m/s
done
clear
D)
2000 m/s
done
clear
View Answer play_arrow
A transistor has an a \[\alpha =0.95,\] then \[\beta \] is equal to
A)
\[\frac{1}{19}\]
done
clear
B)
19
done
clear
C)
1.5
done
clear
D)
0.95
done
clear
View Answer play_arrow
A coil of circular cross-section having 1000 turns and \[4\,\,c{{m}^{2}}\] face area is placed with its axis parallel to a magnetic field which decreases by \[{{10}^{-2}}\] Wb \[{{m}^{-2}}\] in \[0.01\,\,s.\] The e.m.f. induced in the coil is:
A)
400 mV
done
clear
B)
200 Mv
done
clear
C)
4 mV
done
clear
D)
0.4 mV
done
clear
View Answer play_arrow
A particle is moving eastwards with a velocity of 5 m/s. hi 10 seconds the velocity changes to 5 m/s northwards. The average acceleration in this time is
A)
\[\frac{1}{2}m{{s}^{-2}}\] North
done
clear
B)
\[\frac{1}{\sqrt{2}}m{{s}^{-2}}\] North - East
done
clear
C)
\[\frac{1}{\sqrt{2}}m{{s}^{-2}}\] North-West
done
clear
D)
zero
done
clear
View Answer play_arrow
In bringing an electron towards another electron, the electrostatic potential energy of system
A)
increases
done
clear
B)
decreases
done
clear
C)
becomes zero
done
clear
D)
remains unchanged
done
clear
View Answer play_arrow
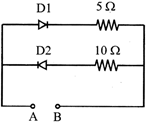
Four bulbs \[{{B}_{1}},\]\[{{B}_{2}},\]\[{{B}_{3}}\]and \[{{B}_{4}}\] of 100 W each are connected to 220 V main as shown in the figure.
The reading in an ideal ammeter will be:
A)
0.45 A
done
clear
B)
0.90 A
done
clear
C)
1.35 A
done
clear
D)
1.80 A
done
clear
View Answer play_arrow
A metal conductor of length 1 m rotates vertically about one of its ends at angular velocity 5 radians per second. If the horizontal component of earth's magnetic field is \[0.2\times {{10}^{-4}}T,\] then the e.m.f. developed between the two ends of the conductor is
A)
\[5\,\,mV\]
done
clear
B)
\[50\,\,\mu V\]
done
clear
C)
\[5\,\,\mu V\]
done
clear
D)
\[50\,\,mV\]
done
clear
View Answer play_arrow
The observed osmotic pressure for a 0.10 M solution of \[Fe{{(N{{H}_{4}})}_{2}}{{(S{{O}_{4}})}_{2}}\] at \[25{}^\circ C\] is 10.8 atm. The expected and experimental (observed) values of van't Hoff factor (i) will be respectively: \[(R=0.082\,\,L\,\,atm\,\,{{K}^{-1}}\,\,mo{{l}^{-1}})\]
A)
5 and 4.42
done
clear
B)
4 and 4.00
done
clear
C)
5 and 3.42
done
clear
D)
3 and 5.42
done
clear
View Answer play_arrow
In the Freidel Craft's acylation reaction, the effective electrophile is
A)
\[RCOC{{l}^{+}}\]
done
clear
B)
\[AlC{{l}_{3}}\]
done
clear
C)
\[RCOCl\]
done
clear
D)
\[RC{{O}^{+}}\]
done
clear
View Answer play_arrow
Match the columns
Column-I Column-II [A] Lanthanide hard as steel, (p) Lu [B] Lanthanide with maximum paramagnetic character in\[L{{n}^{4+}}\] state. (q) Tb [C] Lanthanide with maximum value of \[E{}^\circ \] for reaction\[L{{n}^{3+}}(aq)+3{{e}^{-}}\to Ln(s).\] (r) Sm [D] Lanthanide whose \[L{{n}^{3+}}\]ion is (s) Eu diamagnetic in nature (s) Eu
A)
A\[\to \](r), B\[\to \](s), C\[\to \](p), D\[\to \](q)
done
clear
B)
A\[\to \](r), B\[\to \](q), C\[\to \](s), D\[\to \](p)
done
clear
C)
A\[\to \](s), B\[\to \](r), C\[\to \](q), D\[\to \](p)
done
clear
D)
A\[\to \](r), B\[\to \](s), C\[\to \](q), D\[\to \](p)
done
clear
View Answer play_arrow
Specific volume of cylindrical virus particle is \[6.02\times {{10}^{-2}}cc/gm.\] whose radius and length \[7\,\,\overset{{}^\circ }{\mathop{A}}\,\] & \[10\,\,\overset{{}^\circ }{\mathop{A}}\,\] respectively If \[{{N}_{A}}=6.02\times {{10}^{23}}mo{{l}^{-1}},\] find molecular weight of virus
A)
\[3.08\times {{10}^{3}}\,kg\text{/}mol\]
done
clear
B)
\[3.08\times {{10}^{4}}\,kg\text{/}mol\]
done
clear
C)
\[1.54\times {{10}^{4}}\,kg\text{/}mol\]
done
clear
D)
\[1.54\,kg\text{/}mol\]
done
clear
View Answer play_arrow
Arrange the following in increasing order of their acidity? [a] o-cresol, [b] salicyclic acid, [c] phenol
A)
c < a < b
done
clear
B)
b < c < a
done
clear
C)
a < b < a
done
clear
D)
a < c < b
done
clear
View Answer play_arrow
Consider the following statements
I. The radius of an anion is larger than that of the parent atom. II. The ionization energy generally increases with increasing atomic number in a period. III. The electronegativity of an element is the tendency of an isolated atom to attract an electron. Which of the above statements is/are correct?
A)
I alone
done
clear
B)
II alone
done
clear
C)
I and II
done
clear
D)
II and III
done
clear
View Answer play_arrow
\[C{{l}_{2}}\] changes to \[C{{l}^{-}}\] and \[Cl{{O}^{-}}\] in cold NaOH. Equivalent weight of \[C{{l}_{2}}\] will be
A)
\[M\]
done
clear
B)
\[\frac{M}{2}\]
done
clear
C)
\[\frac{M}{3}\]
done
clear
D)
\[\frac{3M}{3}\]
done
clear
View Answer play_arrow
Pick out the wrong statement.
A)
Nitrogen has the ability to form \[p\pi -p\pi \] bonds with itself.
done
clear
B)
Bismuth forms metallic bonds in elemental state.
done
clear
C)
Catenation tendency is higher in nitrogen when compared with other elements of the same group.
done
clear
D)
Nitrogen has higher first ionisation enthalpy when compared with other elements of the same group.
done
clear
View Answer play_arrow
Chlorine cannot displace
A)
fluorine from NaF
done
clear
B)
iodine from Nal
done
clear
C)
bromine from NaBr
done
clear
D)
None of these
done
clear
View Answer play_arrow
Arrange the following phenols in order of their increasing acidity.
A)
I > III > II
done
clear
B)
I > II > III
done
clear
C)
III > I > II
done
clear
D)
III > II > I
done
clear
View Answer play_arrow
The average molecular speed is greatest in which of the following gas samples?
A)
1.0 mole of \[{{O}_{2}}\] at 560 K.
done
clear
B)
0.50 mole of Ne at 500 K.
done
clear
C)
0.20 mole of \[C{{O}_{2}}\] at 440 K.
done
clear
D)
2.0 mole of He at 140 K.
done
clear
View Answer play_arrow
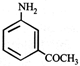
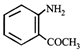
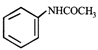
Aniline is an activated system for electrophilic substitution. The compound formed on heating aniline with acetic anhydride is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The formula mass of Mohr's salt is 392. The iron present in it is oxidised by \[KMn{{O}_{4}}\] in acid medium. The equivalent mass of Mohr's salt is
A)
392
done
clear
B)
31.6
done
clear
C)
278
done
clear
D)
156
done
clear
View Answer play_arrow
Sewage mostly constitutes :
A)
Non-biodegradable pollutants
done
clear
B)
Biodegradable pollutants
done
clear
C)
Effluents
done
clear
D)
Air pollutants
done
clear
View Answer play_arrow
The Langmuir adsorption isotherm is deduced using the assumption
A)
the adsorption sites are equivalent in their ability to adsorb the particles.
done
clear
B)
the heat of adsorption varies with coverage.
done
clear
C)
the adsorbed molecules interact with each other.
done
clear
D)
the adsorption takes place in multilayers.
done
clear
View Answer play_arrow
Which of the following can be repeatedly soften on heating?
(i) Polystyrene (ii) Melamine (iii) Polyesters (iv) Polyethylene (v) Neoprene
A)
(i) and (iii)
done
clear
B)
(i) and (iv)
done
clear
C)
(iii),(iv) and (v)
done
clear
D)
(ii) and (iv)
done
clear
View Answer play_arrow
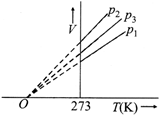
The volume-temperature graphs of a given mass of an ideal gas at constant pressure are shown below.
What is the correct order of pressures?
A)
\[{{p}_{1}}>{{p}_{3}}>{{p}_{2}}\]
done
clear
B)
\[{{p}_{1}}>{{p}_{2}}>{{p}_{3}}\]
done
clear
C)
\[{{p}_{2}}>{{p}_{3}}>{{p}_{1}}\]
done
clear
D)
\[{{p}_{2}}>{{p}_{1}}>{{p}_{3}}\]
done
clear
View Answer play_arrow
Which of the following is used for making optical instruments?
A)
\[Si{{O}_{2}}\]
done
clear
B)
\[Si\]
done
clear
C)
\[Si{{H}_{4}}\]
done
clear
D)
\[SiC\]
done
clear
View Answer play_arrow
he correct order of acid strength of oxyacids is:
A)
\[HClO>HCl{{O}_{2}}>HCl{{O}_{3}}>HCl{{O}_{4}}\]
done
clear
B)
\[HCl{{O}_{2}}>HCl{{O}_{3}}>HClO>HCl{{O}_{4}}\]
done
clear
C)
\[HCl{{O}_{4}}>HCl{{O}_{3}}>HCl{{O}_{2}}>HClO\]
done
clear
D)
\[HCl{{O}_{3}}>HCl{{O}_{4}}>HClO>HCl{{O}_{2}}\]
done
clear
View Answer play_arrow
25.3 g of sodium carbonate, \[N{{a}_{2}}C{{O}_{3}}\] is dissolved in enough water to make 250 mL of solution. If sodium carbonate dissociates completely, molar concentration of sodium ions,\[N{{a}^{+}}\] and carbonate ions, \[CO_{3}^{2-}\] are respectively \[(\]Molar mass of \[N{{a}_{2}}C{{O}_{3}}=106\,g\,mo{{l}^{-1}})\]
A)
0.955 M and 1.910 M
done
clear
B)
1.910 M and 0.955 M
done
clear
C)
1.90 M and 1.910M
done
clear
D)
0.477 M and 0.477 M
done
clear
View Answer play_arrow
In which alkyl halide, \[{{S}_{N}}2\] mechanism is favoured maximum?
A)
\[C{{H}_{3}}Cl\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}Cl\]
done
clear
C)
\[{{(C{{H}_{3}})}_{2}}CHCl\]
done
clear
D)
\[{{(C{{H}_{3}})}_{3}}C-Cl\]
done
clear
View Answer play_arrow
Equilibrium constant K changes with temperature. At 300 K, equilibrium constant is 25 and at 400 K it is 10. Hence, backward reaction will have energy of activation
A)
equal to that of forward reaction.
done
clear
B)
less than that of forward reaction.
done
clear
C)
greater than that of forward reaction.
done
clear
D)
given values are not sufficient to explain given statement.
done
clear
View Answer play_arrow
Which of the two ions from the list given below that have the geometry that is explained by the same hybridization of orbitals, \[N{{O}_{2}}^{-},\] \[N{{O}_{3}}^{-},\]\[N{{H}_{2}},\] \[N{{H}_{4}}^{+},\] \[SC{{N}^{-}}?\]
A)
\[N{{O}_{2}}^{-}\]and\[N{{O}_{3}}^{-}\]
done
clear
B)
\[N{{H}_{4}}^{+}\]and\[N{{O}_{3}}^{-}\]
done
clear
C)
\[SC{{N}^{-}}\] and \[N{{H}_{2}}^{-}\]
done
clear
D)
\[N{{O}_{2}}^{-}\]and\[N{{H}_{2}}^{-}\]
done
clear
View Answer play_arrow
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
White lead is.
A)
\[P{{b}_{3}}{{O}_{4}}\]
done
clear
B)
\[PbO\]
done
clear
C)
\[2PbC{{O}_{3}}.Pb{{(OH)}_{2}}\]
done
clear
D)
\[Pb{{(C{{H}_{3}}COO)}_{2}}.Pb{{(OH)}_{2}}\]
done
clear
View Answer play_arrow
The commonest method for the extraction of metals from oxide ores involves:
A)
Reduction with carbon and carbon monoxide
done
clear
B)
Reduction with aluminium
done
clear
C)
Reduction with hydrogen
done
clear
D)
Electrolytic method
done
clear
View Answer play_arrow
\[N{{H}_{4}}Cl\] crystallises in a bcc lattice with edge length of unit cell equal to 387 pm. If the radius of the \[C{{l}^{-}}\] ion is 181 pm, the radius of \[\overset{+}{\mathop{N}}\,{{H}_{4}}\] ions is
A)
366.3 pm
done
clear
B)
154.1 pm
done
clear
C)
92.6 pm
done
clear
D)
None of these
done
clear
View Answer play_arrow
IUPAC name of \[[Pt{{(N{{H}_{3}})}_{3}}(Br)(N{{O}_{2}})Cl]\] is
A)
Triamminechlorobromonitroplatinum (IV) chloride
done
clear
B)
Triamminebromonitrochloroplatinum (IV) chloride
done
clear
C)
Triamminebromochloronitroplatinum (IV) chloride
done
clear
D)
Triamminenitrochlorobromoplatinum (IV) chloride
done
clear
View Answer play_arrow
A molecule M associates in a given solvent according to the equation\[M{{(M)}_{n}}.\] For a certain concentration of M, the van't Hoff factor was found to be 0.9 and the fraction of associated molecules was 0.2. The value of n is:
A)
3
done
clear
B)
5
done
clear
C)
2
done
clear
D)
4
done
clear
View Answer play_arrow
Which of the following compound will undergo self aldol condensation in the presence of cold dilute alkali?
A)
\[C{{H}_{2}}=CH-CHO\]
done
clear
B)
\[CH=C-CHO\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}CHO\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}CHO.\]
done
clear
View Answer play_arrow
When one mole of an ideal gas is compressed to half its initial volume and simultaneously heated to twice in initial temperature, the change in entropy \[(\Delta S)\] is
A)
\[{{C}_{V}}\,\ln \,2\]
done
clear
B)
\[{{C}_{p}}\,\ln \,2\]
done
clear
C)
\[R\,\ln \,2\]
done
clear
D)
\[({{C}_{V}}-R)\,\ln \,2\]
done
clear
View Answer play_arrow
If x is amount of adsorbate and m is amount of adsorbent, which of the following relations is not related to adsorption process?
A)
\[x/m=K(p)\] at very low pressure.
done
clear
B)
\[x/m=K{{p}^{\text{l}/n}}\] at inter mediate pressure
done
clear
C)
\[\frac{x}{m}=K\] at very high pressure
done
clear
D)
\[\frac{x}{m}=p\]
done
clear
View Answer play_arrow
For the reaction of one mole of zinc dust with one mole of sulphuric acid in a bomb calorimeter, \[\Delta U\]and w corresponds to
A)
\[\Delta U<0,\]\[w=0\]
done
clear
B)
\[\Delta U<0,\]\[w<0\]
done
clear
C)
\[\Delta U>0,\]\[w=0\]
done
clear
D)
\[\Delta U>0,\]\[w>0\]
done
clear
View Answer play_arrow
Reactivity of hydrogen atoms attached to different carbon atoms in alkanes has the order
A)
Tertiary > Primary > Secondary
done
clear
B)
Primary > Secondary > Tertiary
done
clear
C)
Both [a] and [b]
done
clear
D)
Tertiary > Secondary > Primary.
done
clear
View Answer play_arrow
In most cases, for a rise of 10K temperature the rate constant is doubled to triple. This is due to the reason that
A)
collision frequency increases by a factor of 2 to 3.
done
clear
B)
fraction of molecules possessing threshold energy increases by a factor of 2 to 3.
done
clear
C)
Activation energy is lowered by a factor of 2 to 3.
done
clear
D)
none of these
done
clear
View Answer play_arrow
The restricted rotation about carbon carbon double bond in 2-butene is due to :
A)
Overlap of one s- and \[s{{p}^{2}}\]-hybridized orbitals.
done
clear
B)
Overlap of two \[s{{p}^{2}}\]- hybridized orbitals.
done
clear
C)
Overlap of one p-and one \[s{{p}^{2}}\]-hybridized orbitals.
done
clear
D)
Sideways overlap of two p-orbitals.
done
clear
View Answer play_arrow
Which of the following colligative property can provide molar mass of proteins (or polymers or colloids) with greatest precision?
A)
Osmotic pressure
done
clear
B)
Elevation of boiling point
done
clear
C)
Depression of freezing point
done
clear
D)
Relative lowering of vapour pressure
done
clear
View Answer play_arrow
Cyclic hydrocarbon 'A' has all the carbon and hydrogen atoms in a single plane. All the carbon carbon bonds have the same length, less than \[1.54\,\overset{{}^\circ }{\mathop{A}}\,,\] but more than \[1.34\,\overset{{}^\circ }{\mathop{A}}\,.\] The\[C-C-C\] bond angle will be
A)
\[109{}^\circ 28'\]
done
clear
B)
\[100{}^\circ \]
done
clear
C)
\[180{}^\circ \]
done
clear
D)
\[120{}^\circ \]
done
clear
View Answer play_arrow
The major product obtained in the photo catalysed bromination of 2-methylbutane is:
A)
1-bromo-2-methylbutane
done
clear
B)
1-bromo-3-methylbutane
done
clear
C)
2-bromo-3-methylbutane
done
clear
D)
2-bromo-2-methylbutane
done
clear
View Answer play_arrow
The reason for double helical structure of DNA is the operation of:
A)
Electrostatic attractions
done
clear
B)
van der Waals forces
done
clear
C)
Dipole - Dipole interactions
done
clear
D)
Hydrogen bonding
done
clear
View Answer play_arrow
Among the following, correct statement is:
A)
Brownian movement is more pronounced for smaller particles than for bigger- particles.
done
clear
B)
Sols of metal sulphides are lyophilic.
done
clear
C)
Hardy Schuize law states that bigger the size of the ions, the greater is its coagulating power.
done
clear
D)
One would expect charcoal to adsorb chlorine more than hydrogen sulphide.
done
clear
View Answer play_arrow
What is the correct order of spin only magnetic moment (in BM) of \[M{{n}^{2+}},\]\[C{{r}^{2+}}\] and \[V{{r}^{2+}}?\]
A)
\[M{{n}^{2+}}>{{V}^{2+}}>C{{r}^{2+}}\]
done
clear
B)
\[{{V}^{2+}}>C{{r}^{2+}}>M{{n}^{2+}}\]
done
clear
C)
\[M{{n}^{2+}}>C{{r}^{2+}}>{{V}^{2+}}\]
done
clear
D)
\[C{{r}^{2+}}>{{V}^{2+}}>M{{n}^{2+}}\]
done
clear
View Answer play_arrow
In which of the following cases, pH is greater than 7?
A)
\[50\,mL\,\,\text{of}\,\,0.1\,MHCl+50\,mL\,\,\text{of}\,\,0.1\,M\,NaCl\]
done
clear
B)
\[50\,mL\,\text{of}\,\,0.1\,M{{H}_{2}}S{{O}_{4}}+50\,mL\,\text{of}\,0.2\,M\,NaOH\]
done
clear
C)
\[50\,mL\,\text{of}\,\,0.1\,MC{{H}_{3}}COOH+50\,mL\,\text{of}\,0.1\,M\,KOH\]
done
clear
D)
\[50\,mL\,\text{of}\,\,0.1\,MHN{{O}_{3}}+50\,mL\,\text{of}\,\,0.1\,M\,N{{H}_{3}}\]
done
clear
View Answer play_arrow
In the presence of platinum catalyst, hydrocarbon A adds hydrogen to form n-hexane. When hydrogen bromide is added to A instead of hydrogen, only a single bromo compound is formed. Which of the following is A?
A)
\[C{{H}_{3}}-C{{H}_{2}}-CH=CH-C{{H}_{2}}-C{{H}_{3}}\]
done
clear
B)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-CH=CH-C{{H}_{3}}\]
done
clear
C)
\[C{{H}_{3}}-CH=CH-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{3}}\]
done
clear
D)
\[C{{H}_{2}}=CH-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{3}}\]
done
clear
View Answer play_arrow
Which of the following statements are correct?
(i) Polynuclear hydrocarbons contain two or more benzene rings fused together. (ii) Polynuclear hydrocarbons have carcinogenic property. (iii) Polynuclear hydrocarbons are formed on incomplete combustion of organic materials like tobacco, coal and petroleum. (iv) They are also produced in human body due to various biochemical reactions.
A)
(i), (ii) and (iv)
done
clear
B)
(i), (iii) and (iv)
done
clear
C)
(ii), (iii) and (iv)
done
clear
D)
(i), (ii) and (iii)
done
clear
View Answer play_arrow
Most plants are green in colour because
A)
the atmosphere filters out all the colours of the visible light spectrum except green.
done
clear
B)
green light is the most effective wavelength region of the visible spectrum in sunlight for photosynthesis.
done
clear
C)
chlorophyll is least effective in absorbing green light.
done
clear
D)
green light allows maximum photosynthesis.
done
clear
View Answer play_arrow
Which of the following organism possesses characteristics of both a plant and an animal?
A)
Bacteria
done
clear
B)
Euglena
done
clear
C)
Mycoplasma
done
clear
D)
Paramecium
done
clear
View Answer play_arrow
Function of companion cells is
A)
providing energy to sieve elements for active transport.
done
clear
B)
providing water to phloem.
done
clear
C)
loading of sucrose into sieve elements by passive transport.
done
clear
D)
loading of sucrose into sieve elements.
done
clear
View Answer play_arrow
In cell cycle, DNA replication takes place in
A)
\[{{G}_{1}}\]phase
done
clear
B)
\[{{G}_{2}}\]phase
done
clear
C)
mitotic metaphase
done
clear
D)
S phase
done
clear
View Answer play_arrow
Select the events that do not occur in interphase stage of cell-cycle
(i) RNA and protein synthesis. (ii) Cytoplasmic growth. (iii) Polymerisation of spindle fibres protein. (iv) Disappearance of Golgi bodies and ER. (v) DNA molecules in highly supercoiled stage.
A)
(iii), (iv) & (v)
done
clear
B)
(iv) & (v) only
done
clear
C)
(ii), (iii) & (iv)
done
clear
D)
(iii) & (iv) only
done
clear
View Answer play_arrow
Organisms which are indicator of \[S{{O}_{2}}\] pollution of air are
A)
Mosses
done
clear
B)
Lichens
done
clear
C)
Mushrooms
done
clear
D)
Puffballs
done
clear
View Answer play_arrow
Choose the correct option for A, B, C and D of a mature embryo sac.
A)
A-Filiform apparatus; B-Antipodal cells; C-Synergids; D - Polar nuclei
done
clear
B)
A-Antipodal cells; B-Filiform apparatus; C- Synergids; D-Polar nuclei
done
clear
C)
A-Antipodal cells; B-Filiform apparatus; C- Polar nuclei; D-Synergids
done
clear
D)
A-Antipodal cells; B-Polar nuclei; C-Filiform apparatus; D-Synergids
done
clear
View Answer play_arrow
In which one pair both the plants can be vegetative propagated by leaf pieces?
A)
Bryophyllum and Kalanchoe
done
clear
B)
Chrysanthemum and Agave
done
clear
C)
Agave and Kalanchoe
done
clear
D)
Asparagus and Bryophyllum
done
clear
View Answer play_arrow
A typical angiospermous ovule is attached to the placenta by means of a stalk called X. Body of the ovule fuses with X in the region called Y. Identify X and Y.
A)
X - Funicle Y - Hihim
done
clear
B)
X - Hilum Y - Funicle
done
clear
C)
X - Funicle Y - Micropyle
done
clear
D)
X - Hilum Y - Chalaza
done
clear
View Answer play_arrow
The greatest threat to global biodiversity is
A)
natural disasters such as storms
done
clear
B)
pollution
done
clear
C)
overexploitation of natural resources
done
clear
D)
alteration of habitats by humans
done
clear
View Answer play_arrow
Secondary sewage treatment is mainly a
A)
physical process
done
clear
B)
mechanical process
done
clear
C)
chemical process
done
clear
D)
biological process
done
clear
View Answer play_arrow
Species interaction with negative influence on both is referred to as
A)
amensalism
done
clear
B)
mutualism
done
clear
C)
commensalism
done
clear
D)
competition
done
clear
View Answer play_arrow
Cotyledons and testa respectively are edible parts in
A)
walnut and tamarind
done
clear
B)
french bean and coconut
done
clear
C)
cashew nut and litchi
done
clear
D)
groundnut and pomegranate
done
clear
View Answer play_arrow
When pollen grains are not transferred from anthers to stigma in a flower, due to the barrier, it is called
A)
herkogamy
done
clear
B)
heterogamy
done
clear
C)
cleistogamy
done
clear
D)
dichogamy
done
clear
View Answer play_arrow
Water potential is equal to
A)
\[\Psi +O.P.\]
done
clear
B)
\[{{\Psi }_{s}}+O.P.\]
done
clear
C)
\[{{\Psi }_{p}}+{{\Psi }_{w}}\]
done
clear
D)
\[{{\Psi }_{s}}+{{\Psi }_{p}}\]
done
clear
View Answer play_arrow
Which of the following is a free living aerobic non-photosynthetic nitrogen-fixer?
A)
Rhizobium
done
clear
B)
Azotobacter
done
clear
C)
Azospirillum
done
clear
D)
Nostoc
done
clear
View Answer play_arrow
What kinds of the structures of proteins are shown in the following figure?
A)
\[A=1{}^\circ \] structure, \[B=2{}^\circ \] structure, \[C=3{}^\circ \] structure, \[D=4{}^\circ \] structure
done
clear
B)
\[A=4{}^\circ \] structure, \[B=2{}^\circ \] structure, \[C=3{}^\circ \] structure, \[D=2{}^\circ \] structure
done
clear
C)
\[A=1{}^\circ \] structure, \[B=4{}^\circ \] structure, \[C=3{}^\circ \] structure, \[D=2{}^\circ \] structure
done
clear
D)
\[A=4{}^\circ \] structure, \[B=3{}^\circ \] structure, \[C=2{}^\circ \] structure, \[D=1{}^\circ \] structure
done
clear
View Answer play_arrow
In soil, the water available for root absorption is
A)
gravitational water
done
clear
B)
capillary water
done
clear
C)
hygroscopic water
done
clear
D)
combined water
done
clear
View Answer play_arrow
Which of the following minerals activate the enzymes involved in respiration?
A)
nitrogen and phosphorus
done
clear
B)
magnesium and manganese
done
clear
C)
potassium and calcium
done
clear
D)
sulphur and iron
done
clear
View Answer play_arrow
\[{{C}_{4}}\] acid, formed in the mesophyll of \[{{C}_{4}}\] plants leaf during photosynthesis is
A)
OAA or malic acid or aspartic acid
done
clear
B)
pyruvic acid
done
clear
C)
succinic acid
done
clear
D)
fumaric acid
done
clear
View Answer play_arrow
AzotobacterandBeijerinckia are the examples of
A)
symbiotic nitrogen-fixers
done
clear
B)
non-symbiotic nitrogen-fixers
done
clear
C)
ammonifying bacteria
done
clear
D)
disease causing bacteria
done
clear
View Answer play_arrow
The first carbon dioxide acceptor in \[{{C}_{4}}\]-plants is
A)
Phosphoenol-pyruvate
done
clear
B)
Ribulose 1, 5-diphosphate
done
clear
C)
Qxalo-acetic acid
done
clear
D)
Phosphoglyceric acid
done
clear
View Answer play_arrow
Select the incorrect statement regarding DNA replication
A)
Leading strand is formed in \[5'\to 3'\] direction
done
clear
B)
Okazaki fragments are formed in \[5'\to 3'\] direction
done
clear
C)
DNA polymerase catalyses polymerization in \[5'\to 3'\] direction
done
clear
D)
DNA polymerase catalyses polymerization in \[3'\to 5'\] direction
done
clear
View Answer play_arrow
Red (RR) Antirrhinum is crossed with white (WW) one. Offspring RW are pink. This is an example of
A)
dominant-recessive
done
clear
B)
incomplete dominance
done
clear
C)
hybrid
done
clear
D)
supplementary genes
done
clear
View Answer play_arrow
Food chain in which micro-organisms breakdown the food formed by primary producers is
A)
parasitic food chain
done
clear
B)
detritus food chain
done
clear
C)
consumer food chain
done
clear
D)
predator food chain
done
clear
View Answer play_arrow
Which of the following pairs is a sedimentary type of biogeochemical cycle?
A)
Oxygen and nitrogen
done
clear
B)
Phosphorus and sulphur
done
clear
C)
Phosphorus and nitrogen
done
clear
D)
Phosphorus and carbon dioxide
done
clear
View Answer play_arrow
Independent assortment of genes does not take place when
A)
genes are located on homologous chromosomes
done
clear
B)
genes are linked and located on same chromosomes
done
clear
C)
genes are located on non-homologous chromosomes
done
clear
D)
All the above
done
clear
View Answer play_arrow
Breeding of crops with high levels of minerals, vitamins and proteins is called
A)
Somatic hybridization
done
clear
B)
Biofortification
done
clear
C)
Biomagnification
done
clear
D)
Micropropagation
done
clear
View Answer play_arrow
If we completely remove the decomposers from an ecosystem, its functioning will be adversely affected, because
A)
energy flow will be blocked
done
clear
B)
herbivores will not receive solar energy
done
clear
C)
mineral movement will be blocked
done
clear
D)
rate of decomposition will be very high
done
clear
View Answer play_arrow
A nucleotide is formed of
A)
Purine, pyrimidine and phosphate
done
clear
B)
Purine, sugar and phosphate
done
clear
C)
Nitrogen base, sugar and phosphate
done
clear
D)
Pyrimidine, sugar and phosphate
done
clear
View Answer play_arrow
Which of the following hormones are responsible for maintaining bud dormancy in deciduous trees?
A)
Auxins
done
clear
B)
Cytokinins
done
clear
C)
Gibberellins
done
clear
D)
Abscisic acid
done
clear
View Answer play_arrow
A small segment of DNA contains the base sequence CGT. If an mRNA transcript is made that includes this DNA sequence, what will be the anticodon on the tRNA that will bind to the corresponding mRNA codon for this DNA triplet?
A)
CGT
done
clear
B)
GCA
done
clear
C)
CGU
done
clear
D)
GCT
done
clear
View Answer play_arrow
Which of the following is a reducing sugar?
A)
Galactose
done
clear
B)
Gluconic acid
done
clear
C)
P-methyl galactoside
done
clear
D)
Sucrose
done
clear
View Answer play_arrow
Which of the following processes make direct use of oxygen?
A)
Glycolysis
done
clear
B)
Fermentation
done
clear
C)
Electron transport
done
clear
D)
Krebs cycle
done
clear
View Answer play_arrow
Phosphofructokinase is inhibited by \[{{H}^{+}}\] ions and hence the rate of glycolysis:
A)
increases when pH falls
done
clear
B)
decreases when pH falls significantly
done
clear
C)
neither increase or decrease
done
clear
D)
either increase or decrease
done
clear
View Answer play_arrow
Which one of the following is incorrect about the activities associated with PS-I and PS-II in non-cyclic photophospho-rylation?
A)
Water is oxidised in PS-II, but not in PS-I.
done
clear
B)
Photons (light) are needed to activate both PS-I and PS-II.
done
clear
C)
Photolysis of water, formation of ATP + NADPH+H occur.
done
clear
D)
Production of NADPH + \[{{H}^{+}}\] is associated with PS-II, but not with PS-I.
done
clear
View Answer play_arrow
Wildlife is destroyed most when
A)
there is lack of proper care
done
clear
B)
mass scale hunting for foreign trade
done
clear
C)
its natural habitat is destroyed
done
clear
D)
natural calamity occurs
done
clear
View Answer play_arrow
Golden rice is a transgenic crop of the future with the following improved trait:
A)
insect resistance
done
clear
B)
high lysine (essential amino acid) content
done
clear
C)
high protein content
done
clear
D)
high vitamin-A content
done
clear
View Answer play_arrow
Which of the following could not be considered an ecosystem?
A)
A small pond
done
clear
B)
All the fish in a coral reef
done
clear
C)
Earth
done
clear
D)
A pile of dung in a pasture
done
clear
View Answer play_arrow
A common biucontrol agcni for the control of plant diseases is
A)
Baculovirus
done
clear
B)
Bacillus thuringiensis
done
clear
C)
Glomus
done
clear
D)
Trichoderma
done
clear
View Answer play_arrow
Which of the following is not caused by deficiency of mineral nutrition -
A)
Necrosis
done
clear
B)
Etiolation
done
clear
C)
Chlorosis
done
clear
D)
Shortening of intern odes.
done
clear
View Answer play_arrow
A number of natural reserves have been created to conserve specific wild life species. Identify the correct combination from the following
A)
Gir Forest - Tiger
done
clear
B)
Kaziranga - Elephants
done
clear
C)
Rann of Kutch - Wild Ass
done
clear
D)
Manas wild life Sancturay - Musk deer
done
clear
View Answer play_arrow
PCR and Restriction Fragment Length Polymorphism are the methods for:
A)
Study of enzymes
done
clear
B)
Genetic transformation
done
clear
C)
DNA sequencing
done
clear
D)
Genetic Fingerprinting
done
clear
View Answer play_arrow
The \[{{C}_{4}}\] plants are photosynthetically more efficient than \[{{C}_{3}}\] plants because:
A)
the \[C{{O}_{2}}\] compensation point is more
done
clear
B)
\[C{{O}_{2}}\] generated during photorespiration is trapped and recycled through PEP carboxylase
done
clear
C)
the \[C{{O}_{2}}\] efflux is not prevented
done
clear
D)
they have more chloroplasts
done
clear
View Answer play_arrow
The enormous diversity of protein molecules is due mainly to the diversity of
A)
amino groups on the amino acids
done
clear
B)
R groups on the amino acids
done
clear
C)
amino acid sequences within the protein molecule
done
clear
D)
peptide bonds
done
clear
View Answer play_arrow
A tooth scraping yields large numbers of corkscrew shaped bacteria. These bacteria are referred to as
A)
Bacilli
done
clear
B)
Cocci
done
clear
C)
Spirffli
done
clear
D)
Helici
done
clear
View Answer play_arrow
Which of the following class is being correctly described by given statements (i - iv)?
(i) All living members of this class are ectoparasites on some fishes, (ii) They have a sucking and circular mouth without jaws. (iii) Circulation is of closed type. (iv) They are marine but migrate for spawning to fresh water. After spawing, within a few days they die.
A)
Cyclostomata
done
clear
B)
Chondrichthyes
done
clear
C)
Osteichthyes
done
clear
D)
Amphibia
done
clear
View Answer play_arrow
Intercalated discs are the communication junctions between the cells of
A)
cardiac muscles
done
clear
B)
striped muscles
done
clear
C)
adipose tissue
done
clear
D)
nerve and striated muscles
done
clear
View Answer play_arrow
In which of the phylum, excretory organ like proboscis gland is present?
A)
Hemichordata
done
clear
B)
Chordata
done
clear
C)
Echinodermata
done
clear
D)
Annelida
done
clear
View Answer play_arrow
Pseudostratified epithelium is found in
A)
oesophagus
done
clear
B)
respiratory tract
done
clear
C)
urinary tract
done
clear
D)
kidney
done
clear
View Answer play_arrow
Male and female cockroaches can be distinguished externally through
A)
anal styles in male
done
clear
B)
anal cerci in female
done
clear
C)
anal style and antennae in females
done
clear
D)
Both [b] and [c]
done
clear
View Answer play_arrow
Nerve cells do not divide because they do not have
A)
nucleus
done
clear
B)
centrosome
done
clear
C)
golgi body
done
clear
D)
mitochondria
done
clear
View Answer play_arrow
Which of the following is not currently a major cause of the global reduction in biodiversity?
A)
Overexploitation
done
clear
B)
Global warming
done
clear
C)
Habitat destruction
done
clear
D)
Introduction of foreign predators and disease
done
clear
View Answer play_arrow
Human population growth in India
A)
tends to follow a sigmoid curve as in case of many other animal species
done
clear
B)
tends to reach a zero population growth as in case of some animal species
done
clear
C)
can be reduced by permitting natural calamities and enforcing birth control measures
done
clear
D)
can be regulated by following the national programme of family planning
done
clear
View Answer play_arrow
The diaphragm, cervical cap and vaults are
A)
Disposable contraceptive devices
done
clear
B)
Reusable contraceptives
done
clear
C)
lUDs
done
clear
D)
Implants
done
clear
View Answer play_arrow
The main function of trophoectoderm in mammalian embryo is
A)
formation of future endoderm
done
clear
B)
formation of the body of developing embryo
done
clear
C)
formation of future ectoderm
done
clear
D)
formation of placenta
done
clear
View Answer play_arrow
Following diagrammatic representation refers the natural selection on different traits. In which all the three graphs A, B and C are identified correctly
A)
A-Directional, B-Stabilising, C-Disruptive
done
clear
B)
A-Stabilising, B-Directional, C-Disruptive
done
clear
C)
A-Stabilising, B-Disruptive, C-Directional
done
clear
D)
A-Directional, B-Disruptive, C-Stabilising
done
clear
View Answer play_arrow
Which one of the following statements about human sperm is correct?
A)
Acrosome has a conical pointed structure used for piercing and penetrating the egg, resulting in fertilization
done
clear
B)
The sperm lysis in the acrosome dissolve the egg envelope facilitating fertilization
done
clear
C)
Acrosome serves as a sensory structure leading the sperm towards the ovum
done
clear
D)
Acrosome serves no particular function
done
clear
View Answer play_arrow
Bulbourethral gland is present in
A)
Males and is another name for Uterus masculine
done
clear
B)
Females and is another name for Bertholin's gland
done
clear
C)
Males and is another name for Cowper's gland
done
clear
D)
None of these
done
clear
View Answer play_arrow
One of the factors required for the maturation of the erythrocytes is
A)
vitamin A
done
clear
B)
vitamin D
done
clear
C)
vitamin \[{{B}_{12}}\]
done
clear
D)
vitamin C
done
clear
View Answer play_arrow
In which of the following would you record the highest blood pressure?
A)
The ventricle supplying blood to the gills of a fish
done
clear
B)
The anterior dorsal artery of an ant
done
clear
C)
The pulmonary vein of a frog
done
clear
D)
The ventricle supplying blood to the systemic circuit of a bird
done
clear
View Answer play_arrow
Oxyntic cells are located in
A)
Islets of Langerhans
done
clear
B)
Gastric epithelium and secrete pepsin
done
clear
C)
Kidneys and secrete renin
done
clear
D)
Gastric epithelium and secrete HCl
done
clear
View Answer play_arrow
If Henle's loop were absent from mammalian nephron which of the following is to be expected?
A)
The urine will be more concentrated
done
clear
B)
The urine will be more dilute
done
clear
C)
There will be no urine formation
done
clear
D)
There will be hardly any change in the quality and quantity of urine formed
done
clear
View Answer play_arrow
Hepatic portal system carries
A)
Oxygenated blood from liver to intestine
done
clear
B)
Deoxygenated blood from liver to intestine
done
clear
C)
Oxygenated blood from intestine to liver
done
clear
D)
Deoxygenated blood from intestine to liver
done
clear
View Answer play_arrow
"Kwashiorkor' and 'Beri-Beri' are
A)
communicable diseases
done
clear
B)
infectious diseases
done
clear
C)
deficiency diseases
done
clear
D)
None of the above
done
clear
View Answer play_arrow
ABO blood group system is due to
A)
multifactor inheritance
done
clear
B)
incomplete dominance
done
clear
C)
multiple allelism
done
clear
D)
epi stasis
done
clear
View Answer play_arrow
Change over time within an evolutionary lineage (phyletic change).
A)
is the only important evolutionary process.
done
clear
B)
rarely occurs without geographic separation of populations.
done
clear
C)
is the main means by which new species arise.
done
clear
D)
cannot account for the proliferation of species.
done
clear
View Answer play_arrow
Bull semen for the purpose of artificial insemination is stored in
A)
Ice
done
clear
B)
Liquid oxygen
done
clear
C)
Liquid nitrogen
done
clear
D)
Liquid \[C{{O}_{2}}\]
done
clear
View Answer play_arrow
The kind of evolution in which two species of different geneology come to resemble one another closely, is termed as
A)
progressive evolution
done
clear
B)
convergent evolution
done
clear
C)
parallel evolution
done
clear
D)
retrogressive evolution
done
clear
View Answer play_arrow
Drosophila flies with XXY genotype are females, but human beings with such genotype are abnormal males. It shows that
A)
Y-chromosome is essential for sex determination in Drosophila.
done
clear
B)
Y-chromosome is female determinant in Drosophila.
done
clear
C)
Y-chromosome is male determination in human beings.
done
clear
D)
Y-chromosome has no role in sex determination either in Drosophila or in human beings.
done
clear
View Answer play_arrow
Hinny is a hybrid of male
A)
Horse and female donkey
done
clear
B)
Donkey and female horse
done
clear
C)
Goat and female lamb
done
clear
D)
Sheep and female goat.
done
clear
View Answer play_arrow
Which of the following is the first step in allopatric speciation?
A)
genetic drift
done
clear
B)
geographical isolation
done
clear
C)
polyploidy
done
clear
D)
hybridization
done
clear
View Answer play_arrow
When \[C{{O}_{2}}\] concentration in blood increases, breathing becomes
A)
shallower and slow
done
clear
B)
there is no effect on breathing
done
clear
C)
slow and deep
done
clear
D)
faster and deeper
done
clear
View Answer play_arrow
Identify A, B, C and D
A)
A-Forebrain, B-Brainstem, C-Corpus callosum, D-Cerebral aqueduct
done
clear
B)
A-Forebrain, B-Brainstem, C-Cerebral aqueduct, D-Corpus callosum
done
clear
C)
A-Brainstem, B-Forebrain, C-Corpus callosum, D-Cerebral aqueduct
done
clear
D)
A-Brainstem, B-Forebrain, C-Cerebral aqueduct, D-Corpus luteum
done
clear
View Answer play_arrow
Which of the following is a single U shaped bone, present at the base of the buccal cavity and it is also included in the skull?
A)
Hyoid
done
clear
B)
Malleus
done
clear
C)
Sacrum
done
clear
D)
Scapula
done
clear
View Answer play_arrow
Which of the following would be most likely to help to slow down the greenhouse effect?
A)
Ensuring that all excess paper packaging is burned to ashes.
done
clear
B)
Promoting the use of private rather than public transport.
done
clear
C)
Converting tropical forests into grazing land for cattle.
done
clear
D)
Re-designing land-fill dumps to allow methane to be collected.
done
clear
View Answer play_arrow
Which gland is involved in the fanction of our ?biological clock"?
A)
pineal gland
done
clear
B)
thyroid gland
done
clear
C)
adrenal glands
done
clear
D)
ovaries
done
clear
View Answer play_arrow
Which of the following statements regarding glucagon is false?
A)
It is secreted by a-cells of Langerhans.
done
clear
B)
It acts antagonistically to insulin.
done
clear
C)
It decreases blood sugar level.
done
clear
D)
The gland responsible for its secretion is heterocrine gland.
done
clear
View Answer play_arrow
Which hormone possesses anti-insulin effect?
A)
Cortisol
done
clear
B)
Calcitonin
done
clear
C)
Qxytocin
done
clear
D)
Aldosterone
done
clear
View Answer play_arrow
Which is the example of conditioned reflex?
A)
Eyes closed when anything enter into it.
done
clear
B)
Hand took up when piercing with needle.
done
clear
C)
Salivation in a hungry dog in response to ringing of a bell.
done
clear
D)
Digestion food goes forward in alimentary canal.
done
clear
View Answer play_arrow
Which one of the following techniques made it possible to genetically engineerd living organism?
A)
Recombinant DNA techniques
done
clear
B)
X-ray diffraction
done
clear
C)
Heavier isotope labelling
done
clear
D)
Hybridization
done
clear
View Answer play_arrow
The diagram opposite shows a homologous (bivalent) pair of chromosomes during meiosis.
Which of the following correctly represents the final products of the second meiotic division?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following is the "bird flu virus"?
A)
\[{{H}_{5}}{{N}_{1}}\]
done
clear
B)
Haemophilus influenza
done
clear
C)
HTV
done
clear
D)
Rhino virus
done
clear
View Answer play_arrow
What is mode of bacterial resistance against antibiotics?
A)
Development of thick mucilaginous layer
done
clear
B)
Alteration of cell membrane
done
clear
C)
Mutation in bacteria
done
clear
D)
All the above
done
clear
View Answer play_arrow
Which one of the following is a wrong matching of a microbe and its industrial product, while the remaining three are correct ?
A)
Yeast - statins
done
clear
B)
Acetobacter aceti - acetic acid
done
clear
C)
Clostridium butylicum - lactic acid
done
clear
D)
Aspergillus niger - citric acid
done
clear
View Answer play_arrow
Interleukins are responsible for activation of
A)
Cell mediated immunity
done
clear
B)
Humoral immunity
done
clear
C)
BothNone
done
clear
D)
done
clear
View Answer play_arrow
Select the correct statement with respect to diseases and immunisation?
A)
If due to some reason B-and T-lymphocytes are damaged, the body will not produce antibodies against a pathogen
done
clear
B)
Injection of dead / inactivated pathogens causes passive immunity
done
clear
C)
Certain protozoans have been used to mass produce hepatitis B vaccine.
done
clear
D)
Injection of snake antivenom against snake bite is an example of active immunization
done
clear
View Answer play_arrow
The simplest amino acid is ______.
A)
glycine
done
clear
B)
proline
done
clear
C)
leucine
done
clear
D)
tryptophan
done
clear
View Answer play_arrow
Which one of the following is the correct matching of three items and their grouping category?
A)
Items - Ilium, ischium, pubis Group - coxal bones of pelvic girdle
done
clear
B)
Items - Actin, myosin, rhodopsin. Group - muscle proteins
done
clear
C)
Items - Cytosine, uracil, thiamine Group - Pyrimidines
done
clear
D)
Items - Malleus, incus, cochlea Group - ear ossicles
done
clear
View Answer play_arrow
What is the first step in the Southern blot technique?
A)
Denaturation of DNA on the gel for hybridization with specific probe.
done
clear
B)
Production of a group of genetically identical cells.
done
clear
C)
Digestion of DNA by restriction enzyme.
done
clear
D)
Denaturation of DNA from a nucleated cell such as the one from the scene of crime.
done
clear
View Answer play_arrow
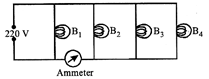 The reading in an ideal ammeter will be:
The reading in an ideal ammeter will be: 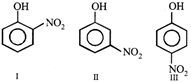
 What is the correct order of pressures?
What is the correct order of pressures? 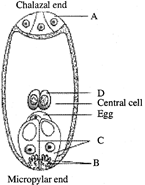
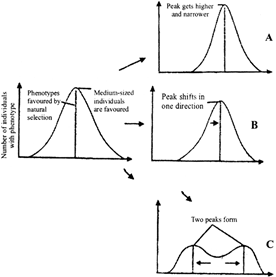 Following diagrammatic representation refers the natural selection on different traits. In which all the three graphs A, B and C are identified correctly
Following diagrammatic representation refers the natural selection on different traits. In which all the three graphs A, B and C are identified correctly 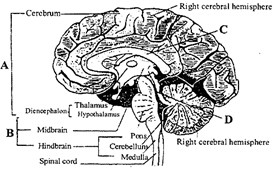
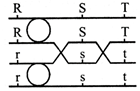 Which of the following correctly represents the final products of the second meiotic division?
Which of the following correctly represents the final products of the second meiotic division?