If the velocity of light C, the universal gravitational constant G and planck's constant h be chosen as fundamental units, the dimensions of mass in this system are:
A)
hCG
done
clear
B)
hCG-1
done
clear
C)
\[{{\operatorname{h}}^{-1}}{{K}^{-1}}G\]
done
clear
D)
\[{{\operatorname{h}}^{{}^{1}/{}_{2}}}{{K}^{{}^{1}/{}_{2}}}{{G}^{{}^{-1}/{}_{2}}}\]
done
clear
View Answer play_arrow
A particle is executing SHM along a straight line its velocities at distance \[{{\operatorname{x}}_{1}} and {{x}_{2}}\]from the mean position are \[{{\operatorname{v}}_{1}} and {{v}_{2}}\]respectively its time period is:
A)
\[2\pi \sqrt{\frac{{{\operatorname{v}}_{1}}^{2}-{{\operatorname{v}}_{2}}^{2}}{{{\operatorname{x}}_{1}}^{2}-{{\operatorname{x}}_{1}}^{2}}}\]
done
clear
B)
\[2\pi \sqrt{\frac{{{\operatorname{x}}_{1}}^{2}+{{\operatorname{x}}_{1}}^{2}}{{{\operatorname{v}}_{1}}^{2}+{{\operatorname{v}}_{2}}^{2}}}\]
done
clear
C)
\[2\pi \sqrt{\frac{{{\operatorname{x}}_{1}}^{2}-{{\operatorname{x}}_{1}}^{2}}{{{\operatorname{v}}_{1}}^{2}-{{\operatorname{v}}_{2}}^{2}}}\]
done
clear
D)
\[2\pi \sqrt{\frac{{{\operatorname{v}}_{1}}^{2}+{{\operatorname{v}}_{2}}^{2}}{{{\operatorname{x}}_{1}}^{2}+{{\operatorname{x}}_{1}}^{2}}}\]
done
clear
View Answer play_arrow
A shell bursts on contact with the ground and fragments fly in all directions with speed upto 39.2 m/s. A man 78.4m way is in danger for:
A)
4scc
done
clear
B)
6sec
done
clear
C)
\[\sqrt{2} sec\]
done
clear
D)
\[4\sqrt{2} sec\]
done
clear
View Answer play_arrow
The following four wires of length L and radius r are made of the same material. Which of these will have the largest extension when the same tension is applied?
A)
L = 50cm, r= 0.25
done
clear
B)
L = 100cm, r = 0.5 mm
done
clear
C)
L = 200cm, r = 1mm
done
clear
D)
L = 300cm, r = 1.5mm
done
clear
View Answer play_arrow
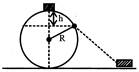
A particle originally at rest at the highest point of smooth circle in a vertical plane, is gently pushed and starts sliding along the circle. It will leave the circle at a vertical distance h below the highest point such that:
A)
h = 2R
done
clear
B)
\[\operatorname{h}=\frac{R}{2}\]
done
clear
C)
h = R
done
clear
D)
\[\operatorname{h}=\frac{R}{3}\]
done
clear
View Answer play_arrow
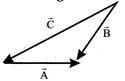
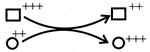
For the figure shown:
A)
\[\vec{A}+\vec{B}=\vec{C}\]
done
clear
B)
\[\vec{B}+\vec{C}=\vec{A}\]
done
clear
C)
\[\vec{C}+\vec{A}=\vec{B}\]
done
clear
D)
\[\vec{A}+\vec{B}+\vec{C}=0\]
done
clear
View Answer play_arrow
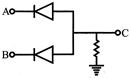
In the circuit below, A and B represents two inputs and C represents the output. The circuit represents:
A)
OR
done
clear
B)
NOR
done
clear
C)
AND
done
clear
D)
NAND
done
clear
View Answer play_arrow
Two metals spheres are falling through a liquid of density \[2\times 1{{0}^{3}} kg / {{m}^{3}}\]with the same uniform speed. The material density of sphere 1 and sphere\[2 are\,\,8\times {{10}^{3}}kg/{{m}^{3}} and 11\times 1{{0}^{3}} kg/{{m}^{3}}\] respectively. The ratio of their radii is:
A)
\[\frac{11}{8}\]
done
clear
B)
\[\sqrt{\frac{11}{8}}\]
done
clear
C)
\[\frac{3}{2}\]
done
clear
D)
\[\sqrt{\frac{3}{2}}\]
done
clear
View Answer play_arrow
Two rain drops falling through air have radii in the ratio 1 : 2. They will have terminal velocity in the ratio:
A)
4:1
done
clear
B)
1:4
done
clear
C)
2:1
done
clear
D)
1: 2
done
clear
View Answer play_arrow
An\[\alpha - particlea\]and a deuteron are moving with velocities v and 2v respectively. What will be the ratio of their de-broglie wave length:
A)
1:1
done
clear
B)
\[\sqrt{2}:1\]
done
clear
C)
\[1:\sqrt{2}\]
done
clear
D)
2:1
done
clear
View Answer play_arrow
In which type of material, the magnetic susceptibility does not depend on temperature?
A)
Diamagnetic
done
clear
B)
Paramagnetic
done
clear
C)
Ferromagnetic
done
clear
D)
Ferrite
done
clear
View Answer play_arrow
A hydrogen atom is in excited state of principal quantum number n. It emits a photon of wavelength\[\lambda \], when returns to ground state. The value of n is:
A)
\[\sqrt{\lambda \operatorname{R}\left( \lambda \operatorname{R}-1 \right)}\]
done
clear
B)
\[\sqrt{\frac{\lambda \operatorname{R}-1}{\lambda \operatorname{R}}}\]
done
clear
C)
\[\sqrt{\frac{\lambda \operatorname{R}}{\lambda \operatorname{R}-1}}\]
done
clear
D)
\[\sqrt{\lambda \left( \operatorname{R}-1 \right)}\]
done
clear
View Answer play_arrow
An electric current passes through a long straight copper wire. At a distance 5cm from the straight wire, the magnetic field is B, The magnetic field at 20cm from the straight wire would be:
A)
\[\frac{B}{6}\]
done
clear
B)
\[\frac{B}{4}\]
done
clear
C)
\[\frac{B}{3}\]
done
clear
D)
\[\frac{B}{2}\]
done
clear
View Answer play_arrow
A quarter horse power motor runs at a speed of 600 rpm. Assuming 40% efficiency, the work done by the motor in one rotation will be:
A)
7.46J
done
clear
B)
7400J
done
clear
C)
7.46erg
done
clear
D)
74.6J
done
clear
View Answer play_arrow
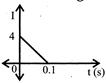
In a coil of resistance\[10\Omega .\], the induced current developed by changing magnetic flux through it, is shown in figure as a function of time. The magnitude of change in flux through the coil is:
A)
8
done
clear
B)
2
done
clear
C)
6
done
clear
D)
4
done
clear
View Answer play_arrow
Monochromatic light frequency \[{{\operatorname{f}}_{1}}\]incident on a photo cell and stopping potential, is found to be\[{{\operatorname{V}}_{1}}\]. What is the new stopping potential of the cell, if it is radiated by monochromatic light of frequency\[{{\operatorname{f}}_{2}}\]?
A)
\[{{\operatorname{V}}_{1}}-\frac{h}{e}\left( {{f}_{2}}-{{f}_{1}} \right)\]
done
clear
B)
\[{{\operatorname{V}}_{1}}+\frac{h}{e}\left( {{f}_{2}}+{{f}_{1}} \right)\]
done
clear
C)
\[{{\operatorname{V}}_{1}}-\frac{h}{e}\left( {{f}_{2}}+{{f}_{1}} \right)\]
done
clear
D)
\[{{\operatorname{V}}_{1}}+\frac{h}{e}\left( {{f}_{2}}-{{f}_{1}} \right)\]
done
clear
View Answer play_arrow
For a common emitter amplifier, the audio frequency voltage across the collector resistance \[2k\Omega \] is 2V. If the current amplification factor of the transistor is 200, and base resistance is \[15k\Omega \], the input signal voltage and base currant:
A)
\[0.1V, 1\mu A\]
done
clear
B)
0.1\[0.15V, 10\mu A\]
done
clear
C)
\[0.015V, 1A\]
done
clear
D)
\[0.0075V, 5\mu A\]
done
clear
View Answer play_arrow
The height at which the weight of a body becomes\[\frac{1}{16}\] its weight on the surface of earth (radius R), is:
A)
5R
done
clear
B)
15R
done
clear
C)
3R
done
clear
D)
4R
done
clear
View Answer play_arrow
Ratio of intensities of two waves is 16:9. If they produce interference then ratio of maximum and minimum will be:
A)
4: 3
done
clear
B)
49:1
done
clear
C)
64 : 27
done
clear
D)
81: 49
done
clear
View Answer play_arrow
Two cells of \[\operatorname{emf} {{E}_{1}} and {{E}_{2}} \left( {{E}_{1}}> {{E}_{2}} \right)\]are connected as shown in figure.
When a potentiometer is connected between A and B, then the balancing length of the potentiometer wire is 300cm. On connecting the same potentiometer between A and C, the balancing length is 100cm. The ratio\[\frac{{{\operatorname{E}}_{1}}}{{{\operatorname{E}}_{2}}}\] is:
A)
3:1
done
clear
B)
1:3
done
clear
C)
2:3
done
clear
D)
3:2
done
clear
View Answer play_arrow
A radioactive nucleus of mass M emits a photon of frequency d and the nucleus recoils. The recoil energy will be:
A)
\[\frac{{{h}^{2}}{{v}^{2}}}{2M{{c}^{2}}}\]
done
clear
B)
Zero
done
clear
C)
hv
done
clear
D)
\[{{\operatorname{Mc}}^{2}}-hv\]
done
clear
View Answer play_arrow
An ideal gas after going through a series of four thermodynamic states in order, reaches the initial state again (cyclic process). The amounts of heat and work involved in these states are:
\[{{\operatorname{Q}}_{1}}=6000J, {{Q}_{2}}=-5500J\] \[{{\operatorname{Q}}_{3}} =-3000J, {{Q}_{4}}= 3500J\] \[{{\operatorname{W}}_{1}} = 2500J, {{W}_{2}} = -1000J\] \[{{\operatorname{W}}_{3}}=1200J,{{W}_{4}}=xJ\] The ratio of net work done by the gas to the total heat absorbed by the gas is n. The value of x and n are nearly.
A)
500, 7.5%
done
clear
B)
700, 10.5%
done
clear
C)
1000, 21%
done
clear
D)
1500,15%
done
clear
View Answer play_arrow
A ball is projected horizontally with a velocity of 4ir/s from the top of a tower. The velocity of the ball after 0.7 sec is:
A)
1 m/s
done
clear
B)
10 m/s
done
clear
C)
8 m/s
done
clear
D)
3 m/s
done
clear
View Answer play_arrow
The escape velocity for the earth is \[{{\operatorname{V}}_{e}}\]The escape velocity for a planet whose radius is\[\frac{{{1}^{th}}}{4}\] the radius of earth and mass is half that of the earth is:
A)
\[\frac{{{\operatorname{V}}_{e}}}{\sqrt{2}}\]
done
clear
B)
\[\sqrt{2}{{\operatorname{V}}_{e}}\]
done
clear
C)
\[2{{\operatorname{V}}_{e}}\]
done
clear
D)
\[\frac{{{\operatorname{V}}_{e}}}{2}\]
done
clear
View Answer play_arrow
A block of mass 10 kg is moving in x-direction with a constant speed of 10m/s. It is subjected to a retarding force F = - 0.1 xJ/m during its travel from x = 20m to x = 30m. It is final kinetic energy will be:
A)
475J
done
clear
B)
450J
done
clear
C)
275J
done
clear
D)
250J
done
clear
View Answer play_arrow
The frequency of the first overtone of a closed pipe of length \[{{l}_{1}}\] is equal to that of the first overtone of an open pipe of length I. The ratio of their lengths \[\left( {{l}_{1}}:{{l}_{2}} \right)\]is:
A)
2:3
done
clear
B)
4:5
done
clear
C)
3:5
done
clear
D)
3:4
done
clear
View Answer play_arrow
A wire of resistance \[{{\operatorname{R}}_{o}}\]is elongated 'n' fold to make a new uniform wire. The resistance of new wire is:
A)
\[{{\operatorname{nR}}_{o}}\]
done
clear
B)
\[{{\operatorname{n}}^{2}}{{R}_{o}}\]
done
clear
C)
\[2{{\operatorname{nR}}_{o}}\]
done
clear
D)
\[2{{n}^{2}}{{R}_{o}}\]
done
clear
View Answer play_arrow
A particle moving along x-axis has acceleration f, at time t, given by\[\operatorname{f} = {{f}_{o}}\left[ 1-\frac{t}{T} \right]\], where \[{{\operatorname{f}}_{o}}\]and T are constants. The particle at t = 0 has zero velocity. In the time interval between t = 0 and the instant when f = 0, the particle's velocity \[\left( {{v}_{x}} \right)\] is:
A)
\[{{\operatorname{f}}_{o}}T\]
done
clear
B)
\[{{\operatorname{f}}_{o}}{{T}^{2}}\]
done
clear
C)
\[{{\operatorname{f}}_{o}}^{2}{{T}^{2}}\]
done
clear
D)
\[\frac{{{\operatorname{f}}_{o}}T}{2}\]
done
clear
View Answer play_arrow
If for Hydrogen \[{{\operatorname{C}}_{p}}- {{C}_{v}}\]= m and for Nitrogen \[{{\operatorname{C}}_{p}}- {{C}_{v}}\] = n where,\[{{\operatorname{C}}_{p}}\,and {{C}_{v}}\]refer to specific heat per unit mass respectively at constant pressure and constant volume, the relation between m and n is: [molar mass of Hydrogen = 2 and molar mass of\[{{\operatorname{N}}_{2}} = 28\]]
A)
n = 14m
done
clear
B)
n = 7m
done
clear
C)
m = 7n
done
clear
D)
m = 14n
done
clear
View Answer play_arrow
Light from two coherent sources of the same amplitude A and wavelength \[\lambda \] illuminates the screen. The intensity of the central maxima is L. If the sources were incoherent, the intensity at the same point will be:
A)
\[4{{I}_{o}}\]
done
clear
B)
\[2{{I}_{o}}\]
done
clear
C)
\[{{I}_{o}}\]
done
clear
D)
\[\frac{{{I}_{o}}}{2}\]
done
clear
View Answer play_arrow
The area of the acceleration-displacement curve of a body gives:
A)
Impulse
done
clear
B)
Change in kinetic energy per unit mass
done
clear
C)
Change in momentum per unit mass
done
clear
D)
Total change in energy
done
clear
View Answer play_arrow
An electric motor operates on a 50V supply and a current of 12A. If the efficiency of the motor is 30%, what is the resistance of the winding of the motor?
A)
\[6\Omega \]
done
clear
B)
\[4\Omega \]
done
clear
C)
\[2.9\Omega \]
done
clear
D)
\[3.1\Omega \]
done
clear
View Answer play_arrow
A glass has refractive index \[\frac{3}{2}\] and water has refractive index \[\frac{4}{3}\] if the speed of light in glass is \[2\times 1{{0}^{8}}m/s\].
A)
\[1.5\times 1{{0}^{6}}\]
done
clear
B)
\[1.78\times 1{{0}^{8}}\]
done
clear
C)
\[2.25\times 1{{0}^{8}}\]
done
clear
D)
\[2.67\times 1{{0}^{8}}\]
done
clear
View Answer play_arrow
A body is moving along a rough horizontal surface with an initial velocity 6m/s. If the body comes to rest after travelling a distance 9m. Then the coefficient of sliding friction will be:
A)
0.4
done
clear
B)
0.2
done
clear
C)
0.6
done
clear
D)
0.8
done
clear
View Answer play_arrow
A \[2\mu F\] condenser is charged upto 200V and then battery is removed. On combining this with another uncharged condenser in parallel, the potential difference between two plates are found to be 40V. The capacity of second condenser is:
A)
\[2\mu F\]
done
clear
B)
\[4\mu F\]
done
clear
C)
\[8\mu F\]
done
clear
D)
\[16\mu F\]
done
clear
View Answer play_arrow
A straight wire conductor of length of 0.4m is moving with a speed of 7m/s perpendicular to a magnetic field B of intensity\[0.9 Wb/{{m}^{2}}\]. The induced emf across the conductor is:
A)
2.52V
done
clear
B)
25.2V
done
clear
C)
5.26V
done
clear
D)
1.26V
done
clear
View Answer play_arrow
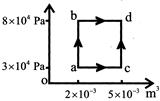
A thermodynamic process is shown in figure in process ab, 600J of heat is added, and in process bd 200J of heat is added. The total heat added in process acd is:
A)
550J
done
clear
B)
650J
done
clear
C)
750J
done
clear
D)
850J
done
clear
View Answer play_arrow
A rigid body rotates about a fixed axis with variable angular velocity equal to (a-bt) at time t where a and b are constants. The angle through which it rotates before it comes to rest is:
A)
\[\frac{{{a}^{2}}}{b}\]
done
clear
B)
\[\frac{{{a}^{2}}}{2b}\]
done
clear
C)
\[\frac{{{a}^{2}}}{4b}\]
done
clear
D)
\[\frac{{{a}^{2}}}{2b}\]
done
clear
View Answer play_arrow
Two opposite and equal charge \[4 \times {{10}^{-8}}\]when placed\[2 \times {{10}^{-2}}\]cm away, form a dipole. If this dipole is placed in an external electric field\[4 \times {{10}^{8}}\]N/C, the value of maximum torque and the work done in rotating it through \[180{}^\circ \] will be:
A)
\[64\times {{10}^{-4}}\operatorname{N}-m and 64\times 1{{0}^{-4}}\operatorname{J}\]
done
clear
B)
\[64\times {{10}^{-4}}\operatorname{N}-m and 32\times 1{{0}^{-4}}\operatorname{J}\]
done
clear
C)
\[32\times {{10}^{-4}}\operatorname{N}-m and 32\times 1{{0}^{-4}}\operatorname{J}\]
done
clear
D)
\[32\times {{10}^{-4}}\operatorname{N}-m and 64\times 1{{0}^{-4}}\operatorname{J}\]
done
clear
View Answer play_arrow
A uniform rope of length \[\ell \] lies on a table. If the coefficient of friction is u, then the maximum length \[{{\ell }_{1}}\] of the part of this rope which can overhang from the edge of the table without slicing down is:
A)
\[\frac{\ell }{\mu }\]
done
clear
B)
\[\frac{\ell }{\mu +1}\]
done
clear
C)
\[\frac{\mu \ell }{\mu +1}\]
done
clear
D)
\[\frac{\mu \ell }{\mu -1}\]
done
clear
View Answer play_arrow
A capacitor of \[20\mu F\] charged upto 500V is connected in parallel with another capacitor of \[10\mu F\]which is charged upto 200V. The common potential is:
A)
250V
done
clear
B)
300V
done
clear
C)
400V
done
clear
D)
600V
done
clear
View Answer play_arrow
A body of mass m rests on a horizontal floor, with which it has a coefficient of static friction u. It is desired to make the body move by applying the minimum possible force F. the magnitude of F is:
A)
\[\mu \operatorname{mg}\]
done
clear
B)
\[\sqrt{\frac{1+{{\mu }^{2}}}{\mu }}\operatorname{mg}\]
done
clear
C)
\[\mu \sqrt{1+{{\mu }^{2}}}\operatorname{mg}\]
done
clear
D)
\[\frac{\mu \operatorname{mg}}{\sqrt{1+{{\mu }^{2}}}}\]
done
clear
View Answer play_arrow
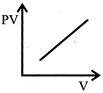
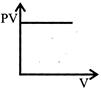
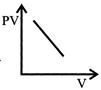
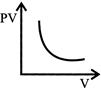
Which one of the following graphs represents the behavior of an ideal gas at constant temperature?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
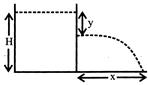
A cylindrical vessel is filled with a liquid up to a height H. A small hole is made in the vessel at a distance y below the liquid surface as shown in figure. The liquid emerging from the hole strike the ground at distance x:
A)
x is equal, if depth = y or H - y
done
clear
B)
x is maximum for \[\operatorname{y}=\frac{H}{2}\]
done
clear
C)
a & b correct
done
clear
D)
None
done
clear
View Answer play_arrow
A block released from rest from the top of a smooth inclined plane of angle \[{{\theta }_{1}}\] reaches the bottom in time t\[{{\operatorname{t}}_{1}}\]. The same block released from rest from the top of another smooth inclined plane of angle\[{{\theta }_{2}}\], reached the bottom in time\[{{\operatorname{t}}_{2}}\]. If the two inclined planes have the same height, the relation between\[{{\operatorname{t}}_{1}} and {{t}_{2}}\] is:
A)
\[\frac{{{t}_{2}}}{{{\operatorname{t}}_{1}}}=1\]
done
clear
B)
\[\frac{{{t}_{2}}}{{{\operatorname{t}}_{1}}}={{\left[ \frac{\sin {{\theta }_{1}}}{\sin {{\theta }_{2}}} \right]}^{{}^{1}/{}_{2}}}\]
done
clear
C)
\[\frac{{{t}_{2}}}{{{\operatorname{t}}_{1}}}=\frac{\sin {{\theta }_{1}}}{\sin {{\theta }_{2}}}\]
done
clear
D)
\[\frac{{{t}_{2}}}{{{\operatorname{t}}_{1}}}=\frac{{{\sin }^{2}}{{\theta }_{1}}}{{{\sin }^{2}}{{\theta }_{2}}}\]
done
clear
View Answer play_arrow
The weight of water molecular present ir a drop of water (volume 0.0018 ml) at room temperature is:
A)
\[6.023\times {{10}^{19}}\]
done
clear
B)
\[1.084\times {{10}^{18}}\]
done
clear
C)
\[4.84\times {{10}^{17}}\]
done
clear
D)
\[6.023\times {{10}^{23}}\]
done
clear
View Answer play_arrow
The wavelength of which series lie towards the ultraviolet:
A)
Lymann
done
clear
B)
Balmer
done
clear
C)
Paschen
done
clear
D)
None
done
clear
View Answer play_arrow
Principal, azimuthal and magnetic quantum numbers are respectively reacted to:
A)
Size, orientation and shape
done
clear
B)
Size, shape and orientation
done
clear
C)
Shape, size and orientation
done
clear
D)
None
done
clear
View Answer play_arrow
Which of the following transitions involves maximum amount of energy?
A)
\[{{\operatorname{M}}^{-}} \left( g \right) \to M \left( g \right)\]
done
clear
B)
\[\operatorname{M} \left( g \right) \to {{M}^{-}} \left( g \right)\]
done
clear
C)
\[{{\operatorname{M}}^{+}} \left( g \right) \to {{M}^{2+}} \left( g \right)\]
done
clear
D)
\[{{\operatorname{M}}^{2+}} \left( g \right) \to {{M}^{3+}} \left( g \right)\]
done
clear
View Answer play_arrow
Arrange the following in order of decreasing bond angle, giving reason: \[{{\operatorname{NO}}_{2}}, N{{O}^{+}}_{2}, N{{O}^{-}}_{2}\]
A)
\[{{\operatorname{NO}}^{+}}_{2}> N{{O}_{2}}> N{{O}^{-}}_{2}\]
done
clear
B)
\[{{\operatorname{NO}}^{-}}_{2}> N{{O}_{2}}> N{{O}^{+}}_{2}\]
done
clear
C)
\[{{\operatorname{NO}}_{2}}> N{{O}_{2}}^{-}>N{{O}^{+}}_{2}\]
done
clear
D)
\[{{\operatorname{NO}}_{2}}> N{{O}_{2}}^{-}>N{{O}^{+}}_{2}\]
done
clear
View Answer play_arrow
Equal weights of ethane and hydrogen are mixed in an empty container at\[25{}^\circ C\]. The fraction of the total pressure exerted by hydrogen is:
A)
1:2
done
clear
B)
1:1
done
clear
C)
1: 16
done
clear
D)
15:16
done
clear
View Answer play_arrow
The number of unpaired electrons in a paramagnetic diatomic molecule of an element with atomic number 16 is:
A)
4
done
clear
B)
1
done
clear
C)
2
done
clear
D)
3
done
clear
View Answer play_arrow
The entropy change in the fusion of one mole of a solid melting at \[27{}^\circ C\] (latent heat of fusion, 2930 J\[{{\operatorname{mol}}^{-1}}\]) is:
A)
\[10.73 J{{K}^{-1}} mo{{l}^{-1}}\]
done
clear
B)
\[2930 J{{K}^{-1}} mo{{l}^{-1}}\]
done
clear
C)
\[108.5 J{{K}^{-1}} mo{{l}^{-1}}\]
done
clear
D)
\[9.77 J{{K}^{-1}} mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
The correct order regarding the electronegativity of hybrid orbitals of carbon is:
A)
\[\operatorname{sp} < s{{p}^{2}} < sp\]
done
clear
B)
\[\operatorname{sp} < s{{p}^{2}} > s{{p}^{3}}\]
done
clear
C)
\[\operatorname{sp} > s{{p}^{2}} < s{{p}^{3}}\]
done
clear
D)
\[\operatorname{sp} > s{{p}^{2}} > s{{p}^{3}}\]
done
clear
View Answer play_arrow
The ONO bond angle is maximum in:
A)
\[{{\operatorname{NO}}_{3}}^{-}\]
done
clear
B)
\[{{\operatorname{NO}}_{2}}^{-}\]
done
clear
C)
\[{{\operatorname{NO}}_{2}}\]
done
clear
D)
\[{{\operatorname{NO}}_{2}}^{+}\]
done
clear
View Answer play_arrow
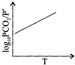
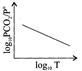
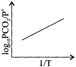
For the chemical equilibrium, \[{{\operatorname{CaCO}}_{3}} \left( s \right)\rightleftharpoons CaO\left( s \right)+C{{O}_{2}} \left( g \right)\] \[\Delta {{H}^{o}}_{r}\]Can be determined from which of the following plots?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The pH of a \[{{10}^{-8}}M\]solution of HCl in water is:
A)
8
done
clear
B)
-8
done
clear
C)
Between 7 and 8
done
clear
D)
Between 6 and 7
done
clear
View Answer play_arrow
The oxidation state of chromium in the final product formed by the reaction between KI and acidified potassium dichromate solution is:
A)
+4
done
clear
B)
+6
done
clear
C)
+2
done
clear
D)
+3
done
clear
View Answer play_arrow
Which of the following pairs of substances on reaction will not evolve H gas?
A)
Fe and \[{{\operatorname{H}}_{2}}S{{O}_{4}}\](aqueous)
done
clear
B)
Copper and HC1 (aquecous)
done
clear
C)
Sodium and ethyl alcohol
done
clear
D)
Iron and steam
done
clear
View Answer play_arrow
The paramagnetic species is:
A)
\[{{\operatorname{KO}}_{2}}\]
done
clear
B)
\[{{\operatorname{SiO}}_{2}}\]
done
clear
C)
\[Ti{{O}_{2}}\]
done
clear
D)
\[{{\operatorname{BaO}}_{2}}\]
done
clear
View Answer play_arrow
The IUPAC name of
is:
A)
Cyclohe xanone
done
clear
B)
Cyclohexylmethanone
done
clear
C)
Oxycyclohexene
done
clear
D)
Cyclohexyli denemethanone
done
clear
View Answer play_arrow
Which of the following Is not a protonic acid?
A)
\[\operatorname{B}{{\left( OH \right)}_{3}}\]
done
clear
B)
\[\operatorname{PO}{{\left( OH \right)}_{3}}\]
done
clear
C)
\[\operatorname{SO}{{\left( OH \right)}_{3}}\]
done
clear
D)
\[{{\operatorname{SO}}_{2}}{{\left( OH \right)}_{2}}\]
done
clear
View Answer play_arrow
In the compound \[{{\operatorname{CH}}_{2}} =CH-C{{H}_{2}}-C{{H}_{2}}-C=CH,\,\,the {{C}_{2}}-{{C}_{3}}\]bond is of the type:
A)
\[\operatorname{sp}-s{{p}^{2}}\]
done
clear
B)
\[{{\operatorname{sp}}^{3}}-s{{p}^{3}}\]
done
clear
C)
\[\operatorname{sp}-s{{p}^{3}}\]
done
clear
D)
\[{{\operatorname{sp}}^{2}}-s{{p}^{3}}\]
done
clear
View Answer play_arrow
In chlorination of benzene, the reactive species is:
A)
\[{{\operatorname{Cl}}^{+}}\]
done
clear
B)
\[{{\operatorname{Cl}}^{-}}\]
done
clear
C)
\[{{\operatorname{Cl}}_{2}}\]
done
clear
D)
\[{{\operatorname{Cl}}_{2}}^{-}\]
done
clear
View Answer play_arrow
Which of the following compounds possesses the C-H bond with the lowest bond dissociation energy?
A)
Toluene
done
clear
B)
Benzene
done
clear
C)
n-Pentane
done
clear
D)
2, 2-dimethylpropane
done
clear
View Answer play_arrow
When 2- butyne is treated with dil. \[{{\operatorname{H}}_{2}}S{{O}_{4}}/\]\[{{\operatorname{HgSO}}_{4}}\], the product formed is:
A)
Butanol-1
done
clear
B)
Butanol-2
done
clear
C)
2 - Butanone
done
clear
D)
Butanol
done
clear
View Answer play_arrow
Which of the following has greatest - affinity for haemoglobin?
A)
CO
done
clear
B)
NO
done
clear
C)
\[{{\operatorname{O}}_{2}}\]
done
clear
D)
\[C{{\operatorname{O}}_{2}}\]
done
clear
View Answer play_arrow
The fraction of the total volume occupied by the atoms present in a simple cube is:
A)
\[\frac{\pi }{4}\]
done
clear
B)
\[\frac{\pi }{6}\]
done
clear
C)
\[\frac{\pi }{3\sqrt{2}}\]
done
clear
D)
\[\frac{\pi }{4\sqrt{2}}\]
done
clear
View Answer play_arrow
The material used in the solar cells contains:
A)
Cs
done
clear
B)
Si
done
clear
C)
Sn
done
clear
D)
Ti
done
clear
View Answer play_arrow
A 500g tooth paste sample has 0.2g fluoride concentration. What is the concentration of fluorine in terms of ppm lavel?
A)
250
done
clear
B)
200
done
clear
C)
400
done
clear
D)
1000
done
clear
View Answer play_arrow
A solution of acetone in ethanol:
A)
Behaves like a near ideal solution
done
clear
B)
Obeys Raoulfs law
done
clear
C)
Shows a negative deviation from Raoult's law
done
clear
D)
Shows a positive deviation from Raoult's law
done
clear
View Answer play_arrow
The rate constant of a chemical reaction has units\[\operatorname{L} mo{{l}^{-1}} {{S}^{-1}}\]. Order of the reaction will be:
A)
O
done
clear
B)
1
done
clear
C)
2
done
clear
D)
3
done
clear
View Answer play_arrow
When a lead storage battery is discharged
A)
\[{{\operatorname{SO}}_{2}} is evolved\]
done
clear
B)
Lead sulphate is consumed
done
clear
C)
Lead is formed
done
clear
D)
Sulphuric acid is consumed
done
clear
View Answer play_arrow
The temperature coefficient of most of the reaction lies between:
A)
1 and 3
done
clear
B)
2 and 3
done
clear
C)
1 and 4
done
clear
D)
2 and 4
done
clear
View Answer play_arrow
Alums purify muddy water by:
A)
Dialysis
done
clear
B)
Coagulation
done
clear
C)
Adsorption
done
clear
D)
Forming a true solution
done
clear
View Answer play_arrow
Which of the following has least bond angle?
A)
\[{{\operatorname{H}}_{2}}O\]
done
clear
B)
\[{{\operatorname{H}}_{2}}S\]
done
clear
C)
\[{{\operatorname{H}}_{2}}Se\]
done
clear
D)
\[{{\operatorname{H}}_{2}}Te\]
done
clear
View Answer play_arrow
Heating mixture of \[{{\operatorname{Cu}}_{2}}O and C{{u}_{2}}S\]will give:
A)
\[\operatorname{Cu} + S{{O}_{2}}\]
done
clear
B)
\[\operatorname{Cu} + S{{O}_{3}}\]
done
clear
C)
\[\operatorname{CuO} + CuS\]
done
clear
D)
\[{{\operatorname{Cu}}_{2}}S{{O}_{3}}\]
done
clear
View Answer play_arrow
\[{{\operatorname{F}}_{2}}\]is formed by reaction of\[{{K}_{2}}Mn{{F}_{6}}\] with
A)
\[{{\operatorname{SbF}}_{3}}\]
done
clear
B)
\[{{\operatorname{MnF}}_{3}}\]
done
clear
C)
\[{{\operatorname{kSbF}}_{6}}\]
done
clear
D)
\[{{\operatorname{MnF}}_{5}}\]
done
clear
View Answer play_arrow
Least paramagnetic property is shown by:
A)
Fe
done
clear
B)
Mn
done
clear
C)
Ni
done
clear
D)
Cu
done
clear
View Answer play_arrow
In the compound, lithiumtetrahydrido aluminate, the ligand is:
A)
\[{{\operatorname{H}}^{+}}\]
done
clear
B)
H
done
clear
C)
\[{{\operatorname{H}}^{-}}\]
done
clear
D)
None
done
clear
View Answer play_arrow
Which of the following compounds shows optical isomerism?
A)
\[{{\left[ \operatorname{Cu}{{\left( {{\operatorname{NH}}_{3}} \right)}_{4}} \right]}^{2+}}\]
done
clear
B)
\[{{\left[ Zn{{\operatorname{Cl}}_{4}} \right]}^{2-}}\]
done
clear
C)
\[{{\left[ \operatorname{Cr}\left( {{\operatorname{C}}_{2}}{{\operatorname{O}}_{3}} \right) \right]}^{3-}}\]
done
clear
D)
\[{{\left[ \operatorname{Co}{{\left( \operatorname{CN} \right)}_{6}} \right]}^{3-}}\]
done
clear
View Answer play_arrow
The addition of \[\operatorname{HBr}\]is easiest with:
A)
\[{{\operatorname{CH}}_{2}} = CHCl\]
done
clear
B)
\[\operatorname{ClCH} = CHCl\]
done
clear
C)
\[{{\operatorname{CH}}_{3}}- CH = C{{H}_{2}}\]
done
clear
D)
\[{{(C{{H}_{3}})}_{2}} C = CH,\]
done
clear
View Answer play_arrow
In the following reaction, \[{{\operatorname{C}}_{2}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}+4H\xrightarrow[\operatorname{HI}]{\operatorname{Red}\,{{p}^{+}}}2X+{{H}_{2}}o, Xis:\]B
A)
Ethane
done
clear
B)
Ethylene
done
clear
C)
Butane
done
clear
D)
Propane
done
clear
View Answer play_arrow
With which of the following reagents carbonyl compound shows addition cun elimination reaction?
A)
\[{{\operatorname{PCl}}_{5}}\]
done
clear
B)
Brady's reagent
done
clear
C)
HCN
done
clear
D)
All of these
done
clear
View Answer play_arrow
Identify the product Y in the following reaction:
A)
Pentane
done
clear
B)
cyclobutane
done
clear
C)
Cyclopentane
done
clear
D)
cyclopentanone
done
clear
View Answer play_arrow
Identify the product C in the series: \[{{\operatorname{CH}}_{3}}CN \xrightarrow{Na/{{C}_{2}}{{H}_{5}}OH}A\xrightarrow{HNO2}B\xrightarrow{CU/573}C\]
A)
\[{{\operatorname{CH}}_{3}}COOH\]
done
clear
B)
\[{{\operatorname{CH}}_{3}}{{\operatorname{CH}}_{2}}NHOH\]
done
clear
C)
\[{{\operatorname{CH}}_{3}}CON{{H}_{2}}\]
done
clear
D)
\[{{\operatorname{CH}}_{3}}CHO\]
done
clear
View Answer play_arrow
The functional group which is found in amino acid is:
A)
-COOH
done
clear
B)
\[-N{{H}_{2}}\]
done
clear
C)
\[-C{{H}_{3}}\]
done
clear
D)
Both [a] and [b]
done
clear
View Answer play_arrow
\[{{\operatorname{F}}_{2}}C = C{{F}_{2}}\]is a monomer of:
A)
Teflon
done
clear
B)
Glyptal
done
clear
C)
Nylon-6
done
clear
D)
Buna - 5
done
clear
View Answer play_arrow
A broad spectrum antibiotic is:
A)
Paracetamol
done
clear
B)
Pencillin
done
clear
C)
Aspirin
done
clear
D)
chloramphenicol
done
clear
View Answer play_arrow
Which is the strongest base in aqueous solution?
A)
Methylamine
done
clear
B)
Trimethylamine
done
clear
C)
Aniline
done
clear
D)
Dimethylamine
done
clear
View Answer play_arrow
The scientific name of man is Homo sapiens, Homo denotes the name of:
A)
Family
done
clear
B)
Genus
done
clear
C)
Species
done
clear
D)
Common name
done
clear
View Answer play_arrow
The organisms are given scientific name so that:
A)
The family to which it belongs can be traced
done
clear
B)
Another biologist does not give the same name
done
clear
C)
Species is recognised by the same name by all taxonomists
done
clear
D)
Diversity of organisms could be increased
done
clear
View Answer play_arrow
A flower that has only one stamen is called:
A)
Unisexual flower
done
clear
B)
Bisexual flower
done
clear
C)
Complete flower
done
clear
D)
Neuter flower
done
clear
View Answer play_arrow
The scientific name should be written in which language:
A)
English
done
clear
B)
Latin
done
clear
C)
Common
done
clear
D)
Non of these
done
clear
View Answer play_arrow
A scion is grafted to a stock the quality of fruits produced will be determined by the gene type of:
A)
Stock
done
clear
B)
Scion
done
clear
C)
Both a & b
done
clear
D)
Neither a nor b
done
clear
View Answer play_arrow
In a culture there is a mixture of haploid and diploid cells. It must be because of:
A)
Generative cells
done
clear
B)
Anther wall
done
clear
C)
Vegetative cells
done
clear
D)
Pollen cells
done
clear
View Answer play_arrow
Arrangement of nuclei in normal dicot embryo sac is:
A)
3+3+2
done
clear
B)
2+4+2
done
clear
C)
3+2+3
done
clear
D)
2+3+3
done
clear
View Answer play_arrow
Unisexuality of flowers prevents:
A)
Autogamy and geitonogamy
done
clear
B)
Both autogamy and geitonogamy
done
clear
C)
Autogamy but not geitonogamy
done
clear
D)
Geitonogamy but not xenogamy
done
clear
View Answer play_arrow
The multicellular decomposes belongs to kingdom:
A)
Monera
done
clear
B)
Protista
done
clear
C)
Fungi
done
clear
D)
Plantae
done
clear
View Answer play_arrow
Which of the following is an amphibian of plant kingdom:
A)
Red algae
done
clear
B)
Fungi
done
clear
C)
Moss
done
clear
D)
Pines
done
clear
View Answer play_arrow
Chilgoza are the seeds of:
A)
A monocot
done
clear
B)
A dicot
done
clear
C)
Conifer
done
clear
D)
False fruit
done
clear
View Answer play_arrow
Archentron is found in:
A)
Morula
done
clear
B)
Blastula
done
clear
C)
Gastrula
done
clear
D)
Blastodermic vesicle
done
clear
View Answer play_arrow
Segmentation cavity is another name for:
A)
Cleavage
done
clear
B)
Blastocoel
done
clear
C)
Archenteron
done
clear
D)
Body cavity
done
clear
View Answer play_arrow
Acrosome of sperms is/are modified:
A)
Lysosome
done
clear
B)
ER
done
clear
C)
Golgi complex
done
clear
D)
Mitochondria
done
clear
View Answer play_arrow
The function of cork cambium is to produce:
A)
Only cork
done
clear
B)
Only secondary cortex
done
clear
C)
Only cambium
done
clear
D)
Both cork and secondary cortex
done
clear
View Answer play_arrow
A non-medullated nerve fibre does not have:
A)
Schwann cells
done
clear
B)
Neurilemma
done
clear
C)
NissFs granvles
done
clear
D)
Nodes of Ranvier
done
clear
View Answer play_arrow
Junction that help to stop substances from leaking across a tissue:
A)
Gap junction
done
clear
B)
Ampullary - isthamic junction
done
clear
C)
Tight junction
done
clear
D)
Adipose tissue
done
clear
View Answer play_arrow
Mammary glands are modified:
A)
Sebacious gland
done
clear
B)
Sweat gland
done
clear
C)
Cutaneous gland
done
clear
D)
Scent gland
done
clear
View Answer play_arrow
The 80S ribosome has two sub units which are:
A)
30S and 50S
done
clear
B)
40S and 60S
done
clear
C)
40S and 40S
done
clear
D)
20S and 60S
done
clear
View Answer play_arrow
Which of the following birth control measures can be considered as the safest.
A)
The rhythm method
done
clear
B)
The use of physical barrier
done
clear
C)
Termination of unwanted pregnancy
done
clear
D)
Sterilisation techniques
done
clear
View Answer play_arrow
Population increases in ratio while food production increase in __ ratio:
A)
Geometric, artithmetic
done
clear
B)
Geometric, logical
done
clear
C)
Arithmetic, analoy
done
clear
D)
Logical, analog
done
clear
View Answer play_arrow
Cilia & flagells both have:
A)
9 + 2 arrangement
done
clear
B)
9 + 3 arrangement
done
clear
C)
Symmetrical beats
done
clear
D)
100 |im length
done
clear
View Answer play_arrow
The width of a DNA molecule is:
A)
\[234 \overset{\operatorname{o}}{\mathop{A}}\,\]
done
clear
B)
\[23.4 \overset{\operatorname{o}}{\mathop{A}}\,\]
done
clear
C)
\[0.34 \overset{\operatorname{o}}{\mathop{A}}\,\]
done
clear
D)
\[20 \overset{\operatorname{o}}{\mathop{A}}\,\]
done
clear
View Answer play_arrow
Which class of enzyme show these type of reaction:
A)
Transferase
done
clear
B)
Lyases
done
clear
C)
Ligases
done
clear
D)
Oxidoreductases
done
clear
View Answer play_arrow
Turner's syndrome is:
A)
XO
done
clear
B)
XXY
done
clear
C)
XXX
done
clear
D)
XYY
done
clear
View Answer play_arrow
A test cross is:
A)
\[\operatorname{Ww} X ww\]
done
clear
B)
\[\operatorname{WW}\,\,X\,\,ww\]
done
clear
C)
\[\operatorname{Ww} X Ww\]
done
clear
D)
\[\operatorname{ww} X ww\]
done
clear
View Answer play_arrow
The bivalent are attached to the spindle at:
A)
Centriole
done
clear
B)
Centromere
done
clear
C)
Chromosomere
done
clear
D)
Chromatids
done
clear
View Answer play_arrow
50% of recombinants in linked gene occur when:
A)
The genes are present on different chromosome
done
clear
B)
The genes are preset for apart on the chromosome
done
clear
C)
The genes are present close together on the same chromosome
done
clear
D)
The genes do not undergo crossing over
done
clear
View Answer play_arrow
Pure water has been given an osmotic potential of:
A)
1
done
clear
B)
-1
done
clear
C)
Zero
done
clear
D)
-10
done
clear
View Answer play_arrow
The water lost during transpiration:
A)
Contains minerals
done
clear
B)
Contains salts
done
clear
C)
Is pure water
done
clear
D)
Contains dissolved nutrients
done
clear
View Answer play_arrow
The water left in the dry soils is generally the:
A)
Gravitational water
done
clear
B)
Hygroscopic water
done
clear
C)
Capillary water
done
clear
D)
Water in water
done
clear
View Answer play_arrow
The genetic materialised in one-gene-one- enzyme hypothesis was:
A)
Diplococcus pneumonia
done
clear
B)
Neurospora
done
clear
C)
E.coli
done
clear
D)
\[{{\operatorname{T}}_{2}} phage\]
done
clear
View Answer play_arrow
The condition stem crack of celery is due to deficiency of:
A)
Zinc
done
clear
B)
Copper
done
clear
C)
borum
done
clear
D)
Molybdnem
done
clear
View Answer play_arrow
The S - strain of bacteria Diplococcus pneumonia:
A)
Are not virulent
done
clear
B)
From rough colonies
done
clear
C)
Are capsulated
done
clear
D)
Are non - capsulated
done
clear
View Answer play_arrow
The first product of\[{{\operatorname{C}}_{4}}\]pathway is:
A)
Oxaloacetic acid
done
clear
B)
RuBP
done
clear
C)
PGA
done
clear
D)
PGAL
done
clear
View Answer play_arrow
A change in the wobble position of a gene means a change in the:
A)
First base of codon
done
clear
B)
Second base of codon
done
clear
C)
Third base of codon
done
clear
D)
Whole of codon
done
clear
View Answer play_arrow
The primary pigment molecule of PS I is:
A)
\[{{\operatorname{P}}_{700}}\]
done
clear
B)
\[{{\operatorname{P}}_{680}}\]
done
clear
C)
\[{{\operatorname{P}}_{670}}\]
done
clear
D)
\[\operatorname{Chl}\,\,B\]
done
clear
View Answer play_arrow
Which of the following scientist tested suggested that amino acid were very important for origin of life:
A)
Van Helmont
done
clear
B)
Oparin
done
clear
C)
Pasteur
done
clear
D)
Redi
done
clear
View Answer play_arrow
The final acceptor of electrons in ETC is:
A)
Oxygen
done
clear
B)
Cytochromes
done
clear
C)
Cytochrome C
done
clear
D)
Water
done
clear
View Answer play_arrow
What is another name of krebs cycle:
A)
ACA cycle
done
clear
B)
GCA cycle
done
clear
C)
TCA cycle
done
clear
D)
AGA cycle
done
clear
View Answer play_arrow
Which of the following is used as a weed killer?
A)
IBA
done
clear
B)
IAA
done
clear
C)
NAA
done
clear
D)
2,4-D
done
clear
View Answer play_arrow
Human appeared on earth in:
A)
Palaeozoic era
done
clear
B)
Mesozoic era
done
clear
C)
Cenozoic era
done
clear
D)
Jurassic era
done
clear
View Answer play_arrow
The flowering hormone 'florigen' is perceived by:
A)
Shoot apex
done
clear
B)
Leaves
done
clear
C)
Buds
done
clear
D)
Flowers
done
clear
View Answer play_arrow
DNA and RNA molecule differ in the following molecule:
A)
Phosphate
done
clear
B)
Sugar
done
clear
C)
Purines
done
clear
D)
Kinds of bonds
done
clear
View Answer play_arrow
Which of the following matched correctly:
A)
Apiculture - Bee
done
clear
B)
Pisiculture - Silk moth
done
clear
C)
Sericulture - Fish
done
clear
D)
Aquaculture - Mosquito
done
clear
View Answer play_arrow
mRNA is a polymer of:
A)
Ribonucleosides
done
clear
B)
Deoxy ribonucleosides,
done
clear
C)
Deoxy ribonucleotides
done
clear
D)
Ribonucleotides
done
clear
View Answer play_arrow
What is the full form of MTOCs:
A)
Microtubule organ centres
done
clear
B)
Microtubule oxygen centres
done
clear
C)
Microtubules organising centres
done
clear
D)
Microtubules oxytocin centres
done
clear
View Answer play_arrow
Ciliated epithelium is found in the lining of:
A)
Stomach
done
clear
B)
Oviduct
done
clear
C)
Salivary glands
done
clear
D)
Bowman's capsule
done
clear
View Answer play_arrow
Callus is:
A)
An organised mass of cells
done
clear
B)
Cells from the root tip
done
clear
C)
Rapidly dividing cells
done
clear
D)
An unorganised mass of cells
done
clear
View Answer play_arrow
Which of the following pair is not correctly matched?
A)
\[\operatorname{Vitamin} {{B}_{1}} - Beri Beri\]
done
clear
B)
\[\operatorname{Vitamin} {{B}_{5}}- Pellagra\]
done
clear
C)
\[\operatorname{Vitamin} {{B}_{12}}- Pernicious anaemia\]
done
clear
D)
\[\operatorname{Vitamin} {{B}_{6}}- Loss of appetite\]
done
clear
View Answer play_arrow
Which one of the following is correctly matched:
A)
UUA - Valine
done
clear
B)
CCE - Alanine
done
clear
C)
AUG - Cysteine
done
clear
D)
AAA - Lycine
done
clear
View Answer play_arrow
Who give one-gene-one-enzyme hypothesis:
A)
Darwin
done
clear
B)
Oparin & Halden
done
clear
C)
Beadle and tatom
done
clear
D)
Miller & urey
done
clear
View Answer play_arrow
The source for sugar for making of beer is:
A)
Grape juice
done
clear
B)
Fruit juice
done
clear
C)
Baoley grains
done
clear
D)
None of above
done
clear
View Answer play_arrow
Enterokinase activates:
A)
Pepsin
done
clear
B)
Trypsin
done
clear
C)
Chymotrypsin
done
clear
D)
Trypsinogen
done
clear
View Answer play_arrow
Which is true according to Chargaff's rule?
A)
A+G=T+C
done
clear
B)
A=C
done
clear
C)
G=T
done
clear
D)
\[\frac{A+T}{C++G} =1\]
done
clear
View Answer play_arrow
Respiratory control centre is present in:
A)
Thorax
done
clear
B)
Aortic arch
done
clear
C)
Medulla oblongata
done
clear
D)
Lungs
done
clear
View Answer play_arrow
\[{{\operatorname{CO}}_{2}}\]is mainly transported in our body as:
A)
Sodium bicarbonate
done
clear
B)
Carbonic acid in plasma
done
clear
C)
Carbaminohaemoglobin
done
clear
D)
Carboxyhaemoglobin
done
clear
View Answer play_arrow
Which of the following neem product used as insect repellent:
A)
Azadirachtin
done
clear
B)
Rotenone
done
clear
C)
Parathione
done
clear
D)
Endrin
done
clear
View Answer play_arrow
Most cultivated plants are:
A)
Autopolyploids
done
clear
B)
Allopolyploids
done
clear
C)
Aneuploids
done
clear
D)
Haploids
done
clear
View Answer play_arrow
Which of the following gas was absent on primitive earth:
A)
\[{{\operatorname{CO}}_{2}}\]
done
clear
B)
\[{{\operatorname{CH}}_{4}}\]
done
clear
C)
\[{{\operatorname{NH}}_{3}}\]
done
clear
D)
\[{{\operatorname{O}}_{2}}\]
done
clear
View Answer play_arrow
Bicuspid and tricuspid valves are closed during:
A)
Ventricular systole
done
clear
B)
Ventricular diastole
done
clear
C)
Artrial systole
done
clear
D)
Late joint diastole
done
clear
View Answer play_arrow
In Bt cotton, the Bt protein:
A)
Increases the protein content
done
clear
B)
Stops the larva to feed further
done
clear
C)
Stops egg laying of adult
done
clear
D)
Produces more cotton
done
clear
View Answer play_arrow
SA node is called the pacemaker of the heart because:
A)
It conducts the impulse from AV node
done
clear
B)
It initiate the cardiac impulse
done
clear
C)
It is present in the right atrium
done
clear
D)
It is made up of specialised muscle fibres
done
clear
View Answer play_arrow
Which of the following is used in bioware:
A)
A pathogen
done
clear
B)
Toxin from a pathogen
done
clear
C)
A delivery system for the bioweapon agent
done
clear
D)
All of the above
done
clear
View Answer play_arrow
The ascending limb of loop of Henie:
A)
Is impermeable to water
done
clear
B)
Has hypotonic filtrate
done
clear
C)
Both a & b
done
clear
D)
None of these
done
clear
View Answer play_arrow
The gene 'cry' is naturally present in:
A)
Bacillus thuringiensis
done
clear
B)
Brassica
done
clear
C)
Tissue culture
done
clear
D)
Spirulina
done
clear
View Answer play_arrow
More ADH means:
A)
Less permeable DCT
done
clear
B)
Descending limb
done
clear
C)
Cone. Urine
done
clear
D)
Fall in blood osmotic pressure
done
clear
View Answer play_arrow
A thin layer of ozone is present in:
A)
Troposphere
done
clear
B)
Stratosphere
done
clear
C)
Stratopause
done
clear
D)
Mesosphere
done
clear
View Answer play_arrow
The principle junction of loop of henie is:
A)
Reabsorption of \[{{\operatorname{Na}}^{+}}\] ion
done
clear
B)
Reabsorption of \[{{\operatorname{Cl}}^{-}}\]ion
done
clear
C)
To concentrate urine
done
clear
D)
Dilution of urine
done
clear
View Answer play_arrow
During summer stralification the upper layer of water is called?
A)
Thermocline
done
clear
B)
Ectotherms
done
clear
C)
homoiotherms
done
clear
D)
Thermoregulators
done
clear
View Answer play_arrow
SynoviaI joints are found at the:
A)
Elbow
done
clear
B)
Skull
done
clear
C)
Symphysis
done
clear
D)
Vertebrae
done
clear
View Answer play_arrow
Biopiracy is related to which of the following:
A)
Traditional knowledge
done
clear
B)
Biomolecules and related bio resources, gene isolated from bio resources
done
clear
C)
Bio resources
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Oligotrophic soils contain:
A)
Low nutrients
done
clear
B)
High nutrients
done
clear
C)
Less oxygen
done
clear
D)
High phosphorus
done
clear
View Answer play_arrow
The Z line is located:
A)
In M line
done
clear
B)
In H line
done
clear
C)
At the centre of I band
done
clear
D)
AT the centre of A band
done
clear
View Answer play_arrow
Total energy stored by green plants is called?
A)
NPD
done
clear
B)
GPP
done
clear
C)
Community productivity
done
clear
D)
Secondary productivity
done
clear
View Answer play_arrow
The number of sacral vertebrae in children are:
A)
1
done
clear
B)
3
done
clear
C)
4
done
clear
D)
5
done
clear
View Answer play_arrow
The IUCN red list gives information about:
A)
Threatened plants
done
clear
B)
Threatened animals
done
clear
C)
Threatened plants & animals
done
clear
D)
Extinct plant & animals only
done
clear
View Answer play_arrow
Ribs that are not attached to sternum are called?
A)
False ribs
done
clear
B)
Floating ribs
done
clear
C)
True ribs
done
clear
D)
Incomplete ribs
done
clear
View Answer play_arrow
Detritus food chain begins with:
A)
Dead organic matter
done
clear
B)
Detritus feeders
done
clear
C)
Earthworms
done
clear
D)
Soil bacteria
done
clear
View Answer play_arrow
Which of the following are pioneer organisms of primary succession on a xerach:
A)
Grasses
done
clear
B)
Lichen
done
clear
C)
Shrubs
done
clear
D)
Trees
done
clear
View Answer play_arrow
The region where the image is sharpest is known as:
A)
Blind spot
done
clear
B)
Rod region
done
clear
C)
Retina
done
clear
D)
Fovea
done
clear
View Answer play_arrow
The following is a 'hotspof region of India:
A)
North Himalayas
done
clear
B)
Western ghats
done
clear
C)
Peninsula region
done
clear
D)
Andaman & nicobar islands
done
clear
View Answer play_arrow
Hotspots are region on earth that:
A)
Have very hot climate
done
clear
B)
Are tropical region
done
clear
C)
Have rare species
done
clear
D)
Are rich in endemic species
done
clear
View Answer play_arrow
The depolarised membrane is:
A)
More permeable to \[{{\operatorname{Na}}^{+}}\]
done
clear
B)
Less permeable to \[{{\operatorname{K}}^{+}}\]
done
clear
C)
More permeable to both \[{{\operatorname{Na}}^{+}} \And {{K}^{+}}\]
done
clear
D)
Less permeable to both \[{{\operatorname{Na}}^{+}} \And {{K}^{+}}\]
done
clear
View Answer play_arrow
Amongest the best known grassland biomes are:
A)
Western ghats of India
done
clear
B)
'Praire' of North America
done
clear
C)
South Africa
done
clear
D)
Western Himalayas
done
clear
View Answer play_arrow
Minamata disease are caused by:
A)
Mercury poisoning
done
clear
B)
Lead poisoning
done
clear
C)
Cadmium
done
clear
D)
Fluorine
done
clear
View Answer play_arrow
Methaemoglobinemia is due to the following pollutant in drinking water.
A)
Nitrates
done
clear
B)
Fluoride
done
clear
C)
Arsenic
done
clear
D)
Mercury
done
clear
View Answer play_arrow
Sketetal fluorosis is caused due to:
A)
Nitrates
done
clear
B)
Stiff joints nerve
done
clear
C)
Harden joints
done
clear
D)
Hardened bones/stiff joints
done
clear
View Answer play_arrow
The number of teeth that grow once in the human life is
A)
4
done
clear
B)
12
done
clear
C)
20
done
clear
D)
28
done
clear
View Answer play_arrow
Radicle tip is derived from
A)
Suspensor
done
clear
B)
Proembryo
done
clear
C)
Basal cell
done
clear
D)
Hypophysis
done
clear
View Answer play_arrow