-
question_answer1)
Which molecule is not linear [CPMT 1994]
A)
\[Be{{F}_{2}}\] done
clear
B)
\[Be{{H}_{2}}\] done
clear
C)
\[C{{O}_{2}}\] done
clear
D)
\[{{H}_{2}}O\] done
clear
View Solution play_arrow
-
question_answer2)
The bond angle in water molecule is nearly or Directed bonds in water forms an angle of [NCERT 1980; EAMCET 1981; MNR 1983, 85; AIIMS 1982; CPMT 1989; MP PET 1994, 96; MP PET/PMT 1998]
A)
\[{{120}^{o}}\] done
clear
B)
\[{{180}^{o}}\] done
clear
C)
\[{{109}^{o}}28'\] done
clear
D)
\[{{104}^{o}}30'\] done
clear
View Solution play_arrow
-
question_answer3)
The central atom in a molecule is in \[s{{p}^{2}}\]hybrid state. The shape of molecule will be [MP PMT 1987; CBSE PMT 1989]
A)
Pyramidal done
clear
B)
Tetrahedral done
clear
C)
Octahedral done
clear
D)
Trigonal planar done
clear
View Solution play_arrow
-
question_answer4)
Which molecule is linear [MP PMT 1984; IIT 1982, 88; EAMCET 1993; CBSE PMT 1992; MP PET 1995; RPMT 1997]
A)
\[N{{O}_{2}}\] done
clear
B)
\[Cl{{O}_{2}}\] done
clear
C)
\[C{{O}_{2}}\] done
clear
D)
\[{{H}_{2}}S\] done
clear
View Solution play_arrow
-
question_answer5)
Which of the following molecules has trigonal planer geometry [CBSE PMT 2005]
A)
\[I{{F}_{3}}\] done
clear
B)
\[PC{{l}_{3}}\] done
clear
C)
\[N{{H}_{3}}\] done
clear
D)
\[B{{F}_{3}}\] done
clear
View Solution play_arrow
-
question_answer6)
A \[s{{p}^{3}}\] hybridized orbital contains [DPMT 1984; BHU 1985; CPMT 1976]
A)
\[\frac{1}{4}s-\]character done
clear
B)
\[\frac{1}{2}s-\]character done
clear
C)
\[\frac{2}{3}s-\]character done
clear
D)
\[\frac{3}{4}s-\]character done
clear
View Solution play_arrow
-
question_answer7)
Structure of ammonia is [MP PMT 1987, 89, 91; CPMT 1975, 82; RPMT 1999; JIPMER 2002]
A)
Trigonal done
clear
B)
Tetrahedral done
clear
C)
Pyramidal done
clear
D)
Trigonal pyramidal done
clear
View Solution play_arrow
-
question_answer8)
The bond angle in ethylene is [CPMT 1987]
A)
\[{{180}^{o}}\] done
clear
B)
\[{{120}^{o}}\] done
clear
C)
\[{{109}^{o}}\] done
clear
D)
\[{{90}^{o}}\] done
clear
View Solution play_arrow
-
question_answer9)
Compound formed by \[s{{p}^{3}}d\]hybridization will have structure [BHU 1982; RPMT 1999]
A)
Planar done
clear
B)
Pyramidal done
clear
C)
Angular done
clear
D)
Trigonal bipyramidal done
clear
View Solution play_arrow
-
question_answer10)
Which of the following formula does not correctly represent the bonding capacity of the atom involved [CBSE PMT 1990]
A)
\[\left[ H-\underset{\underset{H}{\mathop{|}}\,}{\overset{\overset{H}{\mathop{|}}\,}{\mathop{P}}}\,-H \right]\] done
clear
B)
C)
D)
View Solution play_arrow
-
question_answer11)
Which of the following statement is not correct [AIIMS 1983]
A)
Hybridization is the mixing of atomic orbitals prior to their combining into molecular orbitals done
clear
B)
\[s{{p}^{2}}\]hybrid orbitals are formed from two \[p\]atomic orbitals and one \[HCl\]atomic orbital done
clear
C)
\[{{d}^{2}}s{{p}^{3}}\]hybrid orbitals are directed towards the corners of a regular octahedron done
clear
D)
\[ds{{p}^{3}}\]hybrid orbitals are all at \[{{90}^{o}}\]to one another done
clear
View Solution play_arrow
-
question_answer12)
The mode of hybridisation of carbon in\[s{{p}^{2}}<sp<s{{p}^{3}}\]is [CPMT 1991]
A)
\[sp\] done
clear
B)
\[s{{p}^{2}}\] done
clear
C)
\[s{{p}^{3}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer13)
In which of the following the central atom does not use \[s{{p}^{3}}\] hybrid orbitals in its bonding [MNR 1992]
A)
\[Be{{F}_{3}}^{-}\] done
clear
B)
\[OH_{3}^{+}\] done
clear
C)
\[N{{H}_{2}}^{-}\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OH\] done
clear
View Solution play_arrow
-
question_answer14)
\[Xe{{F}_{2}}\] involves hybridisation [DPMT 1990]
A)
\[s{{p}^{3}}\] done
clear
B)
\[s{{p}^{3}}d\] done
clear
C)
\[s{{p}^{3}}{{d}^{2}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer15)
Which of the following hybridisation results in non-planar orbitals [CBSE PMT 1991]
A)
\[s{{p}^{3}}\] done
clear
B)
\[ds{{p}^{2}}\] done
clear
C)
\[s{{p}^{2}}\] done
clear
D)
\[sp\] done
clear
View Solution play_arrow
-
question_answer16)
Octahedral molecular shape exists in .......... hybridisation [DPMT 1990]
A)
\[s{{p}^{3}}d\] done
clear
B)
\[s{{p}^{3}}{{d}^{2}}\] done
clear
C)
\[s{{p}^{3}}{{d}^{3}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer17)
The electronic structure of molecule \[O{{F}_{2}}\] is a hybrid of
A)
\[sp\] done
clear
B)
\[s{{p}^{2}}\] done
clear
C)
\[s{{p}^{3}}\] done
clear
D)
\[s{{d}^{3}}\] done
clear
View Solution play_arrow
-
question_answer18)
Percentage of s-character in \[s{{p}^{3}}\]hybrid orbital is
A)
25 done
clear
B)
50 done
clear
C)
66 done
clear
D)
75 done
clear
View Solution play_arrow
-
question_answer19)
Shape of \[Xe{{F}_{4}}\]molecule is [BHU 1987; AFMC 1992; CET Pune 1998; Roorkee Qualifying 1998; DCE 2002]
A)
Linear done
clear
B)
Pyramidal done
clear
C)
Tetrahedral done
clear
D)
Square planar done
clear
View Solution play_arrow
-
question_answer20)
For which of the following hybridisation the bond angle is maximum [CBSE PMT 1991]
A)
\[s{{p}^{2}}\] done
clear
B)
\[sp\] done
clear
C)
\[s{{p}^{3}}\] done
clear
D)
\[ds{{p}^{2}}\] done
clear
View Solution play_arrow
-
question_answer21)
The \[C-H\]bond distance is the longest in [MNR 1990]
A)
\[{{C}_{2}}{{H}_{2}}\] done
clear
B)
\[{{C}_{2}}{{H}_{4}}\] done
clear
C)
\[{{C}_{2}}{{H}_{4}}B{{r}_{2}}\] done
clear
D)
\[{{C}_{6}}{{H}_{6}}\] done
clear
View Solution play_arrow
-
question_answer22)
The nature of hybridization in \[C{{H}_{2}}Cl-C{{H}_{2}}Cl\] for carbon is
A)
\[sp\] done
clear
B)
\[s{{p}^{2}}\] done
clear
C)
\[s{{p}^{3}}\] done
clear
D)
\[s{{p}^{2}}d\] done
clear
View Solution play_arrow
-
question_answer23)
Shape of methane molecule is [MNR 1983]
A)
Tetrahedral done
clear
B)
Pyramidal done
clear
C)
Octahedral done
clear
D)
Square planer done
clear
View Solution play_arrow
-
question_answer24)
Which one amongst the following possesses an \[sp\] hybridized carbon in its structure [CBSE PMT 1989]
A)
\[Ca{{I}_{2}}\] done
clear
B)
\[C.C{{l}_{2}}=C.C{{l}_{2}}\] done
clear
C)
\[C{{H}_{2}}=C=C{{H}_{2}}\] done
clear
D)
\[C{{H}_{2}}=CH-CH=C{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer25)
Which of the following is the correct electronic formula of chlorine molecule [CPMT 1971]
A)
\[:\,\underset{.\,.}{\overset{\,\,\,.\,.}{\mathop{Cl}}}\,\,:\,\underset{.\,.}{\overset{\,\,\,.\,.}{\mathop{Cl}}}\,\,:\] done
clear
B)
\[:\,\underset{.\,.}{\overset{\,\,\,.\,.}{\mathop{C{{l}^{-}}}}}\,\,:\,\,:\,\,\underset{.\,.}{\overset{\,.\,.}{\mathop{C{{l}^{+}}}}}\,\,:\] done
clear
C)
\[:\,\underset{{}}{\overset{\,\,\,.\,.}{\mathop{Cl}}}\,\,:\,\underset{{}}{\overset{\,\,\,.\,.}{\mathop{Cl}}}\,\,:\] done
clear
D)
\[:\,\underset{{}}{\overset{\,\,\,.\,.}{\mathop{Cl}}}\,\,:\,\underset{{}}{\overset{\,\,\,.\,.}{\mathop{\,:\,Cl}}}\,\,:\] done
clear
View Solution play_arrow
-
question_answer26)
In \[Xe{{F}_{4}}\] hybridization is
A)
\[s{{p}^{3}}{{d}^{2}}\] done
clear
B)
\[s{{p}^{3}}\] done
clear
C)
\[s{{p}^{3}}d\] done
clear
D)
\[s{{p}^{2}}d\] done
clear
View Solution play_arrow
-
question_answer27)
In \[HCHO,\,\,'C'\] has hybridization [AIIMS 1987]
A)
\[sp\] done
clear
B)
\[s{{p}^{2}}\] done
clear
C)
\[s{{p}^{3}}\] done
clear
D)
All the above done
clear
View Solution play_arrow
-
question_answer28)
Which has the shortest \[C-C\] bond length [NCERT 1982; CPMT 1989]
A)
\[{{C}_{2}}{{H}_{5}}OH\] done
clear
B)
\[{{C}_{2}}{{H}_{6}}\] done
clear
C)
\[{{C}_{2}}{{H}_{2}}\] done
clear
D)
\[{{C}_{2}}{{H}_{4}}\] done
clear
View Solution play_arrow
-
question_answer29)
The hybridization of \[Ag\] in the linear complex \[{{\left[ Ag\,{{\left( N{{H}_{3}} \right)}_{2}} \right]}^{+}}\] is [CPMT 1985; BHU 1981]
A)
\[ds{{p}^{2}}\] done
clear
B)
\[sp\] done
clear
C)
\[s{{p}^{2}}\] done
clear
D)
\[CC{{l}_{4}}\] done
clear
View Solution play_arrow
-
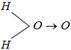
question_answer30)
Experiment shows that \[{{H}_{2}}O\]has a dipole moment while \[C{{O}_{2}}\] has not. Point out the structures which best illustrate these facts [DPMT 1984; NCERT 1983; CPMT 1984]
A)
B)
\[O=C=O\,;\,\,\,H-O-H\] done
clear
C)
D)
\[\underset{\underset{C=O\,;}{\mathop{||\,\,\,\,\,\,\,\,\,\,}}\,}{\mathop{O\,\,\,\,\,\,\,}}\,\,\,\,\underset{\underset{O-H}{\mathop{|\,\,\,\,\,\,\,\,\,\,\,\,\,}}\,}{\mathop{H\,\,\,\,\,\,\,}}\,\] done
clear
View Solution play_arrow
-
question_answer31)
Which species do not have \[s{{p}^{3}}\]hybridization [DPMT 1985]
A)
Ammonia done
clear
B)
Methane done
clear
C)
Water done
clear
D)
Carbon dioxide done
clear
View Solution play_arrow
-
question_answer32)
As compared to pure atomic orbitals, hybrid orbitals have
A)
Low energy done
clear
B)
Same energy done
clear
C)
High energy done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer33)
The compound 1, 2-butadiene has [IIT 1983; MP PMT 1996]
A)
Only \[sp\]hybridized carbon atoms done
clear
B)
Only \[s{{p}^{2}}\]hybridized carbon atoms done
clear
C)
Both \[sp\]and \[s{{p}^{2}}\]hybridized carbon atoms done
clear
D)
\[sp,\,\,s{{p}^{2}}\] and \[s{{p}^{3}}\]hybridized carbon atoms done
clear
View Solution play_arrow
-
question_answer34)
The number of unpaired electrons in \[{{O}_{2}}\] molecule is [MNR 1983; Kerala PET 2002]
A)
0 done
clear
B)
1 done
clear
C)
2 done
clear
D)
3 done
clear
View Solution play_arrow
-
question_answer35)
In the following molecule, the two carbon atoms marked by asterisk (*) possess the following type of hybridized orbitals \[{{H}_{3}}C-{{C}^{*}}\equiv {{C}^{*}}-C{{H}_{3}}\] [NCERT 1984]
A)
\[s{{p}^{3}}\]orbital done
clear
B)
\[s{{p}^{2}}\]orbital done
clear
C)
\[sp\]orbital done
clear
D)
\[s\]orbital done
clear
View Solution play_arrow
-
question_answer36)
The bond angle in carbon tetrachloride is approximately [MNR 1981; MP PMT 1987]
A)
\[{{90}^{o}}\] done
clear
B)
\[{{109}^{o}}\] done
clear
C)
\[{{120}^{o}}\] done
clear
D)
\[{{180}^{o}}\] done
clear
View Solution play_arrow
-
question_answer37)
When two pairs of electrons are shared, bond is [MNR 1979]
A)
Single covalent bond done
clear
B)
Double covalent bond done
clear
C)
Dative bond done
clear
D)
Triple bond done
clear
View Solution play_arrow
-
question_answer38)
The nature of hybridization in the \[BC{{l}_{3}}\] molecule is [EAMCET 1982]
A)
\[sp\] done
clear
B)
\[s{{p}^{2}}\] done
clear
C)
\[s{{p}^{3}}\] done
clear
D)
\[s{{p}^{3}}d\] done
clear
View Solution play_arrow
-
question_answer39)
Which one of the following compounds has bond angle as nearly \[{{90}^{o}}\] [MP PMT 1985]
A)
\[N{{H}_{3}}\] done
clear
B)
\[{{H}_{2}}S\] done
clear
C)
\[{{H}_{2}}O\] done
clear
D)
\[C{{H}_{4}}\] done
clear
View Solution play_arrow
-
question_answer40)
In ethene, the bond angle(s) is/are [CPMT 1976; AMU 1984; MP PMT 1985]
A)
\[{{109}^{o}}28'\] done
clear
B)
\[{{120}^{o}}\] done
clear
C)
\[{{180}^{o}}\] done
clear
D)
Different done
clear
View Solution play_arrow
-
question_answer41)
Structure formula of \[{{H}_{2}}{{O}_{2}}\] is [CPMT 1993]
A)
B)
\[H-O-O-H\] (straight line) done
clear
C)
D)
View Solution play_arrow
-
question_answer42)
Number of shared electrons in between carbon-carbon atoms in ethylene molecule is [MADT Bihar 1983]
A)
2 done
clear
B)
4 done
clear
C)
6 done
clear
D)
3 done
clear
View Solution play_arrow
-
question_answer43)
The structural formula of a compound is \[C{{H}_{3}}-CH=C=C{{H}_{2}}.\] The type of hybridization at the four carbons from left to right are [CBSE PMT 1989]
A)
\[s{{p}^{2}},\,\,sp,\,\,s{{p}^{2}},\,\,s{{p}^{3}}\] done
clear
B)
\[s{{p}^{2}},\,\,s{{p}^{3}},\,\,s{{p}^{2}},\,\,sp\] done
clear
C)
\[s{{p}^{3}},\,\,s{{p}^{2}},\,\,sp,\,\,s{{p}^{2}}\] done
clear
D)
\[s{{p}^{3}},\,\,s{{p}^{2}},\,\,s{{p}^{2}},\,\,s{{p}^{2}}\] done
clear
View Solution play_arrow
-
question_answer44)
Acetate ion contains [AMU 1983]
A)
One \[C,\,\,O\]single bond and one \[C,\,\,O\] double bond done
clear
B)
Two \[C,\,\,O\] single bonds done
clear
C)
Two \[C,\,\,O\]double bonds done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer45)
The two carbon atoms in acetylene are [AMU 1984; MADT Bihar 1982]
A)
\[s{{p}^{3}}\]hybridized done
clear
B)
\[s{{p}^{2}}\]hybridized done
clear
C)
\[sp\]hybridized done
clear
D)
Unhybridized done
clear
View Solution play_arrow
-
question_answer46)
Among the following compounds which is planar in shape [AMU 1992]
A)
Methane done
clear
B)
Acetylene done
clear
C)
Benzene done
clear
D)
Isobutene done
clear
View Solution play_arrow
-
question_answer47)
In methane the bond angle is [AMU 1983]
A)
\[{{180}^{o}}\] done
clear
B)
\[{{90}^{o}}\] done
clear
C)
\[{{120}^{o}}\] done
clear
D)
\[{{109}^{o}}\] done
clear
View Solution play_arrow
-
question_answer48)
The angle between \[s{{p}^{2}}\]orbitals in ethylene is [BHU 1987, 95; AMU 1985]
A)
\[{{90}^{o}}\] done
clear
B)
\[{{120}^{o}}\] done
clear
C)
\[{{180}^{o}}\] done
clear
D)
\[{{109.5}^{o}}\] done
clear
View Solution play_arrow
-
question_answer49)
The species in which the central atom uses \[s{{p}^{2}}\]hybrid orbitals in its bonding is [IIT 1988]
A)
\[P{{H}_{3}}\] done
clear
B)
\[N{{H}_{3}}\] done
clear
C)
\[{{H}_{3}}{{C}^{+}}\] done
clear
D)
\[Sb{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer50)
Carbon atoms in diamond are bonded to each other in a configuration [CPMT 1981]
A)
Tetrahedral done
clear
B)
Planar done
clear
C)
Linear done
clear
D)
Octahedral done
clear
View Solution play_arrow
-
question_answer51)
Which of the following molecules can central atom said to adopt \[s{{p}^{2}}\]hybridization [CBSE PMT 1989; MP PET 1994]
A)
\[Be{{F}_{2}}\] done
clear
B)
\[BC{{l}_{3}}\] done
clear
C)
\[{{C}_{2}}{{H}_{2}}\] done
clear
D)
\[N{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer52)
In \[\left[ Cu\,{{\left( N{{H}_{3}} \right)}_{4}} \right]\,S{{O}_{4}},\,\,;\,Cu\] has following hybridization [AIIMS 1988; UPSEAT 2001]
A)
\[ds{{p}^{2}}\] done
clear
B)
\[s{{p}^{3}}\] done
clear
C)
\[s{{p}^{2}}\] done
clear
D)
\[s{{p}^{3}}{{d}^{2}}\] done
clear
View Solution play_arrow
-
question_answer53)
The hybridization of carbon atoms in \[C-C\] single bond of \[HC\equiv C-CH=C{{H}_{2}}\] is [IIT 1991; MP PET 1995]
A)
\[s{{p}^{3}}-s{{p}^{3}}\] done
clear
B)
\[s{{p}^{2}}-s{{p}^{3}}\] done
clear
C)
\[sp-s{{p}^{2}}\] done
clear
D)
\[s{{p}^{3}}-sp\] done
clear
View Solution play_arrow
-
question_answer54)
The compound in which \[{{C}^{*}}\] uses \[s{{p}^{3}}\]hybrids for bond formation is [IIT 1989]
A)
\[H\overset{+}{\mathop{C}}\,OOH\] done
clear
B)
\[{{(N{{H}_{2}})}_{2}}\overset{+}{\mathop{C}}\,O\] done
clear
C)
\[{{(N{{H}_{3}})}_{3}}\overset{+}{\mathop{C}}\,OH\]\[HgC{{l}_{2}}\] done
clear
D)
\[C{{H}_{3}}\overset{+}{\mathop{C}}\,HO\] done
clear
View Solution play_arrow
-
question_answer55)
In diborane, the \[H-B-H\]bond angle is \[{{120}^{o}}\]. The hybridization of boron is likely to be [BHU 1981; CBSE PMT 1999]
A)
\[sp\] done
clear
B)
\[s{{p}^{2}}\] done
clear
C)
\[s{{p}^{3}}\] done
clear
D)
\[ds{{p}^{2}}\] done
clear
View Solution play_arrow
-
question_answer56)
The number of shared pairs of electrons in propane is [BHU 1981]
A)
2 done
clear
B)
4 done
clear
C)
6 done
clear
D)
10 done
clear
View Solution play_arrow
-
question_answer57)
s-character in \[sp\]hybridised orbitals are
A)
\[\frac{1}{3}\] done
clear
B)
\[\frac{1}{2}\] done
clear
C)
\[\frac{1}{4}\] done
clear
D)
\[\frac{2}{3}\] done
clear
View Solution play_arrow
-
question_answer58)
The two types of bonds present in \[{{B}_{2}}{{H}_{6}}\] are covalent and [IIT 1994]
A)
Three centre bond done
clear
B)
Hydrogen bond done
clear
C)
Two centre bond done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer59)
In the compound \[C{{H}_{3}}OCl,\] which type of orbitals have been used by the circled carbon in bond formation [MP PET 1994]
A)
\[s{{p}^{3}}valuva\] done
clear
B)
\[s{{p}^{2}}\] done
clear
C)
\[sp\] done
clear
D)
p done
clear
View Solution play_arrow
-
question_answer60)
The correct order of the \[O-O\] bond length in \[{{O}_{2}},\,\,{{H}_{2}}{{O}_{2}}\] and \[{{O}_{3}}\] is [CBSE PMT 1995]
A)
\[{{O}_{2}}>{{O}_{3}}>{{H}_{2}}{{O}_{2}}\] done
clear
B)
\[{{O}_{3}}>{{H}_{2}}{{O}_{2}}>{{O}_{2}}\] done
clear
C)
\[{{H}_{2}}{{O}_{2}}>{{O}_{3}}>{{O}_{2}}\] done
clear
D)
\[{{O}_{2}}>{{H}_{2}}{{O}_{2}}>{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer61)
The structure of \[P{{F}_{5}}\]molecule is [AFMC 1995; JIPMER 2001]
A)
Tetrahedral done
clear
B)
Trigonal bipyramidal done
clear
C)
Square planar done
clear
D)
Pentagonal bipyramidal done
clear
View Solution play_arrow
-
question_answer62)
Which of the following hybridisation has maximum s-characters [MP PET 1995]
A)
\[s{{p}^{3}}\] done
clear
B)
\[s{{p}^{2}}\] done
clear
C)
\[sp\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer63)
The \[PC{{l}_{5}}\] molecule is a result of the hybridisation of [MP PET 1995; DCE 2000; MP PMT 2002]
A)
\[s{{p}^{2}}{{d}^{2}}\] done
clear
B)
\[s{{p}^{3}}d\] done
clear
C)
\[sp{{d}^{3}}\] done
clear
D)
\[s{{p}^{2}}{{d}^{3}}\] done
clear
View Solution play_arrow
-
question_answer64)
Hybridisation involves [MP PMT 1996]
A)
Addition of an electron pair done
clear
B)
Mixing up of atomic orbitals done
clear
C)
Removal of an electron pair done
clear
D)
Separation of orbitals done
clear
View Solution play_arrow
-
question_answer65)
The geometry of sulphur trioxide molecule is
A)
Tetrahedral done
clear
B)
Trigonal planar done
clear
C)
Pyramidal done
clear
D)
Square planar done
clear
View Solution play_arrow
-
question_answer66)
The shapes of \[BC{{l}_{3}},\,\,PC{{l}_{3}}\]and \[IC{{l}_{3}}\]molecules are all
A)
Triangular done
clear
B)
Pyramidal done
clear
C)
\[T-\]shaped done
clear
D)
All above are incorrect done
clear
View Solution play_arrow
-
question_answer67)
In benzene molecule all \[C-C\] bond lengths are equal because
A)
All carbon atoms are equivalent done
clear
B)
All carbon atoms are \[s{{p}^{2}}\] hybridised done
clear
C)
All \[C-C\] bonds in benzene, have same order done
clear
D)
All \[C-C\] bonds are single covalent bond done
clear
View Solution play_arrow
-
question_answer68)
Which one is false in the following statements [MP PET 1997]
A)
Each carbon in ethylene is in \[s{{p}^{2}}\] hybridisation done
clear
B)
Each carbon in acetylene is in \[s{{p}^{3}}\] hybridization done
clear
C)
Each carbon in benzene is in \[s{{p}^{2}}\] hybridisation done
clear
D)
Each carbon in ethane is in \[s{{p}^{3}}\] hybridization done
clear
View Solution play_arrow
-
question_answer69)
Out of the following hybrid orbitals, the one which forms the bond at angle\[{{120}^{o}}\], is [MP PMT 1997]
A)
\[{{d}^{2}}s{{p}^{3}}\] done
clear
B)
\[s{{p}^{3}}\] done
clear
C)
\[s{{p}^{2}}\] done
clear
D)
\[sp\] done
clear
View Solution play_arrow
-
question_answer70)
As the \[p-\]character increases, the bond angle in hybrid orbitals formed by \[s\] and atomic orbitals [MP PMT 1997]
A)
Decreases done
clear
B)
Increases done
clear
C)
Doubles done
clear
D)
Remains unchanged done
clear
View Solution play_arrow
-
question_answer71)
\[s{{p}^{3}}\] hybridization leads to which shape of the molecule [MP PET/PMT 1998]
A)
Tetrahedron done
clear
B)
Octahedron done
clear
C)
Linear done
clear
D)
Plane triangle done
clear
View Solution play_arrow
-
question_answer72)
Which of the following will be octahedral [MP PET 1999]
A)
\[S{{F}_{6}}\] done
clear
B)
\[B{{F}_{4}}^{-}\] done
clear
C)
\[PC{{l}_{5}}\] done
clear
D)
\[BO_{3}^{3-}\] done
clear
View Solution play_arrow
-
question_answer73)
The hybrid orbitals used by central atoms in \[BeC{{l}_{2}},\,\,BC{{l}_{3}}\] and \[CC{{l}_{4}}\] molecules are respectively [MP PMT 1999]
A)
\[s{{p}^{2}},\,\,s{{p}^{3}}\] and \[sp\] done
clear
B)
\[s{{p}^{{}}},\,\,s{{p}^{2}}\] and \[s{{p}^{3}}\] done
clear
C)
\[s{{p}^{2}},\,\,s{{p}^{3}}\] and \[s{{p}^{2}}\] done
clear
D)
\[s{{p}^{2}},\,\,sp\] and \[s{{p}^{3}}\] done
clear
View Solution play_arrow
-
question_answer74)
The structure of \[{{H}_{2}}{{O}_{2}}\] is [CBSE PMT 1999; AFMC 2003]
A)
Planar done
clear
B)
Non-planar done
clear
C)
Spherical done
clear
D)
Linear done
clear
View Solution play_arrow
-
question_answer75)
Which of the following is isoelectronic as well as has same structure as that of \[{{N}_{2}}O\] [CPMT 1999]
A)
\[{{N}_{3}}H\] done
clear
B)
\[{{H}_{2}}O\] done
clear
C)
\[N{{O}_{2}}\] done
clear
D)
\[C{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer76)
\[CC{{l}_{4}}\] has the hybridisation [DPMT 1996]
A)
\[s{{p}^{3}}d\] done
clear
B)
\[ds{{p}^{2}}\] done
clear
C)
\[sp\] done
clear
D)
\[s{{p}^{3}}\] done
clear
View Solution play_arrow
-
question_answer77)
Compound having planar symmetry is [DPMT 1996]
A)
\[{{H}_{2}}S{{O}_{4}}\] done
clear
B)
\[{{H}_{2}}O\] done
clear
C)
\[HN{{O}_{3}}\] done
clear
D)
\[CC{{l}_{4}}\] done
clear
View Solution play_arrow
-
question_answer78)
Which of the following compounds is not linear [CPMT 1996]
A)
\[SnC{{l}_{2}}\] done
clear
B)
\[HCl\] done
clear
C)
\[C{{O}_{2}}\] done
clear
D)
\[HgC{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer79)
Which one of the following statements is true for ammonium ion [EAMCET 1997]
A)
All bonds are ionic done
clear
B)
All bonds are coordinate covalent done
clear
C)
\[H\]atoms are situated at the corners of a square done
clear
D)
\[H\]atoms are situated at the corners of a tetrahedron done
clear
View Solution play_arrow
-
question_answer80)
The bond angle in \[s{{p}^{2}}\] hybridisation is [RPMT 1997]
A)
\[{{180}^{o}}\] done
clear
B)
\[{{120}^{o}}\] done
clear
C)
\[{{90}^{o}}\] done
clear
D)
\[{{109}^{o}}2'\] done
clear
View Solution play_arrow
-
question_answer81)
The correct order towards bond angle is [RPMT 1997]
A)
\[sp<s{{p}^{2}}<s{{p}^{3}}\] done
clear
B)
\[s{{p}^{2}}<sp<s{{p}^{3}}\] done
clear
C)
\[s{{p}^{3}}<s{{p}^{2}}<sp\] done
clear
D)
Bond angle does not depend on hybridization done
clear
View Solution play_arrow
-
question_answer82)
The geometry and the type of hybrid orbital present about the central atom in \[B{{F}_{3}}\]is [IIT 1998; BHU 2001]
A)
Linear, \[sp\] done
clear
B)
Trigonal planar, \[s{{p}^{2}}\] done
clear
C)
Tetrahedral, \[s{{p}^{3}}\] done
clear
D)
Pyramidal, \[s{{p}^{3}}\] done
clear
View Solution play_arrow
-
question_answer83)
In graphite, electrons are [CBSE PMT 1997]
A)
Localised on every third \[C\] atom done
clear
B)
Present in antibonding orbital done
clear
C)
Localised on each \[C\] atom done
clear
D)
Spread out between the structure done
clear
View Solution play_arrow
-
question_answer84)
The ammonium ion is [CET Pune 1998]
A)
Tetrahedral done
clear
B)
Trigonal pyramidal done
clear
C)
Square planar done
clear
D)
Square pyramidal done
clear
View Solution play_arrow
-
question_answer85)
In \[sp\]hybridisation, shape is [Bihar MEE 1997]
A)
Angular done
clear
B)
Tetrahedral done
clear
C)
Bipyramidal done
clear
D)
Linear done
clear
E)
None of these done
clear
View Solution play_arrow
-
question_answer86)
When the hybridisation state of carbon atom changes from \[s{{p}^{3}}\]to \[s{{p}^{2}}\]to \[sp,\] the angle between the hybridised orbitals [AIIMS 1998]
A)
Decreases gradually done
clear
B)
Increases gradually done
clear
C)
Decreases considerably done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer87)
The structure and hybridisation of \[Si{{(C{{H}_{3}})}_{4}}\] is [CBSE PMT 1996]
A)
Bent, \[sp\] done
clear
B)
Trigonal, \[s{{p}^{2}}\] done
clear
C)
Octahedral, \[s{{p}^{3}}d\] done
clear
D)
Tetrahedral, \[s{{p}^{3}}\] done
clear
View Solution play_arrow
-
question_answer88)
The type of hybridisation of boron in diborane is [BHU 1999]
A)
sp - hybridisation done
clear
B)
\[s{{p}^{2}}\]- hybridisation done
clear
C)
\[s{{p}^{3}}\]- hybridisation done
clear
D)
\[s{{p}^{3}}{{d}^{2}}\] - hybridization done
clear
View Solution play_arrow
-
question_answer89)
Which compound does not possess linear geometry [RPET 1999]
A)
\[C{{H}_{2}}=C{{H}_{2}}\] done
clear
B)
\[HC\equiv CH\] done
clear
C)
\[BeC{{l}_{2}}\] done
clear
D)
\[C{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer90)
Which of the following molecule does not show tetrahedral shape [RPET 1999]
A)
\[CC{{l}_{4}}\] done
clear
B)
\[SiC{{l}_{4}}\] done
clear
C)
\[S{{F}_{4}}\] done
clear
D)
\[C{{F}_{4}}\] done
clear
View Solution play_arrow
-
question_answer91)
Pyramidal shape would be of [RPET 1999]
A)
\[NO_{3}^{-}\] done
clear
B)
\[{{H}_{2}}O\] done
clear
C)
\[{{H}_{3}}{{O}^{+}}\] done
clear
D)
\[NH_{4}^{+}\] done
clear
View Solution play_arrow
-
question_answer92)
What is the correct mode of hybridization of the central atom in the following compounds : \[NO_{2}^{+},S{{F}_{4,}}P{{F}_{6}}^{-}\] [AMU 1999]
A)
\[s{{p}^{2}},\,\,s{{p}^{3}},\,\,{{d}^{2}}s{{p}^{3}}\] done
clear
B)
\[s{{p}^{3}},\,\,s{{p}^{3}}{{d}^{2}},\,\,s{{p}^{3}}{{d}^{2}}\] done
clear
C)
\[sp,\,\,s{{p}^{3}}d,\,\,s{{p}^{3}}{{d}^{2}}\] done
clear
D)
\[sp,\,\,s{{p}^{2}},\,\,s{{p}^{3}}\] done
clear
View Solution play_arrow
-
question_answer93)
The hybridization in \[P{{F}_{3}}\] is [DCE 2000]
A)
\[s{{p}^{3}}\] done
clear
B)
\[s{{p}^{2}}\] done
clear
C)
\[ds{{p}^{3}}\] done
clear
D)
\[{{d}^{2}}s{{p}^{3}}\] done
clear
View Solution play_arrow
-
question_answer94)
Which of the following molecule is linear [MP PMT 2000]
A)
\[S{{O}_{2}}\] done
clear
B)
\[NO_{2}^{+}\] done
clear
C)
\[NO_{2}^{-}\] done
clear
D)
\[SC{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer95)
The geometry of the molecule with \[s{{p}^{3}}{{d}^{2}}\] hybridised central atom is [NCERT 1981; AFMC 1982; RPMT 2000]
A)
Square planar done
clear
B)
Trigonal bipyramidal done
clear
C)
Octahedral done
clear
D)
Square pyramidal done
clear
View Solution play_arrow
-
question_answer96)
The bond angle in \[P{{H}_{3}}\] is [RPMT 2000]
A)
Much less than \[N{{H}_{3}}\] done
clear
B)
Equal to that of \[N{{H}_{3}}\] done
clear
C)
Much greater than \[N{{H}_{3}}\] done
clear
D)
Slightly greater than \[N{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer97)
Which of the following has tetrahedral structure [CPMT 2000]
A)
\[CO_{3}^{-}\] done
clear
B)
\[NH_{4}^{+}\] done
clear
C)
\[{{K}_{4}}[Fe{{(CN)}_{6}}]\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer98)
The single, double and triple bond lengths of carbon in carbon dioxide are respectively [AIIMS 2000]
A)
1.15, 1.22 and 1.10 Å done
clear
B)
1.22, 1.15 and 1.10 Å done
clear
C)
1.10, 1.15 and 1.22 Å done
clear
D)
1.15, 1.10 and 1.22 Å done
clear
View Solution play_arrow
-
question_answer99)
Shape of \[B{{F}_{3}}\] molecule is [CPMT 2000; Pb. CET 2002]
A)
Linear done
clear
B)
Planar done
clear
C)
Tetrahedral done
clear
D)
Square pyramidal done
clear
View Solution play_arrow
-
question_answer100)
In the complex \[{{[Sb{{F}_{5}}]}^{\,2-}},\]\[s{{p}^{3}}d\] hybridization is present. Geometry of the complex is [Pb. PMT 2000]
A)
Square done
clear
B)
Square pyramidal done
clear
C)
Square bipyramidal done
clear
D)
Tetrahedral done
clear
View Solution play_arrow
-
question_answer101)
The bond angle is minimum in [Pb. PMT 2001; MP PET 2003; UPSEAT 2004]
A)
\[{{H}_{2}}Te\] done
clear
B)
\[{{H}_{2}}Se\] done
clear
C)
\[{{H}_{2}}O\] done
clear
D)
\[{{H}_{2}}S\] done
clear
View Solution play_arrow
-
question_answer102)
The correct order of hybridization of the central atom in the following species \[N{{H}_{3}}\] \[{{[PtC{{l}_{4}}]}^{2-}},\,PC{{l}_{5}}\] and \[BC{{l}_{3}}\] is [IIT Screening 2001; BHU 2005]
A)
\[ds{{p}^{2}},\,\,ds{{p}^{3}},\,\,s{{p}^{2}}\]and \[s{{p}^{3}}\] done
clear
B)
\[s{{p}^{3}},\,\,ds{{p}^{2}},\,\,ds{{p}^{3}},\,\,s{{p}^{2}}\] done
clear
C)
\[ds{{p}^{2}},\,\,s{{p}^{2}},\,\,s{{p}^{3}},\,\,ds{{p}^{3}}\] done
clear
D)
\[ds{{p}^{2}},\,\,s{{p}^{3}},\,\,s{{p}^{2}},\,\,ds{{p}^{3}}\] done
clear
View Solution play_arrow
-
question_answer103)
Which of the following pairs has same structure [BHU 2001]
A)
\[P{{H}_{3}}\] and \[BC{{l}_{3}}\] done
clear
B)
\[S{{O}_{2}}\] and \[N{{H}_{3}}\] done
clear
C)
\[PC{{l}_{5}}\] and \[S{{F}_{6}}\] done
clear
D)
\[NH_{4}^{+}\] and \[SO_{4}^{2-}\] done
clear
View Solution play_arrow
-
question_answer104)
The smallest bond angle is found in [AIIMS 2001]
A)
\[I{{F}_{7}}\] done
clear
B)
\[C{{H}_{4}}\] done
clear
C)
\[Be{{F}_{2}}\] done
clear
D)
\[B{{F}_{3}}\] done
clear
View Solution play_arrow
-
question_answer105)
Which of the following is not linear [DCE 2001]
A)
\[C{{O}_{2}}\] done
clear
B)
\[Cl{{O}_{2}}\] done
clear
C)
\[I_{3}^{-}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer106)
Which of the following is not tetrahedral [MP PMT 2001]
A)
\[SC{{l}_{4}}\] done
clear
B)
\[SO_{4}^{2-}\] done
clear
C)
\[Ni{{(CO)}_{4}}\] done
clear
D)
\[NiCl_{4}^{2-}\] done
clear
View Solution play_arrow
-
question_answer107)
As the s-character of hybridisation orbital increases, the bond angle [BHU 2002; RPMT 2002]
A)
Increases done
clear
B)
Decreases done
clear
C)
Becomes zero done
clear
D)
Does not change done
clear
View Solution play_arrow
-
question_answer108)
The shape of \[I{{F}_{7}}\]molecule is [AFMC 2002; MHCET 2003]
A)
Octahedral done
clear
B)
Pentagonal bipyramidal done
clear
C)
Trigonal bipyramidal done
clear
D)
Tetrahedral done
clear
View Solution play_arrow
-
question_answer109)
A completely filled d orbital \[({{d}^{10}})\] [UPSEAT 2002]
A)
Spherically symmetrical done
clear
B)
Has octahedral symmetry done
clear
C)
Has tetrahedral symmetry done
clear
D)
Depends on the atom done
clear
View Solution play_arrow
-
question_answer110)
Which has \[s{{p}^{3}}\] hybridization of central atom [UPSEAT 2002]
A)
\[PC{{l}_{3}}\] done
clear
B)
\[S{{O}_{3}}\] done
clear
C)
\[B{{F}_{3}}\] done
clear
D)
\[NO{}_{3}^{-}\] done
clear
View Solution play_arrow
-
question_answer111)
In which of the following species is the interatomic bond angle is \[{{109}^{o}}2{8}'\] [AIEEE 2002]
A)
\[N{{H}_{3}},\,\,{{(B{{F}_{4}})}^{-1}}\] done
clear
B)
\[{{(N{{H}_{4}})}^{+}},\,\,B{{F}_{3}}\] done
clear
C)
\[N{{H}_{3}},\,\,B{{F}_{4}}\] done
clear
D)
\[{{(N{{H}_{2}})}^{-1}},\,\,B{{F}_{3}}\] done
clear
View Solution play_arrow
-
question_answer112)
A square planar complex is formed by hybridisation of which atomic orbitals [AIEEE 2002]
A)
\[s,\,\,{{p}_{x}},\,\,{{p}_{y}},\,\,{{d}_{yz}}\] done
clear
B)
\[s,\,\,{{p}_{x}},\,\,{{p}_{y}},\,\,{{d}_{{{x}^{2}}-{{y}^{2}}}}\] done
clear
C)
\[s,\,\,{{p}_{x}},\,\,{{p}_{y,\,\,}}{{d}_{{{z}^{2}}}}\] done
clear
D)
\[s,\,\,{{p}_{y}},\,\,{{p}_{z}},{{d}_{xy}}\] done
clear
View Solution play_arrow
-
question_answer113)
In benzene, all the six \[CC\] bonds have the same length because of [MP PET 2002]
A)
Tautomerism done
clear
B)
\[s{{p}^{2}}\]hybridization done
clear
C)
Isomerism done
clear
D)
Inductive effect done
clear
View Solution play_arrow
-
question_answer114)
The bond energies of \[H-H\] and \[ClCl\] are 430 kJ \[\text{mo}{{\text{l}}^{-1}}\]and 242 kJ \[\text{mo}{{\text{l}}^{-1}}\] respectively, \[\Delta {{H}_{t}}\] for HCl is 91 kJ mol-1. The bond energy of HCl will be [MP PET 2003]
A)
427 kJ done
clear
B)
766 kJ done
clear
C)
285 kJ done
clear
D)
245 kJ done
clear
View Solution play_arrow
-
question_answer115)
Which of the following has \[ds{{p}^{2}}\]hybridization [MP PET 2003]
A)
\[NiCl_{4}^{2-}\] done
clear
B)
\[SC{{l}_{4}}\] done
clear
C)
\[NH_{4}^{+}\] done
clear
D)
\[PtCl_{4}^{2-}\] done
clear
View Solution play_arrow
-
question_answer116)
Which one of the following is a planar molecule [EAMCET 2003]
A)
\[N{{H}_{3}}\] done
clear
B)
\[{{H}_{3}}{{O}^{+}}\] done
clear
C)
\[BC{{l}_{3}}\] done
clear
D)
\[PC{{l}_{3}}\] done
clear
View Solution play_arrow
-
question_answer117)
Which one of the following is a correct set with respect to molecule, hybridisation and shape [EAMCET 2003]
A)
\[BeC{{l}_{2}}\], \[s{{p}^{2}}\], linear done
clear
B)
\[BeC{{l}_{2}}\], \[s{{p}^{2}}\], triangular planar done
clear
C)
\[BC{{l}_{3}}\], \[s{{p}^{2}}\], triangular planar done
clear
D)
\[BC{{l}_{3}}\], \[s{{p}^{3}}\], tetrahedral done
clear
View Solution play_arrow
-
question_answer118)
Which of the following compounds doesn?t have linear structure [RPET 1997, 2003]
A)
\[C{{O}_{2}}\] done
clear
B)
\[S{{O}_{2}}\] done
clear
C)
\[BeC{{l}_{2}}\] done
clear
D)
\[{{C}_{2}}{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer119)
Which of the following bonds require the largest amount of bond energy to dissociate the atom concerned [UPSEAT 2003]
A)
\[HH\] bond in \[{{H}_{2}}\] done
clear
B)
\[CC\] bond in \[C{{H}_{4}}\] done
clear
C)
\[N\equiv N\] bond in \[{{N}_{2}}\] done
clear
D)
\[O=O\] bond in \[{{O}_{2}}\] done
clear
E)
\[CC\] bond in ethane done
clear
View Solution play_arrow
-
question_answer120)
The percentage s-character of the hybrid orbitals in methane, ethene and ethyne are respectively [KCET 2003]
A)
25, 33, 50 done
clear
B)
25, 50, 75 done
clear
C)
50, 75, 100 done
clear
D)
10, 20, 40 done
clear
View Solution play_arrow
-
question_answer121)
Arrange the hydra-acids of halogens in increasing order of acidity [Orissa JEE 2003]
A)
\[HF<HCl<HBr<HI\] done
clear
B)
\[HI<HBr<HCl<HF\] done
clear
C)
\[HF<HBr<HI<HCl\] done
clear
D)
\[HF<HI<HBr<HCl\] done
clear
View Solution play_arrow
-
question_answer122)
Which one has \[s{{p}^{2}}-\]hybridisation [MP PMT 2004]
A)
\[C{{O}_{2}}\] done
clear
B)
\[{{N}_{2}}O\] done
clear
C)
\[S{{O}_{2}}\] done
clear
D)
\[CO\] done
clear
View Solution play_arrow
-
question_answer123)
Among the following compounds the one that is polar and has central atom with \[s{{p}^{2}}-\]hybridization is [MP PMT 2004; IIT 1997]
A)
\[{{H}_{2}}C{{O}_{3}}\] done
clear
B)
\[B{{F}_{3}}\] done
clear
C)
\[Si{{F}_{4}}\] done
clear
D)
\[HCl{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer124)
The molecule which is pyramid shape is [MP PMT 2004; EAMCET 1985; IIT 1989]
A)
\[PC{{l}_{3}}\] done
clear
B)
\[CO_{3}^{2-}\] done
clear
C)
\[S{{O}_{3}}\] done
clear
D)
\[NO_{3}^{-}\] done
clear
View Solution play_arrow
-
question_answer125)
Which of the following has a linear structure [MP PMT 2004]
A)
\[CC{{l}_{4}}\] done
clear
B)
\[{{C}_{2}}{{H}_{2}}\] done
clear
C)
\[S{{O}_{2}}\] done
clear
D)
\[{{C}_{2}}{{H}_{4}}\] done
clear
View Solution play_arrow
-
question_answer126)
In a regular octahedral molecule, \[M{{X}_{6}}\], the number \[X-M-X\] bonds at 180° is [CBSE PMT 2004]
A)
Six done
clear
B)
Four done
clear
C)
Three done
clear
D)
Two done
clear
View Solution play_arrow
-
question_answer127)
\[s{{p}^{3}}{{d}^{2}}\] hybrid orbitals are [MP PET 2004]
A)
Linear bipyramidal done
clear
B)
Pentagonal done
clear
C)
Trigonal bipyramidal done
clear
D)
Octahedral done
clear
View Solution play_arrow
-
question_answer128)
In an octahedral structure, the pair of d orbitals involved in \[{{d}^{2}}s{{p}^{3}}\] hybridization is [CBSE PMT 2004]
A)
\[{{d}_{{{x}^{2}}}},{{d}_{xz}}\] done
clear
B)
\[{{d}_{xy}},{{d}_{yz}}\] done
clear
C)
\[{{d}_{{{x}^{2}}-{{y}^{2}}}},{{d}_{{{z}^{2}}}}\] done
clear
D)
\[{{d}_{xz}},{{d}_{{{x}^{2}}-{{y}^{2}}}}\] done
clear
View Solution play_arrow
-
question_answer129)
The correct order of bond angles (smallest first) in \[{{H}_{2}}S,N{{H}_{3}},B{{F}_{3}}\]and \[Si{{H}_{4}}\]is [AIEEE 2004]
A)
\[{{H}_{2}}S<N{{H}_{3}}<Si{{H}_{4}}<B{{F}_{3}}\] done
clear
B)
\[N{{H}_{3}}<{{H}_{2}}S<Si{{H}_{4}}<B{{F}_{3}}\] done
clear
C)
\[{{H}_{2}}S<Si{{H}_{4}}<N{{H}_{3}}<B{{F}_{3}}\] done
clear
D)
\[{{H}_{2}}S<N{{H}_{3}}<B{{F}_{3}}<Si{{H}_{4}}\] done
clear
View Solution play_arrow
-
question_answer130)
Which one of the following has the regular tetrahedral structure [AIEEE 2004]
A)
\[BF_{4}^{-}\] done
clear
B)
\[S{{F}_{4}}\] done
clear
C)
\[Xe{{F}_{4}}\] done
clear
D)
\[{{\left[ Ni{{(CN)}_{4}} \right]}^{2-}}\] (Atomic no. : \[B=5,S=16,Ni=28,Xe=54\]) done
clear
View Solution play_arrow
-
question_answer131)
The states of hybridazation of boron and oxygen atoms in boric acid \[({{H}_{3}}B{{O}_{3}})\] are respectively [AIEEE 2004]
A)
\[s{{p}^{3}}\] and \[s{{p}^{2}}\] done
clear
B)
\[s{{p}^{2}}\] and \[s{{p}^{3}}\] done
clear
C)
\[s{{p}^{2}}\] and \[s{{p}^{2}}\] done
clear
D)
\[s{{p}^{3}}\]and \[s{{p}^{3}}\] done
clear
View Solution play_arrow
-
question_answer132)
The hybridisation in \[B{{F}_{3}}\] molecule is [Pb. PMT 2004]
A)
\[sp\] done
clear
B)
\[s{{p}^{2}}\] done
clear
C)
\[s{{p}^{3}}\] done
clear
D)
\[s{{p}^{3}}d\] done
clear
View Solution play_arrow
-
question_answer133)
Among the compounds, \[B{{F}_{3}},NC{{l}_{3}},{{H}_{2}}S,S{{F}_{4}}\]and \[BeC{{l}_{2}}\], identify the ones in which the central atom has the same type of hybridisation [Kerala PMT 2004]
A)
\[B{{F}_{3}}\] and \[NC{{l}_{3}}\] done
clear
B)
\[{{H}_{2}}S\]and \[BeC{{l}_{2}}\] done
clear
C)
\[B{{F}_{3}},NC{{l}_{3}}\]and \[{{H}_{2}}S\] done
clear
D)
\[S{{F}_{4}}\]and \[BeC{{l}_{2}}\] done
clear
E)
\[NC{{l}_{3}}\] and \[{{H}_{2}}S\] done
clear
View Solution play_arrow
-
question_answer134)
The molecule of \[C{{O}_{2}}\] has 180° bond angle. It can be explanid on the basis of [AFMC 2004]
A)
\[s{{p}^{3}}\] hybridisation done
clear
B)
\[s{{p}^{2}}\] hybridisation done
clear
C)
\[sp\] hybridisation done
clear
D)
\[{{d}^{2}}s{{p}^{3}}\] hybridization done
clear
View Solution play_arrow
-
question_answer135)
\[s{{p}^{3}}\] hybridisation is found in [Pb. CET 2003; Orissa JEE 2005]
A)
\[CO_{3}^{2-}\] done
clear
B)
\[B{{F}_{3}}\] done
clear
C)
\[NO_{3}^{-}\] done
clear
D)
\[N{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer136)
Which set hydridisation is correct for the following compounds [Pb. CET 2003]
A)
\[N{{O}_{2}}\]-\[sp\], \[S{{F}_{4}}\]-\[s{{p}^{2}}\], \[PF_{6}^{-}\]-\[s{{p}^{3}}\] done
clear
B)
\[N{{O}_{2}}\]-\[sp\], \[S{{F}_{4}}\]-\[s{{p}^{3}}d\], \[PF_{6}^{-}\]-\[s{{p}^{3}}{{d}^{2}}\] done
clear
C)
\[N{{O}_{2}}\]-\[s{{p}^{2}}\], \[S{{F}_{4}}\]-\[s{{p}^{3}}\], \[PF_{6}^{-}\]-\[{{d}^{2}}s{{p}^{3}}\] done
clear
D)
\[N{{O}_{2}}\]-\[s{{p}^{3}}\], \[S{{F}_{4}}\]-\[s{{p}^{3}}{{d}^{2}}\], \[PF_{6}^{-}\]-\[s{{p}^{3}}{{d}^{2}}\] done
clear
View Solution play_arrow
-
question_answer137)
The state of hybridisation of \[B\] in \[BC{{l}_{3}}\] is [Pb. CET 2000; BHU 2004]
A)
\[sp\] done
clear
B)
\[s{{p}^{2}}\] done
clear
C)
\[s{{p}^{3}}\] done
clear
D)
\[s{{p}^{2}}{{d}^{2}}\] done
clear
View Solution play_arrow
-
question_answer138)
The hybrid state of sulphur in \[S{{O}_{3}}\] molecule is [DCE 2004]
A)
\[s{{p}^{3}}d\] done
clear
B)
\[s{{p}^{3}}\] done
clear
C)
\[s{{p}^{3}}{{d}^{2}}\] done
clear
D)
\[s{{p}^{2}}\] done
clear
View Solution play_arrow
-
question_answer139)
Which of the following molecules has pyramidal shape [DCE 2004; J&K CET 2005]
A)
\[PC{{l}_{3}}\] done
clear
B)
\[S{{O}_{3}}\] done
clear
C)
\[CO_{3}^{2-}\] done
clear
D)
\[NO_{3}^{-}\] done
clear
View Solution play_arrow
-
question_answer140)
The hybrdization of \[I{{F}_{7}}\] is [Pb. CET 2001]
A)
\[s{{p}^{3}}{{d}^{3}}\] done
clear
B)
\[s{{p}^{2}}d\] done
clear
C)
\[{{d}^{2}}s{{p}^{3}}\] done
clear
D)
\[s{{p}^{3}}\] done
clear
View Solution play_arrow
-
question_answer141)
In which compound, the hydrogen bonding is the strongest in its liquid phase [Pb. CET 2001]
A)
\[HF\] done
clear
B)
\[HI\] done
clear
C)
\[C{{H}_{4}}\] done
clear
D)
\[P{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer142)
Geometry of ammonia molecule and the hybridization of nitrogen involved in it are [MH CET 2004]
A)
\[s{{p}^{3}}\]-hybridization and tetrahedral geometry done
clear
B)
\[s{{p}^{3}}\]-hybridization and distorted tetrahedral geometry done
clear
C)
\[s{{p}^{2}}\]-hybridization and triangular geometry done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer143)
\[Be\] in \[BeC{{l}_{2}}\] undergoes [MH CET 2004]
A)
Diagonal hybridization done
clear
B)
Trigonal hybridization done
clear
C)
Tetrahedral hybridization done
clear
D)
No hybridization done
clear
View Solution play_arrow
-
question_answer144)
Which of the following is non-linear molecule [DCE 2003]
A)
\[C{{O}_{3}}\] done
clear
B)
\[C{{O}_{2}}\] done
clear
C)
\[C{{S}_{2}}\] done
clear
D)
\[BeC{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer145)
The trigonal bipyramidal geometry results from the hybridisation [UPSEAT 2004]
A)
\[ds{{p}^{3}}\] or \[s{{p}^{3}}d\] done
clear
B)
\[ds{{p}^{2}}\] or \[s{{p}^{2}}d\] done
clear
C)
\[{{d}^{2}}s{{p}^{3}}\] or \[s{{p}^{3}}{{d}^{2}}\] done
clear
D)
\[{{d}^{3}}s{{p}^{2}}\] or \[{{d}^{2}}s{{p}^{3}}\] done
clear
View Solution play_arrow
-
question_answer146)
The valency of carbon is four. On what principle it can be explained in a better way
A)
Resonance done
clear
B)
Hybridization done
clear
C)
Electron transfer done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer147)
Hybridization is due to the overlapping of [MADT Bihar 1983]
A)
Orbitals of different energy levels done
clear
B)
Orbitals of different energy content done
clear
C)
Orbitals of same energy content done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer148)
If a molecule \[M{{X}_{3}}\] has zero dipole moment, the sigma bonding orbital used by M are [IIT 1981; MP PMT 1994; Kerala PMT 2004]
A)
\[s{{p}^{3}}d-\]hybrid done
clear
B)
\[sp-\]hybrid done
clear
C)
\[s{{p}^{3}}{{d}^{2}}-\]hybrid done
clear
D)
\[s{{p}^{2}}-\]hybrid done
clear
View Solution play_arrow
-
question_answer149)
The linear structure is assumed by [IIT 1991]
A)
\[SnC{{l}_{2}}\] done
clear
B)
\[NC{{O}^{-}}\] done
clear
C)
\[C{{S}_{2}}\] done
clear
D)
\[NO_{2}^{+}\] done
clear
View Solution play_arrow
-
question_answer150)
Hybridisation of central atom in \[N{{F}_{3}}\] is [Orissa JEE 2005]
A)
\[s{{p}^{3}}\] done
clear
B)
\[sp\] done
clear
C)
\[s{{p}^{2}}\] done
clear
D)
\[ds{{p}^{2}}\] done
clear
View Solution play_arrow
-
question_answer151)
The pair having similar geometry is [J&K CET 2005]
A)
\[PC{{l}_{3}},\ N{{H}_{3}}\] done
clear
B)
\[BeC{{l}_{2}},\ {{H}_{2}}O\] done
clear
C)
\[C{{H}_{4}},CC{{l}_{4}}\] done
clear
D)
\[I{{F}_{5}},\ P{{F}_{5}}\] done
clear
View Solution play_arrow
-
question_answer152)
The d-orbital involved in \[s{{p}^{3}}d\] hybridisation is [J&K CET 2005]
A)
\[{{d}_{{{x}^{2}}-{{y}^{2}}}}\] done
clear
B)
\[{{d}_{xy}}\] done
clear
C)
\[{{d}_{{{z}^{2}}}}\] done
clear
D)
\[{{d}_{zx}}\] done
clear
View Solution play_arrow
 done
clear
done
clear
