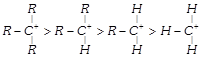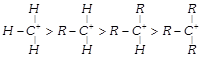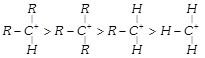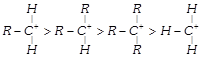Three masses are placed on the x-axis, mass of 300 g at origin, 500 g at x = 40 cm and 400 g at x = 70 cm. The distance of the centre of mass from the origin is
A)
40 cm
done
clear
B)
45 cm
done
clear
C)
50 cm
done
clear
D)
30 cm
done
clear
View Answer play_arrow
A balloon contains \[1500\,\,{{m}^{3}}\] of helium at \[27{}^\circ C\] and 4 atmospheric pressure. The volume of helium at \[-3{}^\circ C\] temperature and 2 atmospheric pressure will be,
A)
\[1500\,\,{{m}^{3}}\]
done
clear
B)
\[1700\,\,{{m}^{3}}\]
done
clear
C)
\[1900\,\,{{m}^{3}}\]
done
clear
D)
\[2700\,\,{{m}^{3}}\]
done
clear
View Answer play_arrow
Two capacitors when connected in series have a capacitance of \[3\,\,\mu F,\] and when connected in parallel have a capacitance of \[16\,\,\mu F.\] Their individual capacities are
A)
\[1\,\,\mu F,\]\[2\,\,\mu F\]
done
clear
B)
\[6\,\,\mu F,\]\[2\,\,\mu F\]
done
clear
C)
\[12\,\,\mu F,\]\[4\,\,\mu F\]
done
clear
D)
\[3\,\,\mu F,\]\[16\,\,\mu F\]
done
clear
View Answer play_arrow
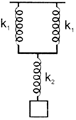
What will be the force constant of the spring system shown in figure?
A)
\[\frac{{{k}_{1}}}{2}+{{k}_{2}}\]
done
clear
B)
\[{{\left[ \frac{1}{2{{k}_{1}}}+\frac{1}{{{k}_{2}}} \right]}^{-1}}\]
done
clear
C)
\[\left[ \frac{1}{2{{k}_{1}}}+\frac{1}{{{k}_{2}}} \right]\]
done
clear
D)
\[{{\left[ \frac{2}{{{k}_{1}}}+\frac{1}{{{k}_{2}}} \right]}^{-1}}\]
done
clear
View Answer play_arrow
The amplitude of magnetic field of an electromagnetic wave is \[2\times {{10}^{-7}}T.\] Its electric field amplitude if the wave is travelling in free space is
A)
\[6\,V{{m}^{-1}}\]
done
clear
B)
\[60\,V{{m}^{-1}}\]
done
clear
C)
\[10/6\,V{{m}^{-1}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
If c, the velocity of light, g the acceleration due to gravity and P the atmospheric pressure be the fundamental quantities in MKS system, then the dimensions of length will be same as that of
A)
\[\frac{c}{g}\]
done
clear
B)
\[\frac{c}{p}\]
done
clear
C)
\[pcg\]
done
clear
D)
\[\frac{{{c}^{2}}}{g}\]
done
clear
View Answer play_arrow
A body having initial velocity of 10 m/s moving on a rough surface comes to rest after moving 50 m. What is coefficient of friction between the body and surface? \[(g=10\,m/{{s}^{2}})\]
A)
0.5
done
clear
B)
0.2
done
clear
C)
0.3
done
clear
D)
0.1
done
clear
View Answer play_arrow
A sonometer wire supports a 4 kg load and vibrates in fundamental mode with a tuning fork of frequency 416 Hz. The length of the wire between the bridges is now doubled. In order to maintain fundamental mode, the load should be changed to
A)
1 kg
done
clear
B)
2 kg
done
clear
C)
4 kg
done
clear
D)
16 kg
done
clear
View Answer play_arrow
Given, \[_{a}{{\mu }_{g}}=\frac{3}{2},\]\[_{a}{{\mu }_{w}}=\frac{4}{3},\] if a convex lens of focal length 10 cm is placed in water, then its focal length in water is
A)
equal to 40 cm
done
clear
B)
equal to 20 cm
done
clear
C)
equal to 10 cm
done
clear
D)
None of these
done
clear
View Answer play_arrow
A large tank filled with water to a height 'h' is to be emptied through a small hole at the bottom. The ratio of time taken for the level of water to fall from h to \[\frac{h}{2}\] and \[\frac{h}{2}\] zero is
A)
\[\sqrt{2}\]
done
clear
B)
\[\frac{1}{\sqrt{2}}\]
done
clear
C)
\[\sqrt{2}-1\]
done
clear
D)
\[\frac{1}{\sqrt{2}-1}\]
done
clear
View Answer play_arrow
A mass is tied to a string and rotated in a vertical circle. The minimum velocity of the body at the top is
A)
\[\sqrt{gr}\]
done
clear
B)
\[g/r\]
done
clear
C)
\[{{\left( \frac{g}{r} \right)}^{3/2}}\]
done
clear
D)
\[gr\]
done
clear
View Answer play_arrow
What is the disintegration constant of radon, if the number of its atoms diminishes by 18% in 24 h?
A)
\[2.1\times {{10}^{-3}}{{s}^{-1}}\]
done
clear
B)
\[2.1\times {{10}^{-4}}{{s}^{-1}}\]
done
clear
C)
\[2.1\times {{10}^{-5}}{{s}^{-1}}\]
done
clear
D)
\[2.1\times {{10}^{-6}}{{s}^{-1}}\]
done
clear
View Answer play_arrow
Hot water cools from \[60{}^\circ C\] to \[50{}^\circ C\] in the first 10 minutes and to \[42{}^\circ C\] in the next 10 minutes. The temperature of the surroundings is:
A)
\[25{}^\circ C\]
done
clear
B)
\[10{}^\circ C\]
done
clear
C)
\[15{}^\circ C\]
done
clear
D)
\[20{}^\circ C\]
done
clear
View Answer play_arrow
Consider the following statement: When jumping from some height, you should bend your knees as you come to rest, instead of keeping your legs stiff. Which of the following relations can be useful in explaining the statement? Where symbols have their usual meanings.
A)
\[\Delta {{\overrightarrow{p}}_{1}}=-\Delta {{\overrightarrow{p}}_{2}}\]
done
clear
B)
\[\Delta E=\Delta (PE+KE)=0\]
done
clear
C)
\[\overrightarrow{F}\Delta t=m\Delta \overrightarrow{v}\]
done
clear
D)
\[\Delta x\propto \Delta F\]
done
clear
View Answer play_arrow
A galvanometer can be changed into an ammeter by using
A)
low resistance shunt in series
done
clear
B)
low resistance shunt in parallel
done
clear
C)
high resistance shunt in series
done
clear
D)
high resistance shunt in parallel
done
clear
View Answer play_arrow
The mass number of He is 4 and that for sulphur is 32. The radius of sulphur nuclei is larger than that of helium by
A)
\[\sqrt{8}\]
done
clear
B)
\[4\]
done
clear
C)
\[2\]
done
clear
D)
\[8\]
done
clear
View Answer play_arrow
The drift current in a p-n junction is
A)
from the n-side to the p-side
done
clear
B)
from the p-side to the n-side
done
clear
C)
from the n-side to the p-side if the junction is forward-baised and in the opposite direction if it is reverse biased
done
clear
D)
from the p-side to the n-side if the junction is forward-baised and in the opposite direction if it is reverse-baised
done
clear
View Answer play_arrow
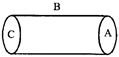
A hollow cylinder has a charge q coulomb within it. If \[\phi \] is the electric flux in units of voltmeter associated with the curved surface B, the flux linked with the plane surface A in units of voltmeter will be
A)
\[\frac{q}{2{{\varepsilon }_{0}}}\]
done
clear
B)
\[\frac{\phi }{3}\]
done
clear
C)
\[\frac{q}{{{\varepsilon }_{0}}}-\phi \]
done
clear
D)
\[\frac{1}{2}\left( \frac{q}{{{\varepsilon }_{0}}}-\phi \right)\]
done
clear
View Answer play_arrow
The fermi energy for a substance is
A)
independent of \[T\]
done
clear
B)
directly proportional to \[\sqrt{T}\]
done
clear
C)
directly proportional to \[T\]
done
clear
D)
directly proportional to \[{{T}^{2}}\]
done
clear
View Answer play_arrow
If the critical angle for total internal reflection from a medium to vacuum is \[30{}^\circ .\] Then velocity of light in the medium is
A)
\[1.5\times {{10}^{8}}m/s\]
done
clear
B)
\[2\times {{10}^{8}}m/s\]
done
clear
C)
\[3\times {{10}^{8}}m/s\]
done
clear
D)
\[0.75\times {{10}^{8}}m/s\]
done
clear
View Answer play_arrow
A metal piece is heated upto \[T{}^\circ \] abs. The temperature of the surrounding is t° abs. The heat in the surrounding due to radiation is proportional to
A)
\[{{(T-T)}^{4}}\]
done
clear
B)
\[{{T}^{4}}-{{t}^{4}}\]
done
clear
C)
\[{{(T-t)}^{1/4}}\]
done
clear
D)
\[{{T}^{2}}-{{t}^{2}}\]
done
clear
View Answer play_arrow
The rain drops are in spherical shape due to
A)
residual pressure
done
clear
B)
thrust on drop
done
clear
C)
surface tension
done
clear
D)
viscosity
done
clear
View Answer play_arrow
An iron rod of length \[2m\] and cross-sectional area of \[50\,m{{m}^{2}}\] is stretched by \[0.5\,mm,\] when a mass of 250 kg is hung from its lower end. Young's modulus of iron rod is
A)
\[19.6\times {{10}^{20}}N/{{m}^{2}}\]
done
clear
B)
\[19.6\times {{10}^{18}}N/{{m}^{2}}\]
done
clear
C)
\[19.6\times {{10}^{10}}N/{{m}^{2}}\]
done
clear
D)
\[19.6\times {{10}^{15}}N/{{m}^{2}}\]
done
clear
View Answer play_arrow
A particle of mass 1 kg is moving in S.H.M. with an amplitude 0.02 and a frequency of 60 Hz. The maximum force acting on the particle is
A)
\[144\,{{\pi }^{2}}\]
done
clear
B)
\[188\,{{\pi }^{2}}\]
done
clear
C)
\[288\,{{\pi }^{2}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
A dip circle is so set that its needle moves freely in the magnetic meridian. In this position, the angle of dip is \[40{}^\circ .\] Now the dip circle is rotated so that the plane in which the needle moves makes an angle of \[30{}^\circ \] with the magnetic meridian. In this position, the needle will dip by an angle
A)
\[40{}^\circ \]
done
clear
B)
\[30{}^\circ \]
done
clear
C)
more than \[40{}^\circ \]
done
clear
D)
less than \[40{}^\circ \]
done
clear
View Answer play_arrow
According to Maxwell's hypothesis, a changing electric field gives rise to
A)
an e.m.f
done
clear
B)
magnetic field
done
clear
C)
electric current
done
clear
D)
pressure gradient
done
clear
View Answer play_arrow
A car moves on a circular road. It describes equal angles about the centre in equal intervals of time. Which of the following statements about the velocity of the car is true?
A)
Magnitude of velocity is not constant
done
clear
B)
Both magnitude and direction of velocity change
done
clear
C)
Velocity is directed towards the centre of the circle
done
clear
D)
Magnitude of velocity is constant but direction changes
done
clear
View Answer play_arrow
A bucket full of hot water is kept in a room and it cools from \[75{}^\circ C\] to \[70{}^\circ C\] in \[{{T}_{1}}\] minutes, from \[70{}^\circ C\] to \[65{}^\circ C\] in \[{{T}_{2}}\] minutes and from \[65{}^\circ C\] to \[60{}^\circ C\] in \[{{T}_{3}}\] minutes. Then
A)
\[{{T}_{1}}={{T}_{2}}={{T}_{3}}\]
done
clear
B)
\[{{T}_{1}}<{{T}_{2}}<{{T}_{3}}\]
done
clear
C)
\[{{T}_{1}}>{{T}_{2}}>{{T}_{3}}\]
done
clear
D)
\[{{T}_{1}}<{{T}_{2}}>{{T}_{3}}\]
done
clear
View Answer play_arrow
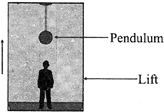
A man measures the period of a simple pendulum inside a stationary lift and finds it to be T s. If the lift accelerates upwards with an acceleration g / 4, then the period of the pendulum will be
A)
\[T\]
done
clear
B)
\[\frac{T}{4}\]
done
clear
C)
\[\frac{2T}{\sqrt{5}}\]
done
clear
D)
\[2T\sqrt{5}\]
done
clear
View Answer play_arrow
The energy of hydrogen atom in the \[{{n}^{th}}\] orbit is \[{{E}_{n}},\] then the energy in the \[{{n}^{th}}\] orbit of single ionised helium atom is
A)
\[\frac{{{E}_{n}}}{2}\]
done
clear
B)
\[2{{E}_{n}}\]
done
clear
C)
\[4{{E}_{n}}\]
done
clear
D)
\[\frac{{{E}_{n}}}{4}\]
done
clear
View Answer play_arrow
A Carnot engine absorbs 1000 J of heat energy from a reservoir at \[127{}^\circ C\] and rejects 600 J of heat energy during each cycle. The efficiency of engine and temperature of sink will be:
A)
\[20%\] and\[-\,43{}^\circ C\]
done
clear
B)
\[40%\]and\[-\,33{}^\circ C\]
done
clear
C)
\[50%\] and\[-\,20{}^\circ C\]
done
clear
D)
\[70%\]and\[-\,10{}^\circ C\]
done
clear
View Answer play_arrow
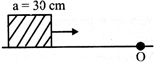
A cubical block of side \[30\,\,cm\] is moving with velocity \[2\,\,m{{s}^{-1}}\] on a smooth horizontal surface. The surface has a bump at a point 0 as shown in figure. The angular velocity (in rad/s) of the block immediately after it hits the bump, is
A)
13.3
done
clear
B)
5.0
done
clear
C)
9.4
done
clear
D)
6.7
done
clear
View Answer play_arrow
A body moves in a circular orbit of radius r under the action of a central force. Potential due to the central force is given by V(r) = kr (k is a positive constant). Period of revolution of the body is proportional to:
A)
\[{{r}^{1/2}}\]
done
clear
B)
\[{{r}^{-1/2}}\]
done
clear
C)
\[{{r}^{-3/2}}\]
done
clear
D)
\[{{r}^{-5/2}}\]
done
clear
View Answer play_arrow
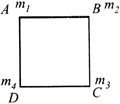
Four particles of masses \[{{m}_{1}},\]\[{{m}_{2}},\]\[{{m}_{3}}\] and \[{{m}_{4}}\] are placed at the vertices A, B, C and D as respectively of a square shown. The COM of the system will lie at diagonal AC if
A)
\[{{m}_{1}}={{m}_{3}}\]
done
clear
B)
\[{{m}_{2}}={{m}_{4}}\]
done
clear
C)
\[{{m}_{1}}={{m}_{2}}\]
done
clear
D)
\[{{m}_{3}}={{m}_{4}}\]
done
clear
View Answer play_arrow
A charge q is moving with a velocity v parallel to a magnetic field B. Force on the charge due to magnetic field is
A)
q v B
done
clear
B)
q B/v
done
clear
C)
zero
done
clear
D)
B v/q
done
clear
View Answer play_arrow
The current in a coil of L = 40 mH is to be increased uniformly from 1A to 11 A in 4 milli sec. The induced e.m.f. will be
A)
100 V
done
clear
B)
0.4 V
done
clear
C)
440 V
done
clear
D)
40 V
done
clear
View Answer play_arrow
A man projects a coin upwards from the gate of a uniformly moving train. The path of coin for the man will be
A)
parabolic
done
clear
B)
inclined straight line
done
clear
C)
vertical straight line
done
clear
D)
horizontal straight line
done
clear
View Answer play_arrow
Time required to boil 2 litres of water initially at \[20{}^\circ C\] by a heater coil which works at 80% efficiency spending 500 joule/s is
A)
82 minutes
done
clear
B)
50 minutes
done
clear
C)
28 minutes
done
clear
D)
37 minutes
done
clear
View Answer play_arrow
The frequencies of X-rays, y-rays and ultraviolet rays are respectively a, b, and c. Then
A)
\[a<b,\] \[b<c\]
done
clear
B)
\[a<b,\] \[b>c\]
done
clear
C)
\[a>b,\] \[b>c\]
done
clear
D)
\[a>b,\] \[b<c\]
done
clear
View Answer play_arrow
A ray of light is incident on the surface of separation of a medium with the velocity of light at an angle \[45{}^\circ \] and is refracted in the medium at an angle \[30{}^\circ .\] Velocity of light in the medium will be (velocity of light in air \[=3\times {{10}^{8}}m/s\])
A)
\[3.8\times {{10}^{8}}m/s\]
done
clear
B)
\[3.38\times {{10}^{8}}m/s\]
done
clear
C)
\[2.12\times {{10}^{8}}m/s\]
done
clear
D)
\[1.56\times {{10}^{8}}m/s\]
done
clear
View Answer play_arrow
A circular coil ABCD carrying a current i is placed in a uniform magnetic field. If the magnetic force' on the segment AB is \[\overrightarrow{F},\] the force on the remaining segment BCDA is
A)
\[\overrightarrow{F}\]
done
clear
B)
\[-\overrightarrow{F}\]
done
clear
C)
\[3\overrightarrow{F}\]
done
clear
D)
\[-3\overrightarrow{F}\]
done
clear
View Answer play_arrow
Which of the following statements is FALSE for a particle moving in a circle with a constant angular speed?
A)
The acceleration vector points to the centre of the circle
done
clear
B)
The acceleration vector is tangent to the circle
done
clear
C)
The velocity vector is tangent to the circle
done
clear
D)
The velocity and acceleration vectors are perpendicular to each other.
done
clear
View Answer play_arrow
In an a.c. circuit V and I are given by \[V=100\,\sin \,(100t)\,volts\] \[I=100\,\sin \,(100t+\pi /3)\,mA\] The power dissipated in the circuit is
A)
\[{{10}^{4}}\]watt
done
clear
B)
\[10\] watt
done
clear
C)
\[2.5\]watt
done
clear
D)
\[5.0\]watt
done
clear
View Answer play_arrow
Two monkeys of masses 10 kg and 8 kg are moving along a vertical rope which is light and inextensible, the former climbing up with an acceleration of \[2m/{{s}^{2}}\] while the latter coming down with a uniform velocity of \[2m/s.\] Find the tension (in newton?s).
A)
200 N
done
clear
B)
150 N
done
clear
C)
300 N
done
clear
D)
100 N
done
clear
View Answer play_arrow
The separation between successive fringes in a double slit arrangement is x. If the whole arrangement is dipped under water what will be the new fringe separation? [The wavelength of light being used is \[5000\,\,\overset{{}^\circ }{\mathop{A}}\,\]]
A)
1.5 x
done
clear
B)
x
done
clear
C)
0.75 x
done
clear
D)
2 x
done
clear
View Answer play_arrow
An acidic solution of 'X' does not give precipitate on passing \[{{H}_{2}}S\] through it. 'X' gives white precipitate when \[N{{H}_{4}}OH\] is added to it. The white precipitate dissolves in excess of \[NaOH\] solution. Pure 'X' fumes in air and dense white fumes are obtained when a glass rod dipped in \[N{{H}_{4}}OH\] is put in the fumes. Compound 'X' can be
A)
\[ZnC{{l}_{2}}\]
done
clear
B)
\[FeC{{l}_{3}}\]
done
clear
C)
\[AlC{{l}_{3}}\]
done
clear
D)
\[SnC{{l}_{2}}\]
done
clear
View Answer play_arrow
\[C{{N}^{-}}\]is a strong field ligand. This is due to the fact that
A)
it carries negative charge.
done
clear
B)
it is a pseudohalide.
done
clear
C)
it can accept electrons from metal species.
done
clear
D)
it forms high spin complexes with metal species.
done
clear
View Answer play_arrow
The amount of arsenic pentasulphide that canbe obtained when 35.5 g arsenic acid is treated with excess \[{{H}_{2}}S\] in the presence of cone. \[HCl\] (assuming 100% conversion) is :
A)
0.25 mole
done
clear
B)
0.50 mole
done
clear
C)
0.333 mole
done
clear
D)
0.125 mole
done
clear
View Answer play_arrow
Which one of the following statement is not true?
A)
pH of drinking water should be between\[5.5-9.5.\]
done
clear
B)
Concentration of DO below 6 ppm is good for the growth of fish.
done
clear
C)
Clean water would have a BOD value of less than 5 ppm.
done
clear
D)
Oxides of sulphur, nitrogen and carbon are the most widespread air pollutant.
done
clear
View Answer play_arrow
Which property of white phosphorus is common to red phosphorous?
A)
It burns when heated in air.
done
clear
B)
It reacts with hot caustic soda solution to give phosphine.
done
clear
C)
It shows chemiluminescence.
done
clear
D)
It is soluble in carbon disulphide.
done
clear
View Answer play_arrow
s The heats of neutralisation of \[C{{H}_{3}}COOH,\] \[HCOOH,\] \[HCN\] and \[{{H}_{2}}S\] are \[-13.2,\] \[-13.4,\] \[-2.9\] and \[-3.8\] kcal per equivalent respectively. Arrange the acids in increasing order of strength
A)
\[HCOOH>C{{H}_{3}}COOH>{{H}_{2}}S>HCN\]
done
clear
B)
\[C{{H}_{3}}COOH>HCOOH>{{H}_{2}}S>HCN\]
done
clear
C)
\[{{H}_{2}}S>HCOOH>C{{H}_{3}}COOH>HCN\]
done
clear
D)
\[HCOOH>{{H}_{2}}S>C{{H}_{3}}COOH>HCN\]
done
clear
View Answer play_arrow
0.4 moles of HCl and 0.2 moles of \[CaC{{l}_{2}}\] were dissolved in water to have 500 mL of solution, the molarity of \[C{{l}^{-}}\]ion is:
A)
0.8 M
done
clear
B)
1.6 M
done
clear
C)
1.2 M
done
clear
D)
10.0M
done
clear
View Answer play_arrow
In sodium fusion test of organic compounds, the nitrogen of the organic compound is converted into
A)
Sodamide
done
clear
B)
Sodium cyanide
done
clear
C)
Sodium nitrite
done
clear
D)
Sodium nitrate
done
clear
View Answer play_arrow
\[A+2B+3C\rightleftharpoons A{{B}_{2}}{{C}_{3}}\] Reaction of 6.0 g of A, \[6.0\times {{10}^{23}}\] atoms of B, and 0.036 mol ofC yields 4.8 g of compound \[A{{B}_{2}}{{C}_{3}}.\] If the atomic mass of A and C are 60 and 80 amu, respectively, the atomic mass of B is (Avogadro no. \[=6\times {{10}^{23}}mo{{l}^{-1}}\]):
A)
50 amu
done
clear
B)
60 amu
done
clear
C)
70 amu
done
clear
D)
40 amu
done
clear
View Answer play_arrow
Inductive effect involves
A)
displacement of \[\sigma \]-electrons.
done
clear
B)
delocalization of \[\pi \]-electrons.
done
clear
C)
delocalisation of \[\sigma \]-electrons.
done
clear
D)
displacement of \[\pi \]-electrons.
done
clear
View Answer play_arrow
Arrange the following elements in the order of ease of detection of wave properties, in the de Broglie experiment. H, Li, Be, B, K
A)
H<Be, B<Li<K.
done
clear
B)
H>Li>K>Be>B
done
clear
C)
H>Li>Be>B>K
done
clear
D)
H<Li<Be<B<K
done
clear
View Answer play_arrow
The decreasing order of stability of alkyl carbonium ion is in the order of:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Aluminium vessels should not be washed with materials containing washing soda since
A)
washing soda is expensive.
done
clear
B)
washing soda is easily decomposed.
done
clear
C)
washing soda reacts with Al to form soluble aluminate.
done
clear
D)
washing soda reacts with Al to form insoluble aluminium oxide.
done
clear
View Answer play_arrow
The following data are for the decomposition of ammonium nitrite in aqueous solution:
Vol. of \[{{N}_{2}}\]in cc Time (min) 6.25 10 9.00 15 11.40 20 13.65 25 35.65 Infinity
The order of rection is:
A)
Zero
done
clear
B)
One
done
clear
C)
Two
done
clear
D)
Three
done
clear
View Answer play_arrow
Which of the following reagents convert propene to 1-propanol?
A)
\[{{H}_{2}}O,\]\[{{H}_{2}}S{{O}_{4}}\]
done
clear
B)
aqueous \[KOH\]
done
clear
C)
\[MgS{{O}_{4}},\]\[NaB{{H}_{4}}/{{H}_{2}}O\]
done
clear
D)
\[{{B}_{2}}{{H}_{6}},\]\[{{H}_{2}}{{O}_{2}},\]\[O{{H}^{-}}\]
done
clear
View Answer play_arrow
A closed container contains equal number of oxygen and hydrogen molecules at a total pressure of 740 mm. If oxygen is removed from the system then pressure will:
A)
Become double of 740 mm.
done
clear
B)
Become half of 740 mm.
done
clear
C)
Become 1/9 of 740 mm.
done
clear
D)
Remains unchanged.
done
clear
View Answer play_arrow
Compound X of molecular formula \[{{C}_{4}}{{H}_{6}}\] takes up one equivalent of hydrogen in presence of Pt to form another compound Y which on ozonolysis gives only ethanoic acid. The compound X can be
A)
\[C{{H}_{2}}=CH-CH=C{{H}_{2}}\]
done
clear
B)
\[C{{H}_{2}}=C=CHC{{H}_{3}}\]
done
clear
C)
\[C{{H}_{3}}C=CC{{H}_{3}}\]
done
clear
D)
All the three
done
clear
View Answer play_arrow
1 M solution of \[C{{H}_{3}}COOH\]should be diluted to............... times so that pH is doubled.
A)
four times
done
clear
B)
\[5.55\times {{10}^{4}}\]times
done
clear
C)
\[5.55\times {{10}^{6}}\]times
done
clear
D)
\[{{10}^{-2}}\]times
done
clear
View Answer play_arrow
The following reactions take place in the blast furnace in the preparation of impure iron. Identify the reaction pertaining to the formation of the slag.
A)
\[F{{e}_{2}}{{O}_{3}}(s)+3\,CO(g)\to 2\,Fe(1)+3\,C{{O}_{2}}(g)\]
done
clear
B)
\[CaC{{O}_{3}}(s)\to CaO(s)+C{{O}_{2}}(g)\]
done
clear
C)
\[CaO(s)+Si{{O}_{2}}(s)\to CaSi{{O}_{3}}(s)\]
done
clear
D)
\[2C(s)+{{O}_{2}}(g)\to 2\,CO(g)\]
done
clear
View Answer play_arrow
Which one of the following is NOT a buffer solution?
A)
\[0.8\,M\,{{H}_{2}}\,S+0.8\,M\,KH{{S}_{+}}\]
done
clear
B)
\[2M{{C}_{6}}{{H}_{5}}N{{H}_{2}}+2M{{C}_{6}}{{H}_{5}}N\,{{H}_{3}}B{{r}^{-}}\]
done
clear
C)
\[3M{{H}_{2}}C{{O}_{3}}+3MKHC{{O}_{3}}\]
done
clear
D)
\[0.05\,M\,KCl{{O}_{4}}+0.05\,M\,HCl{{O}_{4}}\]
done
clear
View Answer play_arrow
Which of the following statements is false?
A)
Radon is obtained from the decay of radium.
done
clear
B)
Helium is inert gas.
done
clear
C)
Xenon is the most reactive among the rare gases.
done
clear
D)
The most abundant rare gas found in the atmosphere is helium.
done
clear
View Answer play_arrow
Which one of the following is expected to exhibit optical isomerism? (en = ethylenediamine)
A)
\[cis-[pt(N{{H}_{3}})\,\,C{{l}_{2}}]\]
done
clear
B)
\[trans-[pt{{(N{{H}_{3}})}_{2}}\,C{{l}_{2}}]\]
done
clear
C)
\[cis-[Co{{(en)}_{2}}\,C{{l}_{2}}]\]
done
clear
D)
\[trans-[Co{{(en)}_{2}}\,C{{l}_{2}}]\]
done
clear
View Answer play_arrow
The bromination of acetone that occurs in acid solution is represented by this equation. \[C{{H}_{3}}COC{{H}_{3}}(aq)+B{{r}_{2}}(aq)\to \] \[C{{H}_{3}}COC{{H}_{2}}Br(aq)+{{H}^{+}}(aq)+B{{r}^{-}}(aq)\] These kinetic data were obtained for given reaction concentrations.
Initial Concentrations, M Initial rate of disappearance of \[B{{r}_{2}},\]\[M{{s}^{-1}}\] \[[C{{H}_{3}}COC{{H}_{3}}]\] \[[B{{r}_{2}}]\] \[[{{H}^{+}}]\] 0.30 0.05 0.05 \[5.7\times {{10}^{-5}}\] 0.30 0.10 0.05 \[5.7\times {{10}^{-5}}\] 0.30 0.10 0.10 \[1.2\times {{10}^{-4}}\] 0.40 0.05 0.20 \[31\times {{10}^{-4}}\]
Based on given data, the rate equations is:
A)
Rate \[=k[C{{H}_{3}}COC{{H}_{3}}][{{H}^{+}}]\]
done
clear
B)
Rate\[=k[C{{H}_{3}}COC{{H}_{3}}][B{{r}_{2}}]\]
done
clear
C)
Rate\[=k[C{{H}_{3}}COC{{H}_{3}}][B{{r}_{2}}]{{[{{H}^{+}}]}^{2}}\]
done
clear
D)
Rate\[=k[C{{H}_{3}}COC{{H}_{3}}][B{{r}_{2}}][{{H}^{+}}]\]
done
clear
View Answer play_arrow
The unpaired electrons in \[Ni{{(CO)}_{4}}\] are
A)
zero
done
clear
B)
one
done
clear
C)
three
done
clear
D)
four
done
clear
View Answer play_arrow
\[[Cr{{({{H}_{2}}O)}_{6}}]C{{l}_{3}}\] (at no. of Cr = 24) has a magnetic moment of 3.83 B.M. The correct distribution of 3d electrons in the Chromium of the complex is
A)
\[3{{d}_{x{{y}^{1}}}},\] \[{{(3{{d}_{{{x}^{2}}-{{y}^{2}}}})}^{1}},\] \[3{{d}_{y{{z}^{1}}}}\]
done
clear
B)
\[3{{d}_{x{{y}^{1}}}},\] \[3{{d}_{y{{z}^{1}}}},\] \[3{{d}_{x{{z}^{1}}}}\]
done
clear
C)
\[3{{d}_{x{{y}^{1}}}},\] \[3{{d}_{y{{z}^{1}}}},\] \[3{{d}_{d{{z}^{2}}}}\]
done
clear
D)
\[{{(3{{d}_{{{x}^{2}}-{{y}^{2}}}})}^{1}},\] \[3{{d}_{\,{{z}^{\,2}}}},\] \[3{{d}_{x{{z}^{1}}}}\]
done
clear
View Answer play_arrow
Which one of the following cannot function as an oxidising agent?
A)
\[{{I}^{-}}\]
done
clear
B)
\[S(s)\]
done
clear
C)
\[NO{}_{3}^{-}(aq)\]
done
clear
D)
\[C{{r}_{2}}O{}_{7}^{2-}\]
done
clear
View Answer play_arrow
Identify the reaction which does not liberate hydrogen :
A)
Reaction of lithium hydride with\[{{B}_{2}}{{H}_{6}}.\]
done
clear
B)
Electrolysis of acidified water using Pt electrodes.
done
clear
C)
Reaction of zinc with aqueous alkali.
done
clear
D)
Allowing a solution of sodium in liquid ammonia to stand.
done
clear
View Answer play_arrow
Which of the following compounds has the highest boiling point?
A)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Cl\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}Cl\]
done
clear
C)
\[C{{H}_{3}}CH(C{{H}_{3}})C{{H}_{2}}Cl\]
done
clear
D)
\[{{(C{{H}_{3}})}_{3}}CCl\]
done
clear
View Answer play_arrow
When concentrated HCl is added to an aqueous solution of \[CoC{{l}_{2}},\] its colour changes from reddish pink to deep blue. Which complex ion gives blue colour in this reaction?
A)
\[{{[CoC{{l}_{4}}]}^{2-}}\]
done
clear
B)
\[{{[CoC{{l}_{6}}]}^{3-}}\]
done
clear
C)
\[{{[CoC{{l}_{6}}]}^{4-}}\]
done
clear
D)
\[{{[Co{{({{H}_{2}}O)}_{6}}]}^{2+}}\]
done
clear
View Answer play_arrow
Hydrogen has an lonisation energy of 1311 kJ \[mo{{l}^{-1}}\] and for chlorine it is 1256 kJ \[mo{{l}^{-1}}.\] Hydrogen forms \[{{H}^{+}}\](aq) ions but chlorine does not form \[C{{l}^{+}}\](aq) ions because.
A)
\[{{H}^{+}}\] has lower hydration enthalpy.
done
clear
B)
\[C{{l}^{+}}\]has lower hydration enthalpy.
done
clear
C)
\[Cl\] has high electron affinity.
done
clear
D)
\[Cl\]has high electronegativity.
done
clear
View Answer play_arrow
The number of enantiomers of the compound \[C{{H}_{3}}CH(Br)CH(Br)COOH\]is:
A)
2
done
clear
B)
3
done
clear
C)
4
done
clear
D)
6
done
clear
View Answer play_arrow
Equivalent weighs of \[KMn{{O}_{4}}\] acidic medium, neutral medium and concentrated alkaline medium respectively are \[\frac{M}{5},\]\[\frac{M}{1},\]\[\frac{M}{3}.\]Reduced products can be
A)
\[Mn{{O}_{2}},\]\[MnO{}_{4}^{2-},\]\[M{{n}^{2+}}\]
done
clear
B)
\[Mn{{O}_{2}},\]\[M{{n}^{2+}},\]\[MnO{}_{4}^{2-}\]
done
clear
C)
\[M{{n}^{2+}},\]\[MnO{}_{4}^{2-},\]\[Mn{{O}_{2}}\]
done
clear
D)
\[M{{n}^{2+}},\]\[Mn{{O}_{2}},\]\[MnO{}_{4}^{2-}\]
done
clear
View Answer play_arrow
Which of these have no unit?
A)
Electronegativity
done
clear
B)
Electron affinity
done
clear
C)
Ionisation energy
done
clear
D)
Excitation potential
done
clear
View Answer play_arrow
Which of the following statements is not correct for sigma and pi-bonds formed between two carbon atoms?
A)
Sigma-bond determines the direction between carbon atoms but a pi-bond has no primary effect in this regard.
done
clear
B)
Sigma-bond is stronger than a pi-bond.
done
clear
C)
Bond energies of sigma- and pi-bonds are of the order of 264 kJ/mol and 347 kJ/mol, respectively.
done
clear
D)
Free rotation of atoms about a sigma-bond is allowed but not in case of a pi-bond.
done
clear
View Answer play_arrow
The reactivity of metals with water is in the order of
A)
\[Na>Mg>Zn>Fe>Cu\]
done
clear
B)
\[Cu>Fe>Zn>Mg>Na\]
done
clear
C)
\[Mg>Zn>Na>Fe>Cu\]
done
clear
D)
\[Zn>Na>Mg>Fe>Cu\]
done
clear
View Answer play_arrow
The emf of Daniell cell at 298 K is \[{{E}_{1}}\] \[Zn|ZnS{{O}_{4}}(0.01\,M)\,\,\parallel \,\,CuS{{O}_{4}}(1.0\,M)|Cu\] When the concentration of \[ZnS{{O}_{4}}\] is 1.0 M and that of \[CuS{{O}_{4}}\] is 0.01 M, the emf changed to \[{{E}_{2}}\] What is the relation between \[{{E}_{1}}\] and \[{{E}_{2}}?\]
A)
\[{{E}_{1}}={{E}_{2}}\]
done
clear
B)
\[{{E}_{2}}=0\ne {{E}_{2}}\]
done
clear
C)
\[{{E}_{1}}>{{E}_{2}}\]
done
clear
D)
\[{{E}_{1}}<{{E}_{2}}\]
done
clear
View Answer play_arrow
In Cannizzaro reaction given below
the slowest step is :
A)
the transfer of proton to the carbonyl group
done
clear
B)
the abstraction of proton from the carboxylic group
done
clear
C)
the deprotonation of Ph
done
clear
D)
the attack of
at the carboxyl group
done
clear
View Answer play_arrow
The correct order of atomic/ionic sizes is
A)
\[N<Li<B\]
done
clear
B)
\[{{F}^{-}}<{{O}^{2-}}<{{N}^{3-}}\]
done
clear
C)
\[C{{a}^{2+}}<{{S}^{2-}}<C{{l}^{-}}\]
done
clear
D)
\[N{{a}^{+}}<M{{g}^{2+}}<C{{l}^{-}}\]
done
clear
View Answer play_arrow
The rapid change of pH near the stoichiometric point of an acid-base titration is the basis of indicator detection. pH of the solution is related to ratio of the concentrations of the conjugate acid (HIn) and base \[(I{{n}^{-}})\] forms of the indicator by the expression
A)
\[\log \frac{\left[ I{{n}^{-}} \right]}{\left[ HIn \right]}=p{{K}_{In}}-pH\]
done
clear
B)
\[\log \frac{\left[ HIn \right]}{\left[ I{{n}^{-}} \right]}=p{{K}_{In}}-pH\]
done
clear
C)
\[\log \frac{\left[ HIn \right]}{\left[ I{{n}^{-}} \right]}=pH-p{{K}_{In}}\]
done
clear
D)
\[\log \frac{\left[ I{{n}^{-}} \right]}{\left[ HIn \right]}=pH-p{{K}_{In}}\]
done
clear
View Answer play_arrow
In the diazotization of arylamines with sodium nitrite and hydrochloric acid, an excess of hydrochloric acid is used primarily to:
A)
Supress the concentration of free aniline available for coupling.
done
clear
B)
Supress hydrolysis of phenol.
done
clear
C)
Ensure a stoichiometric amount of nitrous acid.
done
clear
D)
Neutralise the base liberated.
done
clear
View Answer play_arrow
In lake test of \[A{{l}^{3+}}\] ion, there is formation of coloured floating lake. It is due to
A)
adsorption of litmus by \[{{H}_{2}}O.\]
done
clear
B)
adsorption of litmus by \[Al{{(OH)}_{3}}.\]
done
clear
C)
adsorption of litmus by \[Al(OH){}_{4}^{-}.\]
done
clear
D)
none of these.
done
clear
View Answer play_arrow
An ideal gas expands in volume from \[1\times {{10}^{-3}}\]to \[1\times {{10}^{-2}}{{m}^{3}}\] at \[300\,\,K\] against a constant pressure of \[1\times {{10}^{5}}N{{m}^{-2}}.\] The work done is
A)
\[270\,\,kJ\]
done
clear
B)
\[-900\,\,kJ\]
done
clear
C)
\[-900\,\,J\]
done
clear
D)
\[900\,\,kJ\]
done
clear
View Answer play_arrow
Which of the following fact(s) explain as to why \[p\]-nitrophenol is more acidic than phenol?
(i) \[-I\] Effect of nitro group. (ii) Greater resonance effect of \[p\]-nitrophenoxy group. (iii) Steric effect of bulky nitro group.
A)
I and ii
done
clear
B)
I and iii
done
clear
C)
ii and iii
done
clear
D)
ii alone
done
clear
View Answer play_arrow
Which of the following reactions will not result in the formation of anisole?
A)
Phenol \[+\] dimethyl sulphate in presence of a base.
done
clear
B)
Sodium phenoxide is treated with methyl iodide.
done
clear
C)
Reaction of diazomethane with phenol.
done
clear
D)
Reaction of methylmagnesium iodide with phenol.
done
clear
View Answer play_arrow
What will be the heat of formation of methane, if the heat of combustion of carbon is \['-x'\] kJ, heat of formation of water is \['-y'\] kJ and heat of combustion of methane is 'z' kJ ?
A)
\[(-x-y+z)\,kJ\]
done
clear
B)
\[(-z-x+2y)\,kJ\]
done
clear
C)
\[(-x-2y-z)\,kJ\]
done
clear
D)
\[(-x-2y+z)\,kJ\]
done
clear
View Answer play_arrow
An animal with same generic, specific and subspecific names is
A)
man
done
clear
B)
gorilla
done
clear
C)
rabbit
done
clear
D)
elephant
done
clear
View Answer play_arrow
Which of the following group of animals belongs to the same phylum?
A)
Earthworm, pinworm, tapeworm
done
clear
B)
Prawn, scorpion, Locusta
done
clear
C)
Sponge, Sea anemone, starfish
done
clear
D)
Malarial parasite, Amoeba, mosquito
done
clear
View Answer play_arrow
Match List I with List II and select the correct answer -
List I List II A. Cyanophyta (i) Chrysolaminarin B. Chlorophyata (ii) Amylopectin C. Phaeophyta (iii) Floridean starch D. Rhodophyta (iv) Starch (v) Laminarin
A)
A\[\to \](ii), B\[\to \](v), C\[\to \](i), D\[\to \](iii)
done
clear
B)
A\[\to \](ii), B\[\to \](iv), C\[\to \](v), D\[\to \](iii)
done
clear
C)
A\[\to \](i), B\[\to \](iv), C\[\to \](iii), D\[\to \](v)
done
clear
D)
A\[\to \](ii), B\[\to \](iii), C\[\to \](v), D\[\to \](i)
done
clear
View Answer play_arrow
Which one of the following is a true fruit?
A)
Apple
done
clear
B)
Pear
done
clear
C)
Cashew nut
done
clear
D)
Coconut
done
clear
View Answer play_arrow
The path of water from soil upto secondary xylem is
A)
Soil \[\to \] Root hair cell wall \[\to \] Cortex\[\to \] Endodermis \[\to \] Pericycle \[\to \] Protoxylem \[\to \] Metaxylem
done
clear
B)
Metaxylem \[\to \] Protoxylem \[\to \]Cortex \[\to \] Soil \[\to \] Root hair
done
clear
C)
Cortex \[\to \] Root hair \[\to \] Endodermis \[\to \] Pericycle \[\to \] Protoxylem \[\to \] Metaxylem
done
clear
D)
Pericycle \[\to \] Soil \[\to \] Root hair \[\to \] Cortex \[\to \] Endodermis \[\to \] Protoxylem \[\to \]Metaxylem
done
clear
View Answer play_arrow
Which of the following algae are suitable for human consumption?
A)
Laminaria and Fucus\
done
clear
B)
Gracilaria and Chondrus
done
clear
C)
Porphyra and Spirogyra
done
clear
D)
Rhodymenia and Porphyra
done
clear
View Answer play_arrow
Which of the following organelles is directly connected to the outer membrane of the nucleus in a eukaryotic cell?
A)
Mitochondrion
done
clear
B)
Lysosome
done
clear
C)
Golgi apparatus
done
clear
D)
Endoplasmic reticulum
done
clear
View Answer play_arrow
Chosse the correct combinations.
I. Hypogynous flower II. Perigynous flower III. Epigynous flower
A)
A-I, B-II, C-III
done
clear
B)
A-I, B-III, C-II
done
clear
C)
A-III, B-II, C-I
done
clear
D)
A-III, B-I, C-II
done
clear
View Answer play_arrow
Sequence of development during the formation of embryo sac is
A)
Archesporium \[\to \] Megaspore \[\to \] Megaspore mother cell \[\to \] Embryo sac.
done
clear
B)
Megasporocyte \[\to \] Archesporium \[\to \] Megaspore \[\to \] Embryo sac.
done
clear
C)
Megaspore \[\to \] Megaspore mother cell \[\to \] Archesporium \[\to \] Embryo sac.
done
clear
D)
Archesporium \[\to \] Megaspore mother cell \[\to \] Megaspore \[\to \] Embryo sac.
done
clear
View Answer play_arrow
In a fertilized ovule, n, 2n and 3n conditions occur respectively in
A)
antipodal, egg and endosperm.
done
clear
B)
egg, nucellus and endosperm.
done
clear
C)
endosperm, nucellus and egg.
done
clear
D)
antipodals, synergids and integuments.
done
clear
View Answer play_arrow
Competition for light, nutrients and space is most severe between
A)
closely related organism growing in different habitats
done
clear
B)
closely related organisms growing in the same habitat
done
clear
C)
distantly related organisms growing in the same habitat
done
clear
D)
distantly related organisms growing in different habitats
done
clear
View Answer play_arrow
From megasporocyte to egg cell, what processes are required?
A)
Meiosis followed by mitosis
done
clear
B)
Mitosis followed by meiosis
done
clear
C)
Several meiotic divisions only
done
clear
D)
Several mitotic divisions only
done
clear
View Answer play_arrow
Which one of the following pairs is not correctly matched?
A)
Production of - Burning of coal sulphur dioxide
done
clear
B)
Depletion of ozone - Release of CFCs layer in the atmosphere
done
clear
C)
Eutrophication - Increase in nitrogen and Phosphorus content in auatic bodies
done
clear
D)
Decrease in BOD - Increase in global of pond water temperature
done
clear
View Answer play_arrow
The electrostatic precipitator is used for removing particulate matter from
A)
Exhaust of the thermal power plant
done
clear
B)
Exhaust from the automobiles
done
clear
C)
Industrial effluents
done
clear
D)
Kitchen waste
done
clear
View Answer play_arrow
Keystone species in an ecosystem are those
A)
present in maximum number
done
clear
B)
that are most frequent
done
clear
C)
attaining a large biomass
done
clear
D)
contributing to ecosystem properties
done
clear
View Answer play_arrow
If the gene encoding the trp repressor is mutated such that it can no longer bind tryptophan, will transcription of the trp operon occur?
A)
Yes, because the trp represser can only bind the trp operon and block transcriptional initiation when it is bound to tryptophan
done
clear
B)
No, because this mutation does not affect the part of the repressor that can bind the operator
done
clear
C)
No, because the trp operon is repressed only when tryptophan levels are high
done
clear
D)
Yes, because the trp operon can allosterically regulate the enzymes needed to synthesize the amino acid tryptophan.
done
clear
View Answer play_arrow
What is the best pH of soil for cultivation of plants?
A)
\[3.4-5.4\]
done
clear
B)
\[6.5-7.5\]
done
clear
C)
\[4.5-8.5\]
done
clear
D)
\[5.6-6.5\]
done
clear
View Answer play_arrow
Telomerase is an enzyme which is a
A)
simple protein
done
clear
B)
RNA
done
clear
C)
ribonucleoprotein
done
clear
D)
repetitive DNA
done
clear
View Answer play_arrow
Mass of living matter at a trophic level in an area at any time is called
A)
standing crop
done
clear
B)
detritus
done
clear
C)
humus
done
clear
D)
standing state
done
clear
View Answer play_arrow
The Triticale is an intergeneric hybrid between :
A)
wheat and maize
done
clear
B)
maize and rye
done
clear
C)
wheat and rye
done
clear
D)
bajra and wheat
done
clear
View Answer play_arrow
Fishes die by sewage because
A)
of its bad smell.
done
clear
B)
it replaces food material of fishes.
done
clear
C)
it increases oxygen competition among fishes.
done
clear
D)
\[C{{O}_{2}}\] is mixed in large amounts in water.
done
clear
View Answer play_arrow
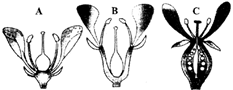
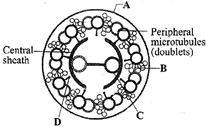
Choose the correct option.
A)
A-Plasma membrane, B-Interdoublet bridge, C-Central microtubule, D-Radial spoke
done
clear
B)
A-Plasmamemhrane, B-Arm, C-Central microtubule, D-Radial spoke
done
clear
C)
A-Plasma membrane, B-Interdoublet bridge, C-Hub, D-Radial spoke
done
clear
D)
A-Plasma membrane, B-Interdoublet bridge, C-Hub, D-Arm
done
clear
View Answer play_arrow
An ecosystem which can be easily damaged but can recover after some time if damaging effect stops will be having
A)
low stability and high resilience
done
clear
B)
high stability and low resilience
done
clear
C)
low stability and low resilience
done
clear
D)
high stability and high resilience
done
clear
View Answer play_arrow
The mode of catching insects in Drosera plants is by means of
A)
sensitive glandular hairs which secrete a sweet, viscous, shining substance.
done
clear
B)
specially sensitive trigger hairs.
done
clear
C)
leaves which are modified into pitcher.
done
clear
D)
leaf segments modified into bladder.
done
clear
View Answer play_arrow
Quantasomes are present in
A)
chloroplast
done
clear
B)
mitochondria
done
clear
C)
golgi body
done
clear
D)
lysosome
done
clear
View Answer play_arrow
What is the minimum number of cell membranes that a water molecule must move through in getting from the soil into a xylem vessel element?
A)
0
done
clear
B)
1
done
clear
C)
2
done
clear
D)
6
done
clear
View Answer play_arrow
Which of the following statements regarding photorespiration are true?
A)
Photorespiration is a metabolically expensive pathway.
done
clear
B)
Photorespiration is avoided when carbon dioxide is abundant.
done
clear
C)
Photorespiration results in a loss of usable carbon dioxide.
done
clear
D)
All of the above.
done
clear
View Answer play_arrow
A free living nitrogen-fixing cyanobacterium which can also form symbiotic association with the water fern Azolla is
A)
Tolypothrix
done
clear
B)
Chlorella
done
clear
C)
Nostoc
done
clear
D)
Anabaena
done
clear
View Answer play_arrow
Which of the following statements correctly char- acterizes changes in \[CaC{{O}_{3}}\] clay and biomass during the long-term aging of soil?
A)
\[CaC{{O}_{3}}\] gradually decreases, clay gradually increases and biomass peaks and then de- clines.
done
clear
B)
\[CaC{{O}_{3}}\]gradually decreases, clay and biom- ass gradually increase.
done
clear
C)
Clay gradually increases, \[CaC{{O}_{3}}\] and biom- ass peak and then decline.
done
clear
D)
All three gradually decrease.
done
clear
View Answer play_arrow
Krebs cycle occurs in
A)
mitochondria
done
clear
B)
cytoplasm
done
clear
C)
chloroplasts
done
clear
D)
ribosomes
done
clear
View Answer play_arrow
Most abundant organic compound on earth is
A)
Protein
done
clear
B)
Cellulose
done
clear
C)
Lipids
done
clear
D)
Steroids
done
clear
View Answer play_arrow
An inhibitor is added to a cell culture so that succinate accumulates. The enzyme catalyzing the formation of which substance has been blocked?
A)
Citrate
done
clear
B)
Oxaloacetate
done
clear
C)
\[\alpha \]-ketoglutarate
done
clear
D)
Fumarate
done
clear
View Answer play_arrow
To avoid excessive water loss during severe drought stress, the closure of stomata is signaled by the production of
A)
IAA
done
clear
B)
NAA
done
clear
C)
ABA
done
clear
D)
IBA
done
clear
View Answer play_arrow
The amount of DNA in a mammalian cell in early prophase I is x. What is the amount of DNA in the same cell in anaphase I of meiosis?
A)
\[\frac{x}{4}\]
done
clear
B)
\[\frac{x}{2}\]
done
clear
C)
\[x\]
done
clear
D)
\[2x\]
done
clear
View Answer play_arrow
The major reason that glycolysis is not as energy productive as respiration is that
A)
\[NA{{D}^{+}}\] is regenerated by alcohol or lactate production, without the high-energy electrons passing through the electron transport chain.
done
clear
B)
it is the pathway common to fermentation and respiration.
done
clear
C)
it does not take place in a specialized membrane-bound organelle.
done
clear
D)
pyruvate is more reduced than \[C{{O}_{2}};\] it still contains much of die energy from glucose.
done
clear
View Answer play_arrow
All the following statements are true with regard to Km value, except:
A)
Km is denned as the substrate concentration at half maximal velocity
done
clear
B)
Then 50% of enzyme molecules are bound with substrate molecules
done
clear
C)
Km is independent of enzyme concentration
done
clear
D)
As numerical value of Km is more, the affinity of enzyme to substrate is more
done
clear
View Answer play_arrow
Biodiversity Act of India was passed by the parliament in the year
A)
1992
done
clear
B)
1996
done
clear
C)
2000
done
clear
D)
2002
done
clear
View Answer play_arrow
Which of the following plants is used as biofertilizer?
A)
Nostoc
done
clear
B)
Funaria
done
clear
C)
Volvox
done
clear
D)
Rhizopus
done
clear
View Answer play_arrow
Which one thing is not true about antibiotics?
A)
The term "antibiotic" was coined by Selman Waksman in 1942
done
clear
B)
First antibiotic was discovered by Alexander Flemming
done
clear
C)
Each antibiotic is effective only against one particular kind of germ
done
clear
D)
Some persons can be allergic to a particular antibiotic
done
clear
View Answer play_arrow
Select false statements for an enzyme promoting a chemical reaction by
(i) lowering the energy of activation. (ii) causing the release of heat, which acts as a primer. (iii) increasing molecular motion. (iv) changing the free energy difference between substrate and product.
A)
(i) and (iv)
done
clear
B)
(ii) and (iii)
done
clear
C)
(ii),(iii) and (iv)
done
clear
D)
(iii) and (iv)
done
clear
View Answer play_arrow
The common nitrogen fixer in paddy fields is
A)
Rhizobium
done
clear
B)
Azospirillum
done
clear
C)
Oscillatoria
done
clear
D)
Frankia
done
clear
View Answer play_arrow
In order to obtain virus- free plants through tissue culture, the best method is
A)
protoplast culture
done
clear
B)
embryo rescue
done
clear
C)
anther culture
done
clear
D)
meristem culture
done
clear
View Answer play_arrow
Rennet is purified by C. Hansen (1874) for commercial use. This enzyme is extracted from
A)
Stomach of goat
done
clear
B)
Stomach of horse
done
clear
C)
Stomach of calf
done
clear
D)
Bacteria
done
clear
View Answer play_arrow
\[{{H}_{3}}\overset{+}{\mathop{N}}\,-\overset{\overset{R}{\mathop{|}}\,}{\mathop{C}}\,\underset{(A)}{\mathop{H}}\,-COOH{{H}_{3}}\overset{+}{\mathop{N}}\,-\overset{R}{\mathop{\overset{|}{\mathop{C}}\,}}\,\underset{(B)}{\mathop{H}}\,-CO{{O}^{-}}\] \[{{H}_{2}}N-\overset{R}{\mathop{\overset{|}{\mathop{C}}\,}}\,\underset{(C)}{\mathop{H}}\,-CO{{O}^{-}}\] Which of the above is Zwitterionic form?
A)
A
done
clear
B)
C
done
clear
C)
B
done
clear
D)
All of these
done
clear
View Answer play_arrow
Restriction endonucleases are enzymes which
A)
make cuts at specific positions within the DNA molecule
done
clear
B)
recognize a specific nucleotide sequence for binding of DNA ligase
done
clear
C)
restrict the action of the enzyme DNA polymerase
done
clear
D)
remove nucleotides from the ends of the DNA molecule
done
clear
View Answer play_arrow
Sex factor in bacteria is
A)
Chromosomal replicon
done
clear
B)
F-replicon
done
clear
C)
RNA
done
clear
D)
Sex-pilus
done
clear
View Answer play_arrow
Animals/organisms floating on the surface of water are
A)
plankton
done
clear
B)
pelagic
done
clear
C)
benthos
done
clear
D)
neritic
done
clear
View Answer play_arrow
Read the following 4-statements (i - iv) and accordingly mark the option that has both correct statements.
(i) Cardiac fibres are branched with one or more nuclei. (ii) Smooth muscles are unbranched and cylindrical. (iii) Striated muscles can be branched or unbranched. (iv) Involuntary muscles are non-striated.
A)
(i) and (iv)
done
clear
B)
(ii) and (iii)
done
clear
C)
(iii) and (iv)
done
clear
D)
(i) and (iii)
done
clear
View Answer play_arrow
Spleen is referred to as
A)
temporary endocrine gland
done
clear
B)
graveyard of RBC
done
clear
C)
largest gland
done
clear
D)
store house of WBC
done
clear
View Answer play_arrow
Given below are four matching?s of an animal and its kind of respiratory organ:
(i) Silver fish trachea (ii) Scorpion book lung (iii) Sea squirt pharyngeal gills (iv) Dolphin skin The correct matchings are
A)
(iii) and (iv)
done
clear
B)
(i) and (iv)
done
clear
C)
(i), (ii) and (iii)
done
clear
D)
(ii) and (iv)
done
clear
View Answer play_arrow
In the mouthparts of the cockroach, the organ of mastication is The correct matchings are
A)
labium
done
clear
B)
maxillae
done
clear
C)
mandibles
done
clear
D)
labrum
done
clear
View Answer play_arrow
Which one of the following characters is not typical of the class Mammalia?
A)
Thecodont dentition
done
clear
B)
Alveolar lungs
done
clear
C)
Ten pairs of cranial nerves
done
clear
D)
Seven cervical vertebrae
done
clear
View Answer play_arrow
Natural parthenogenesis occurs in:
A)
Protozoans
done
clear
B)
Earthworm
done
clear
C)
All insects
done
clear
D)
Honeybee
done
clear
View Answer play_arrow
Consider the statements given below regarding contraception and answer as directed thereafter:
(i) Medical Termination of Pregnancy (MTP) during first trimester is generally safe (ii) Generally chances of conception are nil until mother breast-feeds the infant upto two years (iii) Intrauterine devices like copper-T are effective contraceptives (iv) Contraception pills may be taken upto one week after coitus to prevent conception Which two of the above statements are correct?
A)
(ii) and (iii)
done
clear
B)
(iii) and (iv)
done
clear
C)
(i) and (iii)
done
clear
D)
(i)and(ii)
done
clear
View Answer play_arrow
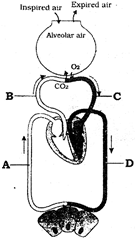
Identify the blood vessels A to D.
A)
A-Systemic vein, B-Pulmonary artery, C-Pulmonary vein, D-Systemic artery
done
clear
B)
A-Systemic artery, B-Pulmonary artery, C- Pulmonary vein, D- Systemic vein
done
clear
C)
A-Pulmonary artery, B-Systemic vein, C-Pulmonary vein, D- Systemic artery
done
clear
D)
A-Systemic vein, B-Pulmonary vein, C- Pulmonary artery, D- Systemic artery
done
clear
View Answer play_arrow
Which pathway of the male reproductive system is correct for the sperms transportation?
A)
Vas enerentia\[\to \]Vas deferens\[\to \]Epididymis
done
clear
B)
Vas deferens\[\to \]Epididymis\[\to \]Seminal vesicle
done
clear
C)
Epididymis\[\to \]Vas deferens\[\to \]Urethra
done
clear
D)
Rete testis\[\to \]Epididymis\[\to \]Vas efferentia
done
clear
View Answer play_arrow
The second maturation division of the mammalian ovum occurs:
A)
in the Graafian follicle following the first maturation division
done
clear
B)
Shortly after ovulation before the ovum makes entry into the fallopian tube
done
clear
C)
Until after the ovum has been penetrated by a sperm
done
clear
D)
Until the nucleus of the sperm has fused with that of the ovum
done
clear
View Answer play_arrow
A force acting against achievement of highest possible level of population growth is
A)
Carrying capacity
done
clear
B)
Environment resistance
done
clear
C)
Population pressure
done
clear
D)
Saturation level
done
clear
View Answer play_arrow
The phase of menstrual cycle in humans that lasts for 7-8 days, is
A)
follicular phase
done
clear
B)
ovulatory phase
done
clear
C)
luteal phase
done
clear
D)
menstruation
done
clear
View Answer play_arrow
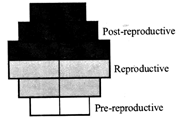
What type of human population is represented by the following age pyramid?
A)
Vanishing population
done
clear
B)
Stable population
done
clear
C)
Declining population
done
clear
D)
Expanding population
done
clear
View Answer play_arrow
The 'Mule' is the result of
A)
Inbreeding depression
done
clear
B)
Out breeding
done
clear
C)
Cross breeding
done
clear
D)
Inter-specific hybridization
done
clear
View Answer play_arrow
Haemophilia is more common in males because it is a
A)
Recessive character carried by Y-chromosome
done
clear
B)
Dominant character carried by Y-chromosome
done
clear
C)
Dominant trait carried by X-chromosome
done
clear
D)
Recessive trait carried by X-chromosome
done
clear
View Answer play_arrow
Theory of inheritance of acquired characters was given by
A)
Wallace
done
clear
B)
Lamarck
done
clear
C)
Darwin
done
clear
D)
De Vries
done
clear
View Answer play_arrow
The animal husbandry deals with the care, breeding and management of
A)
Domesticated animals
done
clear
B)
Fishes
done
clear
C)
Honey bees and silk worms
done
clear
D)
All of these
done
clear
View Answer play_arrow
'Inland fishery' refers to
A)
Culturing fish in fresh water
done
clear
B)
Trapping and capturing fishes from sea coast
done
clear
C)
Deep sea fishing
done
clear
D)
Extraction of oil from fishes
done
clear
View Answer play_arrow
Sphincter of Oddi controls the flow of digestive juice by guarding which duct?
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
Which two ducts are responsible for the formation of a duct that carry bile from the gall bladder and conduct it into the first section of the small intestine?
A)
1 and 2
done
clear
B)
2 and 3
done
clear
C)
3 and 4
done
clear
D)
4 and 1
done
clear
View Answer play_arrow
Pacemaker of heart is
A)
AV node
done
clear
B)
Bundle of His
done
clear
C)
SA node
done
clear
D)
Purkinje fibres
done
clear
View Answer play_arrow
Uricotelism is found in
A)
Frogs and toads
done
clear
B)
Mammals and birds
done
clear
C)
Birds, reptiles and insects
done
clear
D)
Fishes and fresh water protozoans
done
clear
View Answer play_arrow
A large proportion of oxygen is left unused in the human blood even after its uptake by the body tissues. This\[{{O}_{2}}\]
A)
acts as a reserve during muscular exercise
done
clear
B)
raise the \[pC{{O}_{2}}\] of blood to 75 mm of Hg.
done
clear
C)
is enough to keep oxyhaemoglobin saturation at 96%
done
clear
D)
helps in releasing more\[{{O}_{2}}\] to the epithelial tissues.
done
clear
View Answer play_arrow
The basic functional unit of the human kidney is
A)
nephron
done
clear
B)
nephridia
done
clear
C)
pyramid
done
clear
D)
Henle's loop
done
clear
View Answer play_arrow
Urea from the blood can be removed by
A)
uremia
done
clear
B)
diuresis
done
clear
C)
dialysis
done
clear
D)
micturition
done
clear
View Answer play_arrow
What prevents molecules with a molecular mass greater than 68000 passing from the glomerular capillaries into Bowman's capsule?
A)
the basement membrane of the epithelial cells of Bowman's capsule
done
clear
B)
the cell surface membrane of the endothelial cells of the capillaries
done
clear
C)
the cell surface membrane of the epithelial cells of Bowman's capsule
done
clear
D)
the spaces between the extensions of the podocytes of Bowman's capsule
done
clear
View Answer play_arrow
The nerve centres which control the body temperature and the urge for eating are contained in:
A)
hypothalamus
done
clear
B)
pons
done
clear
C)
cerebellum
done
clear
D)
thalamus
done
clear
View Answer play_arrow
Rods and cones of eyes are modified
A)
multipolar neuron
done
clear
B)
unipolar neuron
done
clear
C)
bipolar neuron
done
clear
D)
None of these
done
clear
View Answer play_arrow
A patient with a pituitary tumor might suffer with polyuria (production of too much urine) as a result of decreased secretion of
A)
MSH
done
clear
B)
FSH
done
clear
C)
aldosterone
done
clear
D)
ADH
done
clear
View Answer play_arrow
The sensation of fatigue in the muscles after prolonged strenuous physical work, is caused by
A)
a decrease in the supply of oxygen
done
clear
B)
minor wear and tear of muscle fibres
done
clear
C)
the depletion of glucose
done
clear
D)
the accumulation of lactic acid
done
clear
View Answer play_arrow
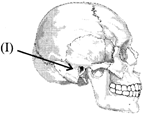
In the given diagram of skull, what does "I" represent?
A)
Frontal bone
done
clear
B)
Temporal bone
done
clear
C)
Occipital bone
done
clear
D)
Parietal bone
done
clear
View Answer play_arrow
External gills, tracheae, and lungs all share which of the following sets of characteristics?
A)
Part of gas-exchange system, exchange both \[C{{O}_{2}}\] and \[{{O}_{2}};\] increase surface area for diffusion.
done
clear
B)
Used by water breathers; based on countercurrent exchange; use negative pressure breathing.
done
clear
C)
Exchange only \[{{O}_{2}};\] are associated with a circulatory system; found in vertebrates.
done
clear
D)
Found in insects; employ positive-pressure pumping based on crosscurrent flow.
done
clear
View Answer play_arrow
Which one of the following does not act as a neurotransmitter?
A)
Epinephrine
done
clear
B)
Norepinephrine
done
clear
C)
Cortisone
done
clear
D)
Acetylcholine
done
clear
View Answer play_arrow
Which one of the following statements is correct?
A)
Neurons regulate endocrine activity, but not vice versa.
done
clear
B)
Endocrine glands regulate neural activity and nervous system regulates endocrine glands.
done
clear
C)
Neither hormones control neural activity nor the neurons control endocrine activity.
done
clear
D)
Endocrine glands regulate neural activity but not vice versa.
done
clear
View Answer play_arrow
'Cloning'is meant for/to
A)
production of hGH gene in E. coli
done
clear
B)
preserve the genotype of organism
done
clear
C)
replace the original gene
done
clear
D)
All of the above
done
clear
View Answer play_arrow
A cell coded protein formed in response to infection with most animal viruses is
A)
Antigen
done
clear
B)
Antibody
done
clear
C)
Interferon
done
clear
D)
Histohe
done
clear
View Answer play_arrow
Which one of the following is not used in organic farming?
A)
Glomus
done
clear
B)
Earthworm
done
clear
C)
Oscillatoria
done
clear
D)
Snail
done
clear
View Answer play_arrow
ELISA is used to detect viruses where the key reagent is
A)
RNase
done
clear
B)
alkaline phosphatase
done
clear
C)
catalase
done
clear
D)
DNA probe
done
clear
View Answer play_arrow
Which bacterium helps in the production of 'Swiss cheese'?
A)
Propionibacterium sharmanii
done
clear
B)
Trichoderma polysporum
done
clear
C)
Saccharomyces cerevisiae
done
clear
D)
Aspergillus niger
done
clear
View Answer play_arrow
Which of the following pairs is not correctly matched?
A)
Dengue fever ? Arbovirus
done
clear
B)
Plague - Yersinia pestis
done
clear
C)
Syphilis - Trichuris trichura
done
clear
D)
Sleeping sickness - Trypanosoma gambiense
done
clear
View Answer play_arrow
Which one of the following depresses brain activity and produced feelings of calmness, relaxation and drowsiness?
A)
Morphine
done
clear
B)
'Valium
done
clear
C)
Amphetamines
done
clear
D)
Hashish
done
clear
View Answer play_arrow
Which one of the following is correctly matched pair of the given secretion and its primary role in human physiology?
A)
Sebum - Sexual attraction
done
clear
B)
Sweat ? Thermoregulation
done
clear
C)
Saliva - Tasting food
done
clear
D)
Tears - Excretion of salts
done
clear
View Answer play_arrow
Consider the following four statements (i-iv) and select the option which includes all the correct ones only.
(i) Single cell Spirulina can produce large quantities of food rich in protein, minerals, vitamins etc. (ii) Body weight-wise the micro-organism Methylophilus methylotrophus may be able to produce several times more proteins than the cows per day. (iii) Common button mushrooms are a very rich source of vitamin C. (iv) A rice variety has been developed which is very rich in calcium.
A)
Statements (ii) and (iv)
done
clear
B)
Statements (i), (iii) and (iv)
done
clear
C)
Statements (ii), (iii) and (iv)
done
clear
D)
Statements (i) and (ii)
done
clear
View Answer play_arrow