The angles which a vector \[\widehat{i}+\widehat{j}+\sqrt{2}\,\widehat{k}\] makes with X, Y and Z axes respectively are
A)
\[60{}^\circ ,\text{ }60{}^\circ ,\text{ }60{}^\circ \]
done
clear
B)
\[45{}^\circ ,\text{ }45{}^\circ ,\text{ }45{}^\circ \]
done
clear
C)
\[60{}^\circ ,\text{ }60{}^\circ ,\text{ }45{}^\circ \]
done
clear
D)
\[45{}^\circ ,\text{ }45{}^\circ ,\text{ }60{}^\circ \]
done
clear
View Answer play_arrow
The surface tension of a liquid is 70 dyne/cm. In MKS system its value is
A)
\[70\,\,N/m\]
done
clear
B)
\[7\,\,\times \,\,{{10}^{-\,2}}\,N/m\]
done
clear
C)
\[7\,\,\times \,\,{{10}^{3}}\,N/m\]
done
clear
D)
\[7\times {{10}^{2}}\,N/m\]
done
clear
View Answer play_arrow
A ball of mass 0.1 kg is suspended by a string. It is displaced through an angle of \[60{}^\circ \] and left. When the ball passes through the mean position, the tension in the string is
A)
19.6 N
done
clear
B)
1.96 N
done
clear
C)
9.8 N
done
clear
D)
zero
done
clear
View Answer play_arrow
A block can slide on a smooth inclined plane of inclination \[\theta \] kept on the floor of a lift. When the lift is descending with a retardation a, the acceleration of the block relative to the incline is
A)
\[\left( g+a \right)\,\sin \,\theta ~\]
done
clear
B)
\[\left( g-a \right)\]
done
clear
C)
\[g\,\,sin\,\,\theta \]
done
clear
D)
\[\left( g-a \right)\text{ }sin\,\theta \]
done
clear
View Answer play_arrow
A force of 19.6 N when applied parallel to the surface just moves a body of mass 10 kg kept on a horizontal surface. If a 5 kg mass is kept on the first mass, the force applied parallel to the surface to just move the combined body is
A)
29.4 N
done
clear
B)
39.2 N
done
clear
C)
18.6 N
done
clear
D)
42.6 N
done
clear
View Answer play_arrow
Work done in time t on a body of mass m which is accelerated from rest to a speed v in time \[{{t}_{1}}\], as a function of time t is given by
A)
\[\frac{1}{2}m\frac{v}{{{t}_{1}}}\,{{t}^{2}}\]
done
clear
B)
\[m\frac{v}{{{t}_{1}}}\,{{t}^{2}}\]
done
clear
C)
\[\frac{1}{2}{{\left( \frac{mv}{{{t}_{1}}} \right)}^{2}}\,{{t}^{2}}\]
done
clear
D)
\[\frac{1}{2}m\frac{{{v}^{2}}}{{{t}_{1}}^{2}}\,\,{{t}^{2}}\]
done
clear
View Answer play_arrow
If a planet consists of a satellite whose mass and radius were both half that of the earth, the acceleration due to gravity at its surface would be \[(g\,on\text{ }earth=9.8\text{ }m/se{{c}^{2}})\]
A)
\[4.9\text{ }m/se{{c}^{2}}\]
done
clear
B)
\[8.9\text{ }m/se{{c}^{2}}\]
done
clear
C)
\[19.6\text{ }m/se{{c}^{2}}\]
done
clear
D)
\[29.4\text{ }m/se{{c}^{2}}\]
done
clear
View Answer play_arrow
A soap film of surface tension \[3\,\,\times \,\,{{10}^{-}}^{2}\,N{{m}^{-}}^{1}\] formed in rectangular frame, can support a straw. The length of the film is 10 cm. Mass of the straw the film can support is
A)
0.06 g
done
clear
B)
0.6 g
done
clear
C)
6 g
done
clear
D)
60 g
done
clear
View Answer play_arrow
Suppose ideal gas equation follows \[V{{P}^{3}}=\]constant. Initial temperature and volume of the gas are T and V respectively. If gas expand to 27 V then its temperature will become
A)
T
done
clear
B)
9T
done
clear
C)
27T
done
clear
D)
T/9
done
clear
View Answer play_arrow
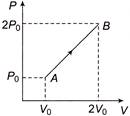
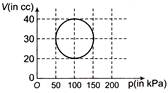
The \[P-V\] diagram of 2 g of helium gas for a certain process \[A\to B\] is shown in the figure. What is the heat given to the gas during the process\[A\to B\]?
A)
\[4\,\,{{P}_{O}}{{V}_{O}}\]
done
clear
B)
\[6\,\,{{P}_{O}}{{V}_{O}}\]
done
clear
C)
\[4.5\,\,{{P}_{O}}{{V}_{O}}\]
done
clear
D)
\[2\,\,{{P}_{O}}{{V}_{O}}\]
done
clear
View Answer play_arrow
Two vessels of different materials are similar in size in every respect. The same quantity of ice filled in them gets melted in 20 minutes and 40 minutes respectively. The ratio of thermal conductivities of the materials is
A)
\[5\,\,:\,\,6\]
done
clear
B)
\[6:5\]
done
clear
C)
\[3 : 1\]
done
clear
D)
\[2 : 1\]
done
clear
View Answer play_arrow
A pendulum has time period T in air. When it is made to oscillate in water, it acquired a time period\[T'=\sqrt{2}T\]. The density of the pendulum bob is equal to (density of water = 1)
A)
\[\sqrt{2}\]
done
clear
B)
2
done
clear
C)
\[2\sqrt{2}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
A wave travelling along positive x-axis is / / given by y == A sin (cot - he). If it is reflected (y from rigid boundary such that 80% amplitude is reflected, then equation of reflected wave is
A)
\[y = A\,\,sin\left( \omega t + kx \right)\]
done
clear
B)
\[y = -0.8\,A\,\,sin\left( \omega t + kx \right)\]
done
clear
C)
\[y = 0.8\,A\,\,sin\left( \omega t + kx \right)\]
done
clear
D)
\[y = A\,\,sin\left( \omega t +0.8 kx \right)\]
done
clear
View Answer play_arrow
Two charges each equal to \[\eta q({{\eta }^{-1}}\,<\,\,\sqrt{3})\] are placed at the comers of an equilateral triangle of side a. The electric field at the third comer is \[{{E}_{3}}\,where\,\left( {{E}_{0}}=q/4\pi {{\varepsilon }_{0}}{{a}^{2}} \right)\]
A)
\[E{{ }_{3}}={{E}_{0}}\]
done
clear
B)
\[E{{ }_{3}}<{{E}_{0}}\]
done
clear
C)
\[E{{ }_{3}}>{{E}_{0}}\]
done
clear
D)
\[E{{ }_{3}}\ge {{E}_{0}}\]
done
clear
View Answer play_arrow
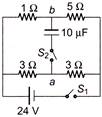
In the circuit shown in figure, switch \[{{S}_{1}}\] is initially closed and \[{{S}_{2}}\] is open. Find\[{{V}_{a}}-{{V}_{b}}\].
A)
4 V
done
clear
B)
8 V
done
clear
C)
(e) 12 V
done
clear
D)
16 V
done
clear
View Answer play_arrow
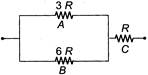
The three resistances A, B and C have values 3R, 6R and R respectively. When some potential difference is applied across the network, the thermal powers dissipated by A, B and C are in ratio
A)
\[2 : 3 : 4\]
done
clear
B)
\[2 : 4 : 3\]
done
clear
C)
\[4 : 2 : 3\]
done
clear
D)
\[3 : 2 : 4\]
done
clear
View Answer play_arrow
In the hydrogen atom, the electron is making\[6.6\,\,\times \,\,{{10}^{15}}r.p.s.\text{ }\]If the radius of the orbit is \[0.53\,\,\times \,\,{{10}^{10}}\,metre\text{ }\] then magnetic field produced the centre of the orbit is
A)
140 Tesla
done
clear
B)
12.5 Tesla
done
clear
C)
1.4 Tesla
done
clear
D)
0.14 Tesla
done
clear
View Answer play_arrow
The distance between the poles of a horse shoe magnet is 0.1 m and its pole strength is 0.01 amp-m. The induction of magnetic field at a point midway between the poles will be
A)
\[2\times {{10}^{-5}}\,T\]
done
clear
B)
\[4\times {{10}^{-6}}\,T\]
done
clear
C)
\[8\times {{10}^{-7}}\,T\]
done
clear
D)
zero
done
clear
View Answer play_arrow
A coil of Cu wire (radius--r, self-inductance---L) is bent in two concentric turns each having radius\[r/2\]. The self-inductance now
A)
2 L
done
clear
B)
L
done
clear
C)
4 L
done
clear
D)
L/2
done
clear
View Answer play_arrow
The r.m.s. current in an ac circuit is 2 A. If the wattless current be\[\sqrt{3}\,A\], what is the power factor?
A)
\[\frac{1}{\sqrt{3}}\]
done
clear
B)
\[\frac{1}{\sqrt{2}}\]
done
clear
C)
\[\frac{1}{2}\]
done
clear
D)
\[\frac{1}{3}\]
done
clear
View Answer play_arrow
An electron is moving through a field. It is moving (i) opposite an electric field (ii) perpendicular to a magnetic field as shown. For each situation the de-Broglie wave length of electron
A)
increasing, increasing
done
clear
B)
increasing, decreasing
done
clear
C)
decreasing, same
done
clear
D)
same, Same
done
clear
View Answer play_arrow
The energy level diagram for a hydrogen like atom is shown in the figure. The radius of its first Bohr orbit is
A)
\[0.265\text{ }\overset{{}^\circ }{\mathop{A}}\,\]
done
clear
B)
\[0.53\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[0.132\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
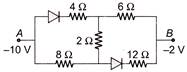
In the following circuit the equivalent resistance between A and B is
A)
\[\frac{20}{3}\,\Omega \]
done
clear
B)
\[10\,\,\Omega \]
done
clear
C)
\[16\,\,\Omega \]
done
clear
D)
\[20\,\,\Omega \]
done
clear
View Answer play_arrow
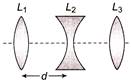
Three lenses \[{{L}_{1}},\,\,{{L}_{2}},\,\,{{L}_{3}}\] are placed co-axially as shown in figure. Focal lengths of lenses are / given 30 cm, 10 cm and 5 cm respectively. If a parallel beam of light falling on lens \[{{L}_{1}}\] emerging \[{{L}_{3}}\] as a convergent beam such that it converges at the focus of\[{{L}_{3}}\] . Distance between \[{{L}_{1}}\,and\,\,{{L}_{2}}\] will be
A)
40 cm
done
clear
B)
30 cm
done
clear
C)
2.0 cm
done
clear
D)
10 cm
done
clear
View Answer play_arrow
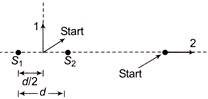
Following figure shows sources \[{{S}_{1}}\,\,and\,\,{{S}_{2}}\] that emit light of wavelength \[\lambda \] in all directions. The sources are exactly in phase and are separated by a distance equal to\[1.5\,\,\lambda \]. If we start at the indicated start point and travel along path 1 and 2, the interference produces a maxima all along
A)
path 1
done
clear
B)
path 2
done
clear
C)
any path
done
clear
D)
none of these
done
clear
View Answer play_arrow
A solid sphere and a hollow sphere of equal mass and radius, are placed over a rough horizontal surface after rotating it about its mass centre with same angular velocity\[{{\omega }_{0}}\]. Once the pure rolling starts let \[{{\nu }_{1}}\,\,and\,\,{{\nu }_{2}}\] be the linear speeds of their centres of mass. Then
A)
\[{{\nu }_{1}}={{\nu }_{2}}\]
done
clear
B)
\[{{\nu }_{1}}>{{\nu }_{2}}\]
done
clear
C)
\[{{\nu }_{1}}<{{\nu }_{2}}\]
done
clear
D)
data is insufficient
done
clear
View Answer play_arrow
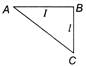
Figure shows a thin metallic triangular sheet ABC. The sides AB and BC are of equal lengths l. The mass of the sheet is M. What is its moment of inertia about AC?
A)
\[\frac{M{{l}^{2}}}{18}\]
done
clear
B)
\[\frac{M{{l}^{2}}}{12}\]
done
clear
C)
\[\frac{M{{l}^{2}}}{6}\]
done
clear
D)
\[\frac{M{{l}^{2}}}{4}\]
done
clear
View Answer play_arrow
A student performs an experiment to determine the Young?s modulus of a wire, exactly 2 m long, by Searle?s method. In a particular reading, the student measures the extension in the length of the wire to be 0.8 mm with an uncertainty of 0.05 mm, at a load of exactly kg. The student also measures the diameter of the wire to be 0.4 mm with an uncertainty of 0.01 mm. Take \[\operatorname{g}= 9.8 m/{{s}^{2}}\] (exact). The Young?s modulus obtained from the reading is
A)
\[(2.0\,\,\pm \,\,0.3)\,\,\times \,\,{{10}^{11}}\,N/{{m}^{2}}\]
done
clear
B)
\[(2.0\,\,\pm \,\,0.2)\,\,\times \,\,{{10}^{11}}\,N/{{m}^{2}}\]
done
clear
C)
\[(2.0\,\,\pm \,\,0.1)\,\,\times \,\,{{10}^{11}}\,N/{{m}^{2}}\]
done
clear
D)
\[(2.0\,\,\pm \,\,0.05)\,\,\times \,\,{{10}^{11}}\,N/{{m}^{2}}\]
done
clear
View Answer play_arrow
A particle is projected from a point O with a velocity u in a direction making an angle \[\theta \] upward with the horizontal. After some time at point P it is moving at right angle with its initial direction of projection. The time of flight from O to P is
A)
\[\frac{u\,\sin \,\alpha }{g}\]
done
clear
B)
\[\frac{u\,\,\cos ec\,\alpha }{g}\]
done
clear
C)
\[\frac{u\,\tan \,\alpha }{g}\]
done
clear
D)
\[\frac{u\,\sec \,\alpha }{g}\]
done
clear
View Answer play_arrow
The rest mass of the deuteron, is equivalent to an energy of 1876 MeV, the rest mass of proto equivalent to 939 MeV and that of a neutron to 40 Mev. A deuteron may disintegrate to a proton and a neutron if it
A)
emits an X-ray photon of energy 2 MeV
done
clear
B)
captures an X-ray photon of energy 2 MeV
done
clear
C)
emits an X-ray photon of energy 3 MeV
done
clear
D)
captures an X-ray photon of energy 3 MeV
done
clear
View Answer play_arrow
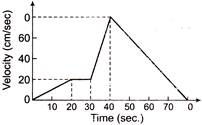
The v-t graph of a moving object is given in figure. The maximum acceleration is
A)
\[1\,\,cm/se{{c}^{2}}\]
done
clear
B)
\[2\,\,cm/se{{c}^{2}}\]
done
clear
C)
\[3\,\,cm/se{{c}^{2}}\]
done
clear
D)
\[6\,cm/se{{c}^{2}}\]
done
clear
View Answer play_arrow
A system is taken through a cyclic process represented by a circle as shown. The heat absorbed by the system is
A)
\[\pi \times {{10}^{3}}J\]
done
clear
B)
\[\frac{\pi }{2}J\]
done
clear
C)
\[4\pi \,\,\times \,\,{{10}^{2}}J\]
done
clear
D)
\[{{\pi }^{2}}J\]
done
clear
View Answer play_arrow
Two capillary tubes of same diameter are put vertically one each in two liquids whose relative densities are 0.8 and 0.6 and surface tensions are 60 and 50 dyne/cm respectively Ratio of heights of liquids in the two tubes \[{{h}_{1}}/{{h}_{2}}\] is
A)
\[\frac{10}{9}\]
done
clear
B)
\[\frac{3}{10}\]
done
clear
C)
\[\frac{10}{3}\]
done
clear
D)
\[\frac{9}{10}\]
done
clear
View Answer play_arrow
The distance of the centres of moon and earth is D. The mass of earth is 81 times the mass of the moon. At what distance from the centre of the earth, the gravitational force will be zero
A)
\[\frac{D}{2}\]
done
clear
B)
\[\frac{2\,D}{3}\]
done
clear
C)
\[\frac{4\,D}{3}\]
done
clear
D)
\[\frac{9\,D}{10}\]
done
clear
View Answer play_arrow
A particle P is moving in a circle of radius ?a? with a uniform speed\[\nu \]. C is the centre of the circle and AB is a diameter. When passing through B the angular velocity of P about A and C are in the ratio
A)
\[1\,\,:\,\,1\]
done
clear
B)
\[1\,\,:\,\,2\]
done
clear
C)
\[2\,\,:\,\,1\]
done
clear
D)
\[4\,\,:\,\,1\]
done
clear
View Answer play_arrow
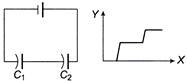
Figure shows two capacitors connected in series and joined to a battery. The graph in figure shows the variation in potential as one moves from left to right on the branch containing the capacitors, if
\[{{C}_{1}}\] \[{{C}_{2}}\]
A)
\[{{C}_{1}}>{{C}_{2}}\]
done
clear
B)
\[{{C}_{1}}={{C}_{2}}\]
done
clear
C)
\[{{C}_{1}}<{{C}_{2}}\]
done
clear
D)
the information is not sufficient to decide the relation between \[{{C}_{1}}\,and\,{{C}_{3}}\]
done
clear
View Answer play_arrow
Electric bulb 50 W-100 V glowing at full power are to be used in parallel with battery 120 V, \[10\,\,\Omega \]. Maximum number of bulbs that can be connected so that they glow in full power is
A)
2
done
clear
B)
8
done
clear
C)
4
done
clear
D)
6
done
clear
View Answer play_arrow
A coil of\[40\,\,\Omega \], resistance has 100 turns and radius 6 mm is connected to ammeter of resistance of 160 ohms. Coil is placed perpendicular to the magnetic field. When coil is taken out of the field, \[32\,\mu C\] charge flows through it. The intensity of magnetic field will be
A)
6.55 T
done
clear
B)
5.66 T
done
clear
C)
0.655 T
done
clear
D)
0.566 T
done
clear
View Answer play_arrow
The focal length of objective and eye lens of a microscope are 4 cm and 8 cm respectively. If the least distance of distinct vision is 24 cm and object distance is 4.5 cm from the objective lens, then the magnifying power of the microscope will be
A)
18
done
clear
B)
32
done
clear
C)
64
done
clear
D)
20
done
clear
View Answer play_arrow
200 Me V of energy may be obtained per fission\[{{U}^{235}}\]. A reactor is generating 1000 kW of power. The rate of nuclear fission in the reactor is
A)
1000
done
clear
B)
\[2\,\,\times {{10}^{8}}\]
done
clear
C)
\[3.125\,\,\times {{10}^{16}}\]
done
clear
D)
931
done
clear
View Answer play_arrow
A beam of light of \[\lambda \,\,=\,\,600\,nm\] from a distant source falls on a single slit 1 mm wide and the resulting diffraction pattern is observed on a screen 2 m away. The distance between first dark fringes on either side of the central bright fringe is
A)
1.2 cm
done
clear
B)
1.2 mm
done
clear
C)
2.4 cm
done
clear
D)
2.4 mm
done
clear
View Answer play_arrow
If particles are moving with the same velocity, then maximum de-Broglie wavelength will be for
A)
Neutron
done
clear
B)
Proton
done
clear
C)
\[\beta -particle\]
done
clear
D)
\[\alpha -particle\]
done
clear
View Answer play_arrow
The momentum of a photon of energy 1 MeV in kg m/s will be
A)
\[0.33 \times 1{{0}^{6}}\]
done
clear
B)
\[7 \times 1{{0}^{-24}}\]
done
clear
C)
\[1{{0}^{-22}}\]
done
clear
D)
\[5 \times 1{{0}^{-22}}\]
done
clear
View Answer play_arrow
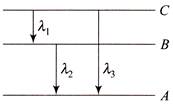
Energy levels A, B, C of a certain atom corresponding to increasing values of energy, i.e., \[{{E}_{A}}<{{E}_{B}}<{{E}_{C}}\]. If \[{{\lambda }_{1}},\,\,{{\lambda }_{2}},\,\,{{\lambda }_{3}}\] are the wavelengths of radiations corresponding to the transitions C to B, B to A and C to A respectively, which of the following statements is correct.
A)
\[{{\lambda }_{3}}=\,\,{{\lambda }_{1}}+\,\,{{\lambda }_{2}}\]
done
clear
B)
\[{{\lambda }_{3}}=\,\,\frac{{{\lambda }_{1}}{{\lambda }_{2}}}{{{\lambda }_{1}}+{{\lambda }_{2}}}\]
done
clear
C)
\[{{\lambda }_{1}}+{{\lambda }_{2}}+{{\lambda }_{3}}=0\]
done
clear
D)
\[\lambda _{3}^{2}\,=\,\lambda _{1}^{2}\,\,+\,\lambda _{2}^{2}\]
done
clear
View Answer play_arrow
Two radioactive materials \[{{X}_{1}}\,\,and\,\,{{X}_{2}}\] have decay constants \[5\lambda \,\,and\,\,\lambda \] respectively. If initially they have the same number of nuclei, then the ratio of the number of nuclei of \[{{X}_{1}}\] to that of \[{{X}_{2}}\] will be \[\frac{1}{e}\] after a time.
A)
\[\lambda \]
done
clear
B)
\[\frac{1}{2}\,\,\lambda \]
done
clear
C)
\[\frac{1}{4\lambda }\]
done
clear
D)
\[\frac{e}{\lambda }\]
done
clear
View Answer play_arrow
Dimensions of pressure are same as that of
A)
energy
done
clear
B)
force
done
clear
C)
energy per unit volume
done
clear
D)
force per unit volume
done
clear
View Answer play_arrow
In the final answer of the expression \[\frac{\left( 29.2-20.2 \right)\left( 1.79\times {{10}^{5}} \right)}{1.37}\]. The number of significant figures is
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
Cathode rays are
A)
protons
done
clear
B)
electrons
done
clear
C)
neutrons
done
clear
D)
\[\alpha -particles\]
done
clear
View Answer play_arrow
Heaviest particle is
A)
meson
done
clear
B)
neutron
done
clear
C)
proton
done
clear
D)
electron
done
clear
View Answer play_arrow
The electronic configuration of four elements L, P, Q and R are given in brackets
\[L\left( 1{{s}^{2}},\text{ }2{{s}^{2}}\,2{{p}^{4}} \right);\text{ }Q\left( 1{{s}^{2}},\,\,2{{s}^{2}}\,\,2{{p}^{6}},\text{ }3{{s}^{2}}\,3{{p}^{5}} \right)\] \[P\left( 1{{s}^{2}},\text{ }2{{s}^{2}}\,\,2{{p}^{6}},\text{ }3{{s}^{1}} \right);\,\,R\left( 1{{s}^{2}},2{{s}^{2}}\,\,2{{p}^{6}},\text{ }3{{s}^{2}} \right)\] The formulae of ionic compounds that can be formed between these elements are
A)
\[{{L}_{2}}P,\,\,RL,\,\,PQ\,\,and\,\,{{R}_{2}}Q\]
done
clear
B)
LP, RL, PQ and RQ
done
clear
C)
\[{{P}_{2}}L,\,\,RL,\,\,PQ\,\,and\,\,R{{Q}_{2}}\]
done
clear
D)
\[LP,\,\,{{R}_{2}}L,\,\,{{P}_{2}}Q\,\,and\,\,RQ\]
done
clear
View Answer play_arrow
Electrovalent bond formation depends on
A)
ionization energy
done
clear
B)
electron affinity
done
clear
C)
lattice energy
done
clear
D)
all the three above
done
clear
View Answer play_arrow
If 1 M and 2.5 litre NaOH solution is mixed with another 0.5 M and 3 litre NaOH solution, then molarity of the resultant solution will be
A)
1.0 M
done
clear
B)
0.73 M
done
clear
C)
0.80 M
done
clear
D)
0.50 M
done
clear
View Answer play_arrow
The molarity of a solution made by mixing 50 ml of cone. \[{{H}_{2}}S{{O}_{4}}\,(36\,\,N)\] with 50 ml of water is
A)
36 M
done
clear
B)
18 M
done
clear
C)
9 M
done
clear
D)
6 M
done
clear
View Answer play_arrow
A crystalline solid
A)
changes abruptly from solid to liquid when heated
done
clear
B)
has no definite melting point
done
clear
C)
undergoes deformation of its geometry easily.
done
clear
D)
has an irregular 3-dimensional arrangements
done
clear
View Answer play_arrow
The existence of a substance in more than one solid modifications is known as or any compound having more than two crystal structure is called
A)
polymorphism
done
clear
B)
isomorphism
done
clear
C)
allotropy
done
clear
D)
enantiomorphism
done
clear
View Answer play_arrow
Kinetic energy of molecules is highest in
A)
gases
done
clear
B)
solids
done
clear
C)
liquids
done
clear
D)
solutions
done
clear
View Answer play_arrow
Pressure of a gas in a vessel can be measured by
A)
barometer
done
clear
B)
manometer
done
clear
C)
stalgometer
done
clear
D)
all the above
done
clear
View Answer play_arrow
Deuterons when bombarded on a nuclide produce \[_{18}A{{g}^{38}}\] and neutrons. The target is
A)
\[_{17}C{{l}^{35}}\]
done
clear
B)
\[_{19}C{{l}^{27}}\]
done
clear
C)
\[_{17}C{{l}^{37}}\]
done
clear
D)
\[_{19}C{{l}^{39}}\]
done
clear
View Answer play_arrow
The radionuclide \[_{90}^{234}Th\] undergoes two successive \[\beta \text{-}decay\] followed by one\[\alpha \text{-}decay\]. The atomic number and the mass number respectively of the resulting radionuclide are
A)
92 and 234
done
clear
B)
94 and 230
done
clear
C)
90 and 230
done
clear
D)
92 and 230
done
clear
View Answer play_arrow
A reversible reaction is one which
A)
proceeds in one direction
done
clear
B)
proceeds in both directions
done
clear
C)
proceeds spontaneously
done
clear
D)
all the statements are wrong
done
clear
View Answer play_arrow
The reaction \[CaC{{O}_{3}}\,\,\rightleftharpoons \,\,CaO+C{{O}_{2}}\,(g)\] goes to completion in lime kiln because
A)
of the high temperature
done
clear
B)
CaO is more stable than \[CaC{{O}_{3}}\]
done
clear
C)
CaO is not dissociated
done
clear
D)
\[C{{O}_{2}}\] escapes continuously
done
clear
View Answer play_arrow
A monoprotic acid in 1.00 M solution is \[0.01\,%\] ionised. The dissociation constant of this acid is
A)
\[1\,\,\times \,\,{{10}^{-}}^{8}\]
done
clear
B)
\[1\,\,\times \,\,{{10}^{-}}^{4}\]
done
clear
C)
\[1\,\,\times \,\,{{10}^{-}}^{6}\]
done
clear
D)
\[{{10}^{-}}^{5}\]
done
clear
View Answer play_arrow
Which one is strongest electrolyte in the following?
A)
NaCl
done
clear
B)
\[C{{H}_{3}}COOH~\]
done
clear
C)
\[N{{H}_{4}}OH\]
done
clear
D)
\[{{C}_{6}}{{H}_{12}}{{O}_{6}}\]
done
clear
View Answer play_arrow
Internal energy of an ideal gas depends on
A)
volume
done
clear
B)
temperature
done
clear
C)
pressure
done
clear
D)
none of these
done
clear
View Answer play_arrow
Any series of operations so carried out that at the end, the system is back to its initial state is called
A)
Boyle?s cycle
done
clear
B)
Reversible process
done
clear
C)
Adiabatic process
done
clear
D)
Cyclic process
done
clear
View Answer play_arrow
In a catalytic conversion of \[{{N}_{2}}\,to\text{ }N{{H}_{3}}\] by Haber?s process, the rate of reaction was expressed as change in the concentration of ammonia per time is\[40\,\,\times \,\,{{10}^{-}}^{3}mol\text{ }litr{{e}^{-}}^{1}{{s}^{-}}^{1}\]. If there are no side reaction, the rate of the reaction as expressed in terms of hydrogen is \[\left( in\text{ }mol\text{ }litr{{e}^{-}}^{1}{{s}^{-}}^{1} \right)\]
A)
\[60\times {{10}^{-}}^{3}\]
done
clear
B)
\[20\times {{10}^{-}}^{3}\]
done
clear
C)
1.200
done
clear
D)
\[10.3\times {{10}^{-}}^{3}\]
done
clear
View Answer play_arrow
If the concentration of the reactants is increased, the rate of reaction
A)
remains unaffected
done
clear
B)
increases
done
clear
C)
decreases
done
clear
D)
may increase or decrease
done
clear
View Answer play_arrow
Which one of the following metals could not be obtained on electrolysis of aqueous solution of its salts
A)
Ag
done
clear
B)
Mg
done
clear
C)
Cu
done
clear
D)
Cr
done
clear
View Answer play_arrow
On the electrolysis of aqueous solution of sodium sulphate, on cathode we get
A)
Na
done
clear
B)
\[{{H}_{2}}\]
done
clear
C)
\[S{{O}_{2}}\]
done
clear
D)
\[S{{O}_{3}}\]
done
clear
View Answer play_arrow
\[H{{ }_{2}}{{O}_{2}}\] reduces \[{{K}_{4}}Fe(CN)6\]
A)
in neutral solution
done
clear
B)
in acidic solution
done
clear
C)
in non-polar solvent
done
clear
D)
in alkaline solution
done
clear
View Answer play_arrow
When sodium metal is dissolved in liquid ammonia, blue colour solution is formed. The blue colour is due to
A)
\[solvated\text{ }N{{a}^{+}}\,\,ions\]
done
clear
B)
solvated electrons
done
clear
C)
\[solvated\text{ }N{{a}^{-}}_{2}\,\,ions\]
done
clear
D)
solvated protons
done
clear
View Answer play_arrow
When the temperature is raised, the viscosity of liquid decreases, this is because of
A)
decreased volume of the solution
done
clear
B)
increase in temperature increases the average kinetic energy of molecules, which overcome the attractive force between them
done
clear
C)
decreased covalent and hydrogen bond forces
done
clear
D)
increased attraction between molecules
done
clear
View Answer play_arrow
According to Langmuir adsorption isotherm, the amount of gas adsorbed at very high pressures
A)
reaches a constant limiting value
done
clear
B)
goes on increasing with pressure
done
clear
C)
goes on decreasing with pressure
done
clear
D)
increases first and decreases later with pressure
done
clear
View Answer play_arrow
The tenth elements in the periodic table resembles with the
A)
first period
done
clear
B)
second period
done
clear
C)
fourth group
done
clear
D)
ninth group
done
clear
View Answer play_arrow
Who developed the long form of periodic table?
A)
Lothar Meyer
done
clear
B)
Niels Bohr
done
clear
C)
Mendeleef
done
clear
D)
Moseley
done
clear
View Answer play_arrow
An example of halide ore is
A)
galena
done
clear
B)
bauxite
done
clear
C)
cinnabar
done
clear
D)
cryolite
done
clear
View Answer play_arrow
Which of the following is not an ore?
A)
Bauxite
done
clear
B)
Malachite
done
clear
C)
Zinc blende
done
clear
D)
Pig iron
done
clear
View Answer play_arrow
Which of the following can adsorb largest volume of hydrogen gas?
A)
Finely divided platinum
done
clear
B)
Finely divided nickel
done
clear
C)
Colloidal palladium
done
clear
D)
Colloidal platinum
done
clear
View Answer play_arrow
Hydrogen does not combine with
A)
antimony
done
clear
B)
sodium
done
clear
C)
bismuth
done
clear
D)
helium
done
clear
View Answer play_arrow
Which statement is true about the transitional elements?
A)
They are highly reactive
done
clear
B)
They show variable oxidation states
done
clear
C)
They have low M.P.
done
clear
D)
They are highly electropositive
done
clear
View Answer play_arrow
Which of the following has highest ionic radii?
A)
\[C{{r}^{+\,3}}\]
done
clear
B)
\[M{{n}^{+\,3}}\]
done
clear
C)
\[F{{e}^{+\,3}}\]
done
clear
D)
\[Co{{\,}^{+\,3}}\]
done
clear
View Answer play_arrow
The coordination number of copper in cuprammonium sulphate is
A)
2
done
clear
B)
6
done
clear
C)
4
done
clear
D)
- 4
done
clear
View Answer play_arrow
The coordination number of cobalt in the complex \[\left[ Co{{(en)}_{2}}\,B{{r}_{2}} \right]\,C{{l}_{2}}\] is
A)
2
done
clear
B)
6
done
clear
C)
5
done
clear
D)
4
done
clear
View Answer play_arrow
To prepare a pure sample of n-hexane using sodium metal as one reactant, the other reactant will be
A)
n-propyl bromide
done
clear
B)
ethyl bromide and n-butyl bromide
done
clear
C)
ethyl chloride and n -butyl chloride
done
clear
D)
methyl bromide and n -pentyl chloride
done
clear
View Answer play_arrow
Sodium acetate can be converted to ethane by
A)
heating with \[LiAl{{H}_{4}}\]
done
clear
B)
electrolysing its aqueous solution
done
clear
C)
heating with sodalime
done
clear
D)
heating with calcium acetate
done
clear
View Answer play_arrow
When chlorine is passed over dry slaked lime at room temperature, the main reaction product is
A)
\[Ca{{\left( Cl{{O}_{2}} \right)}_{2}}\]
done
clear
B)
\[CaC{{l}_{2}}\]
done
clear
C)
\[CaOC{{l}_{2}}\]
done
clear
D)
\[Ca{{\left( OC{{l}_{2}} \right)}_{2}}\]
done
clear
View Answer play_arrow
The noble gas which forms maximum number of compounds is
A)
Ar
done
clear
B)
He
done
clear
C)
Xe
done
clear
D)
Ne
done
clear
View Answer play_arrow
Among the following series of transition metal ions, the one where all metal ions have \[3{{d}^{2}}-\]electronic configuration is
A)
\[T{{i}^{3\,+}},\,\,{{V}^{2\,+}},\,\,C{{r}^{3\,+}},\,\,M{{n}^{4\,+}}\]
done
clear
B)
\[T{{i}^{\,+}},\,\,{{V}^{4\,+}},\,\,C{{r}^{6\,+}},\,\,M{{n}^{7\,+}}\]
done
clear
C)
\[T{{i}^{\,4+}},\,\,{{V}^{3\,+}},\,\,C{{r}^{2\,+}},\,\,M{{n}^{3\,+}}\]
done
clear
D)
\[T{{i}^{\,2+}},\,\,{{V}^{3\,+}},\,\,C{{r}^{4\,+}},\,\,M{{n}^{5\,+}}\]
done
clear
View Answer play_arrow
\[KMn{{O}_{4}}\] can be prepared from \[{{K}_{2}}Mn{{O}_{4}}\] as per the reaction \[3Mn{{O}_{4}}^{2}~\,+\,\,2{{H}_{2}}O\,\,\rightleftharpoons \,\,Mn{{O}_{4}}^{-}+Mn{{O}_{2}}\,+\,\,4O{{H}^{-}}\] The reaction can go to completion by removing \[O{{H}^{-}}\] ions by adding.
A)
KLOH
done
clear
B)
CO
done
clear
C)
\[S{{O}_{2}}\]
done
clear
D)
HCl
done
clear
View Answer play_arrow
Which is the correct order of increasing energy of the listed orbitals in the atom of titanium?
A)
3s 3p 3d 4s
done
clear
B)
3s 3p 4s 3d
done
clear
C)
3s 4s 3p 3d
done
clear
D)
4s 3s 3p 3d
done
clear
View Answer play_arrow
Cartilage is formed by
A)
chondrocytes
done
clear
B)
osteoblasts
done
clear
C)
osteoclasts
done
clear
D)
fibroblasts
done
clear
View Answer play_arrow
Achondroplasia is a disease related with the defect in the formation of
A)
membrane
done
clear
B)
cartilage
done
clear
C)
bone
done
clear
D)
mucosa
done
clear
View Answer play_arrow
Philadelphia chromosome is found in the patient suffering from
A)
albinism
done
clear
B)
insomnia
done
clear
C)
myelocytic leukaemia
done
clear
D)
hepatitis
done
clear
View Answer play_arrow
Diabetes insipidus occurs due to the hyposecretion of
A)
thymosine
done
clear
B)
oxytocin
done
clear
C)
insulin
done
clear
D)
vasopressin
done
clear
View Answer play_arrow
Steroid hormones are almost similar in structure to
A)
triglyceride
done
clear
B)
tyrosine
done
clear
C)
coenzyme-A
done
clear
D)
cholesterol
done
clear
View Answer play_arrow
The golden age of reptiles was
A)
mesozoic era
done
clear
B)
palaeozoic era
done
clear
C)
proterozoic era
done
clear
D)
coenozoic era
done
clear
View Answer play_arrow
Colour blindness, in which all colours are perceived as gray, is termed as
A)
monochromasia
done
clear
B)
chromasia
done
clear
C)
dichromasia
done
clear
D)
all of these
done
clear
View Answer play_arrow
Which of the following layer of epidermis m man provides the main protection of body against water loss and the entry of disease causing organisms?
A)
stratum lucidium
done
clear
B)
stratum spirosum
done
clear
C)
stratum germinativum
done
clear
D)
stratum comeum
done
clear
View Answer play_arrow
Which of the following cell type is capable of giving rise to other cell types in sponges?
A)
archaeocytes
done
clear
B)
pinacocytes
done
clear
C)
collehcytes
done
clear
D)
thesocytes
done
clear
View Answer play_arrow
Outer covering of cartilage is known as
A)
endosteum
done
clear
B)
perichondrium
done
clear
C)
peritoneum
done
clear
D)
periosteum
done
clear
View Answer play_arrow
Which of the following is made up of a single bone in mammals?
A)
lower jaw
done
clear
B)
hyoid
done
clear
C)
zygomatic arch
done
clear
D)
upper jaw
done
clear
View Answer play_arrow
The daughter born to haemophilic father and normal mother could be
A)
haemophilic
done
clear
B)
normal
done
clear
C)
carrier
done
clear
D)
all of these
done
clear
View Answer play_arrow
Schuffner?s dots are seen in red blood corpuscles of man due to which of the following disease?
A)
malaria
done
clear
B)
kala-azar
done
clear
C)
diabetes
done
clear
D)
filaria
done
clear
View Answer play_arrow
The contractile element present in a striated muscle fibril, between two successive Z-lines, is called
A)
sarcomere
done
clear
B)
sarcoplasm
done
clear
C)
sarcosomes
done
clear
D)
all of these
done
clear
View Answer play_arrow
Which of the following is a vestigial structure in python?
A)
hind limbs
done
clear
B)
teeth
done
clear
C)
poison glands
done
clear
D)
scales
done
clear
View Answer play_arrow
Which of the following cranial nerve of man is both sensory and motor?
A)
olfactory
done
clear
B)
optic
done
clear
C)
trigeminal
done
clear
D)
vagus
done
clear
View Answer play_arrow
Which of the following is respiratory organ of scorpion?
A)
gills
done
clear
B)
lungs
done
clear
C)
ctenidia
done
clear
D)
book lungs
done
clear
View Answer play_arrow
Which of the following is an essential fatty acid in mammals?
A)
palmitic acid
done
clear
B)
stearic acid
done
clear
C)
Gamma-linolenic acid
done
clear
D)
Acetic acid
done
clear
View Answer play_arrow
The state, during which the respiratory centre is inhibited, is termed as
A)
anoxia
done
clear
B)
asphyxia
done
clear
C)
suffocation
done
clear
D)
choking
done
clear
View Answer play_arrow
Glissonian cirrhosis is a disease related with
A)
liver
done
clear
B)
lung
done
clear
C)
pancreas
done
clear
D)
spleen
done
clear
View Answer play_arrow
Which of the following does not produce any digestive enzyme?
A)
pancreas
done
clear
B)
mouth
done
clear
C)
gastric mucosa
done
clear
D)
liver
done
clear
View Answer play_arrow
Zonula adherens is a kind of
A)
filament
done
clear
B)
desmosome
done
clear
C)
membrane
done
clear
D)
mesosome
done
clear
View Answer play_arrow
Lymphoid tissue is found in
A)
lymph nodes
done
clear
B)
thymus
done
clear
C)
tonsils
done
clear
D)
all of these
done
clear
View Answer play_arrow
Hamburger?s phenomenon is also known as
A)
chloride shift mechanism
done
clear
B)
sodium-potassium pump
done
clear
C)
carbonic acid shift mechanism
done
clear
D)
Hydrogen shift mechanism
done
clear
View Answer play_arrow
Which of the following carries blood rich in food materials, such as glucose and ammo acids, from intestine to liver?
A)
renal portal vein
done
clear
B)
dorsal aorta
done
clear
C)
hepatic portal vein
done
clear
D)
Mesenteric artery
done
clear
View Answer play_arrow
Bacteria with flagella all over its body, is called
A)
monotrichous
done
clear
B)
amphitrichous
done
clear
C)
lophotrichous
done
clear
D)
peritrichous
done
clear
View Answer play_arrow
Which of the following terms represents a pair of contrasting character?
A)
homozygous
done
clear
B)
allele
done
clear
C)
(e) heterozygous
done
clear
D)
phenotypes
done
clear
View Answer play_arrow
?Genera Plantarum? was written by
A)
Hutchinson
done
clear
B)
Bessey
done
clear
C)
Bentham and Hooker
done
clear
D)
Linnaeus
done
clear
View Answer play_arrow
A set of bacterial clones, each containing a plasmid or phage, is called
A)
gene library
done
clear
B)
gene pool
done
clear
C)
genophore
done
clear
D)
genome
done
clear
View Answer play_arrow
The transfer of genetic material of one bacterium to another by virus is called
A)
transcription
done
clear
B)
translation
done
clear
C)
replication
done
clear
D)
transduction
done
clear
View Answer play_arrow
Maize has ten pairs of chromosomes. How many linkage groups will be present, if all the genes are mapped?
A)
20
done
clear
B)
5
done
clear
C)
40
done
clear
D)
10
done
clear
View Answer play_arrow
The complete of chromosome, inherited as a single unit, from one parent, is known as
A)
genome
done
clear
B)
linkage
done
clear
C)
gene pool
done
clear
D)
genotype
done
clear
View Answer play_arrow
Which of the following are initiator codons?
A)
UGA and UAG
done
clear
B)
UUU and UUC
done
clear
C)
AUG and GUG
done
clear
D)
UAA and UAG
done
clear
View Answer play_arrow
A pigment, which absorbs red and far red light.
A)
cytochrome
done
clear
B)
phytochrome
done
clear
C)
xanthophyll
done
clear
D)
carotene
done
clear
View Answer play_arrow
The epistgatic effect, in which the dihybrid cross \[9:3:3:1\] between Aa Bb? Aa bb is modified as
A)
dominance of one allele on another allele of both loci
done
clear
B)
interaction between two alleles of different loci
done
clear
C)
dominance of one allele on another allele of same loci
done
clear
D)
interaction between two alleles of same loci
done
clear
View Answer play_arrow
Which of the following is known as resurrection plant?
A)
Rafflesia
done
clear
B)
Selaginella
done
clear
C)
Chlorella
done
clear
D)
Welwitschia
done
clear
View Answer play_arrow
The jumping genes are called
A)
cistrons
done
clear
B)
mutons
done
clear
C)
transposons
done
clear
D)
recons
done
clear
View Answer play_arrow
The leaves of Mimosa pudica droop when touched due to
A)
seismonasty
done
clear
B)
photonasty
done
clear
C)
epinasty
done
clear
D)
nyctinasty
done
clear
View Answer play_arrow
When pollen grains are not transferred from anthers to stigma in a flower, due to the barrier, it is called
A)
cleistogamy
done
clear
B)
herkogamy
done
clear
C)
dichogamy
done
clear
D)
heterogamy
done
clear
View Answer play_arrow
The pairing of homologous chromosomes in meiosis is known as
A)
bivalent
done
clear
B)
synapsis
done
clear
C)
disjunction
done
clear
D)
synergids
done
clear
View Answer play_arrow
Nodules with nitrogen fixing bacteria are present in
A)
wheat
done
clear
B)
cotton
done
clear
C)
mustard
done
clear
D)
gram
done
clear
View Answer play_arrow
Which of the following explains, how progeny can possess the combinations of traits that none of the parent possessed?
A)
law of segregation
done
clear
B)
chromosome theory
done
clear
C)
Law of independent assortment
done
clear
D)
polygenic inheritance
done
clear
View Answer play_arrow
The presence of continuous phenotypic variation in an \[{{F}_{1}}\]-generation suggests that a character is inherited by
A)
gene linkage
done
clear
B)
epistasis
done
clear
C)
polygenic inheritance
done
clear
D)
recombination
done
clear
View Answer play_arrow
The new strand synthesised, in small pieces and then joined together during DNA replication, is called
A)
dead strand
done
clear
B)
lagging strand
done
clear
C)
leading stand
done
clear
D)
all of these
done
clear
View Answer play_arrow
Parkinsonia is a good example of
A)
winged fruit
done
clear
B)
phyllode
done
clear
C)
parachute mechanism
done
clear
D)
phylloclade
done
clear
View Answer play_arrow
Inflorescence, which begins as a dichasial cyme and ends in a monochasial cyme, is called
A)
cyathium
done
clear
B)
biparous
done
clear
C)
verticillaster
done
clear
D)
thyrsus
done
clear
View Answer play_arrow
Cyanobacteria is a new name for
A)
mycoplasma
done
clear
B)
Nostoc
done
clear
C)
myxophyceae
done
clear
D)
myxomycetes
done
clear
View Answer play_arrow
The thallus of Volvox is called
A)
coenocyte
done
clear
B)
filament
done
clear
C)
heterotrichous
done
clear
D)
coenobium
done
clear
View Answer play_arrow
The similarity between bacterium and cyanobacterium is in the presence of
A)
chloroplast
done
clear
B)
flagella
done
clear
C)
80 S ribosomes
done
clear
D)
nucleoid
done
clear
View Answer play_arrow
The five-kingdom classification was suggested by
A)
Engler and Prantl
done
clear
B)
Eichler
done
clear
C)
Bentham and Hooker
done
clear
D)
Whittaker
done
clear
View Answer play_arrow
Male and female gametophytes are independent and free living in:
A)
Mustard
done
clear
B)
Castor
done
clear
C)
Finns
done
clear
D)
Sphagnum
done
clear
View Answer play_arrow
Which one of the following pairs is wrongly matched?
A)
Salvinia?Prothallus
done
clear
B)
Viroids?RNA
done
clear
C)
Mustard?Synergids
done
clear
D)
Ginkgo?Archegonia
done
clear
View Answer play_arrow
Which one of the following is a matching set of phylum and its three examples?
A)
Cnidaria?Bonellia, Physalia, Aurelia
done
clear
B)
Platyhelminthes?Planaria, Schistosoma, Enterobius
done
clear
C)
Mollusca?Loligo, Teredo, Octopus
done
clear
D)
Porifera?Spongilla, Euplectella, Pennatula
done
clear
View Answer play_arrow
Which one of the following pairs of animals comprises ?Jawless fishes??
A)
Guppies and hag fishes
done
clear
B)
Lampreys and eels
done
clear
C)
Mackerels and rohu
done
clear
D)
Lampreys and hag fishes
done
clear
View Answer play_arrow
Which of the following animals is not viviparous?
A)
Whale
done
clear
B)
Flying fox (Bat)
done
clear
C)
Elephant
done
clear
D)
Platypus
done
clear
View Answer play_arrow
An example of axile placentation is
A)
Marigold
done
clear
B)
Argemone
done
clear
C)
Dianthus
done
clear
D)
Lemon
done
clear
View Answer play_arrow
Among bitter gourd, mustard, brinjal, pumpkin, China rose, lupin, cucumber, sunn hemp, gram, guava, bean, chili, plum, petunia, tomato, rose, withania, potato, onion, aloe and tulip how many plants have hypogynous flower?
A)
Six
done
clear
B)
Ten
done
clear
C)
Fifteen
done
clear
D)
Eighteen
done
clear
View Answer play_arrow
Among china rose, mustard, brinjal, potato, guava, cucumber, onion and tulip, how many plants have superior ovary?
A)
Four
done
clear
B)
Five
done
clear
C)
Six
done
clear
D)
Three
done
clear
View Answer play_arrow
One very special feature in the earthworm pheretima is that:
A)
It has a long dorsal tubular heart
done
clear
B)
Fertilisation of eggs occurs inside the body
done
clear
C)
The typhlosole greatly increases the effective absorption area of the digested food in the intestine
done
clear
D)
The S-shaped state embedded in the integument are the defensive weapons used against the enemies
done
clear
View Answer play_arrow
Select the option which is not correct with respect to enzyme action:
A)
Substrate binds with enzyme at its active site.
done
clear
B)
Addition of lot of succinate does not reverse the inhibition of succinic dehy-drogenase by malonate
done
clear
C)
A non-competitive inhibitor binds the enzyme at a site distinct from that which binds the substrate
done
clear
D)
Malonate is a competitive inhibitor of succinic dehydrogenase
done
clear
View Answer play_arrow
During the formation of bread it becomes porous due to release of \[C{{O}_{2}}\] by the action of:
A)
Yeast
done
clear
B)
Bacteria
done
clear
C)
Virus
done
clear
D)
Protozoans
done
clear
View Answer play_arrow
Viruses are no more ?alive? than isolated chromosomes because:
A)
they require both RNA and DNA
done
clear
B)
they both need food molecules
done
clear
C)
they both require oxygen for respiration
done
clear
D)
both require the environment of a cell to replicate
done
clear
View Answer play_arrow
Auxospores and hormocysts are formed respectively by
A)
Some cyanobacteria and many diatoms
done
clear
B)
Several diatoms and a few cyanobacteria
done
clear
C)
Several cyanobacteria and several diatoms
done
clear
D)
Some diatoms and several cyanobacteria
done
clear
View Answer play_arrow
Thermococcus, Methanococcus and Metha- nobacterium exemplify:
A)
bacteria whose DNA is relaxed or positively supercoiled but which have cytoskeleton as well as mitochondria
done
clear
B)
bacteria that contain a cytoskeleton and ribosomes
done
clear
C)
archaebacteria that contain protein homologous to eukaryotic core histones
done
clear
D)
archaebacteria that lack any histones resembling those found in eukaryotes but whose DNA is negatively supercoiled
done
clear
View Answer play_arrow
Single-celled eukaryotes are included in:
A)
Protista
done
clear
B)
Fungi
done
clear
C)
Archaea
done
clear
D)
Monera
done
clear
View Answer play_arrow
Poly some is formed by:
A)
a ribosome with several subunits
done
clear
B)
ribosomes attached to each other in a linear arrangement
done
clear
C)
several ribosomes attached to a single mRNA
done
clear
D)
many ribosomes attached to a strand endoplasmic reticulum
done
clear
View Answer play_arrow
Given below is a sample of a portion of DNA strand giving the base sequence on the opposite strands. What is so special shown in it?
5?................. GAATTC................. 3? 3?................. CTTAAG................. 5?
A)
Palindromic sequence of base pairs
done
clear
B)
Replication completed
done
clear
C)
Deletion mutation
done
clear
D)
Start codon at the 5? end
done
clear
View Answer play_arrow
Which structures perform the function of mitochondria in bacteria?
A)
Nucleoid
done
clear
B)
Ribosomes
done
clear
C)
Cell wall
done
clear
D)
Mesosomes
done
clear
View Answer play_arrow
A protoplast is a cell:
A)
without cell wall
done
clear
B)
without plasma membrane
done
clear
C)
without nucleus
done
clear
D)
undergoing division
done
clear
View Answer play_arrow
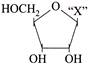
Given below is the diagrammatic representation of one of the categories of small molecular weight organic compounds in the living tissues. Identify the category shown and the one blank component ?X? in it.
A)
Category
Component
Nucleotide
Adenine
done
clear
B)
Category
Component
Nucleotide
Uracil
done
clear
C)
Category
Component
Cholesterol
Guanine
done
clear
D)
Category
Component
Amino acid
\[N{{H}_{2}}\]
done
clear
View Answer play_arrow
Nitrogen fixation in root nodules of Alnus is brought about by:
A)
Frankia
done
clear
B)
Azorhizobium
done
clear
C)
Bradyrhizobium
done
clear
D)
Clostridium
done
clear
View Answer play_arrow
During biological nitrogen fixation, inactivation of nitrogenase by oxygen poisoning prevented by:
A)
Cytochrome
done
clear
B)
Leghaemoglobin
done
clear
C)
Xanthophyll
done
clear
D)
Carotene
done
clear
View Answer play_arrow
In the leaves of \[{{C}_{4}}\] plants, malic acid formation during \[C{{O}_{2}}\] fixation occurs in the cells of:
A)
Epidermis
done
clear
B)
Mesophyll
done
clear
C)
Bundle sheath
done
clear
D)
Phloem
done
clear
View Answer play_arrow
Anoxygenic photosynthesis is characteristic of:
A)
Rhodospirillum
done
clear
B)
Spirogyra
done
clear
C)
Chlamydomonas
done
clear
D)
Ulva
done
clear
View Answer play_arrow
?Foolish Seedling? disease of rice led to the discovery of:
A)
IAA
done
clear
B)
GA
done
clear
C)
ABA
done
clear
D)
2, 4-D
done
clear
View Answer play_arrow
What causes a green plant exposed to the light on only one side to bend toward the source of light as it grows?
A)
Auxin accumulates on the shaded side, stimulating greater cell elongation there.
done
clear
B)
Green plants need light to perform photosynthesis.
done
clear
C)
Green plants seek light because they are phototropic.
done
clear
D)
Light stimulates plant cells on the lighted side to grow faster.
done
clear
View Answer play_arrow
A young infant may be feeding entirely on mother?s milk which is white in colour but the stools which the infant passes out is quite yellowish. What is this yellow colour due to?
A)
Pancreatic juice poured into duodenum
done
clear
B)
Intestinal juice
done
clear
C)
Bile pigments passed through bile juice
done
clear
D)
Undigested milk protein casein
done
clear
View Answer play_arrow
The enzymes that is not present in succus entericus is:
A)
lipase
done
clear
B)
maltase
done
clear
C)
nucleases
done
clear
D)
nucleosidase
done
clear
View Answer play_arrow
'Bundle of His' is a part of which one of the following organs in humans?
A)
Pancreas
done
clear
B)
Brain
done
clear
C)
Heart
done
clear
D)
Kidney
done
clear
View Answer play_arrow
Blood pressure in the mammalian aorta is maximum during:
A)
Diastole of the right atrium
done
clear
B)
Systole of the left atrium
done
clear
C)
Diastole of the right ventricle
done
clear
D)
Systole of the left ventricle
done
clear
View Answer play_arrow
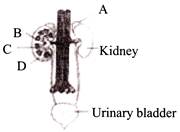
Figure shows human urinary system with structures labeled A to D. Select option which correctly identifies them and gives their characteristics and/or functions.
A)
A-Adrenal gland-located at the anterior part of Kidney. Secrete catecholamines which stimulate glycogen breakdown
done
clear
B)
B-Pelvis-broad funnel shaped space inner to hilum, directly connected to loop of Henle.
done
clear
C)
C-Medulla-inner zone of kidney and contains complete nephrons.
done
clear
D)
D-Cortex-outer part of kidney and do not contain any part of nephrons.
done
clear
View Answer play_arrow
Cornea transplant in humans is almost never rejected. This is because:
A)
It is composed of enucleated cells
done
clear
B)
It is a non-living layer
done
clear
C)
Its cells are least penetrable by bacteria
done
clear
D)
It has no blood supply
done
clear
View Answer play_arrow
The human hind brain comprises three parts, one of which is
A)
Cerebellum
done
clear
B)
Hypothalamus
done
clear
C)
Spinal
done
clear
D)
Corpus callosum
done
clear
View Answer play_arrow
Destruction of the anterior horn cells of the spinal cord would result in loss of:
A)
Integrating impulses
done
clear
B)
Sensory impulses
done
clear
C)
voluntary motor impulses
done
clear
D)
Commissural impulses
done
clear
View Answer play_arrow
Feeling the tremors of an earthquake a scared resident of seventh floor of a multistoried building starts climbing down the stairs rapidly. Which hormone initiated this action?
A)
Gastrin
done
clear
B)
Thyroxin
done
clear
C)
Adrenaline
done
clear
D)
Glucagon
done
clear
View Answer play_arrow
Which of the following statements is correct in relation to the endocrine system?
A)
Adenohypophysis is under direct neural regulation of the hypothalamus
done
clear
B)
Organs in the body like gastrointestinal tract, heart, kidney and liver do not produce any hormones
done
clear
C)
Non-nutrient chemicals produced by the body in trace amount that act as intercellular messenger are known as hormones
done
clear
D)
Releasing and inhibitory hormones are produced by the pituitary gland
done
clear
View Answer play_arrow
Cotyledons and testa respectively are edible parts in:
A)
Cashew nut and litchi
done
clear
B)
Groundnut and pomegranate
done
clear
C)
Walnut and tamarind
done
clear
D)
French bean and coconut
done
clear
View Answer play_arrow
Product of sexual reproduction generally generates:
A)
Longer viability of seeds
done
clear
B)
Prolonged dormancy
done
clear
C)
New genetic combination leading to variation
done
clear
D)
Large biomass
done
clear
View Answer play_arrow
The hilum is a scar on the:
A)
Seed, where micropyle was present
done
clear
B)
Seed, where funicle was attached
done
clear
C)
Fruit, where it was attached to pedicel
done
clear
D)
Fruit, where style was present
done
clear
View Answer play_arrow
The wheat grain has an embryo with one large, shield-shaped cotyledon known as:
A)
Coleoptile
done
clear
B)
Epiblast
done
clear
C)
Coleorrhiza
done
clear
D)
Scutellum
done
clear
View Answer play_arrow

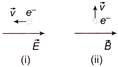

 \[{{C}_{1}}\] \[{{C}_{2}}\]
\[{{C}_{1}}\] \[{{C}_{2}}\]