To get an output Y = 1 from the circuit shown in the figure, the input must be
A)
A-0 B-1 C-0
done
clear
B)
A-0 B-0 C-1
done
clear
C)
A-1 B-0 C-1
done
clear
D)
A-1 B-0 C-0
done
clear
View Answer play_arrow
The ratio of the frequencies of the long wavelength limits of the Lyman and Balmer series of hydrogen is:
A)
27 : 5
done
clear
B)
5 : 27
done
clear
C)
4 : 1
done
clear
D)
1 : 4
done
clear
View Answer play_arrow
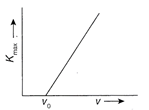
The maximum energy\[{{K}_{\max }}\]of photoelectrons emitted in a photoelectric cell is measured using lights of various frequencies v. The graph in figure shows how\[{{K}_{\max }}\] varies with v. The slope of the graph is equal to
A)
the charge of an electron
done
clear
B)
the charge to mass ratio of an electron
done
clear
C)
the work function of the emitter in the cell
done
clear
D)
Planck's constant.
done
clear
View Answer play_arrow
Which energy state of the triply ionized beryllium\[(B{{e}^{+++}})\]has the same electron orbital radius as that of the ground state of hydrogen? Given Z for beryllium = 4.
A)
n = 1
done
clear
B)
n = 2
done
clear
C)
n = 3
done
clear
D)
n = 4
done
clear
View Answer play_arrow
In a hydrogen atom, the electron is in nth excited state. It comes down to first excited state by emitting ten different wavelengths. The value of n is
A)
6
done
clear
B)
7
done
clear
C)
8
done
clear
D)
9
done
clear
View Answer play_arrow
A small sphere of radius R held against the inner surface of a smooth spherical shell of radius 6R as shown in the figure. The masses of the shell and small spheres are 4M and M, respectively. This arrangement is placed on a smooth horizontal table. The small sphere is now released. The x-coordinate of the center of the shell when the smaller sphere reaches the other extreme position is
A)
R
done
clear
B)
2R
done
clear
C)
3R
done
clear
D)
4R
done
clear
View Answer play_arrow
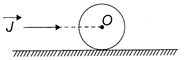
An impulse J is applied on a ring of mass m along a line passing through its center O. The ring is placed on a rough horizontal surface. The linear velocity of center of ring once it starts rolling without slipping is
A)
\[J/m\]
done
clear
B)
\[J/2m\]
done
clear
C)
\[J/4m\]
done
clear
D)
\[J/3m\]
done
clear
View Answer play_arrow
The binding energies per nucleon of deuteron \[({{\,}_{1}}{{H}^{2}})\] and helium atom \[({{\,}_{2}}H{{e}^{4}})\] are 1.1 MeV and 7 MeV If two deuteron atoms react to form a single helium atom, then the energy released is:
A)
13.9 MeV
done
clear
B)
26.9 MeV
done
clear
C)
23.6 MeV
done
clear
D)
19.2 MeV
done
clear
View Answer play_arrow
The half-life period of a radioactive element X is same as the mean-life time of another radioactive element Y. Initially both of them have the same number of atoms. Then
A)
X and Y have the same decay rate initially
done
clear
B)
X and Y decay at the same rate always
done
clear
C)
Y will decay at a faster rate than X
done
clear
D)
X will decay at a faster rate than Y
done
clear
View Answer play_arrow
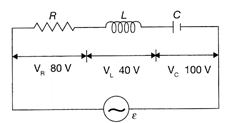
The value of alternating emf \[\varepsilon \] in the given circuit will be
A)
220 V
done
clear
B)
140 V
done
clear
C)
100 V
done
clear
D)
20 V
done
clear
View Answer play_arrow
An electron in hydrogen atom after absorbing a photon jumps from energy state\[{{n}_{1}}\] to\[{{n}_{2}}\]. Then it returns to ground state after emitting six different wavelength in emission spectrum. The energy of emitted photons is either equal to less than or greater than the absorbed photons. Then \[{{n}_{1}}\]and \[{{n}_{2}}\]are
A)
\[{{n}_{2}}=4,{{n}_{1}}=3\]
done
clear
B)
\[{{n}_{2}}=5,{{n}_{1}}=3\]
done
clear
C)
\[{{n}_{2}}=4,{{n}_{1}}=2\]
done
clear
D)
\[{{n}_{2}}=4,{{n}_{1}}=1\]
done
clear
View Answer play_arrow
\[{{K}_{\alpha }}\]wavelength emitted by an atom of atomic number Z = 11 is \[\lambda .\]Find the atomic number for an atom that emits \[{{K}_{\alpha }}\]radiation with wavelength \[4\lambda .\]
A)
Z = 6
done
clear
B)
Z = 4
done
clear
C)
Z = 11
done
clear
D)
Z = 44
done
clear
View Answer play_arrow
Let\[{{K}_{1}}\]be the maximum kinetic energy of photo electrons emitted by light of wavelength\[{{\lambda }_{1}}\]and\[{{K}_{2}}\]corresponding to wavelength\[{{\lambda }_{2}}.\]If \[{{\lambda }_{1}}=2{{\lambda }_{2}},\] then
A)
\[2{{K}_{1}}={{K}_{2}}\]
done
clear
B)
\[{{K}_{1}}=2{{K}_{2}}\]
done
clear
C)
\[{{K}_{1}}<{{K}_{2}}/2\]
done
clear
D)
\[{{K}_{1}}>2{{K}_{2}}\]
done
clear
View Answer play_arrow
100 g of water is heated from Ignoring the slight expansion of the water, the change in its internal energy is (specific heat of water is 4184 J Kg/K)
A)
4.2 kJ
done
clear
B)
8.4 kJ
done
clear
C)
84 kJ
done
clear
D)
2.1 kJ
done
clear
View Answer play_arrow
When a plane electromagnetic wave travels in vacuum, the average electric energy density is given by (here \[{{E}_{0}}\]is the amplitude of the electric field of the wave)
A)
\[\frac{1}{4}{{\varepsilon }_{0}}E_{0}^{2}\]
done
clear
B)
\[\frac{1}{2}{{\varepsilon }_{0}}E_{0}^{2}\]
done
clear
C)
\[2{{\varepsilon }_{0}}E_{0}^{2}\]
done
clear
D)
\[4{{\varepsilon }_{0}}E_{0}^{2}\]
done
clear
View Answer play_arrow
A block floats in a liquid contained in a beaker. The beaker is placed on the floor of an elevator. If the elevator descends with acceleration a (< g), the up thrust on the block due to the liquid
A)
is equal to the weight of the liquid displaced.
done
clear
B)
is greater than the weight of the liquid displaced.
done
clear
C)
is less than the weight of the liquid displaced.
done
clear
D)
becomes equal to zero.
done
clear
View Answer play_arrow
The potential difference between points A and B in the circuit shown in the figure will be:
A)
1 V
done
clear
B)
2 V
done
clear
C)
3V
done
clear
D)
4 V
done
clear
View Answer play_arrow
Two condensers of capacity C and 2C are connected in parallel and are charged to a potential difference V. If now the battery is removed and a material of dielectric constant K is placed between the plates of condenser of capacity C, then the potential difference across the condensers will be
A)
\[\frac{3V}{K+2}\]
done
clear
B)
\[3V(K+2)\]
done
clear
C)
\[\frac{K+2}{3V}\]
done
clear
D)
\[\frac{3(K+2)}{V}\]
done
clear
View Answer play_arrow
The dimensions of specific heat are
A)
\[ML{{T}^{-2}}{{K}^{-1}}\]
done
clear
B)
\[M{{L}^{2}}{{T}^{-2}}{{K}^{-1}}\]
done
clear
C)
\[{{M}^{0}}{{L}^{2}}{{T}^{-2}}{{K}^{-1}}\]
done
clear
D)
\[{{M}^{0}}L{{T}^{-2}}{{K}^{-1}}\]
done
clear
View Answer play_arrow
The image of a small electric bulb fixed on the wall of a room is to be obtained on the opposite wall 3 m away by means of a convex lens. What is the maximum focal length of the lens for this purpose?
A)
0.5 m
done
clear
B)
0.75 m
done
clear
C)
1.0 m
done
clear
D)
1.5 m
done
clear
View Answer play_arrow
The diameter of a Plano-convex lens is 6 cm and the thickness at the center is 3 mm. If the speed of light in the material of the lens is \[2\times {{10}^{8}}\,m/s,\]the focal length of the lens is
A)
15 cm
done
clear
B)
20 cm
done
clear
C)
30 cm
done
clear
D)
10 cm
done
clear
View Answer play_arrow
A uniform rod of length 2L is placed with one end in contact with the horizontal and is then inclined at an angle a to the horizontal and allowed to fall without slipping at contact point When it becomes horizontal, its angular velocity will be
A)
\[\omega =\sqrt{\frac{3g\sin \alpha }{2L}}\]
done
clear
B)
\[\omega =\sqrt{\frac{2L}{3g\,\sin \alpha }}\]
done
clear
C)
\[\omega =\sqrt{\frac{6g\sin \alpha }{L}}\]
done
clear
D)
\[\omega =\sqrt{\frac{L}{g\sin \alpha }}\]
done
clear
View Answer play_arrow
A body of radius R and mass m is rolling horizontally without slipping with speed u. It then rolls up a hill to a maximum height \[h=\frac{3{{u}^{2}}}{4g}.\]The body might be
A)
solid sphere
done
clear
B)
hollow sphere
done
clear
C)
disc
done
clear
D)
ring
done
clear
View Answer play_arrow
A capacitor of capacitance C is charged to a potential difference V from a cell and then disconnected from it. A charge +Q is now given to its positive plate. The potential difference across the capacitor is now
A)
V
done
clear
B)
\[V+\frac{Q}{C}\]
done
clear
C)
\[V+\frac{Q}{2C}\]
done
clear
D)
\[V-\frac{Q}{C},\]if \[V<CV\]
done
clear
View Answer play_arrow
A wheel having metal spokes 1 m long between its axle and rim is rotating in a magnetic field of flux density \[5\times {{1}^{-5}}T\]normal to the plane of the wheel. An emf of 22/7 mV is produced between the rim and the axle of the wheel. The rate of rotation of the wheel in radians per second is:
A)
10
done
clear
B)
20
done
clear
C)
30
done
clear
D)
40
done
clear
View Answer play_arrow
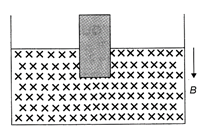
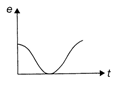
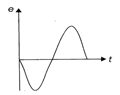
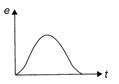
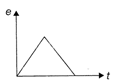
The variation of induced emf (e) with time (t) in a coil, if a short bar magnet in moved along its axis with a constant velocity is best represented as follows:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
In the Young's double slit experiment, separation between the two slits is 0.9 mm and the fringes are observed one meter away. If it produces the second dark fringe at a distance of 1 mm from the central fringe, then wavelength of the monochromatic source of light used is:
A)
400 nm
done
clear
B)
450 nm
done
clear
C)
500 nm
done
clear
D)
600 nm
done
clear
View Answer play_arrow
A liquid cools from \[50{}^\circ C\] to \[45{}^\circ C\] in 5 minutes and from \[45{}^\circ C\] to \[41.5{}^\circ C\] in the next 5 minutes. The temperature of the surrounding is
A)
\[27{}^\circ C\]
done
clear
B)
\[40.3{}^\circ C\]
done
clear
C)
\[23.3{}^\circ C\]
done
clear
D)
\[33.3{}^\circ C\]
done
clear
View Answer play_arrow
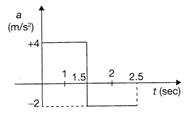
A particle starts from rest at t = 0 and moves in a straight line with an acceleration as shown in figure. The velocity of the particle at t = 2.5 s is
A)
2 m/s
done
clear
B)
4 m/s
done
clear
C)
6 m/s
done
clear
D)
8 m/s
done
clear
View Answer play_arrow
The displacement\[x\]of a particle moving along a straight line varies with time t as, \[x=a{{e}^{-\alpha t}}+b{{e}^{-\beta t}},\]where\[a,b,\alpha \]and\[\beta \]are positive constants. The velocity of the particle will
A)
go on decreasing with time
done
clear
B)
be independent of and \[\beta \]
done
clear
C)
drop to zero when \[\alpha =\beta \]
done
clear
D)
go on increasing with time
done
clear
View Answer play_arrow
The vector that must be added to the vectors \[\hat{j}-3\hat{j}+2\hat{k}\]and \[3\hat{i}-6\hat{j}-7\hat{k}\]so that, the resultant vector is a unit-vector along the Y-axis is
A)
\[4\hat{i}+2\hat{j}+5\hat{k}\]
done
clear
B)
\[-4\hat{i}-2\hat{j}+5\hat{k}\]
done
clear
C)
\[3\hat{i}+4\hat{j}+5\hat{k}\]
done
clear
D)
Null vector
done
clear
View Answer play_arrow
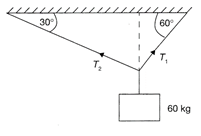
A 60 kg block is suspended by two cables as shown in the figure. The ratio of tensions \[{{T}_{1}}\] and \[{{T}_{2}}\]is
A)
\[\sqrt{3}:1\]
done
clear
B)
\[1:\sqrt{3}\]
done
clear
C)
\[2:\sqrt{3}\]
done
clear
D)
\[\sqrt{3}:2\]
done
clear
View Answer play_arrow
The speed of projectile at its maximum height is half of its initial speed. The angle of projection is
A)
\[{{60}^{o}}\]
done
clear
B)
\[{{15}^{o}}\]
done
clear
C)
\[{{30}^{o}}\]
done
clear
D)
\[{{45}^{o}}\]
done
clear
View Answer play_arrow
A body is thrown horizontally from the top of a tower of height 5 m. It touches the ground at a distance of 10m from the foot of the tower. The initial velocity of the body is\[(g=10\text{ }m/{{s}^{2}})\]
A)
2.5 m/s
done
clear
B)
5 m/s
done
clear
C)
10 m/s
done
clear
D)
20 m/s
done
clear
View Answer play_arrow
A block of mass \[{{m}_{1}}\]is suspended from the ceiling of a room through a rope of mass\[{{m}_{2}}.\] The ceiling pulls the chain by a force
A)
\[{{m}_{1}}g\]
done
clear
B)
\[({{m}_{1}}+{{m}_{2}})g\]
done
clear
C)
\[\frac{({{m}_{1}}{{m}_{2}})g}{2}\]
done
clear
D)
\[{{m}_{2}}g\]
done
clear
View Answer play_arrow
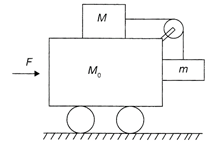
Two blocks of mass M and m are kept on the trolley whose all surfaces are smooth. Select the correct statement
A)
If F = 0 blocks cannot remain stationary
done
clear
B)
For one unique value of F, blocks will be stationary
done
clear
C)
Blocks cannot be stationary for any value of F because all surfaces are smooth
done
clear
D)
Both (1) and (2)
done
clear
View Answer play_arrow
A plane metallic sheet is placed with its face parallel to the lines of magnetic field B of a uniform field, A particle of mass m and charge q is projected with a velocity u from a distance d from the plane normal to the magnetic field lines. Then the maximum velocity of projection for which the particle does not hit the plate is:
A)
2Bqd/m
done
clear
B)
Bqd/m
done
clear
C)
Bqd/2 m
done
clear
D)
Bqm/d
done
clear
View Answer play_arrow
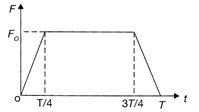
A particle of mass m moving with a velocity u makes an elastic one dimensional collision with a stationary particle of mass m establishing a contact with it for an extremely small time T. Their force of contact increases from 0to\[{{F}_{0}}\]linearly in time\[T/4,\] remains constant for a further time\[T/2\]and decreases linearly from\[{{F}_{0}}\]to 0 in further time \[T/4\] as shown in the figure. The magnitude possessed by\[{{F}_{0}}\] is
A)
\[\frac{mu}{T}\]
done
clear
B)
\[\frac{2mu}{T}\]
done
clear
C)
\[\frac{4mu}{3T}\]
done
clear
D)
\[\frac{3mu}{4T}\]
done
clear
View Answer play_arrow
In an elastic collision
A)
the initial kinetic energy is equal to the final kinetic energy.
done
clear
B)
the final kinetic energy is less than the initial kinetic energy.
done
clear
C)
the kinetic energy remains constant.
done
clear
D)
the kinetic energy first increases then decreases.
done
clear
View Answer play_arrow
A spring of force constant 800 N/m has an extension of 5 cm. The work done in extending it from 5 cm to 15 cm is
A)
16 J
done
clear
B)
8 J
done
clear
C)
32 J
done
clear
D)
24 J
done
clear
View Answer play_arrow
A rectangular tank is filled to the brim with water. When a hole at its bottom is unplugged, the tank is emptied in time T. If the tank is half-filled with water, it will be emptied in time
A)
\[\frac{T}{\sqrt{2}}\]
done
clear
B)
\[\frac{T}{\sqrt{3}}\]
done
clear
C)
\[\frac{T}{2}\]
done
clear
D)
\[\frac{T}{2\sqrt{2}}\]
done
clear
View Answer play_arrow
A particle is executing simple harmonic motion along the \[x-\]axis with amplitude 4 cm and time period 1.2 s. The minimum time taken by the particle to move from \[x=+2\,cm\] to \[x=+4\,cm\]and back again is
A)
0.6 s
done
clear
B)
0.4 s
done
clear
C)
0.3 s
done
clear
D)
0.2 s
done
clear
View Answer play_arrow
Two rods of the same length, same cross sectional area and same material transfer a given amount of heat, in 12 seconds when they are joined end to end. But when they are joined lengthwise, they will transfer the same amount of heat, in the same conditions, in
A)
24 s
done
clear
B)
\[3\,s\]
done
clear
C)
\[1.5\,s\]
done
clear
D)
\[48\,s\]
done
clear
View Answer play_arrow
A parallel plate capacitor is made by stacking 10 identical metallic plates equally spaced from one another and having the same dielectric between plates. The alternate plates are then connected. If the capacitor formed by two neighboring plates has a capacitance C, the total capacitance of the combination will be
A)
\[\frac{C}{10}\]
done
clear
B)
\[\frac{C}{9}\]
done
clear
C)
\[~9\,C\]
done
clear
D)
\[10\,C\]
done
clear
View Answer play_arrow
In a certain organ pipe three successive resonance frequencies are observed at 425,595 and 765 Hz respectively. If the speed of sound in air is 340 m/s, the length of the pipe is:
A)
2 m
done
clear
B)
1 m
done
clear
C)
1.5 m
done
clear
D)
2.5 m
done
clear
View Answer play_arrow
Heat of neutralization of strong acid and strong base is constant and equal to
A)
8.7 kcal
done
clear
B)
13.7 kcal
done
clear
C)
23.7 kcal
done
clear
D)
30.7 kcal
done
clear
View Answer play_arrow
The first law of thermodynamics is represented by
A)
\[\Delta E=\Delta Q+\Delta W\]
done
clear
B)
\[\Delta E=\Delta Q+\Delta W\]
done
clear
C)
\[\Delta E =\Delta Q+\Delta W\]
done
clear
D)
\[\Delta E =Q-W\]
done
clear
View Answer play_arrow
The hybridization shown by six carbon atoms in benzene is
A)
\[~3sp\text{ }3s{{p}^{2}}\]
done
clear
B)
\[~3s{{p}^{3}}\text{ }3s{{p}^{2}}\]
done
clear
C)
\[~3s{{p}^{3}}\text{ }3sp\]
done
clear
D)
\[~6s{{p}^{2}}\]
done
clear
View Answer play_arrow
The metal that forms a self-protecting film of oxide to prevent corrosion is
A)
Ag
done
clear
B)
Au
done
clear
C)
Al
done
clear
D)
Cu
done
clear
View Answer play_arrow
Which of the following explains the sequence of filling the electrons in different shell?
A)
Aufbau's principle
done
clear
B)
Octate rule
done
clear
C)
Hund's rule
done
clear
D)
All the above
done
clear
View Answer play_arrow
\[{{H}_{2}}{{O}_{2}}\]is a reducing agent in the reaction
A)
\[A{{g}_{2}}O+{{H}_{2}}{{O}_{2}}\to 2Ag+{{H}_{2}}O+{{O}_{2}}\]
done
clear
B)
\[{{H}_{2}}{{O}_{2}}+S{{O}_{2}}\to {{H}_{2}}S{{O}_{4}}\]
done
clear
C)
\[2KI+{{H}_{2}}{{O}_{2}}\to 2KOH+{{I}_{2}}\]
done
clear
D)
\[PbS+4{{H}_{2}}{{O}_{2}}\to PbS{{O}_{4}}+4{{H}_{2}}O\]
done
clear
View Answer play_arrow
The molarity of a solution containing 5 g of \[NaOH\] in 250 ml solution
A)
0.5
done
clear
B)
1.0
done
clear
C)
1.3
done
clear
D)
4.0
done
clear
View Answer play_arrow
A gas expands isothermally against a constant external pressure of I atmosphere from a volume of \[10\text{ }d{{m}^{3}}\]to a volume of \[20\text{ }d{{m}^{3}}.\]In this process it absorbs 800 J of thermal energy from its surroundings. The \[\Delta U\]for the process in Joules is
A)
-113 J
done
clear
B)
+113 J
done
clear
C)
-213 J
done
clear
D)
+313 J
done
clear
View Answer play_arrow
The most stable is
A)
\[KH\]
done
clear
B)
\[NaH\]
done
clear
C)
\[RbH\]
done
clear
D)
\[LiH\]
done
clear
View Answer play_arrow
Nitrobenzene and hydrogen in the presence of zinc combines to form
A)
Azoxybenzene
done
clear
B)
Azobenzene
done
clear
C)
Benzene
done
clear
D)
Aniline
done
clear
View Answer play_arrow
The gas used in air ships is
A)
\[He\]
done
clear
B)
\[Xe\]
done
clear
C)
\[Ar\]
done
clear
D)
\[Ne\]
done
clear
View Answer play_arrow
Ethyl alcohol is soluble in water due to
A)
Basic nature
done
clear
B)
Acidic nature
done
clear
C)
Formation of hydrogen bond
done
clear
D)
Dissociation in water
done
clear
View Answer play_arrow
In a chemical equilibrium, the rate constant of forward reaction is \[75\times {{10}^{-4}}\]and the equilibrium constant is 1.5. The rate constant backward reaction is
A)
\[4.5\times {{10}^{4}}\]
done
clear
B)
\[5\times {{10}^{-4}}\]
done
clear
C)
\[2.5\times {{10}^{-4}}\]
done
clear
D)
\[6\times {{10}^{4}}\]
done
clear
View Answer play_arrow
Natural rubber is a polymer of
A)
Ethylene
done
clear
B)
1, 4-butadiene
done
clear
C)
Isoprene
done
clear
D)
Terephthalic acid
done
clear
View Answer play_arrow
The wavelength of\[\,{{\,}_{6}}{{C}^{14}}\] is\[2.31\times {{10}^{-4}}.\]Its half-life is
A)
\[2\times {{10}^{3}}\,years\]
done
clear
B)
\[1.5\times {{10}^{3}}\,years\]
done
clear
C)
\[3\times {{10}^{3}}\,years\]
done
clear
D)
\[2.5\times {{10}^{3}}\,years\]
done
clear
View Answer play_arrow
Conversion of starch into maltose takes place by the enzyme
A)
Zymase
done
clear
B)
Invertase
done
clear
C)
Diastase
done
clear
D)
Maltose
done
clear
View Answer play_arrow
The oxide which forms dimer is
A)
\[{{N}_{2}}{{O}_{3}}\]
done
clear
B)
\[{{N}_{2}}{{O}_{5}}\]
done
clear
C)
\[N{{O}_{2}}\]
done
clear
D)
\[{{N}_{2}}O\]
done
clear
View Answer play_arrow
The hardest naturally occurring substance is
A)
Graphite
done
clear
B)
Iron
done
clear
C)
Diamond
done
clear
D)
Astatine
done
clear
View Answer play_arrow
Wax belong to the class
A)
Acid
done
clear
B)
Esters
done
clear
C)
Alcohol
done
clear
D)
Ether
done
clear
View Answer play_arrow
The heat of formation of \[C{{O}_{2}}\]is -95 kcal. The amount of carbon which on burning will evolve 1000 kcal is
A)
15.63 g
done
clear
B)
12.95 g
done
clear
C)
126.3 g
done
clear
D)
160.5 g
done
clear
View Answer play_arrow
The normality of orthophosphoric acid having purity of 70 per cent by weight and specific gravity 1.54 is
A)
UN
done
clear
B)
15 N
done
clear
C)
33 N
done
clear
D)
30 N
done
clear
View Answer play_arrow
Which particle contains 2 neutrons and 1 proton?
A)
11 N
done
clear
B)
15 N
done
clear
C)
33 N
done
clear
D)
30 N
done
clear
View Answer play_arrow
Philosopher's wool on heating with \[BaO\] at \[1100{{\,}^{o}}C\] produces
A)
\[BaZn{{O}_{2}}\]
done
clear
B)
\[BaCd{{O}_{2}}\]
done
clear
C)
\[Ba{{O}_{2}}+Zn\]
done
clear
D)
\[Ba+Zn{{O}_{2}}\]
done
clear
View Answer play_arrow
Chloroform and concentrated\[HN{{O}_{3}}\]reacts to produce
A)
\[CHC{{l}_{2}}N{{O}_{3}}\]
done
clear
B)
\[CHC{{l}_{2}}NH{{O}_{3}}\]
done
clear
C)
\[CC{{l}_{3}}N{{O}_{2}}\]
done
clear
D)
\[CHCIN{{O}_{3}}\]
done
clear
View Answer play_arrow
Gold number is associated with
A)
Protective colloids
done
clear
B)
Amount of pure gold
done
clear
C)
Purple of Cassius
done
clear
D)
Electrophoresis
done
clear
View Answer play_arrow
Amino acids are the building blocks of
A)
Vitamins
done
clear
B)
Fats
done
clear
C)
Carbohydrates
done
clear
D)
Proteins
done
clear
View Answer play_arrow
The number of atoms in 4.25 g of\[N{{H}_{3}}\] is
A)
\[2.054\times {{10}^{23}}\]
done
clear
B)
\[5.043\times {{10}^{25}}\]
done
clear
C)
\[8.044\times {{10}^{23}}\]
done
clear
D)
\[6.023\times {{10}^{23}}\]
done
clear
View Answer play_arrow
The conjugate acid of \[N{{H}_{3}}\]is
A)
\[N{{H}_{4}}^{+}\]
done
clear
B)
\[{{N}_{2}}{{H}_{4}}\]
done
clear
C)
\[N{{H}_{2}}OH\]
done
clear
D)
\[N{{H}_{3}}\]
done
clear
View Answer play_arrow
Adiabatic process involves
A)
\[\Delta E =0\]
done
clear
B)
\[\Delta W=0\]
done
clear
C)
\[\Delta q=0\]
done
clear
D)
\[\Delta V=0\]
done
clear
View Answer play_arrow
The rate of reaction depends upon
A)
Molar concentration
done
clear
B)
Equivalent mass
done
clear
C)
Atomic mass
done
clear
D)
None of the above
done
clear
View Answer play_arrow
The purest form of iron is
A)
Steel
done
clear
B)
Wrought Iron
done
clear
C)
Pig Iron
done
clear
D)
None of the above
done
clear
View Answer play_arrow
The largest size of the ion is
A)
\[C{{l}^{-}}\]
done
clear
B)
\[{{K}^{+}}\]
done
clear
C)
\[C{{a}^{++}}\]
done
clear
D)
\[{{S}^{--}}\]
done
clear
View Answer play_arrow
The empirical formula of a compound is \[C{{H}_{2}}O.\]Its molecular weight is 180. The molecular formula of the compound is
A)
\[{{C}_{4}}{{H}_{8}}{{O}_{4}}\]
done
clear
B)
\[{{C}_{6}}{{H}_{12}}{{O}_{6}}\]
done
clear
C)
\[{{C}_{3}}{{H}_{6}}{{O}_{3}}\]
done
clear
D)
\[{{C}_{5}}{{H}_{10}}{{O}_{5}}\]
done
clear
View Answer play_arrow
The Bohr's orbit radius for the hydrogen atom (n = 1) is approximately \[0.530\,\overset{\text{o}}{\mathop{A}}\,.\]The radius for the first excited state (n = 2) orbit is
A)
1.08 A
done
clear
B)
2.12 A
done
clear
C)
5.24 A
done
clear
D)
6.36 A
done
clear
View Answer play_arrow
The first ionization potential is maximum for
A)
Iron
done
clear
B)
Hydrogen
done
clear
C)
Lithium
done
clear
D)
Uranium
done
clear
View Answer play_arrow
Roasting of an ore is done in
A)
Presence of air
done
clear
B)
Vacuum
done
clear
C)
Absence of air
done
clear
D)
None of the above
done
clear
View Answer play_arrow
The dissociation constant of two acids \[\text{H}{{\text{A}}_{\text{1}}}\]and \[\text{H}{{\text{A}}_{2}}\]are\[3.0\times {{10}^{-4}}\]and \[1.8\times {{10}^{-5}}\] respectively. The relative strength is
A)
10 : 1
done
clear
B)
1 : 12
done
clear
C)
4 : 1
done
clear
D)
1 : 5
done
clear
View Answer play_arrow
The alkyl halide is converted into alcohol by
A)
Addition
done
clear
B)
Elimination
done
clear
C)
Halogenation
done
clear
D)
Substitution
done
clear
View Answer play_arrow
An aqueous solution of ammonium acetate is
A)
Acidic
done
clear
B)
Basic
done
clear
C)
Neutral
done
clear
D)
None of the above
done
clear
View Answer play_arrow
By passing a beam of light through a colloidal solution, it is
A)
Adsorbed
done
clear
B)
Refracted
done
clear
C)
Scattered
done
clear
D)
Reflected
done
clear
View Answer play_arrow
The highest melting halide is
A)
\[NaF\]
done
clear
B)
\[Nal\]
done
clear
C)
\[NaCl\]
done
clear
D)
\[NaBr\]
done
clear
View Answer play_arrow
The pH of a \[{{10}^{-10}}\]molar solution of \[HCl\] in water is
A)
6.0
done
clear
B)
7.0
done
clear
C)
8
done
clear
D)
13
done
clear
View Answer play_arrow
The product formed by the reduction of glycol acid with HI is
A)
Glycol
done
clear
B)
Acetic acid
done
clear
C)
Formic acid
done
clear
D)
lodoacetic acid
done
clear
View Answer play_arrow
Hydrogen can be differentiated by other alkali meta due to its
A)
Non-metallic character
done
clear
B)
Electropositive character
done
clear
C)
Reducing character
done
clear
D)
Affinity for non-metals
done
clear
View Answer play_arrow
Copper turning on heating with concentrated \[{{\text{H}}_{\text{2}}}\text{S}{{\text{O}}_{\text{4}}}\]produces
A)
\[\text{S}{{\text{O}}_{3}}\]
done
clear
B)
\[{{H}_{2}}S\]
done
clear
C)
\[{{O}_{2}}\]
done
clear
D)
\[S{{O}_{2}}\]
done
clear
View Answer play_arrow
Evaporation of water is
A)
Endothermic change
done
clear
B)
No heat change takes place
done
clear
C)
Exothermic change
done
clear
D)
Chemical reaction
done
clear
View Answer play_arrow
The Bayer's reagent is
A)
Alkaline\[KMn{{O}_{4}}\]solution
done
clear
B)
Neutral\[KMn{{O}_{4}}\]solution
done
clear
C)
Acidic \[KMn{{O}_{4}}\]solution
done
clear
D)
Saturated \[KMn{{O}_{4}}\]solution
done
clear
View Answer play_arrow
Ethyl amine reacts with chloroform and potassium hydroxide to produce
A)
Ethyl cyanide
done
clear
B)
Ethyl isocyanide
done
clear
C)
Ethyl chloride
done
clear
D)
Ethyl alcohol
done
clear
View Answer play_arrow
The number of double bonds in gammexane is
A)
0
done
clear
B)
2
done
clear
C)
3
done
clear
D)
1
done
clear
View Answer play_arrow
The chloramphenicol is an example of
A)
Analgesics
done
clear
B)
Antibiotic
done
clear
C)
Antiseptic
done
clear
D)
Antipyretics
done
clear
View Answer play_arrow
Amoeba is a/an
A)
Unicellular animal
done
clear
B)
Octacellular animal
done
clear
C)
Multicellular animal
done
clear
D)
All of the above
done
clear
View Answer play_arrow
A herbarium sheet
A)
Carries a label providing information about the date and place of collection.
done
clear
B)
Also provides English, local and botanical names.
done
clear
C)
The family name of specimen and the collector's name is also mentioned.
done
clear
D)
More than one option is correct.
done
clear
View Answer play_arrow
The enzyme present in saliva is
A)
Maltase
done
clear
B)
Ptyalin
done
clear
C)
Sucrase
done
clear
D)
Invertase
done
clear
View Answer play_arrow
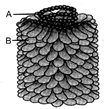
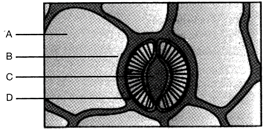
Identify the virus and name the structures A and B:
A)
\[TMV,\text{ }A=ssRNA,B=Capsid\]
done
clear
B)
\[TMV,\,A=dsRNA,\text{ }B=Capsid\]
done
clear
C)
\[~TMV,A=Capsid,B=ssRNA\]
done
clear
D)
\[TMV,A=Capsid,B=dsRNA\]
done
clear
View Answer play_arrow
Fungi in a forest ecosystem is
A)
Producer
done
clear
B)
Decomposer
done
clear
C)
Top consumer
done
clear
D)
Autotroph
done
clear
View Answer play_arrow
Thermococcus, Methanococcus and Methanobacterium exemplify
A)
Bacteria whose DNA is relaxed or positively supercoiled but which has a cytoskeleton as well as mitochondria.
done
clear
B)
Bacteria that contain a cytoskeleton and ribosome.
done
clear
C)
Archaebacteria that contain proteins homologous to eukaryotic core histones.
done
clear
D)
Archaebacteria that lack any histones resembling those found in eukaryotes but whose DNA is negatively supercoiled.
done
clear
View Answer play_arrow
Cyanobacteria is an
A)
Algae having blue-green pigment
done
clear
B)
Algae having red pigment
done
clear
C)
Algae having brown pigment
done
clear
D)
Algae having yellow-brown pigment
done
clear
View Answer play_arrow
Cu is present in
A)
Plasmalemma
done
clear
B)
Plastoquinone
done
clear
C)
Plastocyanin
done
clear
D)
Ferredoxin
done
clear
View Answer play_arrow
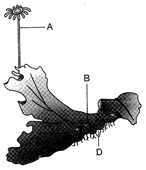
Identify the structure marked as A, B and C for Marchantia:
A)
A = Antheridiophore, B = Gemma cup, C = Rhizoids
done
clear
B)
A = Rhizoids, B = Gemma cup, C = Archegoniophore
done
clear
C)
A = Antheridiophore, B = Rhizoids, C = Gemma cup
done
clear
D)
A = Archegoniophore, B = Gemma cup, C = Rhizoids
done
clear
View Answer play_arrow
The largest phylum in respect of number of species is
A)
Coelenterate
done
clear
B)
Arthropoda
done
clear
C)
Protozoa
done
clear
D)
Porifera
done
clear
View Answer play_arrow
Chronic disturbance in hormone secretion of thyroid gland causes
A)
Goitre
done
clear
B)
Diabetes
done
clear
C)
Addison's disease
done
clear
D)
Colour blindness
done
clear
View Answer play_arrow
The formation of egg and sperm is affected by
A)
LH
done
clear
B)
MH
done
clear
C)
TSH
done
clear
D)
FSH
done
clear
View Answer play_arrow
ACTH is secreted by
A)
Thyroid gland
done
clear
B)
Thymus gland
done
clear
C)
Pituitary gland
done
clear
D)
Islets of Langerhans
done
clear
View Answer play_arrow
Enzymes of electron transport system are present in
A)
Inner mitochondrial membrane
done
clear
B)
Matrix
done
clear
C)
Intermembrane space
done
clear
D)
Endoplasmic reticulum
done
clear
View Answer play_arrow
Ozone hole results in
A)
UV radiation reaching the earth
done
clear
B)
Cataract
done
clear
C)
Increase in skin cancer
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Greenhouse effect is due to the increased concentration of
A)
\[C{{O}_{2}}\]
done
clear
B)
Ne
done
clear
C)
\[S{{O}_{2}}\]
done
clear
D)
\[N{{O}_{2}}\]
done
clear
View Answer play_arrow
Gymnosperms are also called soft wood spermatophytes because they lack
A)
Cambium
done
clear
B)
Phloem fibres
done
clear
C)
Thick-walled tracheids
done
clear
D)
Xylem fibres
done
clear
View Answer play_arrow
Which of the following metal is present in vitamin\[{{B}_{12}}\]?
A)
Cobalt
done
clear
B)
Copper
done
clear
C)
Zinc
done
clear
D)
Magnesium
done
clear
View Answer play_arrow
Primary blood cells are formed in
A)
Plasma
done
clear
B)
Bone marrow
done
clear
C)
Liver
done
clear
D)
Spleen
done
clear
View Answer play_arrow
Secondary host of schistosoma is
A)
Hydra
done
clear
B)
Euglena
done
clear
C)
Snail
done
clear
D)
Pheretima
done
clear
View Answer play_arrow
Find the incorrect match with respect to the floral formula of the concerned families:
A)
Fabaceae -
done
clear
B)
Liliaceae -
done
clear
C)
Solanaceae -
done
clear
D)
Brassicaceae -
done
clear
View Answer play_arrow
Sertoli cells help in
A)
Maturation of eggs
done
clear
B)
Maturation of sperms
done
clear
C)
Enzyme production
done
clear
D)
Ovulation
done
clear
View Answer play_arrow
Opium is extracted from
A)
Atropa belladonna
done
clear
B)
Pap aver somniferum
done
clear
C)
Vinca rosea
done
clear
D)
Azadirachta indica
done
clear
View Answer play_arrow
Quinine is obtained from
A)
Bark of Cinchona
done
clear
B)
Root of Cinchona
done
clear
C)
Wood of Cinchona
done
clear
D)
Leaves of Cinchona
done
clear
View Answer play_arrow
When we peel the skin of a potato tuber, we remove
A)
Periderm
done
clear
B)
Epidermis
done
clear
C)
Cuticle
done
clear
D)
Sapwood
done
clear
View Answer play_arrow
Genes are present in
A)
Chromosomes
done
clear
B)
Lamellae
done
clear
C)
Plasma membrane
done
clear
D)
Mesosomes
done
clear
View Answer play_arrow
In the process of photosynthesis
A)
\[{{O}_{2}}\]is taken and \[C{{O}_{2}}\]is evolved.
done
clear
B)
\[{{O}_{2}}\]is taken but \[C{{O}_{2}}\]is not evolved.
done
clear
C)
\[C{{O}_{2}}\]is taken and \[{{O}_{2}}\] is evolved.
done
clear
D)
\[C{{O}_{2}}\]is taken and is evolved.
done
clear
View Answer play_arrow
Bacteria do not have
A)
Ribosome
done
clear
B)
Protein synthesizing apparatus
done
clear
C)
Mitochondria
done
clear
D)
Cell wall
done
clear
View Answer play_arrow
Which of the following plant hormone is extracted from fungus?
A)
Auxin
done
clear
B)
Gibberellin
done
clear
C)
Cytokinin
done
clear
D)
2, 4-D
done
clear
View Answer play_arrow
The highest concentration of auxin is found in
A)
Root and shoot tips
done
clear
B)
Leaves and fruits
done
clear
C)
Mid stem portion
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Ethylene is used for
A)
Decreasing the senescence
done
clear
B)
Increasing the height of stem
done
clear
C)
Ripening of fruits
done
clear
D)
Prevention of leaf fall
done
clear
View Answer play_arrow
Meristematic tissues are
A)
Premature having the ability of division.
done
clear
B)
Mature does not have the ability of division.
done
clear
C)
Premature not having the ability of division.
done
clear
D)
Complex differentiating in xylem, phloem and cambium.
done
clear
View Answer play_arrow
The function of polymerase chain reaction (PCR) is
A)
Translation
done
clear
B)
Transduction
done
clear
C)
DNA amplification
done
clear
D)
None of these
done
clear
View Answer play_arrow
A codon is made up of
A)
Single nucleotide
done
clear
B)
Two nucleotides
done
clear
C)
Three nucleotides
done
clear
D)
Four nucleotides
done
clear
View Answer play_arrow
Protein in silk thread is
A)
Fibroin
done
clear
B)
Keratin
done
clear
C)
Albumin
done
clear
D)
Globulin
done
clear
View Answer play_arrow
Histamine is secreted by
A)
Goblet cells
done
clear
B)
Nerve cells
done
clear
C)
Kupffer cells
done
clear
D)
Mast cells
done
clear
View Answer play_arrow
Which of the following hormones are secreted by a pancreas?
A)
Insulin and glucagon
done
clear
B)
Epinephrine and norepinephrine
done
clear
C)
Thyroxine and melanin
done
clear
D)
Lactocin and oxytocin
done
clear
View Answer play_arrow
Starfish belongs to phylum
A)
Porifera
done
clear
B)
Coelenterate
done
clear
C)
Echinodermata
done
clear
D)
Arthropoda
done
clear
View Answer play_arrow
Yeast belongs to
A)
Zygomycetes
done
clear
B)
Basidiomycetes
done
clear
C)
Ascomycetes
done
clear
D)
Phycomycetes
done
clear
View Answer play_arrow
Mycorrhiza helps in
A)
Nutrition absorbing
done
clear
B)
Food manufacturing
done
clear
C)
Disease resistance
done
clear
D)
Disease prevention
done
clear
View Answer play_arrow
The conversion of \[NO_{3}^{-}\] to \[{{N}_{2}}\] is called
A)
Nitrification
done
clear
B)
Denitrification
done
clear
C)
Ammonification
done
clear
D)
Nitrogen fixation
done
clear
View Answer play_arrow
Cuscuta is a
A)
Parasitic plant
done
clear
B)
Symbiotic plant
done
clear
C)
Predator
done
clear
D)
Decomposer
done
clear
View Answer play_arrow
Binary fission is a mode of
A)
Micropropagation
done
clear
B)
Vegetative propagation
done
clear
C)
Macropropagation
done
clear
D)
Sexual reproduction
done
clear
View Answer play_arrow
Humidity in atmosphere decreases the rate of
A)
Transpiration
done
clear
B)
Photosynthesis
done
clear
C)
Glycolysis
done
clear
D)
Growth
done
clear
View Answer play_arrow
A phosphoglyceride is always made up of
A)
A saturated or unsaturated fatty acid esterified to a glycerol molecule to which a phosphate group is also attached.
done
clear
B)
A saturated or unsaturated fatty acid esterified to a phosphate group which is also attached to a glycerol molecule.
done
clear
C)
Only a saturated fatty acid esterified to glycerol molecule to which a phosphate group is also attached.
done
clear
D)
Only an unsaturated fatty acid esterified to a glycerol molecule to which a phosphate group is also attached.
done
clear
View Answer play_arrow
Jumping genes in maize were discovered by
A)
Hugo de Vries
done
clear
B)
T. H. Morgan
done
clear
C)
Barbara McClintock
done
clear
D)
Mendel
done
clear
View Answer play_arrow
Sea horse belongs to
A)
Mammals
done
clear
B)
Amphibians
done
clear
C)
Aves
done
clear
D)
Pisces
done
clear
View Answer play_arrow
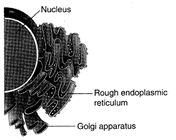
Which one of the following organelle in the figure correctly matches with its functions?
A)
RER, protein synthesis
done
clear
B)
RER, glycoproteins
done
clear
C)
Golgi apparatus, protein synthesis
done
clear
D)
Golgi apparatus, formation of glycolipids
done
clear
View Answer play_arrow
Ontogeny repeats phylogeny as a part of
A)
Recapitulation theory
done
clear
B)
Darwin's evolution theory
done
clear
C)
Lamarck's evolution theory
done
clear
D)
Weismann's theory
done
clear
View Answer play_arrow
A stage where cell division is shown in the figure Select the answer which gives the correct identification of the stage with its characteristics:
A)
Late anaphase Chromosomes move away from equatorial plate, Golgi complex not present.
done
clear
B)
Cytokinesis Cell plate formed, Mitochondria distributed between two daughter cells.
done
clear
C)
Telophase Endoplasmic reticulum and nucleolus not reformed yet.
done
clear
D)
Telophase Nuclear envelop reforms, Golgi complex reforms.
done
clear
View Answer play_arrow
Universal blood recipient is
A)
Blood group-O
done
clear
B)
Blood group-AB
done
clear
C)
Blood group-A
done
clear
D)
Blood group-B
done
clear
View Answer play_arrow
Plant like nutrition is present in
A)
Amoeba
done
clear
B)
Paramecium
done
clear
C)
Euglena
done
clear
D)
Plasmodium
done
clear
View Answer play_arrow
Which of the following is the connecting link between reptiles and birds?
A)
Archaeopteryx
done
clear
B)
Euglena
done
clear
C)
Neopilina
done
clear
D)
Latimeria
done
clear
View Answer play_arrow
Which of the following connects glycolysis to Krebs cycle?
A)
Acetyl-CoA
done
clear
B)
Ribozyme
done
clear
C)
Cytochrome oxidase
done
clear
D)
N-Acetylglucosamine
done
clear
View Answer play_arrow
Pyruvic acid is the end product of
A)
Krebs cycle
done
clear
B)
Electron transport system
done
clear
C)
Photosynthesis
done
clear
D)
Glycolysis
done
clear
View Answer play_arrow
Given below is the diagram of stomatal apparatus. Which option identifies the below labelling A, B, C, D correctly?
A)
A = Subsidiary cell, B = Epidermal cell, C Guard cell, D = Stomatal aperture.
done
clear
B)
A = Guard cell, B = Stomatal aperture, C Subsidiary cell, D = Epidermal cell.
done
clear
C)
A = Epidermal cell, B = Guard cell, C = Stomatal aperture, D = Subsidiary cell.
done
clear
D)
A = Epidermal cell, B = Subsidiary cell, C = Stomatal aperture, D = Guard cell.
done
clear
View Answer play_arrow
Which of the following is a viral disease?
A)
Typhoid
done
clear
B)
Polio
done
clear
C)
Tuberculosis
done
clear
D)
Leprosy
done
clear
View Answer play_arrow
Which one is a sex linked disease7
A)
Colour blindness
done
clear
B)
Haemophilia
done
clear
C)
Syphilis
done
clear
D)
Both (a) and (b)
done
clear
View Answer play_arrow
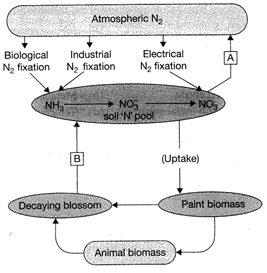
In the given diagram, A and B represents.
A)
Mineralization, Uemmeralisauon
done
clear
B)
Ammonification, Denitrification
done
clear
C)
Denitrification, Ammonification
done
clear
D)
Denitrification, Mineralization
done
clear
View Answer play_arrow
Which of the following has no specific organ for respiration but do respire?
A)
Rabbit
done
clear
B)
Cockroach
done
clear
C)
Earthworm
done
clear
D)
Frog
done
clear
View Answer play_arrow
Match the following and choose the correct option from those give below:
Column I Column II A. Molecular oxygen i. a-Ketoglutaric acid B. Electron acceptor ii. Hydrogen acceptor C. Pyruvate dehydrogenase iii. Cytochrome C D. Decarboxylation iv. Acetyl-CoA
A)
\[A\to ii,\,B\to iii,\,C\to iv,\,D\to i\]
done
clear
B)
\[A\to iii,\,B\to iv,\,C\to ii,\,D\to i\]
done
clear
C)
\[A\to ii,\,B\to i,\,C\to iii,\,D\to iv\]
done
clear
D)
\[A\to iv,\,B\to iii,\,C\to i,\,D\to ii\]
done
clear
View Answer play_arrow
Tendons and ligaments are
A)
Epithelial tissue
done
clear
B)
Fibrous connective tissue
done
clear
C)
Nerve tissue
done
clear
D)
Muscular tissue
done
clear
View Answer play_arrow
Mushroom belongs to
A)
Ascomycetes
done
clear
B)
Basidiomycetes
done
clear
C)
Phycomycetes
done
clear
D)
Zygomycetes
done
clear
View Answer play_arrow
The wall of bacteria consists of
A)
N-Acetylglucosamine
done
clear
B)
N-Acetylmuramic acid
done
clear
C)
Both (a) and (b)
done
clear
D)
Cellulose
done
clear
View Answer play_arrow
The movement of pollen tube is called
A)
Chemotropism
done
clear
B)
Thermotaxis
done
clear
C)
Thermonastic
done
clear
D)
Hydrotropism
done
clear
View Answer play_arrow
What will be the gametic chromosome number of a cell, if the somatic cell has 40 chromosomes?
A)
10
done
clear
B)
20
done
clear
C)
30
done
clear
D)
40
done
clear
View Answer play_arrow
Who coined the term 'cell'?
A)
Purkinje
done
clear
B)
Robert Brown
done
clear
C)
Robert Hooke
done
clear
D)
Hugo von Mohl
done
clear
View Answer play_arrow
Which of the following animals is widely used in genetics experiments?
A)
Butterfly
done
clear
B)
Fruit fly
done
clear
C)
Housefly
done
clear
D)
Dragonfly
done
clear
View Answer play_arrow
In which of the following stage, the chromosomes are arranged at equatorial plate?
A)
Anaphase
done
clear
B)
Metaphase
done
clear
C)
Prophase
done
clear
D)
Telophase
done
clear
View Answer play_arrow
Match the following:
Column I Column II A. IAA i. Herring sperm DNA B. ABA ii. Bolting C. Ethylene iii. Stomatal closure D. GA iv. Weed-free lawns E. Cytokinins v. Ripening of fruits
A)
\[A\to iv,B\to iii,C\to v,D\to ii,E-i\]
done
clear
B)
\[A\to v,B\to iii,C\to iv,D\to ii,E-i\]
done
clear
C)
\[A\to iv,B\to i,C\to v,D\to iii,E-ii\]
done
clear
D)
\[A\to v,B\to iii,C\to ii,D\to i,E-iv\]
done
clear
View Answer play_arrow
Who first conducted the experiment on evolution to prove biochemical origin of life?
A)
Miller and Urey
done
clear
B)
Darwin
done
clear
C)
Lamarck
done
clear
D)
Weismann
done
clear
View Answer play_arrow
Maturation of sperm before penetration is called
A)
Spermatogenesis
done
clear
B)
Spermatogenesis
done
clear
C)
Capacitation
done
clear
D)
Spermatid
done
clear
View Answer play_arrow
Echolocation is found in
A)
Bat
done
clear
B)
Cat
done
clear
C)
Dog
done
clear
D)
Horse
done
clear
View Answer play_arrow
Life span of RBC is
A)
50 days
done
clear
B)
70 days
done
clear
C)
120 days
done
clear
D)
220 days
done
clear
View Answer play_arrow
The phenomenon observed in some plants wherein parts of the sexual apparatus are used for forming embryos without fertilization is called
A)
Parthenocarpy
done
clear
B)
Apomixis
done
clear
C)
Vegetative propagation
done
clear
D)
Sexual reproduction
done
clear
View Answer play_arrow
In the \[{{F}_{2}}\]generation of a Mendelian dihybrid cross, the number of phenotypes and genotypes are
A)
Phenotypes - 4; genotypes - 16
done
clear
B)
Phenotypes - 9; genotypes - 4
done
clear
C)
Phenotypes - 4; genotypes - 8
done
clear
D)
Phenotypes - 4; genotypes - 9
done
clear
View Answer play_arrow
The back bone of RNA consists of which of the following sugar?
A)
Deoxyribose
done
clear
B)
Ribose
done
clear
C)
Sucrose
done
clear
D)
Maltose
done
clear
View Answer play_arrow
Phloem conducts food by
A)
Perforated sieve plates
done
clear
B)
Bast fibres
done
clear
C)
Xylem parenchyma
done
clear
D)
Xylem fibres
done
clear
View Answer play_arrow
The purpose of crop rotation is to
A)
Increase the fertility of soil
done
clear
B)
Decrease the fertility of soil
done
clear
C)
Prevent soil erosion
done
clear
D)
Prevent water erosion
done
clear
View Answer play_arrow
Which of the following is a nitrogen fixing organism?
A)
BGA
done
clear
B)
Rhizobium
done
clear
C)
Both (a) and (b)
done
clear
D)
Aspergillus
done
clear
View Answer play_arrow
To maintain constant body temperature is called
A)
Homeothermic
done
clear
B)
Poikilothermic
done
clear
C)
Homozygous
done
clear
D)
Heterozygous
done
clear
View Answer play_arrow
African sleeping sickness is caused by
A)
Trypanosoma
done
clear
B)
Leishmania
done
clear
C)
Latimeria
done
clear
D)
Plasmodium
done
clear
View Answer play_arrow
During blood clotting which of the following is used?
A)
Co
done
clear
B)
\[C{{a}^{2+}}\]
done
clear
C)
\[N{{a}^{+}}\]
done
clear
D)
\[C{{I}^{-}}\]
done
clear
View Answer play_arrow
The chromosome showing L-shaped structure by the presence of centromere is termed as
A)
Acentric
done
clear
B)
Metacentric
done
clear
C)
Sub-metacentric
done
clear
D)
Telocentric
done
clear
View Answer play_arrow
The infected stage of malarial parasite is
A)
Sporozoite stage
done
clear
B)
Schizozoite stage
done
clear
C)
Merozoite stage
done
clear
D)
Metacryptozoite stage
done
clear
View Answer play_arrow
If Meselson and Stahl?s experiment is continued for four generations in bacteria, the ratio of \[{{15}_{N}}/{{15}_{N}}:{{15}_{N}}/{{14}_{N}}:{{14}_{N}}/{{14}_{N}}\] containing DNA in the fourth generation would be
A)
1 : 1 : 0
done
clear
B)
1 : 4 : 0
done
clear
C)
0 : 1 : 3
done
clear
D)
0 : 1 : 7
done
clear
View Answer play_arrow
In RNAi, the genes are silenced using
A)
ssDNA
done
clear
B)
dsDNA
done
clear
C)
dsRNA
done
clear
D)
ssRNA
done
clear
View Answer play_arrow
Which of the following is concerned with \[C{{O}_{2}}\]fixation?
A)
Krebs cycle
done
clear
B)
Calvin cycle
done
clear
C)
Ornithine cycle
done
clear
D)
Glycolysis
done
clear
View Answer play_arrow
In photosynthesis there is
A)
Reduction of \[{{H}_{2}}O\]
done
clear
B)
Oxidation of \[{{H}_{2}}O\]
done
clear
C)
Oxidation of \[C{{O}_{2}}\]
done
clear
D)
Oxidation of \[N{{O}_{2}}\]
done
clear
View Answer play_arrow
ELISA test is used for the detection of
A)
Antibodies
done
clear
B)
Viral disease
done
clear
C)
AIDS
done
clear
D)
All of these
done
clear
View Answer play_arrow