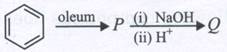-
question_answer1)
Which of the following reagents cannot be used to oxidise primary alcohols to aldehydes?
A)
\[Cr{{O}_{3}}\] in anhydrous medium. done
clear
B)
\[KMn{{O}_{4}}\] in acidic medium. done
clear
C)
Pyridinium chlorochromate. done
clear
D)
Heat in the presence of Cu at 573 K. done
clear
View Solution play_arrow
-
question_answer2)
A primary alcohol, \[{{C}_{3}}{{H}_{8}}O\] [A] on heating with sulphuric acid undergo dehydration to give an alkene, B. B when reacted with HCl gave C, which on treatment with aqueous KOH gives compound D, \[{{C}_{3}}{{H}_{8}}O\]. A and D are
A)
functional isomers done
clear
B)
position isomers done
clear
C)
chain isomers done
clear
D)
stereo isomers. done
clear
View Solution play_arrow
-
question_answer3)
Which of the following reactions will not yield phenol?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer4)
IUPAC name of the compound \[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\mathop{CH}}\,-OC{{H}_{3}}\]is
A)
1-methoxy-l-methylethane done
clear
B)
2-methoxy-2-methylethane done
clear
C)
2-methoxypropane done
clear
D)
isopropylmethyl ether. done
clear
View Solution play_arrow
-
question_answer5)
| Arrange the following alcohols in order of |
| (i) \[{{\left( C{{H}_{3}} \right)}_{3}}C-OH\] |
| (ii) \[{{\left( C{{H}_{3}} \right)}_{2}}CH-OH\] |
| (iii) \[C{{H}_{3}}C{{H}_{2}}OH\] |
A)
(iii) < (ii) < (i) done
clear
B)
(ii) < (i) < (iii) done
clear
C)
(i) < (ii) < (iii) done
clear
D)
(iii) < (i) < (ii) done
clear
View Solution play_arrow
-
question_answer6)
Which of the following compounds will be most easily attacked by an electrophile?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer7)
| Which reducing agent is used for the following conversion? |
| \[RCOOH\to RC{{H}_{2}}OH\] |
A)
\[LiAl{{H}_{4}}\] done
clear
B)
\[NaB{{H}_{4}}\] done
clear
C)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] done
clear
D)
\[KMn{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer8)
Which of the following alcohols reacts most readily with Lucas reagent?
A)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OH\] done
clear
B)
\[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\mathop{CH}}\,-C{{H}_{3}}\] done
clear
C)
\[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\overset{\begin{smallmatrix} C{{H}_{3}} \\ | \end{smallmatrix}}{\mathop{C}}}\,-OH\] done
clear
D)
\[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\mathop{CH}}\,-C{{H}_{2}}OH\] done
clear
View Solution play_arrow
-
question_answer9)
Phenol when treated with excess of bromine water gives a white precipitate of
A)
2, 4, 6-tribromophenol done
clear
B)
o-bromophenol done
clear
C)
p-bromophenol done
clear
D)
bromobenzene. done
clear
View Solution play_arrow
-
question_answer10)
One mole of ethyl acetate on treatment with an excess of \[LiAI{{H}_{4}}\] in dry ether and subsequent acidification produces
A)
1 mol acetic acid + 1 mol ethyl alcohol done
clear
B)
1 mol ethyl alcohol + 1 mol methyl alcohol done
clear
C)
2 moles of ethyl alcohol done
clear
D)
1 mol of 2-butanol. done
clear
View Solution play_arrow
-
question_answer11)
Which of the following are the products shown by the reaction of methoxyethane with HI?
A)
\[{{C}_{2}}{{H}_{5}}I+C{{H}_{3}}OH\] done
clear
B)
\[C{{H}_{3}}I+{{H}_{2}}O\] done
clear
C)
\[{{C}_{2}}{{H}_{5}}OH+{{H}_{2}}O\] done
clear
D)
\[{{C}_{2}}{{H}_{5}}OH+C{{H}_{3}}I\] done
clear
View Solution play_arrow
-
question_answer12)
p-Nitrophenol is a stronger acid than phenol while p-cresol is a weaker acid. This can be explained as
A)
\[C{{H}_{3}}\] group decreases the electron density on oxygen of OH group making p-cresol a weaker acid done
clear
B)
\[N{{O}_{2}}\] group decreases electron density on oxygen of OH group making p-nitrophenol a stronger acid done
clear
C)
\[C{{H}_{3}}\] group increases the electron density on oxygen of OH group making release of \[{{H}^{+}}\]easier done
clear
D)
\[N{{O}_{2}}\] group increases the electron density on oxygen of OH group making release of \[{{H}^{+}}\] easier. done
clear
View Solution play_arrow
-
question_answer13)
Which of the following alcohols will give the most stable carbocation during dehydration?
A)
2-Methyl-1-propanol done
clear
B)
2-Methyl-2-propanol done
clear
C)
1-Butanol done
clear
D)
2-Butanol done
clear
View Solution play_arrow
-
question_answer14)
An organic compound with molecular formula \[{{C}_{4}}{{H}_{10}}O\] does not react with sodium. With excess of HI it gives only one type of alkyl halide. The compound is
A)
\[{{C}_{2}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}\] done
clear
B)
\[C{{H}_{3}}\underset{\begin{smallmatrix} | \\ OC{{H}_{3}} \end{smallmatrix}}{\mathop{CH}}\,C{{H}_{3}}\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OC{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}OH\] done
clear
View Solution play_arrow
-
question_answer15)
Unlike phenol, 2, 4-dinitrophenol is soluble in sodium carbonate solution in water because
A)
presence of two \[N{{O}_{2}}\] groups in the ring makes 2, 4-dinitrophenol a stronger acid than phenol done
clear
B)
presence of two \[N{{O}_{2}}\] groups in the ring makes 2, 4-dinitrophenol a weaker acid than phenol done
clear
C)
presence of two \[N{{O}_{2}}\] groups make the hydrogen bonding easier, making 2, 4-dinitrophenol soluble done
clear
D)
nitro group reacts with \[N{{a}_{2}}C{{O}_{3}}\] while OH group does not. done
clear
View Solution play_arrow
-
question_answer16)
Which of the following is not true in case of reaction with heated copper at \[300{}^\circ C\]?
A)
Phenol\[\to \] benzyl alcohol done
clear
B)
Secondary alcohol \[\to \] ketone done
clear
C)
Primary alcohol\[\to \] aldehyde done
clear
D)
Tertiary alcohol\[\to \] olefin. done
clear
View Solution play_arrow
-
question_answer17)
Which of the following compounds will give tribromo derivative on treatment with bromine water?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer18)
A compound X with the molecular formula \[{{C}_{3}}{{H}_{8}}O\] can be oxidised to another compound Y whose molecular formula is \[{{C}_{3}}{{H}_{6}}{{O}_{2}}\]. The compound X may be
A)
\[C{{H}_{3}}C{{H}_{2}}OC{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}CHO\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OH\] done
clear
D)
\[C{{H}_{3}}CHOHC{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer19)
Which of the following products are not correctly matched in the given reactions?
A)
\[{{C}_{2}}{{H}_{5}}OC{{H}_{3}}+HBr\xrightarrow{373\,K}{{C}_{2}}{{H}_{5}}OH+C{{H}_{3}}Br\] done
clear
B)
\[{{C}_{2}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}+\underset{Excess}{\mathop{2HI}}\,\to {{C}_{2}}{{H}_{5}}I+{{C}_{2}}{{H}_{5}}OH\] done
clear
C)
\[{{C}_{2}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}+HCl\xrightarrow{Cold}\left[ {{\left( {{C}_{2}}{{H}_{5}} \right)}_{2}}{{O}^{+}}H \right]C{{l}^{-}}\] done
clear
D)
\[{{\left( C{{H}_{3}} \right)}_{3}}CO{{C}_{2}}{{H}_{5}}\xrightarrow{HI}{{\left( C{{H}_{3}} \right)}_{3}}CI+{{C}_{2}}{{H}_{5}}OH\] done
clear
View Solution play_arrow
-
question_answer20)
Phenyl methyl ether (anisole) reacts with HI to give phenol and methyl iodide and not iodobenzene and methyl alcohol because
A)
ion prefers to combine with the smaller group in order to minimise steric hindrance done
clear
B)
\[{{I}^{-}}\] ion is not reactive towards benzene done
clear
C)
phenol is formed as a result of hydrolysis of iodobenzene done
clear
D)
methyl alcohol formed during reaction reacts with \[{{I}^{-}}\] to form methyl iodide. done
clear
View Solution play_arrow
-
question_answer21)
| Identify the final product of the reaction sequence. |
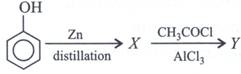 |
A)
Benzophenone done
clear
B)
Acetophenone done
clear
C)
Diphenyl done
clear
D)
Methyl salicylate done
clear
View Solution play_arrow
-
question_answer22)
The COH bond angle in alcohols slightly less than the tetrahedral angle whereas the COC bond angle in ether is slightly greater because
A)
of repulsion between the two bulky R groups. done
clear
B)
O atom in both alcohols and ethers is \[s{{p}^{3}}\]- hybridised. done
clear
C)
lone pair - lone pair repulsion is greater than bond pair-bond pair replusion. done
clear
D)
None of these. done
clear
View Solution play_arrow
-
question_answer23)
The most suitable reagent for the conversion of \[RC{{H}_{2}}OH\to RCHO\] is
A)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] done
clear
B)
\[Cr{{O}_{3}}\] done
clear
C)
\[KMn{{O}_{4}}\] done
clear
D)
PCC. done
clear
View Solution play_arrow
-
question_answer24)
The major product of acid catalyzed dehydration of 2-methylcyclohexanol and butan- 1-ol are respectively
A)
1-methylcyclohexene and but-1-ene done
clear
B)
2-methylcyclohexene and but-2-ene done
clear
C)
2-methylcyclohexene and butane done
clear
D)
1-methylcyclohexene and but-2-ene. done
clear
View Solution play_arrow
-
question_answer25)
Ethers are prepared by the reaction of sodium alkoxides and alkyl halides. Which of the following reagents should be taken to prepare methyl tert-butyl ether?
A)
\[{{\left( C{{H}_{3}} \right)}_{3}}C-Br+NaOC{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}Br+NaOC{{\left( C{{H}_{3}} \right)}_{3}}\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}Br+NaOC{{\left( C{{H}_{3}} \right)}_{2}}\] done
clear
D)
\[{{\left( C{{H}_{3}} \right)}_{2}}C-Br+NaOC{{H}_{2}}C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer26)
IUPAC name of \[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}\underset{\begin{smallmatrix} | \\ C{{H}_{2}}OH \end{smallmatrix}}{\mathop{CH}}\,-C{{H}_{2}}C{{H}_{3}}\]is
A)
3-propylbutan-1-ol done
clear
B)
2-ethylpentan-1-ol done
clear
C)
3-methyl hydroxyhexane done
clear
D)
2-ethyl-2-propyl ethanol done
clear
View Solution play_arrow
-
question_answer27)
Compound \[{{C}_{2}}{{H}_{6}}O\] has two isomers X and Y. On reaction with HI, X gives alkyl iodide and water while Y gives alkyl iodide and alcohol. Compounds X and Y are respectively
A)
\[{{C}_{2}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}\]and \[C{{H}_{3}}O{{C}_{2}}{{H}_{5}}\] done
clear
B)
\[C{{H}_{3}}OC{{H}_{3}}\] and \[{{C}_{2}}{{H}_{5}}OC{{H}_{3}}\] done
clear
C)
\[{{C}_{2}}{{H}_{5}}OH\] and \[C{{H}_{3}}OC{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}OH\] and \[C{{H}_{3}}OC{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer28)
| In the following sequence of reactions, |
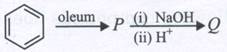 |
| the compound Q formed will be |
A)
aniline done
clear
B)
phenol done
clear
C)
benzaldehyde done
clear
D)
benzene sulphonic acid. done
clear
View Solution play_arrow
-
question_answer29)
Out of benzene and phenol, phenol is more easily nitrated because
A)
presence of OH group in phenol increases the electron density at ortho and para-position done
clear
B)
presence of OH gorup in phenol decreases the electron density at ortho and para-position done
clear
C)
nitration being electrophilic substitution requires less density at ortho and para-position done
clear
D)
phenol is more reactive than benzene due to-R effect. done
clear
View Solution play_arrow
-
question_answer30)
The reaction between phenol and chloroform in the presence of aqueous NaOH is
A)
nucleophilic substitution reaction done
clear
B)
electrophilic addition reaction done
clear
C)
electrophilic substitution reaction done
clear
D)
nucleophilic addition reaction done
clear
View Solution play_arrow
-
question_answer31)
Which of the following statements is correct?
A)
The reaction of methyl magnesium iodide with acetone followed by hydrolysis gives secondary butanol. done
clear
B)
Primary alcohols are dehydrated easily than secondary and tertiary alcohols. done
clear
C)
Tertiary alcohol is more acidic than primary alcohol. done
clear
D)
Tertiary butyl alcohol gives turbidity fastest with Lucas reagent. done
clear
View Solution play_arrow
-
question_answer32)
For the reaction, \[{{C}_{2}}{{H}_{5}}OH+HX\to {{C}_{2}}{{H}_{5}}X+{{H}_{2}}O\]; the order of reactivity is
A)
\[HCl>HBr>HI\] done
clear
B)
\[HI\text{ }>\text{ }HBr\text{ }>\text{ }HCl\] done
clear
C)
\[HBr\text{ }>\text{ }HCl\text{ }>\text{ }HI\] done
clear
D)
\[HI\text{ }>\text{ }HCl\text{ }>\text{ }HBr\] done
clear
View Solution play_arrow
-
question_answer33)
Arrange the following compounds in increasing order of boiling points. Propan-1-ol butan-1-ol, butan-2-ol, pentan-1-ol.
A)
Propan-1-ol < butan-2-ol < butan-1-ol < pentan-1-ol done
clear
B)
Propan-1-ol < butan-1-ol < butan-2-ol < pentan-1-ol done
clear
C)
Pentan-1-ol < butan-2-ol < butan-1-ol < propan-1-ol done
clear
D)
Pentan-1-ol < butan-1-ol < butan-2-ol < propan-1-ol done
clear
View Solution play_arrow
-
question_answer34)
The total number of isomers possible for compound with molecular formula \[{{C}_{5}}{{H}_{12}}O\] having \[-OH\] as functional group is
A)
8 done
clear
B)
6 done
clear
C)
4 done
clear
D)
12 done
clear
View Solution play_arrow
-
question_answer35)
An equimolar quantities of ethanol and propanol is heated with cone. \[{{H}_{2}}S{{O}_{4}}\]. The products formed are
A)
\[{{C}_{2}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}\] done
clear
B)
\[{{C}_{3}}{{H}_{7}}O{{C}_{3}}{{H}_{7}}\] done
clear
C)
\[{{C}_{2}}{{H}_{5}}O{{C}_{3}}{{H}_{7}}\] done
clear
D)
all of these. done
clear
View Solution play_arrow
-
question_answer36)
Which of the following is not a characteristic of alcohol?
A)
They are lighter than water. done
clear
B)
Their boiling points rise fairly uniformly with rising molecular weight. done
clear
C)
Lower members are insoluble in water and organic solvents but the solubility regularly increases with molecular mass. done
clear
D)
Lower members have a pleasant smell and burning taste, higher members are colourless and tasteless. done
clear
View Solution play_arrow
-
question_answer37)
Conversion of phenol to salicylic acid and to salicyaldehyde are known as (respectively)
A)
Reimer-Tiemann reaction and Kolbe's reaction done
clear
B)
Williamson's synthesis and Hydroboration- oxidation done
clear
C)
Kolbe's reaction and Williamson's synthesis done
clear
D)
Kolbe's reaction and Reimer-Tiemann reaction done
clear
View Solution play_arrow
-
question_answer38)
\[{{\left( C{{H}_{3}} \right)}_{3}}C-C{{H}_{2}}OH\xrightarrow[170{}^\circ C]{Conc.\,{{H}_{2}}S{{O}_{4}}}X\] In the reaction, X is
A)
\[{{\left( C{{H}_{3}} \right)}_{2}}C=CHC{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}C\equiv CH\] done
clear
C)
\[{{\left( C{{H}_{3}} \right)}_{2}}CHC{{H}_{2}}C{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}-C{{H}_{2}}-\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\mathop{C}}\,=C{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer39)
Cumene on reaction with oxygen followed by hydrolysis gives
A)
\[C{{H}_{3}}OH\] and \[{{C}_{6}}{{H}_{5}}COC{{H}_{3}}\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}OH\] and \[{{\left( C{{H}_{3}} \right)}_{2}}O\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}OC{{H}_{3}}\] and \[C{{H}_{3}}OH\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}OH\] and \[C{{H}_{3}}COC{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer40)
Benzoquinone is prepared by reaction of phenol with
A)
\[N{{a}_{2}}C{{r}_{2}}{{O}_{7}}\], \[{{H}_{2}}S{{O}_{4}}\] done
clear
B)
\[KMn{{O}_{4}}\], \[{{H}_{2}}S{{O}_{4}}\] done
clear
C)
\[N{{a}_{2}}Cr{{O}_{4}}\], HCl done
clear
D)
\[{{K}_{2}}Mn{{O}_{4}}\], \[{{H}_{2}}S{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer41)
| Which of the following is the proper method to prepare n-hexane from n-propyl alcohol? |
| \[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OH\xrightarrow{(X)}C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Br\xrightarrow{(Y)}\] |
| \[C{{H}_{3}}{{\left( C{{H}_{2}} \right)}_{4}}C{{H}_{3}}\] |
A)
(X) \[\to \]HBr, (Y) \[\to \]HCN done
clear
B)
(X) \[\to \]HBr, (Y) \[\to \]Na, ether done
clear
C)
(X) \[\to \]\[B{{r}_{2}}\], (Y) \[\to \]\[C{{H}_{3}}CN\] done
clear
D)
(X) \[\to \]\[B{{r}_{2}}\], (Y) \[\to \]\[KMn{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer42)
Which of the following alcohols gives the best yield of dialkyl ether on being heated with a trace of sulphuric acid?
A)
2-Pentanol done
clear
B)
2-Methyl-2-butanol done
clear
C)
1-Pentanol done
clear
D)
2-Propanol done
clear
View Solution play_arrow
-
question_answer43)
Which of the following alcohols is dehydrated most easily with cone. \[{{H}_{2}}S{{O}_{4}}\]?
A)
\[p-{{O}_{2}}N{{C}_{6}}{{H}_{4}}CH\left( OH \right)C{{H}_{3}}\] done
clear
B)
\[p-Cl{{C}_{6}}{{H}_{4}}CH(OH)C{{H}_{3}}\] done
clear
C)
\[p-C{{H}_{3}}O{{C}_{6}}{{H}_{4}}CH(OH)C{{H}_{3}}\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}CH\left( OH \right)C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer44)
Propanone on reaction with alkyl magnesium bromide followed by hydrolysis will produce
A)
primary alcohol done
clear
B)
secondary alcohol done
clear
C)
tertiary alcohol done
clear
D)
carboxylic acid. done
clear
View Solution play_arrow
-
question_answer45)
Complete the missing links. \[C{{H}_{3}}CHBrC{{H}_{3}}\xrightarrow{alc.\,KOH}X\xrightarrow[Peroxide]{HBr}Y\xrightarrow{C{{H}_{3}}ONa}Z\]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer46)
The decreasing order of boiling points of the following alcohols is
A)
3-methylbutan-2-ol > 2-methylbutan-2-ol > pentan-1-ol done
clear
B)
pentan-1-ol > 3-methylbutan-2-ol > 2 -methylbutan-2-ol done
clear
C)
2-methylbutan-2-ol > 3-methylbutan-2-ol > pentan-1-ol done
clear
D)
2-methylbutan-2-ol > pentan-1-ol > 3-methylbutan-2-ol done
clear
View Solution play_arrow
-
question_answer47)
Which of the following compounds does not react with NaOH?
A)
\[C{{H}_{3}}COOH\] done
clear
B)
\[C{{H}_{3}}CON{{H}_{2}}\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}OH\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}OH\] done
clear
View Solution play_arrow
-
question_answer48)
Anisole on reaction with chloromethane in presence of anhydrous \[AlC{{l}_{3}}\] gives
A)
o-methyl anisole and p-methoxy anisole done
clear
B)
p-methyl anisole and p-methoxy anisole done
clear
C)
o-methyl anisole and p-methyl anisole done
clear
D)
o-methoxy acetophenone and p-methoxy acetophenone. done
clear
View Solution play_arrow
-
question_answer49)
Vapours of an alcohol X when passed over hot reduced copper, produce an alkene, the alcohol is
A)
primary alcohol done
clear
B)
secondary alcohol done
clear
C)
tertiary alcohol done
clear
D)
dihydric alcohol. done
clear
View Solution play_arrow
-
question_answer50)
Acid catalysed dehydration of t-butanol is faster than that of n-butanol because
A)
tertiary carbocation is more stable than primary carbocation done
clear
B)
primary carbocation is more stable than tertiary carbocation done
clear
C)
t-butanol has a higher boiling point done
clear
D)
rearrangement takes place during dehydration of t-butanol. done
clear
View Solution play_arrow
-
question_answer51)
A)
[A] \[\to \] (ii), [B] \[\to \](iii), [C] \[\to \] (iv), [D] \[\to \](i) done
clear
B)
[A] \[\to \] (i), [B] \[\to \](ii), [C] \[\to \] (iii), [D]\[\to \](iv) done
clear
C)
[A] \[\to \](iv), [B] \[\to \] (iii), [C] \[\to \] (ii), [D] \[\to \] (i) done
clear
D)
[A] \[\to \] (ii), [B] \[\to \] (iv), [C] \[\to \] (i), [D] \[\to \](iii) done
clear
View Solution play_arrow
-
question_answer52)
Ether is obtained from ethyl alcohol in presence of \[{{H}_{2}}S{{O}_{4}}\] at
A)
113 K done
clear
B)
443 K done
clear
C)
413 K done
clear
D)
213 K done
clear
View Solution play_arrow
-
question_answer53)
How many alcohols with molecular formula \[{{C}_{4}}{{H}_{10}}O\] are chiral in nature?
A)
1 done
clear
B)
2 done
clear
C)
3 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer54)
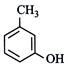
IUPAC name of m-cresol is
A)
3-methylphenol done
clear
B)
3-chlorophenol done
clear
C)
3-methoxyphenol done
clear
D)
benzene-1, 3-diol. done
clear
View Solution play_arrow
-
question_answer55)
Which of the following is most acidic?
A)
Benzyl alcohol done
clear
B)
Cyclohexanol done
clear
C)
Phenol done
clear
D)
m-Chlorophenol done
clear
View Solution play_arrow
-
question_answer56)
Boiling point of ethyl alcohol is greater than ether due to
A)
van der Waals forces done
clear
B)
London forces done
clear
C)
polarity done
clear
D)
hydrogen bonding. done
clear
View Solution play_arrow
-
question_answer57)
Which of the following is phenol?
A)
Cresol done
clear
B)
Catechol done
clear
C)
Benzenol done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer58)
Propene, \[C{{H}_{3}}CH=C{{H}_{2}}\] can be converted into 1-propanol by oxidation. Indicate which set of reagents amongst the following is ideal for the above conversion.
A)
\[KMn{{O}_{4}}\](alkaline) done
clear
B)
Osmium tetroxide \[\left( Os{{O}_{4}}/C{{H}_{2}}C{{l}_{2}} \right)\] done
clear
C)
\[{{B}_{2}}{{H}_{6}}\]and alk.\[{{H}_{2}}{{O}_{2}}\] done
clear
D)
\[{{O}_{3}}/Zn\] done
clear
View Solution play_arrow
-
question_answer59)
Among the following sets of reactants which one produces anisole?
A)
\[C{{H}_{3}}CHO\]; RMgX done
clear
B)
\[{{C}_{6}}{{H}_{5}}OH\]; NaOH; \[C{{H}_{3}}I\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}OH\]; neutral \[FeC{{l}_{3}}\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}C{{H}_{3}}\], \[C{{H}_{3}}COCl\], \[AlC{{l}_{3}}\] done
clear
View Solution play_arrow
-
question_answer60)
Primary, secondary and tertiary alcohols can be distinguished by
A)
Borsche's test done
clear
B)
Lucas test done
clear
C)
Hinsberg's test done
clear
D)
Tollens'test done
clear
View Solution play_arrow
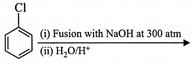 done
clear
done
clear
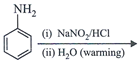 done
clear
done
clear
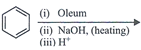 done
clear
done
clear
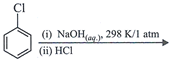 done
clear
done
clear
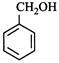 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear