A) \[C{{H}_{3}}CHO\]; RMgX
B) \[{{C}_{6}}{{H}_{5}}OH\]; NaOH; \[C{{H}_{3}}I\]
C) \[{{C}_{6}}{{H}_{5}}OH\]; neutral \[FeC{{l}_{3}}\]
D) \[{{C}_{6}}{{H}_{5}}C{{H}_{3}}\], \[C{{H}_{3}}COCl\], \[AlC{{l}_{3}}\]
Correct Answer: B
Solution :
[a] \[\underset{~2{}^\circ \,alcohol}{\mathop{C{{H}_{3}}CHO+RMgX\xrightarrow{ether}C{{H}_{3}}-\overset{\begin{smallmatrix} R \\ | \end{smallmatrix}}{\mathop{CH}}\,-OH}}\,\] [b] \[\underset{\begin{smallmatrix} Sodium~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~ \\ Phenoxide \end{smallmatrix}}{\mathop{{{C}_{6}}{{H}_{5}}OH+NaOH\xrightarrow[-{{H}_{2}}O]{}{{C}_{6}}{{H}_{5}}ONa}}\,\] \[\underset{\begin{smallmatrix} Methyl\text{ }phenyl\text{ }ether\text{ }\left( Williamsons\text{ }synthesis \right) \\ \left( Anisole \right) \end{smallmatrix}}{\mathop{{{C}_{6}}{{H}_{5}}OC{{H}_{3}}+NaI\xleftarrow[\Delta ]{C{{H}_{3}}I}}}\,\] [c]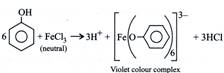 [d]
[d] 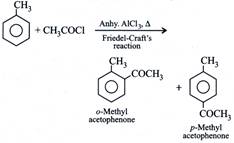
You need to login to perform this action.
You will be redirected in
3 sec
