The velocity of water waves may depend upon their wavelength\[\lambda \], the density of water p and the acceleration due to gravity g. The method of dimensions gives the relation between these quantities as:
A)
\[{{v}^{2}}\propto \lambda {{g}^{-1}}{{\rho }^{-1}}\]
done
clear
B)
\[{{v}^{2}}\propto \,g\lambda \rho \]
done
clear
C)
\[{{v}^{2}}\propto \,g\lambda \]
done
clear
D)
\[{{v}^{2}}\propto \,{{g}^{-1}}\lambda {{\rho }^{-3}}\]
done
clear
View Answer play_arrow
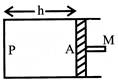
A cylindrical piston of mass M slides smoothly inside a long cylinder closed at one end, enclosing a certain mass of gas. The cylinder is kept with its axis horizontal. If the piston is disturbed from its equilibrium position, it oscillates simple harmonically period of oscillation will be:
A)
\[T=2\pi \,\sqrt{\frac{Mh}{PA}}\]
done
clear
B)
\[T=2\pi \,\sqrt{\frac{Mh}{Ph}}\]
done
clear
C)
\[T=2\pi \,\sqrt{\frac{Mh}{PAh}}\]
done
clear
D)
\[T=2\pi \,\sqrt{MPhA}\]
done
clear
View Answer play_arrow
A body is projected horizontally from the top of a tower with a speed of 20m/s. After 4 sec the displacement of the body is:
A)
\[40m\]
done
clear
B)
\[80m\]
done
clear
C)
\[80\sqrt{2m}\]
done
clear
D)
\[\frac{80}{\sqrt{2}}m\]
done
clear
View Answer play_arrow
The bulk modulus of rubber is\[9.1\times {{10}^{8}}\text{ }N/{{m}^{2}}\]. To what depth a rubber ball be taken in a lake so that its volume is decreased by 0.1%?
A)
25m
done
clear
B)
93m
done
clear
C)
200m
done
clear
D)
500m
done
clear
View Answer play_arrow
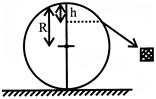
A particle originally at rest at the highest point of a smooth vertical circle is slightly displaced. It will leave the circle at a vertical distance 'h' below the highest point, such that:
A)
\[h=R\]
done
clear
B)
\[h=\frac{R}{2}\]
done
clear
C)
\[h=\frac{R}{3}\,\]
done
clear
D)
\[h=\,2R\]
done
clear
View Answer play_arrow
A particle starting from the origin (0,0) moves in straight line in the (x, y) plane. Its coordinates at a later time are\[\left( \sqrt{3},3 \right)\]. The path of the particle makes with the x-axis an angle of:
A)
\[\frac{\pi }{4}\]
done
clear
B)
\[\frac{\pi }{6}\]
done
clear
C)
\[\frac{\pi }{3}\]
done
clear
D)
\[0{}^\circ \]
done
clear
View Answer play_arrow
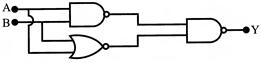
The circuit as shown in figure is equivalent to:
A)
OR gate
done
clear
B)
NOR gate
done
clear
C)
NAND gate
done
clear
D)
AND gate
done
clear
View Answer play_arrow
A rectangular film of liquid is extended from \[(4cm\times 2cm)\]to\[(5cm\times 4cm)\]If the work done is \[3\times {{10}^{-4}}J,\]the value of the surface tension of the liquid is:
A)
8N/m
done
clear
B)
0.250N/m
done
clear
C)
0.125N/m
done
clear
D)
0.2N/m
done
clear
View Answer play_arrow
An electron and a photon possess the same de-broglie wavelength. If and \[{{E}_{e}}\] are the\[{{E}_{p}}\]. energies of electron and photon respectively and v and c are their respective velocities, the is \[\frac{{{E}_{e}}}{{{E}_{p}}}\]:
A)
\[\frac{v}{c}\]
done
clear
B)
\[\frac{v}{2c}\]
done
clear
C)
\[\frac{v}{3c}\]
done
clear
D)
\[\frac{v}{4c}\]
done
clear
View Answer play_arrow
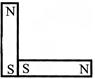
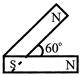
Following figures show the arrangement of bar magnets in different configuration. Each magnet has magnetic dipole moment m. Which configuration has highest net magnetic dipole moment?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Electron in hydrogen atom first jumps from third excited state to second excited state and then from second excited to first excited state. The ratio of the wavelength \[{{\lambda }_{1}}:{{\lambda }_{2}}\] emitted in two cases is:
A)
7:5
done
clear
B)
27:20
done
clear
C)
27:5
done
clear
D)
20:7
done
clear
View Answer play_arrow
A proton carrying 1MeV kinetic energy is moving in a circular path of radius R in uniform magnetic field. What should be the energy of an\[\alpha \]particle to describe a circle a circle of same radius in the same field?
A)
2MeV
done
clear
B)
1 MeV
done
clear
C)
0.5MeV
done
clear
D)
4 MeV
done
clear
View Answer play_arrow
A mercury drop of radius 1cm is broker into 106 droplets of equal size. The work done is \[(surface\text{ }tension\text{ }=\text{ }35\times {{10}^{-2}}\text{ }N/m)\]
A)
\[4.\,35\times {{10}^{-2}}J\]
done
clear
B)
\[4.35\times {{10}^{-3}}J\]
done
clear
C)
\[4.35\times {{10}^{-6}}J\]
done
clear
D)
\[4.35\times {{10}^{-8}}J\]
done
clear
View Answer play_arrow
If m is the mass of an electron and C is the speed of the light, the ratio of the wavelength of a photon of energy E to that of the electron of the same energy is:
A)
\[C\sqrt{\frac{2m}{E}}\]
done
clear
B)
\[\sqrt{\frac{2m}{E}}\]
done
clear
C)
\[\sqrt{\frac{2m}{CE}}\]
done
clear
D)
\[\sqrt{\frac{m}{E}}\]
done
clear
View Answer play_arrow
A bullet of mass m moving with velocity v block strikes a suspended wooden block of mass M. If the block rises to a height h. The initial velocity of the block will be:
A)
\[\sqrt{2gh}\]
done
clear
B)
\[\frac{M+m}{m}\sqrt{gh}\]
done
clear
C)
\[\frac{m}{m+M}(2gh)\]
done
clear
D)
\[\frac{M+m}{M}\sqrt{2gh}\]
done
clear
View Answer play_arrow
A junction diode has a resistance of 25ft when forward biased and 2500ft when reverse biased. The current in the diode for the arrangement shown will be:
A)
\[\frac{1}{15}A\]
done
clear
B)
\[\frac{1}{7}A\]
done
clear
C)
\[\frac{1}{25}A\]
done
clear
D)
\[\frac{1}{480}A\]
done
clear
View Answer play_arrow
The height vertically above the earth's surface at which the acceleration due to gravity becomes 1% of its value at the surface is:
A)
8R
done
clear
B)
9R
done
clear
C)
10R
done
clear
D)
20R
done
clear
View Answer play_arrow
A long solenoid has 1000 turns. When a current of 4A flows through it, the magnetic flux linked with each turn of the solenoid is\[4\times {{10}^{-3}}\,Wb\]. The self-inductance of the solenoid is:
A)
4H
done
clear
B)
3H
done
clear
C)
2H
done
clear
D)
1H
done
clear
View Answer play_arrow
The ratio of velocity of sound in Hydrogen and oxygen at STP is:
A)
\[16:1~\]
done
clear
B)
\[8:1\]
done
clear
C)
\[4:1\]
done
clear
D)
\[2:1\]
done
clear
View Answer play_arrow
In an ammeter, 0.2% of main current passes through the galvanometer. If resistance of galvanometer is G, then the resistance of ammeter will be:
A)
\[\frac{G}{499}\]
done
clear
B)
\[\frac{499G}{500}\]
done
clear
C)
\[\frac{G}{500}\]
done
clear
D)
\[\frac{500G}{499}\]
done
clear
View Answer play_arrow
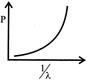
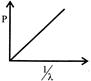
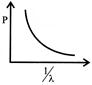
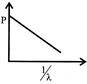
Which of the following figures represent the variation of particle momentum and the associated de-broglie wavelength:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
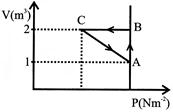
An ideal gas is taken through the cycle\[A\to B\to C\to A,\] as shown in figure. If the net heat supplied to the gas in the cycle is 5J, the work done by the gas in the process \[C\to \text{ }A\]is:
A)
- 5J
done
clear
B)
- 10J
done
clear
C)
- 15J
done
clear
D)
- 20J
done
clear
View Answer play_arrow
A police jeep is chasing with velocity of 45km/h, a their in another jeep moving with velocity 153km/h. Police fires a bullet with muzzle velocity of 180m/s. The velocity with which it will strike the car of the their is:
A)
150m/s
done
clear
B)
27m/s
done
clear
C)
450m/s
done
clear
D)
250m/s
done
clear
View Answer play_arrow
**A satellite of mass m revolves around the earth of radius R at a height x from its surface. If "g" is the acceleration due to gravity on the surface of the earth, the orbital speed of the satellite is:
A)
\[\sqrt{gR}\]
done
clear
B)
\[\sqrt{\frac{g{{R}^{2}}}{R+x}}\]
done
clear
C)
\[\sqrt{\frac{g{{R}^{3}}}{R+x}}\]
done
clear
D)
\[\sqrt{\frac{gR}{g+x}}\]
done
clear
View Answer play_arrow
A body of mass 2kg is thrown up vertically with kinetic energy of 490J. It g = 10m/s2, the height at which the kinetic energy of the body becomes half of the original value is:
A)
50m
done
clear
B)
25m
done
clear
C)
12.5m
done
clear
D)
19.6m
done
clear
View Answer play_arrow
A person is observing two trains one coming towards him and other leaving with the same velocity 4m/s. If their whistling frequencies are 240Hz each, then the number of beats per second are heard by the person will be: (velocity of sound = 320m/s)
A)
3
done
clear
B)
6
done
clear
C)
9
done
clear
D)
Zero
done
clear
View Answer play_arrow
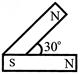
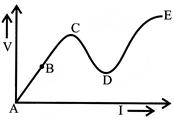
Which part represents the negative dynamic resistance?
A)
AB
done
clear
B)
BC
done
clear
C)
CD
done
clear
D)
DE
done
clear
View Answer play_arrow
The acceleration of a particle is increasing linearly with time t as bt. The particle starts from the origin with an initial velocity\[{{V}_{o}}\]. The distance travelled by the particle in time twill be:
A)
\[{{V}_{o}}t+\frac{1}{3}b{{t}^{2}}\]
done
clear
B)
\[{{V}_{o}}t+\frac{1}{3}b{{t}^{3}}\]
done
clear
C)
\[{{V}_{o}}t+\frac{1}{6}b{{t}^{2}}\]
done
clear
D)
\[{{V}_{o}}t+\frac{1}{2}b{{t}^{2}}\]
done
clear
View Answer play_arrow
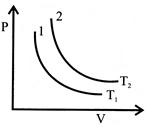
For a certain mass of gas, the isothermal curves between P and \[{{T}_{1}}\] at \[{{T}_{2}}\]and temperatures are 1 and 2 as shown in figure. Then.
A)
\[{{T}_{1}}={{T}_{2}}\]
done
clear
B)
\[{{T}_{1}}>{{T}_{2}}\]
done
clear
C)
\[{{T}_{2}}>{{T}_{1}}\]
done
clear
D)
Can?t predicted
done
clear
View Answer play_arrow
Two coherent sources of intensity ratio a interfere In interference pattern, \[\frac{I{{\,}_{\max }}-{{I}_{\min }}}{{{I}_{\max }}+{{I}_{\min }}}\] is equal to:
A)
\[\frac{2\alpha }{1+\alpha }\]
done
clear
B)
\[\frac{2\sqrt{\alpha }}{1+\alpha }\]
done
clear
C)
\[\frac{2\alpha }{1+\sqrt{\alpha }}\]
done
clear
D)
\[\frac{1+\alpha }{2\alpha }\]
done
clear
View Answer play_arrow
A circuit contains an ammeter, a battery of 30V and a resistance \[40.8\Omega \]all connected in series. If the ammeter has a coil of resistance \[480\Omega \] and a shunt of\[20\Omega \], the reading in the ammeter will be:
A)
2A
done
clear
B)
1A
done
clear
C)
0.5A
done
clear
D)
0.25A
done
clear
View Answer play_arrow
In vacuum, to travel distance d, light takes time t and in medium to travel 5d, it takes time T. The critical angle of the medium is:
A)
\[{{\sin }^{-1}}\,\left( \frac{5T}{t} \right)\]
done
clear
B)
\[{{\sin }^{-1}}\,\left( \frac{5t}{3T} \right)\]
done
clear
C)
\[{{\sin }^{-1}}\,\left( \frac{5t}{T} \right)\]
done
clear
D)
\[{{\sin }^{-1}}\,\left( \frac{3t}{5T} \right)\]
done
clear
View Answer play_arrow
A block is pushed momentarily on a horizontal surface with initial velocity 'V'. If \[\mu \] is the coefficient of sliding friction between the block and surface, the block will come to rest after time.
A)
\[\frac{v}{\mu g}\]
done
clear
B)
(b)\[\frac{\mu g}{v}\]
done
clear
C)
\[\frac{v\mu }{g}\]
done
clear
D)
\[\frac{\mu g}{v}\]
done
clear
View Answer play_arrow
By sucking through a straw, a student can reduce the pressure in his lungs to 750mm of Hg\[(density\text{ }=\text{ }13.6\text{ }g/c{{m}^{3}})\]. Using the straw he can drink water from a glass upto a maximum depth of:
A)
10cm
done
clear
B)
75cm
done
clear
C)
13.6cm
done
clear
D)
1.36cm
done
clear
View Answer play_arrow
At a distance of 1m from a fixed charge of ImC, a particle of mass 2g and charge \[1\mu C\] is held stationary. Both the charges are placed on a smooth horizontal surface. If the particle is made free to move, then its speed at distance of 10m from the fixed charge will be:
A)
10m/s
done
clear
B)
20m/s
done
clear
C)
60m/s
done
clear
D)
90m/s
done
clear
View Answer play_arrow
In an AC circuit the reactance of a coil is \[\sqrt{3}\] times its resistance, the phase difference between the voltage across the coil to the current through the coil will be:
A)
\[\frac{\pi }{3}\]
done
clear
B)
\[\frac{\pi }{2}\]
done
clear
C)
\[\frac{\pi }{4}\]
done
clear
D)
\[\frac{\pi }{6}\]
done
clear
View Answer play_arrow
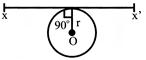
A thin wire of length \[\ell \] and uniform linear mass density \[\rho \] is bent into a circular loop with centre \[o\] and radius r as shown in figure. The moment of inertia of the loop about the axis xx' is:
A)
\[\frac{3\rho {{\ell }^{3}}}{8{{\pi }^{2}}}\]
done
clear
B)
\[\frac{\rho {{\ell }^{3}}}{16{{\pi }^{2}}}\]
done
clear
C)
\[\frac{3\rho {{\ell }^{3}}}{8{{\pi }^{2}}r}\]
done
clear
D)
\[\frac{\rho {{\ell }^{3}}}{8{{\pi }^{2}}r}\]
done
clear
View Answer play_arrow
Eight small drops, each of radius r and having same charge q are combined to form a big drop. The ratio between the potentials of the bigger drop and the smaller drop is:
A)
8 : 1
done
clear
B)
4 : 1
done
clear
C)
2 : 1
done
clear
D)
1 : 8
done
clear
View Answer play_arrow
A mass of 10gm moving horizontally with a velocity of 100 cm/sec, strikes a pendulum bob of mass 10gm. The two masses sticks together. The maximum height reached by the system now is: \[[g=10m/{{s}^{2}}]\]
A)
Zero
done
clear
B)
5cm
done
clear
C)
2.5cm
done
clear
D)
1.25cm
done
clear
View Answer play_arrow
D. A step up transformer operates on a 230V line and supplies current of 2A to a load. The ratio of the primary and secondary windings is 1: 25. The current in the primary is:
A)
15A
done
clear
B)
50A
done
clear
C)
25A
done
clear
D)
12.5A
done
clear
View Answer play_arrow
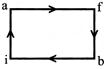
When a system is taken from state i to state f along the path iaf, it is found that Q=50 cal and W = 20 cal. along the path ibf Q = 36 cal. W along the path ibf is:
A)
14 cal
done
clear
B)
6cal
done
clear
C)
16 cal
done
clear
D)
66 cal
done
clear
View Answer play_arrow
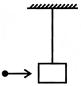
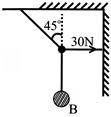
In the figure shown, the tension in the horizontal cord is 30N. Find the weight of the body B:
A)
40N
done
clear
B)
30N
done
clear
C)
20N
done
clear
D)
10N
done
clear
View Answer play_arrow
A wooden ball of density \[\rho \] is immersed in water of density \[\sigma \] to a depth h and then released. The height H above the surface of water upto which the ball will jump out of water is:
A)
\[\frac{\sigma h}{\rho }\]
done
clear
B)
\[\left[ \frac{\sigma }{\rho }+1 \right]h\]
done
clear
C)
\[h\]
done
clear
D)
\[\left[ \frac{\sigma }{\rho }-1 \right]h\]
done
clear
View Answer play_arrow
If \=100cm, \=90 cm and velocity of sound = 396m/s. The number of beats (in Hz) are:
A)
4
done
clear
B)
2
done
clear
C)
3
done
clear
D)
44
done
clear
View Answer play_arrow
A marble block of mass 2kg lying on ice when given a velocity of 6m/s is stopped by friction in 10s. Then the coefficient of friction is:
A)
0.02
done
clear
B)
0.03
done
clear
C)
0.06
done
clear
D)
0.01
done
clear
View Answer play_arrow
The specific heat of a metal in 0.16. Its approximate atomic weight would be:
A)
32
done
clear
B)
16
done
clear
C)
40
done
clear
D)
64
done
clear
View Answer play_arrow
A proton is about 1840 times heavier than an electron. When it is accelerated by a potential difference of IkV, its kinetic energy will be:
A)
1840 KeV
done
clear
B)
1/1840KeV
done
clear
C)
1 KeV
done
clear
D)
920 KeV
done
clear
View Answer play_arrow
The correct order of radii is:
A)
\[N<Be<B~\]
done
clear
B)
\[{{F}^{-}}<{{O}^{2}}-<\text{ }{{N}^{3-}}\]
done
clear
C)
\[Na<Li<K~\]
done
clear
D)
\[F{{e}^{2+}}\,<F{{e}^{2-}}<F{{e}^{4+}}\]
done
clear
View Answer play_arrow
According to Bohr's theory, the angular momentum of an electron in 5th orbit is:
A)
10h/Ti
done
clear
B)
2.5h/7i
done
clear
C)
25h/7i
done
clear
D)
1.0h/Ti
done
clear
View Answer play_arrow
Which among the following has largest dipole moment?
A)
\[N{{H}_{3}}\]
done
clear
B)
\[{{H}_{2}}O\]
done
clear
C)
\[HI\]
done
clear
D)
\[S{{O}_{3}}\]
done
clear
View Answer play_arrow
Which contains both polar and non-polar bonds?
A)
\[N{{H}_{3}}Cl\]
done
clear
B)
\[HCN\]
done
clear
C)
\[{{H}_{2}}{{O}_{2}}\]
done
clear
D)
\[C{{H}_{4}}\]
done
clear
View Answer play_arrow
Number of sigma bonds in\[{{P}_{4}}{{O}_{10}}\] is:
A)
6
done
clear
B)
7
done
clear
C)
17
done
clear
D)
16
done
clear
View Answer play_arrow
Match the lists I and II and write the correct matching:
List I (species) List II (Geometry) A. \[{{H}_{3}}{{O}^{+}}\] 1. Planar B. \[{{H}_{2}}C=NH\] 2. Angular C. \[ClO_{2}^{-}\] 3. Tetrahedral D. \[NH_{4}^{+}\] 4. Trigonal bi-pyramidal E. \[PC{{l}_{5}}\] 5. Pyramidal
A)
A-2, B-1, C-3, D-5, E-4
done
clear
B)
A-1, B-5, C-2, D-3, E-4
done
clear
C)
A-5, B-1, C-2, D-3, E-4
done
clear
D)
A-3, B-1, C-4, D-5, E-2
done
clear
View Answer play_arrow
Triple point of water is:
A)
273K
done
clear
B)
373K
done
clear
C)
203K
done
clear
D)
193K
done
clear
View Answer play_arrow
The bond energy of an O-H bond is 109 k cal/ mol. When a mole of water is formed, then:
A)
109 k cal is released
done
clear
B)
218 k cal is released
done
clear
C)
109 k cal is absorbed
done
clear
D)
218 k cal is absorbed
done
clear
View Answer play_arrow
1 mole of \[{{N}_{2}}\] and 2 moles of \[{{H}_{2}}\] are allowed to react in \[d{{m}^{3}}\]vessel. At equilibrium, 0.8 mole of \[N{{H}_{3}}\]is formed. The concentration of \[{{H}_{2}}\] in the vessel is:
A)
0.6 mole
done
clear
B)
0.8 mole
done
clear
C)
0.2 mole
done
clear
D)
0.4 mole
done
clear
View Answer play_arrow
pH of a solution produced when an aqueous solution of pH 6 is mixed with an equal volume of an aqueous solution of \[p{{H}_{3}}\] is about:
A)
4.5
done
clear
B)
4.0
done
clear
C)
4.3
done
clear
D)
3.3
done
clear
View Answer play_arrow
The products formed when an aqueous solution of NaBr is electrolyzed in a cell having inert electrodes are:
A)
\[Na\,\,and\,\,B{{r}_{2}}\]
done
clear
B)
\[Na\,\,and\,\,{{O}_{2}}\]
done
clear
C)
\[{{H}_{2}},\,B{{r}_{2}},\,\,NaOH\]
done
clear
D)
\[{{H}_{2}}\,and\,\,{{O}_{2}}\]
done
clear
View Answer play_arrow
Heavy water is obtained by:
A)
Boiling water
done
clear
B)
Heating \[{{H}_{2}}{{O}_{2}}\]
done
clear
C)
Prolonged electrolysis of \[{{H}_{2}}O\]
done
clear
D)
Fractional distillation of \[{{H}_{2}}O\]
done
clear
View Answer play_arrow
A sodium salt of unknown anion when treated with \[MgC{{l}_{2}}\] gives white precipitates only-on boiling. The anion is:
A)
\[SO_{4}^{2-}\]
done
clear
B)
\[HCO_{3}^{-}\]
done
clear
C)
\[CO_{3}^{2-}\]
done
clear
D)
\[NO_{3}^{-}\]
done
clear
View Answer play_arrow
A metal, M forms chlorides in +2 and +4 oxidation states. Which of the following statement about these chlorides is correct7
A)
\[MC{{l}_{2}}\] is more volatile than \[MC{{l}_{4}}\]
done
clear
B)
\[MC{{l}_{2}}\]is more soluble in anhydrous ethanol than \[MC{{l}_{4}}\]
done
clear
C)
\[MC{{l}_{2}}\] is more ionic than \[MC{{l}_{4}}\]
done
clear
D)
\[MC{{l}_{2}}\] is more easily hydrolyzed than \[MC{{l}_{4}}\]
done
clear
View Answer play_arrow
How many acyclic isomers are possible for \[{{C}_{4}}{{H}_{10}}O?\]
A)
7
done
clear
B)
4
done
clear
C)
5
done
clear
D)
6
done
clear
View Answer play_arrow
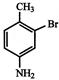
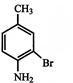
The IUPAC name of following compound:
is
A)
3-ethyl-2-hexene
done
clear
B)
3-propyl-2-hexene
done
clear
C)
3-propyl-3-hexene
done
clear
D)
4-ethyl-4-hexene
done
clear
View Answer play_arrow
The increasing order of stability for the following free radicals is:
A)
\[{{(C{{H}_{3}})}_{2}}\,\overset{\bullet }{\mathop{C}}\,H<{{(C{{H}_{3}})}_{3}}\,\overset{\bullet }{\mathop{C}}\,<{{({{C}_{6}}{{H}_{5}})}_{2}}\overset{\bullet }{\mathop{C}}\,H<{{({{C}_{6}}{{H}_{5}})}_{3}}\overset{\bullet }{\mathop{C}}\,\]
done
clear
B)
\[{{({{C}_{6}}{{H}_{5}})}_{3}}\,\overset{\bullet }{\mathop{C}}\,<{{({{C}_{6}}{{H}_{5}})}_{2}}\,\overset{\bullet }{\mathop{C}}\,H<{{(C{{H}_{3}})}_{3}}\overset{\bullet }{\mathop{C}}\,<{{(C{{H}_{3}})}_{2}}\overset{\bullet }{\mathop{C}}\,H\]
done
clear
C)
\[{{({{C}_{6}}{{H}_{5}})}_{2}}\,\overset{\bullet }{\mathop{CH}}\,<{{({{C}_{6}}{{H}_{5}})}_{3}}\,\overset{\bullet }{\mathop{C}}\,<{{(C{{H}_{3}})}_{3}}\overset{\bullet }{\mathop{C}}\,<{{(C{{H}_{3}})}_{2}}\overset{\bullet }{\mathop{C}}\,H\]
done
clear
D)
\[{{(C{{H}_{3}})}_{2}}\,\overset{\bullet }{\mathop{CH}}\,<{{(C{{H}_{3}})}_{3}}\,\overset{\bullet }{\mathop{C}}\,<{{({{C}_{6}}{{H}_{5}})}_{3}}\overset{\bullet }{\mathop{C}}\,<{{({{C}_{6}}{{H}_{5}})}_{2}}\overset{\bullet }{\mathop{C}}\,H\]
done
clear
View Answer play_arrow
The treatment of benzene with isobutene in the presence of Sulphuric acid gives:
A)
Isobutyl benzene
done
clear
B)
Tert -butyl benzene
done
clear
C)
n-butyl benzene
done
clear
D)
No reaction
done
clear
View Answer play_arrow
Which of the following has the minimum boiling point?
A)
n - butane
done
clear
B)
1-Butyne
done
clear
C)
1-Butene
done
clear
D)
1-Isobutene
done
clear
View Answer play_arrow
The most abundant hydrocarbon pollutant is:
A)
Methane
done
clear
B)
Ethane
done
clear
C)
Propane
done
clear
D)
Butane
done
clear
View Answer play_arrow
The number of atoms in 100g of a fee crystal with density = 10.0g/cm3 and cell edge equal to 200pm is equal to:
A)
\[5\times {{10}^{24}}\]
done
clear
B)
\[5\times {{10}^{25}}\]
done
clear
C)
\[6\times {{10}^{23}}\]
done
clear
D)
\[2\times {{10}^{25}}\]
done
clear
View Answer play_arrow
Due to freckle defect, the density of the ionic solids:
A)
Increases
done
clear
B)
Decreases
done
clear
C)
Does not change
done
clear
D)
Charges
done
clear
View Answer play_arrow
A solution of urea \[(mol.\text{ }mass\text{ }56g\text{ }mo{{l}^{-1}})\] boils at \[100.18{}^\circ \,C\,\,at\]at atmospheric pressure. If \[{{K}_{f}}\] and \[{{K}_{b}}\] for water are 1.86 and 0.512k kg\[mo{{l}^{-1}}\] respectively, the above solution will freeze at:
A)
\[-\,6.54{}^\circ C\]
done
clear
B)
\[-\,0.654{}^\circ C\]
done
clear
C)
\[6.54{}^\circ C\]
done
clear
D)
\[0.654{}^\circ C\]
done
clear
View Answer play_arrow
Which has minimum osmotic pressure?
A)
200ml of 2 M NaCI solution
done
clear
B)
200ml of 1 M glucose solution
done
clear
C)
200ml of 2 M urea solution
done
clear
D)
All have same
done
clear
View Answer play_arrow
The sequence of ionic mobility in the aqueous solution is:
A)
\[{{K}^{+}}>N{{a}^{+}}>R{{b}^{+}}>C{{s}^{+}}\]
done
clear
B)
\[C{{s}^{+}}>R{{b}^{+}}>{{K}^{+}}>N{{a}^{+}}\]
done
clear
C)
\[R{{b}^{+}}>{{K}^{+}}>C{{s}^{+}}>N{{a}^{+}}\]
done
clear
D)
\[N{{a}^{+}}>{{K}^{+}}>R{{b}^{+}}>C{{s}^{+}}\]
done
clear
View Answer play_arrow
For the reaction, \[{{N}_{2}}+3{{H}_{2}}\to 2N{{H}_{3}}\] if \[\frac{\Delta \,[N{{H}_{3}}]}{\Delta \,t}=2\times {{10}^{-4}}\,\,mol\,\,{{L}^{-1}}{{S}^{-1}}\] the value of \[-\frac{\Delta \,[{{H}_{3}}]}{\Delta \,t}\] would be:
A)
\[1\times {{10}^{-4}}\,mol{{L}^{-1}}{{S}^{-1}}\]
done
clear
B)
\[3\times {{10}^{-4}}\,mol{{L}^{-1}}{{S}^{-1}}\]
done
clear
C)
\[4\times {{10}^{-4}}\,mol{{L}^{-1}}{{S}^{-1}}\]
done
clear
D)
\[6\times {{10}^{-4}}\,mol{{L}^{-1}}{{S}^{-1}}\]
done
clear
View Answer play_arrow
For reaction \[\alpha A\to x\,P,\]when\[[A]=2.2\text{ }mM\], the rate was found to be\[2.4\text{ }mM{{s}^{-1}}\]. On reducing the concentration of A to half, the rate changes to\[0.4\text{ }mM{{s}^{-1}}\]. The order of reaction will respect to A is:
A)
1.5
done
clear
B)
2.0
done
clear
C)
2.5
done
clear
D)
3.0
done
clear
View Answer play_arrow
The coagulation of 200 ml of a positive colloid took place when 0.73 g HCl was added to it without changing the volume much. The flocculation value of HCl for the colloid is:
A)
0.365
done
clear
B)
36.5
done
clear
C)
100
done
clear
D)
150
done
clear
View Answer play_arrow
Sulphide ores of metals are usually concentrated by froth flotation process. Which one of the following sulphide ores offers an exception and is concentrated by chemical leaching?
A)
Sphalerite
done
clear
B)
Argentine
done
clear
C)
Galena
done
clear
D)
Copper pyrite
done
clear
View Answer play_arrow
Which of the following halides is most acidic?
A)
\[PC{{l}_{3}}\]
done
clear
B)
\[SbC{{l}_{3}}\]
done
clear
C)
\[BiC{{l}_{3}}\]
done
clear
D)
\[CC{{l}_{4}}\]
done
clear
View Answer play_arrow
Which of the following is the increasing order of enthalpy of vaporization?
A)
\[N{{H}_{3}},\,\,P{{H}_{3}}\text{, }\,As{{H}_{3}}\]
done
clear
B)
\[As{{H}_{3}},\,P{{H}_{3}}\text{, }N{{H}_{3}}\]
done
clear
C)
\[N{{H}_{3}}\text{, }As{{H}_{3}}\text{, }P{{H}_{3}}\]
done
clear
D)
\[P{{H}_{3}},As{{H}_{3}}\text{, }N{{H}_{3}}\]
done
clear
View Answer play_arrow
The "spin-only" magnetic moment \[[in\text{ }units\text{ }of\text{ }Bohr\text{ }magneton,\,({{\mu }_{B}})]\]of \[N{{i}^{2+}}\] in aqueous solution would be: \[(At.\text{ }no.\text{ }Ni=28)\]
A)
0
done
clear
B)
1.73
done
clear
C)
2.84
done
clear
D)
4.90
done
clear
View Answer play_arrow
When \[Mn{{O}_{2}}\] is fused with KOH, a coloured compound is formed. The product and its colour is:
A)
\[{{K}_{2}}Mn{{O}_{4}}\],purple green
done
clear
B)
\[KMn{{O}_{4}}\], purple
done
clear
C)
\[Mn2O3\], brown
done
clear
D)
\[M{{n}_{2}}{{O}_{4}}\], black
done
clear
View Answer play_arrow
Which of the following will given a pair of enantiomers?
A)
\[[P+(N{{H}_{3}})][PtC{{l}_{6}}]\]
done
clear
B)
\[[Co{{(N{{H}_{3}})}_{4}}C{{l}_{2}}]\,N{{O}_{2}}\]
done
clear
C)
\[[Cr\,{{(N{{H}_{3}})}_{4}}[CO{{(CN)}_{6}}J\]
done
clear
D)
\[[CO\,{{(en)}_{2}}C{{l}_{2}}]\,Cl\] \[(en=N{{H}_{2}}C{{H}_{2}}C{{H}_{2}}N{{H}_{3}})\]
done
clear
View Answer play_arrow
HBr reacts with \[C{{H}_{2}}CH-OC{{H}_{3}}\]under anhydrous condition at room temperature of give:
A)
\[C{{H}_{3\,}}CHO\,\,and\,\,C{{H}_{3}}\,Br\]
done
clear
B)
\[Br\,C{{H}_{2}}CHO\,\,and\,\,C{{H}_{3}}OH\]
done
clear
C)
\[Br\,C{{H}_{2}}-C{{H}_{2}}-OC{{H}_{3}}\]
done
clear
D)
\[C{{H}_{3}}-CHBr-OC{{H}_{3}}\]
done
clear
View Answer play_arrow
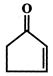
In the following sequence of reactions,
The compound 'D' is:
A)
Propanal
done
clear
B)
Butanal
done
clear
C)
n-butyl alcohol
done
clear
D)
n-propyi alcohol
done
clear
View Answer play_arrow
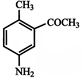
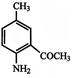
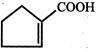
The compound B is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
\[C{{H}_{3}}\,\,C{{H}_{2}}\,COCH\xrightarrow[red\,P]{C{{l}_{2}}}A\xrightarrow{alc\,KOH}B\]
A)
\[C{{H}_{3}}C{{H}_{2}}COCl\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}CHO\]
done
clear
C)
\[C{{H}_{2}}=CHCOOH\]
done
clear
D)
\[CIC{{H}_{2}}C{{H}_{2}}COOH\]
done
clear
View Answer play_arrow
Among the following, weakest base is:
A)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}N{{H}_{2}}\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}NHC{{H}_{3}}\]
done
clear
C)
\[{{O}_{2}}N-C{{H}_{2}}\,N{{H}_{2}}\]
done
clear
D)
\[C{{H}_{3}}NHCHO\]
done
clear
View Answer play_arrow
What is X?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
RNA and DNA are chiral molecules, their chirality is due to:
A)
D-sugar component
done
clear
B)
L-sugar component
done
clear
C)
Chiral bases
done
clear
D)
Chiral phosphate ester unit
done
clear
View Answer play_arrow
Which of the following monomere gives polymer neoprene on polymerization?
A)
\[C{{H}_{2}}=CHCl\]
done
clear
B)
\[C{{H}_{2}}=C(Cl)-CH=C{{H}_{2}}\]
done
clear
C)
\[CC{{l}_{2}}=CC{{l}_{2}}\]
done
clear
D)
\[C{{F}_{2}}=C{{F}_{2}}\]
done
clear
View Answer play_arrow
Which of the following is used as a "morning after pill"?
A)
Norethindrone
done
clear
B)
Ethynylestradiol
done
clear
C)
Mifepristone
done
clear
D)
Bithional
done
clear
View Answer play_arrow
Which of the following is incorrect with respect to museum?
A)
Specimens preserved in preservative solutions in containers or jars
done
clear
B)
Insects preserved in insect boxes after pinning, collecting and killing
done
clear
C)
Stuffed larger animals like birds and mammals
done
clear
D)
Plants and animal specimens preserved as dry specimens
done
clear
View Answer play_arrow
The name virus was given by:
A)
D. J Ivanowsky
done
clear
B)
Pasteur
done
clear
C)
M. W. Beijerinek
done
clear
D)
W.M. Stanley
done
clear
View Answer play_arrow
Deueromycetes also known as imperfect fungi because:
A)
Because only the sexual or vegetative phase of these fungi are known
done
clear
B)
The mycelium is aseptate and coenocytic
done
clear
C)
Because only the asexual or vegetative phase of these fungi are known
done
clear
D)
They occur in aquatic habitats and on decaying wood in moist and damp places or as obligate parasites on plants
done
clear
View Answer play_arrow
Bacteriophages (viruses that infect bacteria) are usually:
A)
ds DNA viruses
done
clear
B)
ss DNA viruses
done
clear
C)
ds RNA viruses
done
clear
D)
ss RNA viruses
done
clear
View Answer play_arrow
The space limited by the inner membrane of the chloroplast is called:
A)
Stroma
done
clear
B)
Stoma
done
clear
C)
Lumen
done
clear
D)
Extra luminal
done
clear
View Answer play_arrow
Which of the following is non-membraneous?
A)
Ribosomes and inclusion bodies
done
clear
B)
Inclusion bodies and plastids
done
clear
C)
Ribosomes and vacuoles
done
clear
D)
Vacuoles and plastids
done
clear
View Answer play_arrow
Select incorrect one:
A)
Golgi apparatus - Camilo Golgi (1898)
done
clear
B)
Nucleus - Robert Brown (1831)
done
clear
C)
Ribosomes - George Palade (1953)
done
clear
D)
Endoplasmic Reticulum (ER) - R. Porter (1950)
done
clear
View Answer play_arrow
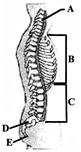
Identify the parts labeled as A to E in given figure of vertebral column and select correct option.
A)
A
B
C
D
E
Thoracic vertebra
Cervical vertebra
Lumber vertebra
Sacrum
Coccyx
done
clear
B)
A
B
C
D
E
Thoracic vertebra
Cervical vertebra
Lumber vertebra
Coccyx
Sacrum
done
clear
C)
A
B
C
D
E
Lumbar vertebra
Thoracic vertebra
Cervical vertebra
Coccyx
Sacrum
done
clear
D)
A
B
C
D
E
Cervical vertebra
Thoracic vertebra
Lumbar vertebra
Sacrum
Coccyx
done
clear
View Answer play_arrow
Which one is not a function of Mesosome:
A)
DNA (Chromosome) replication
done
clear
B)
Distribution of chromosomes to daughter cells.
done
clear
C)
Respiration and secretion processes
done
clear
D)
Decrese the surface area of the plasma membrane and enzymatic content
done
clear
View Answer play_arrow
Non flagellate algae:
A)
Porphyra
done
clear
B)
Ulothrix
done
clear
C)
Volvox
done
clear
D)
Dictyota
done
clear
View Answer play_arrow
Development of the zygotes into young embryos takes place within female gametophytes. This events is a precuruor to the?
A)
Prothallus
done
clear
B)
Seed habit
done
clear
C)
New organism
done
clear
D)
Buds
done
clear
View Answer play_arrow
Gemma cups are:
A)
Unicellular
done
clear
B)
Multicellular
done
clear
C)
Multicellular
done
clear
D)
Yellowish in color
done
clear
View Answer play_arrow
Mono carpellary unilocular with many ovules condition is found in:
A)
Indigofera
done
clear
B)
Tomato
done
clear
C)
Aloe
done
clear
D)
Potato
done
clear
View Answer play_arrow
Cymose type of inflorescence is found in:
A)
Solatium
done
clear
B)
Gloriosa
done
clear
C)
Asparagus
done
clear
D)
Trifolium
done
clear
View Answer play_arrow
One margin of the appendage overlaps tha of the next one and so on present:
A)
Brinjal
done
clear
B)
Lady's finger
done
clear
C)
Cassia
done
clear
D)
Calotropis
done
clear
View Answer play_arrow
Gynoecium occupies the highest position in:
A)
Brinjal
done
clear
B)
Plum
done
clear
C)
Rose
done
clear
D)
Peach
done
clear
View Answer play_arrow
In some leguminous plants the leaf base may become swollen which is called:
A)
Rachis
done
clear
B)
Pulinus
done
clear
C)
Pulvinus
done
clear
D)
Pedicel
done
clear
View Answer play_arrow
The montreal protocol was signed to control the emission of:
A)
Nuclear wastes
done
clear
B)
Non-biodegrable waste
done
clear
C)
Ozone depleting substance
done
clear
D)
Global warming gas
done
clear
View Answer play_arrow
Chipko movement of Garhwal Himalays held on:
A)
1984
done
clear
B)
1974
done
clear
C)
1991
done
clear
D)
1997
done
clear
View Answer play_arrow
In a scrubber, the exhaust is passed through:
A)
Spray of water
done
clear
B)
Lime
done
clear
C)
Both [a] and [b]
done
clear
D)
Calcium carbonate
done
clear
View Answer play_arrow
FOAM is belong to which country:
A)
America
done
clear
B)
Arcata
done
clear
C)
Switzerland
done
clear
D)
Peru
done
clear
View Answer play_arrow
Most abundant protein in the biosphere is:
A)
RuBisco
done
clear
B)
Catalase
done
clear
C)
Collagen
done
clear
D)
Urease
done
clear
View Answer play_arrow
The Cu ions:
A)
Increase the motility and fertilizing capacity of sperms
done
clear
B)
Suppress motility and fertilizing capacity of sperms
done
clear
C)
No role in sperm motility
done
clear
D)
Inhibits the process of coagulation of semen after discharge from penis
done
clear
View Answer play_arrow
Technique is useful for the male partner having inability to inseminate female or low sperm counts:
A)
Intra Uterine Transfer (IUT)
done
clear
B)
Artificial insemination (AI) technique
done
clear
C)
Gemete intra fallopian transfer
done
clear
D)
Surrogacy
done
clear
View Answer play_arrow
Which of the following is incorrect:
A)
In paddy fields, cyanobacteria serve as an important biofertiliser
done
clear
B)
Trichoderma are free livings that are seen in the shoot ecosystems
done
clear
C)
Baculoviruses (especially genus Nucleopolyhedro virus): Attack insects and other arthropods
done
clear
D)
Bacillus thuringiensis (Bt) to control butterfly caterpillar
done
clear
View Answer play_arrow
Pick wrong about statins:
A)
Blood - cholesterol lowering agents
done
clear
B)
Bacteria
done
clear
C)
Inhibits the enzymes responsible for
done
clear
D)
Produces by monascus purpureus
done
clear
View Answer play_arrow
Synaptonemal complex occurs in which stage:
A)
Diakinesis
done
clear
B)
Diplotene
done
clear
C)
Zygotene
done
clear
D)
Leptotene
done
clear
View Answer play_arrow
The nuclear envelope, nucleolus, Golgi complexes and endoplasmic reticulum disappear in:
A)
Early prophase
done
clear
B)
Late prophase
done
clear
C)
Telophase
done
clear
D)
Metaphase
done
clear
View Answer play_arrow
Role in growth of axial hair, pubic hair and facial has during puberty:
A)
Mineral corticoids
done
clear
B)
Androgenic corticoids
done
clear
C)
Glucocorticoids
done
clear
D)
Testosterone
done
clear
View Answer play_arrow
Acts on mammary glands to stimulate formation of alveoli (sacs to store milk) and milk secretion:
A)
Progesterone
done
clear
B)
Oxytocin
done
clear
C)
Prolactin
done
clear
D)
LH
done
clear
View Answer play_arrow
Cretinism is due to:
A)
Hypothyroidism
done
clear
B)
Hyperthyroidism
done
clear
C)
Lack of GH
done
clear
D)
Lack of testosterone in body
done
clear
View Answer play_arrow
Select incorrect statement:
A)
In the resting state a subunit of troponin moska the binding sites for myosin on the actin filaments
done
clear
B)
G-actin is a polymer of monomeric Globular (G) actin
done
clear
C)
A think dark line (M - line) runs through the centre of H - zone
done
clear
D)
Ca binds with a subunits of troponin on actin filaments
done
clear
View Answer play_arrow
Fibrous joints present in:
A)
Knee joint
done
clear
B)
Elbow prints
done
clear
C)
Joints between carpals
done
clear
D)
Between skull bones
done
clear
View Answer play_arrow
Least number of chromosomes are found in:
A)
Apple
done
clear
B)
Onion
done
clear
C)
Drosophilla
done
clear
D)
Rat
done
clear
View Answer play_arrow
Some cells left behind from shoot apical meristem, constitute the:
A)
Axillary bud
done
clear
B)
Primary meristem
done
clear
C)
Intercalary meristems
done
clear
D)
Sclereids
done
clear
View Answer play_arrow
Gymnosperms lacks:
A)
Tracheids
done
clear
B)
Vessels
done
clear
C)
Xylem fibres
done
clear
D)
Xylem parenchyma
done
clear
View Answer play_arrow
Perform cementing to keep neighbouring cell together:
A)
Tight junction
done
clear
B)
Adhering junctions
done
clear
C)
Gap junctions
done
clear
D)
Cemented junctions
done
clear
View Answer play_arrow
Tendoms and ligaments are:
A)
Dense regular connective tissues
done
clear
B)
Dense irregular connective tissues
done
clear
C)
Cartilage
done
clear
D)
Bones
done
clear
View Answer play_arrow
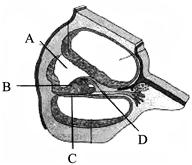
Identify the parts labeled A, B, C and D in the figure and match the correct names from the list (i-viii) given below.
(i) Reissner's membrane
(ii) Basilar membrane
(iii) Tectorial membrane
(iv) Organ of corti
(v) Hair cells
(vi) Otolith organ
(vii) Scala media
(Viii) Scala vestibule
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
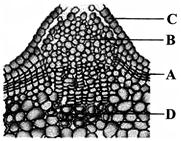
Identify the parts labeled A, B, C, D and select the correct option.
A)
A
B
C
D
Secondary Cortex
Epidermis
Cork Cambium
Comple- mentary cell
done
clear
B)
A
B
C
D
Comple- mentary
Secondary Cortex
Cork Cambium
Epider- mis
done
clear
C)
A
B
C
D
Cork Cam- bium
Comple- mentary cell
Epider- mis
Sec- ondary Cortex
done
clear
D)
A
B
C
D
Comple- tementary Cell
Cork Cambium
Sec- ondary Cortex
Epider- mis
done
clear
View Answer play_arrow
Select the incorrect statement:
A)
During inspiration inter-costal muscles and diaphragm relax causing a decrease in thoracic volume and there by pulmonary volume
done
clear
B)
Part of respiratory tract (from nostrils to terminal bronchi) not involved in gaseous exchange is called dead space
done
clear
C)
Every 100 ml of oxygenated blood can deliver around 5 ml of \[{{O}_{2}}\] to the tissues under normal physiological conditions
done
clear
D)
In asthma difficulty in breathig causing wheezing due to inflammation of bronchi and bronchioles
done
clear
View Answer play_arrow
Each trophic level has a certain mass of living material at a particular time called as the:
A)
Standing tower
done
clear
B)
Standing biomass
done
clear
C)
Standing material
done
clear
D)
Standing crop
done
clear
View Answer play_arrow
The entire sequence of communities that successively change in a given area are called:
A)
Serial
done
clear
B)
Ecosystem
done
clear
C)
Sere
done
clear
D)
Biomass
done
clear
View Answer play_arrow
The cost of climate regulation and habitat for wildlife are about:
A)
8% each
done
clear
B)
7% each
done
clear
C)
6% each
done
clear
D)
10% each
done
clear
View Answer play_arrow
At night and early morning evaporation is:
A)
High
done
clear
B)
Moderate
done
clear
C)
Low
done
clear
D)
Very high
done
clear
View Answer play_arrow
Water potential term was coined by:
A)
Dixon and jolly
done
clear
B)
Hamburger
done
clear
C)
P.S. Chatterjee
done
clear
D)
Slatyer and Taylor
done
clear
View Answer play_arrow
Pici out incorrect one:
A)
Liver lobule is covered by Glisson's capsule
done
clear
B)
Arising from the caecum is a finger-like vestigial organ, the vermiform appendix
done
clear
C)
Fully digested semi fluid and alkaline food is called chyle
done
clear
D)
Ptyalin converts polysaccharide into monosaccharide
done
clear
View Answer play_arrow
Which of the following is absent in pancreatic juice?
A)
Amylase
done
clear
B)
Lipase
done
clear
C)
Sucrose
done
clear
D)
Nuclease
done
clear
View Answer play_arrow
Suppression of FSH secretion is due to:
A)
LH
done
clear
B)
Testosterone
done
clear
C)
Estrogen
done
clear
D)
Oxytocin
done
clear
View Answer play_arrow
Endometrium attains maximum vascularity, thickness and softness during:
A)
Luteal phase
done
clear
B)
Proliferative phase
done
clear
C)
Ovulatory phase
done
clear
D)
Follicular phase
done
clear
View Answer play_arrow
The major organs are well developed during:
A)
End of second month
done
clear
B)
End of 12 weeks
done
clear
C)
End of 24 weeks
done
clear
D)
End of 9 months
done
clear
View Answer play_arrow
Loss of protoplasm to form a treachery element is an example of:
A)
Dedifferentiation
done
clear
B)
Differentiation
done
clear
C)
Redifferentiation
done
clear
D)
Heterophylly
done
clear
View Answer play_arrow
Phytohormone helps in sprouting of potato tubers:
A)
Auxin
done
clear
B)
ABA
done
clear
C)
Gibberellin
done
clear
D)
Ethylene
done
clear
View Answer play_arrow
Select the incorrect statement:
A)
The atria are separated by an inter-atrial septum and the ventricles are separated by inter-venticular septum
done
clear
B)
A bicuspid (mitral) valve guards the opening between left atrium and left ventricle
done
clear
C)
P-wave represents the excitation (depolarization) of atria which causes atrial systole
done
clear
D)
Cardiac arrest is the condition in which heart is not pumping blood enough to meet the needs of the body
done
clear
View Answer play_arrow
Erythroblastosis foetalis can be avoided by:
A)
Administering anti-Rh antibodies to the mother immediately after intercourse
done
clear
B)
Administering ant-Rh antibodies to the mother immediately after the delivery of the first child
done
clear
C)
Administering Rh antibodies to the mother immediately after delivery of first body
done
clear
D)
Administering anti-Rh antibodies to the mother immediately after the delivery of second child
done
clear
View Answer play_arrow
Species affected in Industrial melanism was:
A)
Biston betularia
done
clear
B)
Biston carbonaria
done
clear
C)
Both [a] and [b]
done
clear
D)
Butterfly
done
clear
View Answer play_arrow
Select the wrong one:
A)
Homoerectus: Ate meat
done
clear
B)
Homohabilis - did not eat meat
done
clear
C)
Australo - pithecus: Ate fruits
done
clear
D)
Homo erectus - Brain 1400 cc
done
clear
View Answer play_arrow
An adult human excretes ____ urea per day:
A)
25 - 50 gm
done
clear
B)
25 - 30 gm
done
clear
C)
25 - 30 kg
done
clear
D)
10 - 15 gm
done
clear
View Answer play_arrow
Select the incorrect option:
A)
An increase in fluid volume switches off the osmo receptors and suppresses the ADH release to complete the feedback
done
clear
B)
Blood drained from a convenient artery is pumped into dialyzing unit after adding on anticoagulant like heparin
done
clear
C)
Aldosterone causes reabsorption of Na+ and water from the distal part of the tubule
done
clear
D)
Collecting duct maintain pH and ionic balance of blood by the secretion of Na+ and 1C ions
done
clear
View Answer play_arrow
In the conversion of fructose 6 ? phosphate to fructose 1 - 6 - diphoaphate:
A)
ATP is released
done
clear
B)
ATP is utilized
done
clear
C)
NADH is released
done
clear
D)
NADH is utilized
done
clear
View Answer play_arrow
A peripheral membrane protein complex:
A)
\[{{F}_{1}}\]
done
clear
B)
\[{{F}_{0}}\]
done
clear
C)
\[{{C}_{0}}\]
done
clear
D)
\[{{T}_{0}}\]
done
clear
View Answer play_arrow
Part of a plant that is grown in a test tube under sterile nutrients media:
A)
Ex plant
done
clear
B)
Somaclones
done
clear
C)
Tx plant
done
clear
D)
Nu plant
done
clear
View Answer play_arrow
Select incorrect combination:
A)
Crop
Variety
Resistance to
Cauliflower
Pusa snowball K-l
Curl blight black rot
done
clear
B)
Crop
Variety
Resistance to
Chilli
Pusa Dadaba- har
Leaf curl
done
clear
C)
Crop
Variety
Resistance to
Brassica
Karan mi
White rust
done
clear
D)
Crop
Variety
Resistance to
Cow pea
Pusa shubhra
Leaf& striperust
done
clear
View Answer play_arrow
Finger - like structures which surrounds the mouth of Coelenterates:
A)
Tentacles
done
clear
B)
Cnidocytes
done
clear
C)
Stringing cells
done
clear
D)
Nematocyst
done
clear
View Answer play_arrow
The membrane which covers visceral hump (visceral mags) of molluscan is:
A)
Montle
done
clear
B)
Pallium
done
clear
C)
Ecdysis
done
clear
D)
Both [a] and [b]
done
clear
View Answer play_arrow
A system (present in Echinodermata) in which sea water enters through a porous plate called:
A)
Watreporite
done
clear
B)
Schizoporite
done
clear
C)
Madreporite
done
clear
D)
Matleproite
done
clear
View Answer play_arrow
Which of the following is not a feature of Cnidarians?
A)
Mostly marine
done
clear
B)
Tentacles with cnidoblasts
done
clear
C)
Respiratory system is through trachea
done
clear
D)
Shows altemption of generation
done
clear
View Answer play_arrow
The world summit on sustainable development held in:
A)
Johannesburg, South Africa 2004
done
clear
B)
Johannesburg, South America 2002
done
clear
C)
Johannesburg, South Africa 2002
done
clear
D)
Johannesburg, South America 2004
done
clear
View Answer play_arrow
Which of the following is not a mode of in sites conservation of biodiversity?
A)
National parks
done
clear
B)
Cultural landscape
done
clear
C)
Natural monuments
done
clear
D)
Zoological park
done
clear
View Answer play_arrow
India has more than 50,000 different strains of rice and 1000 varieties of mango come under:
A)
Species diversity
done
clear
B)
Genetic diversity
done
clear
C)
Ecological diversity
done
clear
D)
All of these
done
clear
View Answer play_arrow
Most common type of neurons are:
A)
Unipolar
done
clear
B)
Bipolar
done
clear
C)
Multipolar
done
clear
D)
All are equally found
done
clear
View Answer play_arrow
Select incorrect one:
A)
Pons varoli helps in equilibration
done
clear
B)
In spinal cord outer portion is white matter and inner portion is grey motter
done
clear
C)
Defect in rod cells results in colour blindness
done
clear
D)
Skin is the largest organ
done
clear
View Answer play_arrow
Orchids show diversity of:
A)
Stem patterns
done
clear
B)
Leaf patterns
done
clear
C)
Floral patterns
done
clear
D)
Root patterns
done
clear
View Answer play_arrow
A parasitic plant that is commonly found growing on hedge plants, has lost its chlorophyll and leaves in the course of evolution:
A)
Acacia
done
clear
B)
Bougainvillea
done
clear
C)
Cuscuta
done
clear
D)
Oryza
done
clear
View Answer play_arrow
Tiger census in national parks and tiger reserves based on:
A)
Tagging system
done
clear
B)
Urine mark
done
clear
C)
Counting
done
clear
D)
Pug marks and fecal pellets
done
clear
View Answer play_arrow
Which one is incorrect:
A)
Class-Amphibia, Reptila, Aves, Mammalia etc. come under phylum chordate
done
clear
B)
Order primata, Carnivore etc. is placed incluss mammalia
done
clear
C)
Order polynomials includes family convol-vulaccae and family solonaceae
done
clear
D)
Family solanaceae includes Genus solanum only
done
clear
View Answer play_arrow
Term contagium vivum fluidium was given by:
A)
D.J. Ivanowsky (1892)
done
clear
B)
Pasteur (1898)
done
clear
C)
M.W. Beijerinek (1898)
done
clear
D)
M.W. Beijerinek (1892)
done
clear
View Answer play_arrow
Provides a structural support to prevent the bacteria from bursting or collapsing:
A)
Cell membrane
done
clear
B)
Cytoskeleton
done
clear
C)
Cell wall
done
clear
D)
Tubulin
done
clear
View Answer play_arrow
Which of the following is not use in Gram staining technique?
A)
Iodine
done
clear
B)
Alcohol - acetone solution
done
clear
C)
Crystal violet stains
done
clear
D)
Methylene blue
done
clear
View Answer play_arrow
A scar on the seed coat through which the developing seeds were attached to the fruit:
A)
Hilum
done
clear
B)
Maiden head
done
clear
C)
Plumule
done
clear
D)
Tegmen
done
clear
View Answer play_arrow
Fruit berry or capsule in:
A)
Lilaceae
done
clear
B)
Fabaceae
done
clear
C)
Solanaceae
done
clear
D)
Brassicaceae
done
clear
View Answer play_arrow
The margins of sepals or petals overlap one another but not in any particular direction present in:
A)
China Rose
done
clear
B)
Lady's finger
done
clear
C)
Cotton
done
clear
D)
Gulmohur
done
clear
View Answer play_arrow
Select the incorrect combination
A)
Disease
Causual organism
Symptom
Pneumonia
Streptococcus pneumonia
Lips & finger nails turn grey to bluish
done
clear
B)
Disease
Causual organism
Symptom
Filariasis
W. bancrofti & W. Malayi
Appearance of dry, scaly lesions on varies body parts
done
clear
C)
Disease
Causual organism
Symptom
Common cold
Rhinoviruses
Infect nose & respiratory passage
done
clear
D)
Disease
Causual organism
Symptom
Amoebians
Entamoeba histolytica
Stool with excess mucous & blood clots
done
clear
View Answer play_arrow
Which of the following is an example of pleiotropic:
A)
Sickle cell anaemia
done
clear
B)
Starch synthesis in pea plant
done
clear
C)
Down syndrome
done
clear
D)
Both [a] and [b]
done
clear
View Answer play_arrow
Identify the correct sequence in the given diagram of water absorption by root.
A)
A
B
C
D
Epidermis
Hypodermis
Cortex
Pericycle
done
clear
B)
A
B
C
D
Epidermis
Hypodermis
Cortex
Pith
done
clear
C)
A
B
C
D
Epidermis
Cortex
Endodermis
Xylem
done
clear
D)
A
B
C
D
Epidermis
Cortex
Endodermis
Pericycle
done
clear
View Answer play_arrow
Which of the following is not false fruit?
A)
Apple
done
clear
B)
Strawberry
done
clear
C)
Banana
done
clear
D)
Cashew
done
clear
View Answer play_arrow
Tree-dwelling animals are known as:
A)
Dweloreal
done
clear
B)
Troreal
done
clear
C)
Choreal
done
clear
D)
Arboreal
done
clear
View Answer play_arrow
Which of the following is/are techniques for early diagnosis?
A)
Recombinant DNA technology
done
clear
B)
PCR
done
clear
C)
ELISA
done
clear
D)
All of these
done
clear
View Answer play_arrow
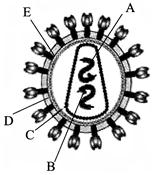
Identify A, B, C, D and E in the below diagram of HIV virus:
A)
A-RNA, B-Reverse transcriptase, C-Capsule protein coat, D-Lipid membrane, E-Envelope protein coat
done
clear
B)
A-RNA, B-Reverse transcriptase, C-Lipid membrane, D-Envelope protein coat, E-Capsule protein coat
done
clear
C)
A-Reverse transcriptase, B-Lipid membrane, C-RNA, D-Capsule protein coat, E-Envelope protein coat
done
clear
D)
A-RNA, B-Reverse transcriptase, C-Envelope protein coat, D-Lipid membrane, E-Capsule protein coat.
done
clear
View Answer play_arrow
Select the incorrect one:
A)
The most commonly used bioreactors are of stirring type
done
clear
B)
The downstream processing and quality control testing same from product to product
done
clear
C)
If a protein encoding gene is expressed in a heterologous host, it is called a recombinant protein
done
clear
D)
Retroviruses in animals can transform normal cells into cancerous cells
done
clear
View Answer play_arrow
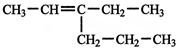 is
is 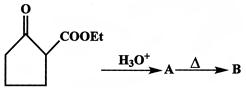 The compound B is:
The compound B is: 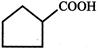
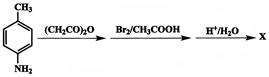 What is X?
What is X?