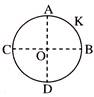
A thin conducting ring of radius R is given a charge +Q. The electric field at the centre O of the ring due to the charge on the part AKB of the ring is E. The electric field at the centre due to the charge on the part ACDB of the ring is-
A)
E along KO
done
clear
B)
3 E along OK
done
clear
C)
3 E along KO
done
clear
D)
E along OK
done
clear
View Answer play_arrow
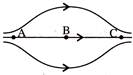
The figure shows some of the electric field lines corresponding to an electric field. The figure suggests-
A)
\[{{E}_{A}}>{{E}_{B}}>{{E}_{C}}\]
done
clear
B)
\[{{E}_{A}}={{E}_{B}}={{E}_{C}}\]
done
clear
C)
\[{{E}_{A}}={{E}_{C}}>{{E}_{B}}\]
done
clear
D)
\[{{E}_{A}}={{E}_{C}}<{{E}_{B}}\]
done
clear
View Answer play_arrow
A charged particle of mass m and charge q is released from rest in a uniform electric field E. Neglecting the effect of gravity, the kinetic energy of the charged particle after 't' second is-
A)
\[\frac{E{{q}^{2}}m}{2{{t}^{2}}}\]
done
clear
B)
\[\frac{2{{E}^{2}}{{t}^{2}}}{mq}\]
done
clear
C)
\[\frac{{{E}^{2}}{{q}^{2}}{{t}^{2}}}{2m}\]
done
clear
D)
\[\frac{Eqm}{t}\]
done
clear
View Answer play_arrow
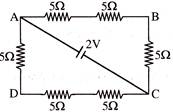
The potential difference between points A and B of adjoining figure is-
A)
\[\frac{2}{3}\,V\]
done
clear
B)
\[\frac{8}{9}\,V\]
done
clear
C)
\[\frac{4}{3}\,V\]
done
clear
D)
\[2\,V\]
done
clear
View Answer play_arrow
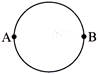
A wire of resistance 12 ohms per meter is bent to form a complete circle of radius 10 cm. The resistance between its two diametrically opposite points A and B as shown in the figure, is
A)
\[0.6\,\pi \,\Omega \]
done
clear
B)
\[3\,\Omega \]
done
clear
C)
\[6\,\pi \,\Omega \]
done
clear
D)
\[6\,\Omega \]
done
clear
View Answer play_arrow
A particle of mass m, charge Q and kinetic energy T enters a transverse uniform magnetic field of induction B. After 3 seconds the kinetic energy of the particle will be-
A)
T
done
clear
B)
4 T
done
clear
C)
3 T
done
clear
D)
2 T
done
clear
View Answer play_arrow
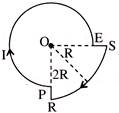
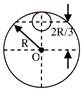
A current I flowing through the loop as shown in figure. The magnetic field at centre O is-
A)
\[\frac{7{{\mu }_{0}}I}{16R}\otimes \]
done
clear
B)
\[\frac{7{{\mu }_{0}}I}{16R}\odot \]
done
clear
C)
\[\frac{5{{\mu }_{0}}I}{16R}\otimes \]
done
clear
D)
\[\frac{5{{\mu }_{0}}I}{16R}\odot \]
done
clear
View Answer play_arrow
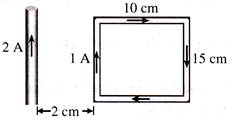
What is the net force on the rectangular coil-
A)
\[25\times {{10}^{-}}^{7}N\text{ }moving\text{ }towards\text{ }wire\]
done
clear
B)
\[25\times {{10}^{-}}^{7}N\text{ }moving\text{ }away\text{ }from\text{ }wire\]
done
clear
C)
\[35\times {{10}^{-}}^{7}N\text{ }moving\text{ }towards\text{ }wire\]
done
clear
D)
\[35\times \,{{10}^{-}}^{7}N\text{ }moving\text{ }away\text{ }from\text{ }wire\]
done
clear
View Answer play_arrow
A bar magnet of length 10 cm and having pole strength equal to \[{{10}^{-3}}\] A x m is kept in a magnetic field of \[4\pi \,\,\times \,\,{{10}^{-}}^{3}\] Tesla. It makes an angle \[30{}^\circ \] with the direction of the field. The torque acting on the magnet is \[\left( newton\text{ }\times \text{ }metre \right)\]-
A)
\[2\pi \,\,\times \,\,{{10}^{-}}^{7}\]
done
clear
B)
\[2\pi \,\,\times \,\,{{10}^{-}}^{5}\]
done
clear
C)
\[0.5\]
done
clear
D)
\[\,50\]
done
clear
View Answer play_arrow
Power dissipated in an LCR series circuit connected to an a.c. source of emf E is-
A)
\[{{E}^{2}}R/\left[ {{R}^{2}}+{{\left( L\omega -\frac{1}{C\omega } \right)}^{2}} \right]\]
done
clear
B)
\[\frac{{{E}^{2}}\sqrt{{{R}^{2}}+{{\left( L\omega -\frac{1}{C\omega } \right)}^{2}}}}{R}\]
done
clear
C)
\[\frac{{{E}^{2}}\left[ {{R}^{2}}+{{\left( L\omega -\frac{1}{C\omega } \right)}^{2}} \right]}{R}\]
done
clear
D)
\[\frac{{{E}^{2}}R}{\sqrt{{{R}^{2}}+{{\left( L\omega -\frac{1}{C\omega } \right)}^{2}}}}\]
done
clear
View Answer play_arrow
In a coil rate of change of area is \[5\text{ }{{m}^{2}}/\text{ }millisecond\] and current becomes 1 amp from 2 amp in \[2\,\,\times \,\,{{10}^{-}}^{3}\] sec. If magnitude of field is 1 tesla then self-inductance of the coil is
A)
2 H
done
clear
B)
5 H
done
clear
C)
20 H
done
clear
D)
10 H
done
clear
View Answer play_arrow
The magnetic susceptibility of a paramagnetic substance at \[-73\text{ }{}^\circ C\] is 0.0060, then its value at \[-173\,{}^\circ C\] will be-
A)
0.0030
done
clear
B)
0.0120
done
clear
C)
0.0180
done
clear
D)
0.0045
done
clear
View Answer play_arrow
Percentage error in measuring the radius and mass of hollow sphere are \[2%\] and \[4%\] respectively. Then error in measurement of moment of inertia with respect to its diameter is-
A)
\[10%\]
done
clear
B)
\[6%\]
done
clear
C)
\[8%\]
done
clear
D)
\[4%\]
done
clear
View Answer play_arrow
Consider slabs of three media A, B and C arranged as shown in figure. R.I of A is 1.5 and that of C is 1.4. If the number of waves in A is equal to the number of waves in the combination B and C then refractive index of B is-
A)
1.4
done
clear
B)
1.5
done
clear
C)
1.6
done
clear
D)
1.7
done
clear
View Answer play_arrow
If the critical angle for total internal reflection from a medium to vacuum is \[30{}^\circ \], the velocity of light in the medium is-
A)
\[3\,\,\times \,\,{{10}^{8}}m/s~\]
done
clear
B)
\[1.5\times {{10}^{8}}m/s\]
done
clear
C)
\[6\,\,\times \,\,{{10}^{8}}m/s\]
done
clear
D)
\[\sqrt{3}\times {{10}^{8}}m/s\]
done
clear
View Answer play_arrow
The refractive index of water is 4/3 and that of glass is 5/3. Then the critical angle for a ray of light entering water from glass will be-
A)
\[si{{n}^{-}}^{1}\left( 4/5 \right)\]
done
clear
B)
\[{{\sin }^{-}}^{1}\,\,\left( 5/4 \right)\]
done
clear
C)
\[si{{n}^{-}}^{1}\left( 20/9 \right)~\]
done
clear
D)
\[si{{n}^{-1}}\,\,\left( 9/20 \right)\]
done
clear
View Answer play_arrow
A long horizontal slit is placed 1 mm above a horizontal plane mirror. The interference between the light coming directly from the slit and that after reflection is seen on a screen 1m away from the slit. If the mirror reflects only \[64%\] of the light falling on it, the ratio of the maximum to the minimum intensity in the interference pattern observed on the screen is-
A)
\[8\text{ }:\text{ }1\]
done
clear
B)
\[3\text{ }:\text{ }1\]
done
clear
C)
\[81\text{ }:\text{ }1\]
done
clear
D)
\[9\text{ }:\text{ }1\]
done
clear
View Answer play_arrow
Two wavelengths of light \[{{\lambda }_{1}}\,\,and\,\,{{\lambda }_{2}}\] are sent through Young's double slit apparatus simultaneously. What must be true about \[{{\lambda }_{1}}\,\,and\,\,{{\lambda }_{2}}\] if the third order bright fringe of \[{{\lambda }_{1}}\] coincides with fifth order dark fringe of\[{{\lambda }_{2}}\]?
A)
\[3{{\lambda }_{1}}=2{{\lambda }_{2}}\]
done
clear
B)
\[2{{\lambda }_{1}}=3{{\lambda }_{2}}\]
done
clear
C)
\[3{{\lambda }_{1}}=5{{\lambda }_{2}}\]
done
clear
D)
\[5{{\lambda }_{1}}=3{{\lambda }_{2}}\]
done
clear
View Answer play_arrow
Two polaroids have their axes at an angle of \[60{}^\circ \] in between. If Io is intensity of unpolarised light falling on first Polaroid then that of emerging light from the second Polaroid is-
A)
\[0.125\text{ }{{I}_{0}}\]
done
clear
B)
\[0.25\,\,{{I}_{0}}\]
done
clear
C)
\[0.375\text{ }{{I}_{0}}\]
done
clear
D)
\[0.50\text{ }{{I}_{0}}\]
done
clear
View Answer play_arrow
Find the de-Broglie wavelength of Earth. Mass of Earth is\[6\times {{10}^{24}}\,kg\]. Mean orbital radius of Earth around Sun is\[150\,\,\times \,\,{{10}^{6}}\,km\].
A)
\[3.7\,m\]
done
clear
B)
\[3.7\,\,\times \,\,{{10}^{-63}}m\]
done
clear
C)
\[3.7\times {{10}^{63}}\,m\]
done
clear
D)
\[3.7\times {{10}^{-63}}cm\]
done
clear
View Answer play_arrow
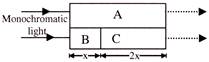
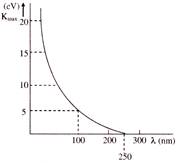
In a photoelectric effect experiment, the maximum kinetic energy of the ejected photoelectrons is measured for various wavelengths of the incident light. Figure shows a graph of this maximum kinetic energy \[{{K}_{max}}\] as a function of the wavelength \[\lambda \] of the light falling on the surface of the metal. Which of the following statement is/are correct?
[a] Threshold frequency for the metal is \[1.2\,\,\times \,\,{{10}^{15}}Hz\] [b] Work function of the metal is \[4.968\text{ }eV\] [c] Maximum kinetic energy of photoelectrons corresponding to light of wavelength 100 nm is nearly \[7.4\text{ }eV\] [d] Photoelectric effect takes place with red light
A)
a, b
done
clear
B)
a, b, c
done
clear
C)
a, b, c, d
done
clear
D)
a, c, d
done
clear
View Answer play_arrow
In the beta decay: \[_{Z}^{A}X\,\,\to \,\,_{Z+1}^{A}Y\,\,+\,\,{{e}^{-}}\]
(i) total energy is conserved (ii) mass number is conserved (iii) charge is conserved (iv) spin angular momentum is conserved
A)
a, b
done
clear
B)
b , c
done
clear
C)
a , c
done
clear
D)
c , d
done
clear
View Answer play_arrow
A radioactive element decays by\[\beta -emission\]. A detector records n beta particles in 2s and in next \[2s\] it records \[0.75n\] beta particle. Find mean life correct to nearest whole number-(Given \[\ell n\,2=0.6931,\text{ }\ell n\text{ }3\,\,=\,\,0986\])
A)
\[17s~\]
done
clear
B)
\[7\,s\]
done
clear
C)
\[5\text{ }s\]
done
clear
D)
\[15\text{ }s\]
done
clear
View Answer play_arrow
The half-life of a radioactive substance is 20 minutes. The approximate time interval \[\left( {{t}_{2}}-{{t}_{1}} \right)\] between the time \[{{t}_{2}}\] when \[\frac{2}{3}\] of it has decayed and time \[{{t}_{1}}\] when \[\frac{1}{3}\] of it had decayed is-
A)
7 min
done
clear
B)
14 min
done
clear
C)
20 min
done
clear
D)
28 min
done
clear
View Answer play_arrow
A thin disc of mass 9M and radius R from which a disc of radius \[R/3\] is cut, as shown in figure. Then moment of inertia of the remaining disc about 0, perpendicular to the plane of disc is-
A)
\[4\text{ }M{{R}^{2}}\]
done
clear
B)
\[9\text{ }M{{R}^{2}}\]
done
clear
C)
\[\frac{37}{9}\,M{{R}^{2}}\]
done
clear
D)
\[\frac{40}{9}M{{R}^{2}}\]
done
clear
View Answer play_arrow
A body moves is a circular path of radius \[r\,\,=\,\,500\,\,\,m\] with tangential acceleration\[{{a}_{t}}=2\text{ }m{{s}^{-}}^{2}\]. When its tangential linear velocity is \[30\text{ }m/s\], the total acceleration will be:
A)
\[5.4\text{ }m{{s}^{-}}^{2}\]
done
clear
B)
\[3.9\text{ }m{{s}^{-}}^{2}\]
done
clear
C)
\[2.7\,\,m{{s}^{-2}}\]
done
clear
D)
\[2.1\,\,m{{s}^{-}}^{2}\]
done
clear
View Answer play_arrow
If three particles each of mass M are placed at the comers of an equilateral triangle of side a, the potential energy of the system and the work done if the side of the triangle is changed from a to \[2a\], are:
A)
\[\frac{3GM}{{{a}^{2}}}\,,\,\,\frac{3GM}{2a}\]
done
clear
B)
\[-\frac{3G{{M}^{2}}}{a}\,,\,\,\frac{3G{{M}^{2}}}{2a}\]
done
clear
C)
\[-\frac{3G{{M}^{2}}}{{{a}^{2}}}\,,\,\,\frac{3G{{M}^{2}}}{4{{a}^{2}}}\]
done
clear
D)
\[-\frac{3G{{M}^{2}}}{a}\,,\,\,\frac{3GM}{2a}\]
done
clear
View Answer play_arrow
A simple pendulum with a solid metal bob has a period T. The metal bob is now immersed in a liquid having density one-tenth that of the metal of the bob. The liquid is non-viscous. Now the period of the same pendulum with its bob remaining all the time in the liquid will be
A)
\[\frac{9}{10}\,T\]
done
clear
B)
\[T\sqrt{\frac{10}{9}}\]
done
clear
C)
unchanged
done
clear
D)
\[T\sqrt{\frac{9}{10}}\]
done
clear
View Answer play_arrow
A sphere and a cube of same material and same volume are heated upto the same temperature and allowed to cool in the same surroundings. The ratio of the amounts of radiations emitted will be-
A)
\[1\,\,:\,\,1\]
done
clear
B)
\[\frac{4\pi }{3}\,\,:\,\,1\]
done
clear
C)
\[{{\left( \frac{\pi }{6} \right)}^{1/3}}\,:\,\,1\,\]
done
clear
D)
\[\frac{1}{2}{{\left( \frac{4\pi }{3} \right)}^{2/3}}\,:\,\,1\,\]
done
clear
View Answer play_arrow
Water is flowing through a cylindrical pipe of cross-section area \[0.0971\text{ }{{m}^{2}}\] at a speed of\[1.0\text{ }m/s\]. If the diameter of the pipe is halved, then find the speed of flow of water through it-
A)
\[4.0\text{ }m/s\]
done
clear
B)
\[6.0\text{ }m/{{s}^{2}}\]
done
clear
C)
\[4.0\text{ }m/{{s}^{2}}\]
done
clear
D)
\[6.0\text{ }m/s\]
done
clear
View Answer play_arrow
Pure silicon at 300 K has equal electron ne and hole \[({{n}_{h}})\] concentration of \[1.5\,\,\times \,\,{{10}^{16}}\,{{m}^{-}}^{3}\]. Doping by indium increases \[{{n}_{h}}\] to \[4.5\,\,\times \,\,{{10}^{22}}\,{{m}^{-}}^{3}\]. The \[{{n}_{e}}\] in the doped silicon is (in per \[{{m}^{3}}\])-
A)
\[9\,\,\times \,\,{{10}^{5}}\]
done
clear
B)
\[5\times {{10}^{9}}\]
done
clear
C)
\[2.25\times {{10}^{11}}\]
done
clear
D)
\[3\times {{10}^{19}}\]
done
clear
View Answer play_arrow
The frequency of a vibrating wire is\[f\]. If tension is doubled, density is halved and diameter is doubled, then the new frequency will be-
A)
\[f\]
done
clear
B)
\[f\sqrt{2}\]
done
clear
C)
\[2f\]
done
clear
D)
\[4f\]
done
clear
View Answer play_arrow
When the listener moves towards a stationary source with a velocity\[{{v}_{1}}\], the apparent frequency of a note emitted by the source is\[f'\]. When the listener moves away from the source with the same velocity, the apparent frequency of the note\[f'/f''\,\,=\,\,3\]. If v the velocity of sound in air, the value of \[v/{{v}_{1}}\] is-
A)
3
done
clear
B)
1 / 2
done
clear
C)
2
done
clear
D)
1
done
clear
View Answer play_arrow
A metallic ball and highly stretched spring are made of the same material and have the same mass. They are heated so that they melt, the latent heat required-
A)
are the same for both
done
clear
B)
Is greater for the ball
done
clear
C)
Is greater for the spring
done
clear
D)
For the two may or may not be the same depending upon the metal
done
clear
View Answer play_arrow
N molecules each of mass m of gas A and 2N molecules each of mass 2m of gas B are contained in the same vessel at temperature T. The mean square of the velocity of molecules of gas B is \[{{V}^{2}}\] and the mean square of x- component of the velocity of molecules of gas A is\[{{w}^{2}}\]. The ratio \[{{w}^{2}}/{{V}^{2}}\,\] is-
A)
1
done
clear
B)
2
done
clear
C)
\[\frac{3}{2}\]
done
clear
D)
\[\frac{2}{3}\]
done
clear
View Answer play_arrow
Two circular discs A and B with equal radii are blackened. They are heated to same temperature and are cooled under identical conditions. What inference do you draw from their cooling curves?
A)
A and B have same specific heats
done
clear
B)
specific heat of A is less
done
clear
C)
specific heat of B is less
done
clear
D)
nothing can be said
done
clear
View Answer play_arrow
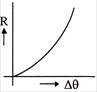
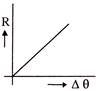
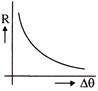
The rate of cooling R with excess of temperature \[\Delta \theta \] varies according to the graph-
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
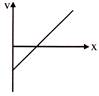
\[\left( v-x \right)\] curve is shown for a particle moving in a straight line. The acceleration of the particle will-
A)
increases with x parabolically
done
clear
B)
remain constant
done
clear
C)
increase linearly with x
done
clear
D)
none of the above
done
clear
View Answer play_arrow
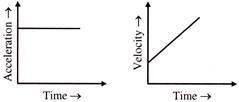
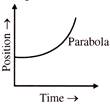
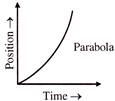
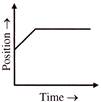
The velocity-time and acceleration-time graphs of a particle are given as
Its position-time graph may be given as
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The equation of projectile is \[y=16x-\frac{5{{x}^{2}}}{4}\] The horizontal range is-
A)
16 m
done
clear
B)
8 m
done
clear
C)
3.2 m
done
clear
D)
12.8 m
done
clear
View Answer play_arrow
A block of mass 20 kg is suspended through two light spring balances as shown in figure, then-
A)
Both the scales show 10 kg reading.
done
clear
B)
Both the scales show 20 kg reading.
done
clear
C)
The upper scale will read 20 kg while the lower zero.
done
clear
D)
Their readings are in between 0 and 20 kg with their sum equal to 20 kg.
done
clear
View Answer play_arrow
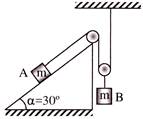
In an arrangement shown in the figure, the acceleration of block A and B are given-
A)
g/3, g/6
done
clear
B)
g/6, g/3
done
clear
C)
g/2, g/2
done
clear
D)
0, 0
done
clear
View Answer play_arrow
Power applied to a particle varies with time as \[P=\left[ 3{{t}^{2}}-2t+1 \right]\] watts. Where t is time in seconds. Then the change in kinetic energy of particle between time \[t=2s\text{ }to\text{ }t=4s\] is-
A)
46 J
done
clear
B)
52 J
done
clear
C)
92 J
done
clear
D)
104 J
done
clear
View Answer play_arrow
A long spring, when stretched by a distance x, has the potential energy U. On increasing the stretching to nx, the potential energy of the spring will be-
A)
\[U/n\]
done
clear
B)
\[nU\]
done
clear
C)
\[{{n}^{2}}U\]
done
clear
D)
\[U/{{n}^{2}}\]
done
clear
View Answer play_arrow
A body is moved along a straight line by a machine delivering constant power. The distance moved by the body in time t is proportional to-
A)
\[{{t}^{1/2}}\]
done
clear
B)
\[{{t}^{3/4}}\]
done
clear
C)
\[{{t}^{3/2}}\]
done
clear
D)
\[{{t}^{2}}\]
done
clear
View Answer play_arrow
The correct order of bond angle is-
A)
\[O{{F}_{2}}<{{H}_{2}}O<C{{l}_{2}}O<Cl{{O}_{2}}\]
done
clear
B)
\[Cl{{O}_{2}}<O{{F}_{2}}<C{{l}_{2}}O<{{H}_{2}}O\]
done
clear
C)
\[Cl{{O}_{2}}<C{{l}_{2}}O<{{H}_{2}}O<O{{F}_{2}}\]
done
clear
D)
\[O{{F}_{2}}<C{{l}_{2}}O<{{H}_{2}}O<Cl{{O}_{2}}\]
done
clear
View Answer play_arrow
Shape and hybridisation of \[I{{F}^{5}}\] respectively are-
A)
\[Sea-saw,\text{ }s{{p}^{3}}d\]
done
clear
B)
\[Trigonal\,\,bipyramidal,s{{p}^{3}}d\]
done
clear
C)
\[Square\,\,pyramidal\,\,s{{p}^{3}}{{d}^{2}}\]
done
clear
D)
\[Pentagonal\,\,pyramidal,s{{p}^{3}}{{d}^{3}}\]
done
clear
View Answer play_arrow
Which of the following does not contain any dative bond-
A)
\[NaB{{F}_{4}}\]
done
clear
B)
\[K{{I}_{3}}\]
done
clear
C)
\[KH{{F}_{2}}\]
done
clear
D)
\[P{{H}_{4}}I\]
done
clear
View Answer play_arrow
The electron affinity of N, O, S and Cl are-
A)
\[\text{O}=Cl<N=S\]
done
clear
B)
\[O<S<Cl<N\]
done
clear
C)
\[N<O<S<Cl\]
done
clear
D)
\[O<N<Cl<S\]
done
clear
View Answer play_arrow
Three elements X, Y, Z are present in the third short period and their oxides are ionic, amphoteric and giant molecules respectively. The correct order of the atomic number of X, Y and Z in the order-
A)
\[Z<Y<X\]
done
clear
B)
\[Y<Z<X\]
done
clear
C)
\[X<Z<Y~\]
done
clear
D)
\[X<Y<Z\]
done
clear
View Answer play_arrow
\[C{{l}_{2}}\] reacts with dilute NaOH and concentrated NaOH to respectively produce?
A)
\[NaCl{{O}_{3}}\,\,\And \,\,NaClO\]
done
clear
B)
\[NaClO\text{ }\And \text{ }NaCl{{O}_{2}}\]
done
clear
C)
\[NaCl\text{ }\And \text{ }NaCl{{O}_{4}}\]
done
clear
D)
\[NaClO\text{ }\And \text{ }NaCl{{O}_{3}}\]
done
clear
View Answer play_arrow
Sulphur react with chlorine in \[1\text{ }:\text{ }2\] ratio and forms (X). (X) on hydrolysis gives a sulphur compound (Y). What is the hydrised state of central atom in the anion of (Y).
A)
\[s{{p}^{3}}d\]
done
clear
B)
\[s{{p}^{2}}\,\]
done
clear
C)
\[s{{p}^{3}}\]
done
clear
D)
\[sp\]
done
clear
View Answer play_arrow
Which of the following set of raw materials are used in the manufacturing of \[N{{a}_{2}}C{{O}_{3}}\] by Solvay process?
A)
\[CaC{{l}_{2}},\,\,N{{H}_{3}},\,\,C{{O}_{2}}\]
done
clear
B)
\[NaOH,\text{ }N{{H}_{3}},\,\,C{{O}_{2}}\]
done
clear
C)
\[NaCl,\,\,N{{H}_{3}},\,\,C{{O}_{2}}\]
done
clear
D)
\[Ca{{\left( OH \right)}_{2}},\,\,N{{H}_{3}},\,\,C{{O}_{2}}\]
done
clear
View Answer play_arrow
Which of the following transition metal oxides is (normally) neither acidic nor amphotonic?
A)
\[Cr{{O}_{3}}\]
done
clear
B)
\[F{{e}_{2}}{{O}_{3}}\]
done
clear
C)
\[M{{n}_{2}}{{O}_{7}}\]
done
clear
D)
\[CuO\]
done
clear
View Answer play_arrow
The possible number of isomers for complex \[\left[ MC{{l}_{2}}B{{r}_{2}} \right]S{{O}_{4}}\] will be-
A)
5
done
clear
B)
4
done
clear
C)
3
done
clear
D)
2
done
clear
View Answer play_arrow
The co-ordination number of cobalt in \[\left[ Co{{\left( en \right)}_{2}}B{{r}_{2}} \right]C{{l}_{2}}\] is
A)
2
done
clear
B)
4
done
clear
C)
8
done
clear
D)
6
done
clear
View Answer play_arrow
Zn gives \[{{H}_{2}}\] gas with \[{{H}_{2}}S{{O}_{4}}\] and HCl but not with \[HN{{O}_{3}}\] because-
A)
in electrochemical series Zn is above hydrogen
done
clear
B)
\[NO_{3}^{\Theta }\] ion is reduced in preference to hydronium ion
done
clear
C)
Zn act as an oxidising agent when react with \[HN{{O}_{3}}\]
done
clear
D)
\[HN{{O}_{3}}\] is weaker acid than \[{{H}_{2}}S{{O}_{4}}\] and \[HCl\]
done
clear
View Answer play_arrow
Which of the following reaction not carried out in Blast Furnace-
A)
\[F{{e}_{2}}{{O}_{3}}+CO\to Fe+C{{O}_{2}}\]
done
clear
B)
\[CaO+Si{{O}_{2}}\to CaSi{{O}_{3}}\]
done
clear
C)
\[FeO+Si{{O}_{2}}\to FeSi{{O}_{3}}\]
done
clear
D)
\[FeO+C\to Fe+C{{O}_{2}}\]
done
clear
View Answer play_arrow
\[Hg{{\left( N{{O}_{3}} \right)}_{2}}\] on heating gives-
A)
\[HgO,\text{ }N{{O}_{2}},\text{ }{{O}_{2}}\]
done
clear
B)
\[HgO,\text{ }{{N}_{2}}O,\text{ }{{O}_{2}}\]
done
clear
C)
\[Hg,\text{ }{{N}_{2}}O,{{O}_{2}}\]
done
clear
D)
\[Hg,\,N{{O}_{2}},{{O}_{2}}\]
done
clear
View Answer play_arrow
10 gms each of \[C{{O}_{2}},\text{ }N{{H}_{3}}\text{ }and\text{ }{{O}_{2}}\] were taken in three separate flasks. What is the correct decreasing order of atoms?
A)
\[C{{O}_{2}},\text{ }N{{H}_{3}},\text{ }{{O}_{2}}\]
done
clear
B)
\[N{{H}_{3}},\text{ }{{O}_{2}},\text{ }C{{O}_{2}}\]
done
clear
C)
\[{{O}_{2}},\text{ }N{{H}_{3}},\text{ }C{{O}_{2}}\]
done
clear
D)
\[N{{H}_{3}},\text{ }C{{O}_{2}},\text{ }{{O}_{2}}\]
done
clear
View Answer play_arrow
To dissolve 3.3g of certain metal 110mL of \[1N{{H}_{2}}S{{O}_{4}}\]. Find the equivalent mass of metal-
A)
15 g
done
clear
B)
30 g
done
clear
C)
20 g
done
clear
D)
10 g
done
clear
View Answer play_arrow
How many electrons in calcium have\[l=0\]?
A)
6
done
clear
B)
8
done
clear
C)
10
done
clear
D)
12
done
clear
View Answer play_arrow
For the reaction \[{{C}_{3}}{{H}_{8}}(g)\,\,+\,\,5{{O}_{2}}(g)\,\,\to 3C{{O}_{2}}(g)\,\,+\,\,4{{H}_{\text{2}}}\text{O(}\ell \text{)}\] at constant temperature, \[\Delta H\,\,-\,\,\Delta U\] is-
A)
\[-RT\]
done
clear
B)
\[RT\]
done
clear
C)
\[-3RT\]
done
clear
D)
\[+3RT\]
done
clear
View Answer play_arrow
How many moles of HCl must be removed from 1 litre of aqueous HCl solution to change its pH from 2 to 3-
A)
1
done
clear
B)
0.02
done
clear
C)
0.009
done
clear
D)
0.01
done
clear
View Answer play_arrow
2 mole of an ideal gas at \[27{}^\circ \,C\] expands isothermally & reversibly from a volume of 4 litre to 40 litre, the work done by the gas is-
A)
\[W=-\,28.72\text{ }kJ\]
done
clear
B)
\[W=-11.488\,kJ\]
done
clear
C)
\[W=-\,5.736\,kJ\]
done
clear
D)
\[W=4.988\,kJ\]
done
clear
View Answer play_arrow
The reduction potential of hydrogen half cell will be most negative if:
A)
\[{{P}_{{{H}_{2}}}}\,=\,\,1\,\,atm,\,[{{H}^{+}}]\,\,=\,\,1\,\,M\]
done
clear
B)
\[{{P}_{{{H}_{2}}}}\,=\,\,1\,\,atm,\,[{{H}^{+}}]\,\,=\,\,2\,\,M\]
done
clear
C)
\[{{P}_{{{H}_{2}}}}\,=\,\,2\,\,atm,\,[{{H}^{+}}]\,\,=\,\,1\,\,M\]
done
clear
D)
\[{{P}_{{{H}_{2}}}}\,=\,\,2\,\,atm,\,[{{H}^{+}}]\,\,=\,\,0.1\,\,M\]
done
clear
View Answer play_arrow
For the cell \[T1|T{{1}^{+}}(0.001\,\,M)||C{{u}^{2+}}(0.1\,\,M)|Cu\]. \[{{E}_{cell}}\,\,at\text{ }25{}^\circ C\text{ }is\text{ }0.83\text{ }V\] which can be increased-
[a] \[by\,\,increasing\,\,C{{u}^{2+}}\] [b] \[by\,\,increasing\,\,[T{{1}^{+}}]\] [c] \[by\,\,decreasing\,\,[C{{u}^{2+}}]\] [d] \[by\,\,decreasing\,\,[T{{1}^{\text{+}}}]\]
A)
a, c
done
clear
B)
a, d
done
clear
C)
a, b, d
done
clear
D)
a, c, d
done
clear
View Answer play_arrow
The unit cell of a metallic element of atomic mass 108 and density \[10.5\text{ }g/c{{m}^{3}}\] is a cube with edge length of 409 pm. The structure of the crystal lattice is-
A)
fcc
done
clear
B)
bcc
done
clear
C)
hcp
done
clear
D)
None of these
done
clear
View Answer play_arrow
In a 0.2 molal aqueous solution of a weak acid HX the degree of ionization is 0.3. Taking \[Kf\] for water as 1.85, the freezing point of the solution will be nearest to-
A)
\[-0.260{}^\circ C\]
done
clear
B)
\[+\,0.480{}^\circ C\]
done
clear
C)
\[-0.480{}^\circ C\]
done
clear
D)
\[-0.360{}^\circ C\]
done
clear
View Answer play_arrow
In a CCP lattice of X and Y, X atoms are present at the comers while Y atoms are at face centres. Then the formula of the compound would be if one of the X atoms from a comer is replaced by Z atoms (also monovalent)?
A)
\[{{X}_{7}}{{Y}_{24}}{{Z}_{2}}~\]
done
clear
B)
\[{{X}_{7}}{{Y}_{24}}Z\]
done
clear
C)
\[{{X}_{24}}{{Y}_{7}}Z\]
done
clear
D)
\[Z{{Y}_{24}}Z\]
done
clear
View Answer play_arrow
Using the data given below the order and rate constant for the reaction: \[C{{H}_{3}}CHO\left( g \right)\to C{{H}_{4}}\left( g \right)+Co\left( g \right)\] would be-
Experiment no. Initial cone. Initial rate \[\left[ mol/\ell \right]\] \[\left[ mol.li{{t}^{-1}}\,{{\sec }^{1}} \right]\] I 0.10 0.020 II 0.20 0.080 III 0.30 0.180 IV 0.40 0.320
Answer is-
A)
\[2,\,\,[k=2.0\,\ell /mol\,\,sec]\]
done
clear
B)
\[0,\,\,[k=2.0\,mol/\ell \,sec]\]
done
clear
C)
\[\,2,\,\,[k=1.5/\ell mol\,\,sec]\]
done
clear
D)
\[\,1,\,\,[k=1.5\,se{{c}^{-1}}]\]
done
clear
View Answer play_arrow
For the reaction \[Sn{{O}_{2}}_{(s)}+2{{H}_{2}}_{(g)}\rightleftharpoons \,\,2{{H}_{2}}{{O}_{(g)}}+S{{n}_{\text{(}\ell \text{)}}}\] at 900 K, the equilibrium steam hydrogen mixture was found to be \[40%\text{ }{{H}_{2}}\] by volume the \[{{K}_{p}}\] is:
A)
1.15
done
clear
B)
2.25
done
clear
C)
7.75
done
clear
D)
10
done
clear
View Answer play_arrow
Which one of the following mixtures will give a solution with pH greater than 7
A)
50 ml of 0.1 M HCl + 50 ml of 0.2 M NaCl
done
clear
B)
50 ml of 0.1 M HCl + 50 ml of 0.1 M \[C{{H}_{3}}COONa\]
done
clear
C)
50 ml of 0.1 M \[C{{H}_{3}}COOH\] + 50 ml of 0.1 M NaOH
done
clear
D)
50 ml of 0.2 M \[{{H}_{2}}S{{O}_{4}}\] + 50 ml of 0.3 M NaOH
done
clear
View Answer play_arrow
Which one of the following statement is not correct
A)
Catalyst participate in Mechanism of reaction
done
clear
B)
Chemical adsorption decrease with temperature
done
clear
C)
Cetyl trimethyl ammonium bromide is +ve micells
done
clear
D)
None of these
done
clear
View Answer play_arrow
1.0 mole of HCl and 1.0 mole of \[C{{H}_{3}}COONa\]are dissolved in water and volume is made one litre. What should be the concentration of \[{{H}^{+}}\] in the solution. If \[{{K}_{a}}\] of \[C{{H}_{3}}COOH\] is \[1.6\,\,\times \,\,{{10}^{-}}^{5}\]
A)
\[1.6\times {{10}^{-}}^{5}\]
done
clear
B)
\[4\times {{10}^{-}}^{3}\]
done
clear
C)
\[16\times {{10}^{-}}^{5}\]
done
clear
D)
\[4\times {{10}^{-}}^{2}\]
done
clear
View Answer play_arrow
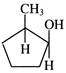
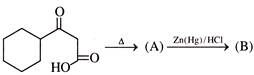
Product of the above reaction is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
\[H-C\equiv C-H\xrightarrow[{{H}_{2}}O]{HgS{{O}_{4}}})\,\,\left( A \right)~\,\,\xrightarrow[(2)\,{{H}_{3}}{{O}^{\oplus }}]{(1)\,HCN}\,\,\left( B \right);\] Product [B] of above reaction is-
A)
Glycine
done
clear
B)
Alanine
done
clear
C)
Acetic acid
done
clear
D)
Lactic acid
done
clear
View Answer play_arrow
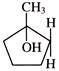
In the above reaction, product is-
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
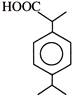
\[\xrightarrow[dil.\,{{H}_{2}}S{{O}_{4}}]{HCN}\,\,\xrightarrow{\operatorname{Re}d\,\,P+HI}\,(Ibuprofen)\,\,\] Ibuprofen is-
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following process involves the cleavage of covalent bond-
A)
Melting of sodium metal
done
clear
B)
Melting of ice
done
clear
C)
Melting of ammonium chloride
done
clear
D)
Formation of atomic hydrogen
done
clear
View Answer play_arrow
Major product of the above reaction is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
In the reaction the structure of the product [X] is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following reaction will not give ether as a major product?
A)
\[C{{H}_{3}}C{{H}_{2}}Cl\,\,+\,\,A{{g}_{2}}O\left( dry \right)\,\,\to \]
done
clear
B)
\[{{\left( C{{H}_{3}} \right)}_{3}}CCl+C{{H}_{3}}C{{H}_{2}}ONa\text{ }\to \]
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The oxidation potentials of Zn, Cu, Ag, \[{{H}_{2}}\] and Ni are 0.76, -0.34, -0.80, 0 and 0.25 volt respectively. Which of the following reactions will provide maximum voltage?
A)
\[Zn+C{{u}^{2+}}\,\to \,\,Cu+Z{{n}^{\text{2+}}}\]
done
clear
B)
\[~Zn+2A{{g}^{+}}\to \text{ }2Ag+Z{{n}^{2+}}\]
done
clear
C)
\[{{H}_{2}}+C{{u}^{2+}}\to \,\,2{{H}^{+}}+Cu\]
done
clear
D)
\[{{H}_{2}}+N{{i}^{2+}}\to 2{{H}^{+}}+Ni\]
done
clear
View Answer play_arrow
Identify product D in the following reaction sequence:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Product will be:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
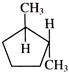
Select pair of chain isomers from the following
A)
I and II
done
clear
B)
II and IV
done
clear
C)
I and IV
done
clear
D)
II and III
done
clear
View Answer play_arrow
Find out correct product of reaction-
A)
done
clear
B)
done
clear
C)
done
clear
D)
No reaction
done
clear
View Answer play_arrow
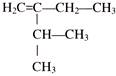
The IUPAC name of the following compound is:
A)
2-ethyl-3-methylbut-l-ene
done
clear
B)
2-isopropylbut-l-ene
done
clear
C)
2-methyl-3-ethylbut-3-ene
done
clear
D)
ethyl ispropyl ethane
done
clear
View Answer play_arrow
Optically active isomer [A] of \[({{C}_{5}}{{H}_{9}}Cl)\] on treatment with one mole of \[{{H}_{2}}\] gives an optically inactive compound [B]. Compound [A] will be:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following is common to both avian and mammalian development?
A)
Primitive streak
done
clear
B)
Trophoblast
done
clear
C)
Holoblastic cleavage
done
clear
D)
Yolk plug
done
clear
View Answer play_arrow
Foetal circulation differs from that of an adult because-
A)
Blood does not circulate through the gastro- intestinal tract, kidneys & lungs.
done
clear
B)
Foetal liver is not developed.
done
clear
C)
Foetus utilizes\[C{{O}_{2}}\], and adult utilizes \[{{O}_{2}}\]
done
clear
D)
Heart is not functional
done
clear
View Answer play_arrow
Major ex-situ conservation of biodiversity is beingmanaged by
A)
National Bureau of Plant, Genetic Resources (NBPGR)
done
clear
B)
Central Arid Zone, Research Institute (CAZRI)
done
clear
C)
Indian Agricultural Research Institute (IARI)
done
clear
D)
International Biological Programme
done
clear
View Answer play_arrow
Acid rain is caused by or recent reports of acid rain in some industrial cities are due to the effect of atmospheric pollution by-
A)
Excessive release of\[C{{O}_{2}}\]. by burning of fuels like wood and charcoal, cutting of forests and increased animal population
done
clear
B)
Excessive release of\[N{{O}_{2}}\], and \[S{{O}_{2}}\] in atmosphere by burning of fossil fuel
done
clear
C)
Excessive release of \[N{{H}_{3}}\] by industrial plants and coal gas
done
clear
D)
Excessive release of CO in atmosphere by incomplete combustion of coke, charcoal and other carbonaceous fuel in paucity of oxygen
done
clear
View Answer play_arrow
The population of an insect species shows an explosive increase in numbers during rainy season followed by its disappearance at the end of the season. What does this show-
A)
S-shaped or sigmoid growth of this insect
done
clear
B)
The food plants mature and die at the end of the rainy season
done
clear
C)
Its population growth curve is of J-type
done
clear
D)
The population of its predators increases enormously
done
clear
View Answer play_arrow
What will happen to a well growing herbaceous plant in the forest if it is transplanted outside the forest in a park?
A)
It will grow normally
done
clear
B)
It will grow well because it is planted in the same locality
done
clear
C)
It may not survive because of change in its microclimate
done
clear
D)
It grows very well because the plant gets more sunlight
done
clear
View Answer play_arrow
A flower is actinomorphic when it is divisible into two-
A)
Halves having equal number of sepals and petals
done
clear
B)
Similar halves by a vertical division in any plane
done
clear
C)
Similar halves by a vertical division in one plane only
done
clear
D)
Halves having similar parts by a transverse division
done
clear
View Answer play_arrow
Total number of meitic division required for forming 100 zygotes/100 grains for wheat is-
A)
100
done
clear
B)
75
done
clear
C)
125
done
clear
D)
50
done
clear
View Answer play_arrow
Flowering in long day plants can be induced in short day condition by treatment of
A)
Auxin
done
clear
B)
Gibberellin
done
clear
C)
Cytokinin
done
clear
D)
ABA
done
clear
View Answer play_arrow
Potometer works on the principle of Amount of water absorbed equals the amount of water transpired
A)
Amount of water absorbed equal the amount of water transpired
done
clear
B)
Osmotic pressure
done
clear
C)
Root pressure
done
clear
D)
Potential difference between the tip of the tube and that of the plant
done
clear
View Answer play_arrow
If number of chromosome in petal is 26 than its pollen grain cell has how many chromosomes-
A)
26
done
clear
B)
13
done
clear
C)
39
done
clear
D)
52
done
clear
View Answer play_arrow
Monoadelphous stamens can be observed in-
A)
Cotton
done
clear
B)
Chilli
done
clear
C)
Asparagus
done
clear
D)
Both [a] & [c]
done
clear
View Answer play_arrow
Biotic succession is caused by-
A)
Competition amongst species
done
clear
B)
Occurrence of diseases
done
clear
C)
Changes is grazing habits
done
clear
D)
Adaptive ability to environmental changes
done
clear
View Answer play_arrow
When environmental conditions are non-limiting what will happen
A)
Maximum mortality and minimum natality
done
clear
B)
Mortality minimum
done
clear
C)
Natality minimum
done
clear
D)
Mortality maximum
done
clear
View Answer play_arrow
Decomposition of detritous plays significant role in humus formation in soil. Which of the following is not the feature of humus-
A)
Colloid nature
done
clear
B)
Nutrient reservoir
done
clear
C)
Easily decomposed
done
clear
D)
Dark coloured
done
clear
View Answer play_arrow
The characteristic associated with community is-
A)
Density independent effect
done
clear
B)
Age pyramid
done
clear
C)
Edge effect
done
clear
D)
Population size
done
clear
View Answer play_arrow
The given kind of population is
A)
Declining
done
clear
B)
Stable
done
clear
C)
Expanding
done
clear
D)
Extincting
done
clear
View Answer play_arrow
Which of the following traits is not characteristic of amphibians?
A)
Skin is moist and without scales
done
clear
B)
Cloaca is present
done
clear
C)
Dioecous, external fertilization, oviparous, indirect development
done
clear
D)
Amniotic egg is present
done
clear
View Answer play_arrow
Parazoa differs from Eumetazoa in
A)
Absence of digestive cavity
done
clear
B)
having intracellular digestion
done
clear
C)
presence of haemocoel
done
clear
D)
having blastocoel
done
clear
View Answer play_arrow
Thorax of Cockroach has segments-
A)
One
done
clear
B)
two
done
clear
C)
Three
done
clear
D)
Four
done
clear
View Answer play_arrow
Mouthparts of cockroach is-
A)
Sponging type
done
clear
B)
Biting and sucking type
done
clear
C)
Biting and chewing type
done
clear
D)
Piercing and sucking type
done
clear
View Answer play_arrow
Sperms undergoing maturation are stored till ejaculation in-
A)
epidydimis
done
clear
B)
ampulla
done
clear
C)
rete testes
done
clear
D)
seminiferous tubule
done
clear
View Answer play_arrow
Onset of menstruation is due to-
A)
Fall in level of progesterone
done
clear
B)
Increase in oestrogen level
done
clear
C)
Increase in FSH level
done
clear
D)
Decrease in oxytocin level
done
clear
View Answer play_arrow
In human embryology, the first cleavage is-
A)
Unequal, Holoblastic
done
clear
B)
Equal, Holoblastic
done
clear
C)
Unequal, Meroblastic
done
clear
D)
Equal, Discoidal
done
clear
View Answer play_arrow
ZIFT-ET is usually applied for-
A)
If female is sterile and provide unfavourable environment for implantation
done
clear
B)
If female is fertile and provide unfavourable environment for implantation
done
clear
C)
If female is fertile and unfavourable environment for fertilization but favourable for implantation
done
clear
D)
Any of the conditions
done
clear
View Answer play_arrow
Hand of man, wing of bat and flipper of seal represent-
A)
Vestigial organs
done
clear
B)
Analogous organs
done
clear
C)
Evolutionary organs
done
clear
D)
Homologous organs
done
clear
View Answer play_arrow
Which statement is correct?
A)
Lamarck theory - Struggle for existence
done
clear
B)
Darwin theory - Use and disuse of organ
done
clear
C)
Biogenetic law - Recapitulatin theory
done
clear
D)
Lamarck theory - Theory of continuity of germplasm
done
clear
View Answer play_arrow
If there is deficiency of surfactant the disease is-
A)
Emphysema
done
clear
B)
Respiratory distress syndrome
done
clear
C)
Bronchitis
done
clear
D)
All above
done
clear
View Answer play_arrow
Which of the following statements correctly defines Bohr effect? Rise in-
A)
\[{{P}_{50}}\text{ }with\text{ }a\text{ }increases\text{ }in\text{ }C{{O}_{2}}\text{ }cone\]
done
clear
B)
\[{{P}_{60}}\text{ }with\text{ }a\text{ }decreases\text{ }in\text{ }pH\]
done
clear
C)
\[~{{P}_{60}}\text{ }with\text{ }a\text{ }decreases\text{ }in\text{ }pH\]
done
clear
D)
\[{{P}_{50}}\text{ }with\text{ }a\text{ }decreases\text{ }in\,C{{O}_{2}}\]
done
clear
View Answer play_arrow
The amount of air remaining in the alveoli at the end of quiet respiration is-
A)
Tidal volume
done
clear
B)
Inspirating reserve volume
done
clear
C)
Inspirating capacity
done
clear
D)
Functional residual capacity
done
clear
View Answer play_arrow
In which point pulmonary artery is different from pulmonary vein:
A)
Its lumen is broad
done
clear
B)
Its wall is thick
done
clear
C)
It have valves
done
clear
D)
It does not possess endothelium
done
clear
View Answer play_arrow
Pulmonary aorta in mammalian embryo communicates with carotico-systemic aorta by a thin vessel called ductus arterious which latter closes and remains in adult as:
A)
Fossa ovalis
done
clear
B)
Ligamentum arteriosus
done
clear
C)
Carotico-pulmonary aperture
done
clear
D)
None of the above
done
clear
View Answer play_arrow
The animal which retains urea for hypertonicity is-
A)
Man
done
clear
B)
Bird
done
clear
C)
Elasmobranch
done
clear
D)
Amphibian
done
clear
View Answer play_arrow
Marine teleost fishes excrete:
A)
Uric acid
done
clear
B)
TMAO
done
clear
C)
Ammonia
done
clear
D)
All of these
done
clear
View Answer play_arrow
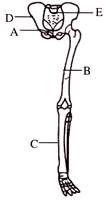
Identify the labeling of A, B, C, D, E in the given diagram-
A)
Femur, Ilium, Tibia, Pubis and Sacrum
done
clear
B)
Pubis, Tibia, Femur, Ilium, and Sacrum
done
clear
C)
Ilium, Femur, Tibia, Pubis and Sacrum
done
clear
D)
Pubis, Femur, Tibia, Ilium, and Sacrum
done
clear
View Answer play_arrow
Joint between the skull and atlas which is responsible for nodding movement is called-
A)
Atlanto-occipetal joint
done
clear
B)
Atlanto-axial joint
done
clear
C)
Occipetal condyle
done
clear
D)
None of these
done
clear
View Answer play_arrow
The energy for muscle contraction is most directly obtained from-
A)
Phosphocreatine
done
clear
B)
ATP
done
clear
C)
Anaerobic respiration
done
clear
D)
Aerobic respiration
done
clear
View Answer play_arrow
Which of these has distinct outer sarcolemma covering?
A)
Striated
done
clear
B)
Unstriated
done
clear
C)
Circular muscle
done
clear
D)
All these
done
clear
View Answer play_arrow
Select the answer which gives correct matching of the end products of digestion in human with site and mechanism of absorption-
Product Site of absorption Mechanism
A)
(a) Galactose Mouth Active absorption
done
clear
B)
(b) Proline Stomach Passive absorption
done
clear
C)
Fructose Small intestine Active absorption
done
clear
D)
Glucose Small intestine Active transport
done
clear
View Answer play_arrow
Which function of the liver results m the production of bile pigments?
A)
Breakdown of haemoglobin
done
clear
B)
Deamination of ammo acids
done
clear
C)
Detoxification of metabolic poisons
done
clear
D)
Release of stores vitamin A
done
clear
View Answer play_arrow
At the point of exit of the optic nerve-
A)
There are no rods and cones
done
clear
B)
There are rods but no cones
done
clear
C)
There are cones but no rods
done
clear
D)
All types of cells are absent
done
clear
View Answer play_arrow
Brain sand is related with-
A)
Thymus
done
clear
B)
Pineal body
done
clear
C)
Islet of langerhans
done
clear
D)
Pituitary gland
done
clear
View Answer play_arrow
In the nerve cells, the reversal of the resting potential is known as depolarization. This occurs due to-
A)
\[influx\,\,of\,\,{{K}^{+}}\]
done
clear
B)
\[influx\,\,of\,\,C{{a}^{2+}}\,\,and\,\,Cl\]
done
clear
C)
\[influx\,\,of\,\,{{K}^{+}}\]
done
clear
D)
\[influx\,\,of\,\,N{{a}^{+}}\]
done
clear
View Answer play_arrow
During the conduction of an impulse on the inside of plasma membrane of a neuron electrical potential changes from-
A)
-ve to +ve and remains +ve
done
clear
B)
-ve to +ve and then to -ve
done
clear
C)
+ve to -ve and then
done
clear
D)
remains -ve +ve to -ve and then to +ve
done
clear
View Answer play_arrow
Smallest formed element in fluid connective tissue is-
A)
Lymphocyte
done
clear
B)
Basophil
done
clear
C)
Thrombocyte
done
clear
D)
Monocyte
done
clear
View Answer play_arrow
Tendon is a structure which connects
A)
a bone with another bone
done
clear
B)
a muscle with a bone
done
clear
C)
a nerve with a muscle or bone
done
clear
D)
a muscle with muscle
done
clear
View Answer play_arrow
ELISA is used to detect viruses, where-
A)
Catalase is the key reagent
done
clear
B)
Alkaline phosphatase is the key reagent
done
clear
C)
Southern blotting is done
done
clear
D)
DNA- Probes are required
done
clear
View Answer play_arrow
Column-I Column-II I. PCR A. Therums aquaticus II. Taq DNA B. Plasmid polymerase III. Extrachoromosomal C. Amplification DNA IV. Ethidium bromide D. DNA staining
A)
I-C, II-A, III-B, IV-D
done
clear
B)
I-B, II-A, III-D, IV-C
done
clear
C)
I-D, II-A, III-C, IV-B
done
clear
D)
I-A, II-D, III-B, IV-C
done
clear
View Answer play_arrow
A human gene product can be produced by genetically engineered bacteria. This is possible because the-
A)
Genetic code is universal
done
clear
B)
Human chromosome can replicate in bacterial cell
done
clear
C)
Bacterial cell can carry out the splicing reaction
done
clear
D)
Mechanism of gene regulation is identical in humans and bacteria
done
clear
View Answer play_arrow
Genetic code consists of-
A)
4codons, each with two nucleotides
done
clear
B)
16 codons, each with four nucleotides
done
clear
C)
64 codons, each with two nucleotides
done
clear
D)
64 codons, each with three nucleotides
done
clear
View Answer play_arrow
The dominantly inherited disorder in human beings is-
A)
Huntington's chorea
done
clear
B)
Tay-sachs disease
done
clear
C)
Phenylketonuria
done
clear
D)
Albinism
done
clear
View Answer play_arrow
Haploids are able to express both recessive and dominant allele/mutations because there are-
A)
Many alleles for each gene
done
clear
B)
Only two allele in a gene
done
clear
C)
Only one allele for each gene in the individual
done
clear
D)
Two alleles for each gene
done
clear
View Answer play_arrow
Satellite DNA is classified into mini and micro satellite DNA on the basis of
A)
A : T / G : C base
done
clear
B)
Length of fragment
done
clear
C)
number of repetitive unit
done
clear
D)
All the above
done
clear
View Answer play_arrow
Match List-I and List-II and select the correct answer using the code given below in the lists-
List-I List-II 1. Light Joins short segments of DNA together 2. DNA Cuts DNA at specific polymerase DNA sequence 3. Helicase Breaks the hydrogen bonds between complementary pairs during DNA replication
Answer codes:
A)
1, 2 and 3 are correct
done
clear
B)
1 and 2 are correct, 3 is false
done
clear
C)
1 is correct, 2 and 3 are false
done
clear
D)
1 and 3 are correct, 2 is false
done
clear
View Answer play_arrow
Match list-I with list-II and select the correct answer using the codes given below the lists-
List-I List-II A DNA structure 1 Muller and stadder B Semiconservative replication of DNA 2 Beader and Tatum C One gene-one enzyme theory 3 Watson and Crick D Induction of mutation 4 Meselson and stahl
Codes:
A)
A-1 B-2 C-3 D-4
done
clear
B)
A-3 B-4 C-1 D-2
done
clear
C)
A-3 B-4 C-2 D-1
done
clear
D)
A-4 B-3 C-2 D-1
done
clear
View Answer play_arrow
Which one statement regarding 'Okazaki fragment' is correct-
A)
These are double helical fragments of DNA
done
clear
B)
These are single stranded fragments of DNA
done
clear
C)
These are fragments of mRNA
done
clear
D)
These are fragments of tRNA
done
clear
View Answer play_arrow
When an albino female plant of maize is crossed with normal green male plant, all plants in the progeny are albino because-
A)
Plastids are inherited through maternal plants
done
clear
B)
Albinism is dominant over green character
done
clear
C)
The crossing results in structural changes in green plastids
done
clear
D)
Green plastids of male parents become mutated
done
clear
View Answer play_arrow
An individual produces following types of gametes: (AbC), (Abc), (abC) and (abc). What can be the possible genotype of this individual?
A)
Aa Bb Cc
done
clear
B)
Aa Bb CC
done
clear
C)
Aa Bb cc
done
clear
D)
Aa bb Cc
done
clear
View Answer play_arrow
A cross of a wild type red-eyed female Drosophila with a violet-eyed male produces all red-eyed offspring. If the gene is sex linked, what should the reciprocal cross (violet-eyed female x red-eyed male) produce? (Assume that the red allele is dominant to the violet allele.)-
A)
All violet-eyed flies
done
clear
B)
3 red-eyed flies to 1 violet-eyed
done
clear
C)
A \[1\text{ }:\text{ }1\] ratio of red and violet eyes in both males and females
done
clear
D)
Red-eyed females and violet-eyed males
done
clear
View Answer play_arrow
Which of the following is not a true difference between IgG and IgM?
A)
IgM is a pentamer while IgG is a monomer
done
clear
B)
IgG is formed early during the primary response, IgM is formed later
done
clear
C)
IgG can cross the placenta, IgM usually does not.
done
clear
D)
IgG has two antigen binding sites, IgM has ten
done
clear
View Answer play_arrow
Which of the following belongs to ?Pain killers??
A)
Sedatives
done
clear
B)
Opiates
done
clear
C)
Hallucinogens
done
clear
D)
Stimulants
done
clear
View Answer play_arrow
Higher plants differ from animals in-
A)
Spindle microtubules
done
clear
B)
Kinetochores
done
clear
C)
Anastral spindle
done
clear
D)
Disappearance of nucleolus during prophase
done
clear
View Answer play_arrow
Mitotic spindle is mainly composed of the protein-
A)
Actin
done
clear
B)
Actomyosin
done
clear
C)
Tubulin
done
clear
D)
Myoglobin
done
clear
View Answer play_arrow
Animal cells undergo cytokinesis by-
A)
Furrowing
done
clear
B)
Cell plate
done
clear
C)
Both [a] and [b]
done
clear
D)
Furrowing followed by deposition of special materials
done
clear
View Answer play_arrow
'Omnis cellula e cellula' - The above statement was given by-
A)
Robert Brown
done
clear
B)
Rudolf Virchow
done
clear
C)
M.J.Schleiden
done
clear
D)
Theodor Schwann
done
clear
View Answer play_arrow
Nucleolus in eukaryotic cells is-
A)
Visible in metaphase
done
clear
B)
Bounded by a membrane
done
clear
C)
The site for synthesis of RNA polymerase
done
clear
D)
The site for packaging of r-RNAs with ribosomal proteins
done
clear
View Answer play_arrow
Match the following and choose the correct combination from the options given below-
Cell organelle Function A. Endoplasmic reticulum I Take part in cellular respiration B. Free ribosome II Take part in osmoregulations and excretions C. Mitochondrion III Synthesis of lipids D. Contractile vacuole Synthesize non-secretory proteins
A)
A=III, B=IV, C=I, D=II
done
clear
B)
A=I, B=II, C=IV, D=III
done
clear
C)
A=III, B=IV, C=II, D=I
done
clear
D)
A=III, B=II, C=I, D=IV
done
clear
View Answer play_arrow
Which of the following is characteristic of phospholipids of plasma membrane?
A)
One non-polar head and two polar tails
done
clear
B)
One polar head and two non-polar tails
done
clear
C)
Two non-polar heads and one polar tail
done
clear
D)
Two polar heads and two polar tails
done
clear
View Answer play_arrow
Mark the correct sequence of layers found in root anatomy-
A)
Epiblema, cortex, endodermis, pericycle
done
clear
B)
Cortex, epiblema, pericycle, endodermis
done
clear
C)
Epiblema, cortex, pericycle, endodermis
done
clear
D)
Cortex, epiblema, endodermis, epidermis
done
clear
View Answer play_arrow
Hardness of seed coats is due to the presence of-
A)
Sclerenchymatous fibres
done
clear
B)
Sclereids
done
clear
C)
Bast
done
clear
D)
Stone cells
done
clear
View Answer play_arrow
The meristematic layer that is responsible for cutting off secondary vascular tissue is known as-
A)
Procambium
done
clear
B)
Vascular cambium
done
clear
C)
Cork cambium
done
clear
D)
both [a] & [b]
done
clear
View Answer play_arrow
Colchicine producing plant belongs to family-
A)
Liliaceae
done
clear
B)
Rubiaceae
done
clear
C)
Malvaceae
done
clear
D)
Solanaceae
done
clear
View Answer play_arrow
Most reduced gametophyte in plant kingdom is
A)
Male gametophyte of lilium
done
clear
B)
Male gametophyte of pinus
done
clear
C)
Female gametophyte of lilium
done
clear
D)
Female gametophyte of pinus
done
clear
View Answer play_arrow
Select the characters, which are not applicable to the family-Solanaceae?
I. Epipetalous and syngenesious anthers II. Bicarpellary and syncarpous ovary III. Oblique ovary with axile placentation IV. Stamens six, arranged in two whorls V. Bicarpellary, syncarpous and inferior ovary
A)
II and III only
done
clear
B)
I, IV and V only
done
clear
C)
II, IV and V only
done
clear
D)
I and III only
done
clear
View Answer play_arrow
The floral formula
is that of-
A)
Tulip
done
clear
B)
Soyabean
done
clear
C)
Sunnhemp
done
clear
D)
Tobacco
done
clear
View Answer play_arrow
Scientific study of diversity of organisms and their evolutionary relationships is-
A)
Morphology
done
clear
B)
Anatomy
done
clear
C)
Classification
done
clear
D)
Systematics
done
clear
View Answer play_arrow
The biological definition of a species depends on-
A)
The geographical distribution of two groups of organisms
done
clear
B)
Reproductive isolation of two groups of organisms
done
clear
C)
Anatomical and developmental differences between the two groups of organisms
done
clear
D)
Difference in the adaptation of two groups of organisms
done
clear
View Answer play_arrow
A bacterium divides after every 35 nun., if a culture containing 105 cells per mL is grown, then cell concentration per mL after 175 min will be-
A)
\[125\times {{10}^{5}}\]
done
clear
B)
\[32\times {{10}^{6}}\]
done
clear
C)
\[3.2\times {{10}^{5}}\]
done
clear
D)
\[32\times {{10}^{5}}\]
done
clear
View Answer play_arrow
Find out the pairs, which are correctly matched?
I. Cyanobacteria Biopesticides II. Mycorrhiza Solubilization of phosphate III. Bacillus thuringeinsis Cry protein IV. Single cell protein Rhizobia
A)
I and II
done
clear
B)
II and III
done
clear
C)
III and IV
done
clear
D)
I and III
done
clear
View Answer play_arrow
Which is correct for the structure of cell wall of bacteria and fungi?
A)
Both are made up of cellulose
done
clear
B)
Both have mucopeptide
done
clear
C)
Both are made up of N-acetylglucosamine
done
clear
D)
None of the above
done
clear
View Answer play_arrow
In the diagram given above, the algae have been labelled as 'A', 'B', 'C', 'D' and 'E'. These algae are respectively identified as-
A)
Dictyota, Polysiphonia, Porphyra, Fucus and Laminaria
done
clear
B)
Porphyra, Dictyota, Laminaria, Fucus and Polysiphonia
done
clear
C)
Dictyota, Polysiphonia, Porphyra, Laminaria and Fucus
done
clear
D)
Laminaria, Polysiphonia, Porphyra, Dictyota and Fucus
done
clear
View Answer play_arrow
Find out the mis-matched pair-
A)
Agar-agar - Polymer of glucose and sulphur containing carbohydrates
done
clear
B)
Chitin - Polymer of glucosamine
done
clear
C)
Peptidoglycan - Polysaccharide linked to peptides
done
clear
D)
Lipopolysaccharides - A complex of lipid and polysaccharide
done
clear
View Answer play_arrow
In Chlorophyceae, the mode of sexual reproduction is-
A)
Anisogamy
done
clear
B)
Oogamy
done
clear
C)
Isogamy
done
clear
D)
All of these
done
clear
View Answer play_arrow
Protonema is the juvenile filamentous state in the life cycle of-
A)
Funaria
done
clear
B)
Riccia
done
clear
C)
Marchantia
done
clear
D)
Laminaria
done
clear
View Answer play_arrow
Swollen placenta is commonly observed in-
A)
Petunia
done
clear
B)
Tulip
done
clear
C)
Sesbania
done
clear
D)
both [a] & [c]
done
clear
View Answer play_arrow
If number of chromosomes in foot of fern embryo is 8, what should be the number in its spores?
A)
4
done
clear
B)
8
done
clear
C)
23
done
clear
D)
16
done
clear
View Answer play_arrow
Winged pollen grains are found in-
A)
Cycas
done
clear
B)
Pinus
done
clear
C)
Pteris
done
clear
D)
Selaginella
done
clear
View Answer play_arrow
The outer covering of endosperm separates the embryo from endosperm by a proteinaceous layer in monocot seed is known as-
A)
Scutellum
done
clear
B)
Aleurone
done
clear
C)
Plumule
done
clear
D)
Seed coat
done
clear
View Answer play_arrow
Which of the following is not an underground stem
A)
Zaminkand
done
clear
B)
Sweet potato
done
clear
C)
Colocassia
done
clear
D)
Turmeric
done
clear
View Answer play_arrow
Name the family having \[\left( 9 \right)\,\,+\,\,1\] arrangement of stamen-
A)
Solanaceae
done
clear
B)
Asteraceae
done
clear
C)
Liliaceae
done
clear
D)
Febaceae
done
clear
View Answer play_arrow
 Its position-time graph may be given as
Its position-time graph may be given as 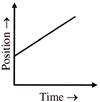
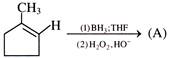 Product of the above reaction is:
Product of the above reaction is: 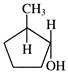
 In the above reaction, product is-
In the above reaction, product is- 
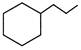
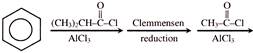 \[\xrightarrow[dil.\,{{H}_{2}}S{{O}_{4}}]{HCN}\,\,\xrightarrow{\operatorname{Re}d\,\,P+HI}\,(Ibuprofen)\,\,\] Ibuprofen is-
\[\xrightarrow[dil.\,{{H}_{2}}S{{O}_{4}}]{HCN}\,\,\xrightarrow{\operatorname{Re}d\,\,P+HI}\,(Ibuprofen)\,\,\] Ibuprofen is- 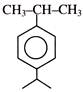
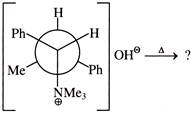 Major product of the above reaction is:
Major product of the above reaction is: 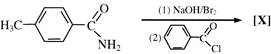
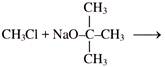
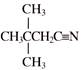
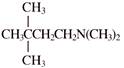
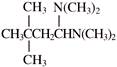
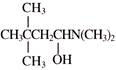
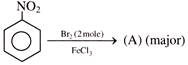 Product will be:
Product will be: 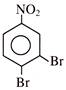
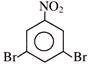
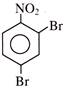
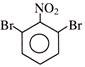
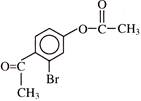
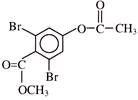
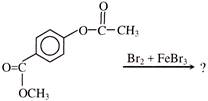 Find out correct product of reaction-
Find out correct product of reaction- 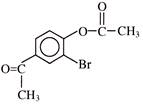
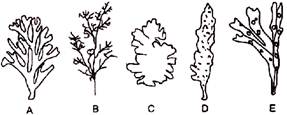 In the diagram given above, the algae have been labelled as 'A', 'B', 'C', 'D' and 'E'. These algae are respectively identified as-
In the diagram given above, the algae have been labelled as 'A', 'B', 'C', 'D' and 'E'. These algae are respectively identified as- 