-
Write the structures of different
chain isomers of alkanes corresponding to the molecular formula, \[{{C}_{6}}{{H}_{14}}\].
Also write their IUPAC names.
View Answer play_arrow
-
Write structures of different
isomeric alkyl groups corresponding to the molecular formula \[{{C}_{5}}{{H}_{11}}\]Write
IUPAC names of alcohols obtained by the attachment of OH groups at different
carbons of the chain.
View Answer play_arrow
-
Write IUPAC names of the following compounds :
(i) \[{{(C{{H}_{3}})}_{3}}CC{{H}_{2}}C{{(C{{H}_{3}})}_{3}}\]
(ii) \[{{(C{{H}_{3}})}_{2}}C{{({{C}_{2}}{{H}_{5}})}_{2}}\]
(iii) Tetra-tert-butylmethane
View Answer play_arrow
-
Write structural formulas of the following compounds :
(i) 3, 4, 4, 5-Tetramethylheptane
(ii) 2, 5-Dimethylhexane
View Answer play_arrow
-
Write structures for each of the following compounds. Why
are the given names incorrect? Write correct IUPAC names.
(i) 2-Ethylpentane
(ii) 5-Ethyl-3-methylheptane
View Answer play_arrow
-
Sodium salt of which acid will be needed
for the preparation of propane? Write chemical equation for the reaction.
View Answer play_arrow
-
Write IUPAC names of the following compounds.
(i) \[{{(C{{H}_{3}})}_{2}}CH-CH=CH-C{{H}_{2}}-CH=CH-\underset{\underset{{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}}}{\mathop{|}}\,}{\mathop{CH}}\,-C{{H}_{5}}\](ii)

(iii) \[{{H}_{2}}C=C{{(C{{H}_{2}}C{{H}_{2}}C{{H}_{3}})}_{2}}\]
(iv) \[C{{H}_{3}}-\overset{\overset{C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}}{\mathop{|}}\,}{\mathop{C}}\,H-CH=\overset{\overset{C{{H}_{2}}C{{H}_{3}}}{\mathop{|}}\,}{\mathop{C}}\,-C{{H}_{2}}-\underset{\underset{C{{H}_{3}}}{\mathop{|}}\,}{\mathop{C}}\,H-C{{H}_{3}}\]
-
Calculate number of sigma (\[\sigma
\]) and pi (\[\pi \]) bonds in
the above structures (i-iv).
View Answer play_arrow
-
Write structures and IUPAC names
ofdifferent structural isomers of alkenes corresponding to\[{{C}_{5}}{{H}_{10}}\].
View Answer play_arrow
-
Draw cis-
and trans-isomers of thefollowing compounds. Also write their IUPAC names:.
(i) \[CHCl=CHCl\]
(ii) \[{{C}_{2}}{{H}_{5}}CC{{H}_{3}}=CC{{H}_{3}}{{C}_{2}}{{H}_{5}}\]
View Answer play_arrow
-
Which of the following compounds willshow cis-trans
isomerism?
(i) \[{{(C{{H}_{3}})}_{2}}C=CH-{{C}_{2}}{{H}_{5}}\]
(ii) \[{{H}_{2}}C=C\,B{{r}_{2}}\]
(iii) \[{{C}_{6}}{{H}_{5}}CH=CH-C{{H}_{3}}\]
(iv) \[C{{H}_{3}}CH=CClC{{H}_{3}}\]
View Answer play_arrow
-
Write
IUPAC names of the productsobtained by addition reactions of \[HBr\]to
hex-1-ene.
(i) In presence of peroxide and
(ii) In the absence of peroxide.
View Answer play_arrow
-
Write structures of different
isomers corresponding to the 5th member of alkyne series. Also write IUPAC
names of all the isomers. What type of isomerism is exhibited by different
pairs of isomers?
View Answer play_arrow
-
View Answer play_arrow
-
How do you account for the
formation of ethane during chlorination of methane?
View Answer play_arrow
-
Write the IUPAC names of the following compounds:
(a) \[C{{H}_{3}}CH=C{{(C{{H}_{3}})}_{2}}\]
(b) \[{{H}_{2}}C=CH-C\equiv C-C{{H}_{3}}\]
(c)

(d)

(e)

(f) \[C{{H}_{3}}{{(C{{H}_{2}})}_{4}}\underset{\underset{\text{C}{{\text{H}}_{\text{2}}}\text{-CH(C}{{\text{H}}_{\text{3}}}{{\text{)}}_{\text{2}}}}{\mathop{|}}\,}{\mathop{C}}\,H{{(C{{H}_{2}})}_{3}}C{{H}_{3}}\]
(g)\[{{H}_{3}}C-CH=CH-C{{H}_{2}}-CH=CH\] \[-\underset{\underset{{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}}}{\mathop{\text{
}\!\!|\!\!\text{ }}}\,}{\mathop{CH}}\,-C{{H}_{2}}-CH=C{{H}_{2}}\]
-
For
the following compounds, write structural formulae and IUPAC names for all
possible isomershaving the number of double or triple bond as indicated:
(a) \[{{C}_{4}}{{H}_{8}}\] (one double bond)
(b) \[{{C}_{5}}{{H}_{8}}\] (one
triple bond)
View Answer play_arrow
-
Write IUPAC names of the products obtained by the ozonolysis
of the following compounds:
(i) Pent-2-ene
(ii) 3, 4-Dimethylhept-3-ene
(ii) 2-Ethylbut-1-ene
(iv) 1-Phenylbut-1-ene
View Answer play_arrow
-
An alkene 'A' on ozonolysis gives
a mixture of ethanal and pentan-3-one. Write structure and IUPAC name of 'A'.
View Answer play_arrow
-
An alkene 'A' contains three C-C,
eight C-H \[\sigma \]bonds and one C-C \[\pi \] bond. 'A' on ozonolysis gives
two moles of an aldehyde of molar mass 44u. Write IUPAC name of 'A'.
View Answer play_arrow
-
Propanal and pentan-3-one are the
ozonolysis products of an alkene? What is the structural formula of the alkene?
View Answer play_arrow
-
Write chemical equations for the combustion reaction of the
following hydrocarbons:
(i) Butane (ii) Pentene (iii)
Hexyne (iv) Toluene
View Answer play_arrow
-
Draw the cis and trans structures
of hex-2-ene. Which isomer will have higher b.p. and why?
View Answer play_arrow
-
Why is benzene extra ordinarily
stable though it contains three double bonds?
View Answer play_arrow
-
What are necessary conditions for
any system to be aromatic?
View Answer play_arrow
-
Explain why the following systems are not aromatic?
(i)

(ii)

(iii)

-
How will you convert benzene into:
(i) \[p-Nitrobromobenzene\]
(ii) \[m\]-Nitrochlorobenzene
(iii) \[p\]-Nitrotoluene
(iv) Acetophenone
View Answer play_arrow
-
In
the alkane \[C{{H}_{3}}-C{{H}_{2}}-C{{(C{{H}_{3}})}_{2}}-C{{H}_{2}}-CH{{(C{{H}_{3}})}_{2}},\]
identify \[1{}^\circ ,\text{ }2{}^\circ ,\text{ }3{}^\circ \] carbon atoms and
give the
H-atoms bonded to each one of
these.
View Answer play_arrow
-
What effect does branching of an
alkane chain hason its boiling point?
View Answer play_arrow
-
Addition of \[HBr\] to propene yields 2-bromopropane, while
in the presence of benzoyl peroxide, the same reaction yields 1-bromopropane.
Explain and give mechanism.
View Answer play_arrow
-
Write down the products of
ozonolysis of 1, 2-dimethylbenzene (o-xylene). How does the result support
Kekule structure of benzene?
View Answer play_arrow
-
Arrange benzene, n-hexme and
ethyne in decreasing order of acidic behaviour. Also give reason for this
behaviour.
View Answer play_arrow
-
Why does benzene undergo
electrophilic substitution reactions easily and nucleophilic substitutions with
difficulty?
View Answer play_arrow
-
How would you convert the
following compounds into benzene: (i) Ethyne (ii) Ethene (iii) Hexane.
View Answer play_arrow
-
Write structures of all the
alkenes which on hydrogenation give 2-methylbutane.
View Answer play_arrow
-
Arrange the following set of compounds in order of their
decreasing relative reactivity with an electrophile,\[{{E}^{+}}\].
(a) Chlorobenzene, 2, 4-dinitrochlorobenzene, p-nitrochlorobenzene
(b) Toluene, \[p-{{H}_{3}}C-{{C}_{6}}{{H}_{4}}-N{{O}_{2}},\]
\[p-{{O}_{2}}N-{{C}_{6}}{{H}_{4}}-N{{O}_{2}}\]
View Answer play_arrow
-
Out of benzene, m-dinitrobenzene
and toluene which will undergo nitration most easily and why?
View Answer play_arrow
-
Suggest the name of a Lewis acid
other than anhydrous aluminium chloride which can be used during ethylation of
benzene.
View Answer play_arrow
-
Why is Wurtz reaction not
preferred for the preparation of alkanes containing odd number of carbon atoms?
Illustrate your answer by taking one example.
View Answer play_arrow
-
question_answer40)
Arrange the following in decreasing
order of their boiling points :
(A) n-butane (B)
2-methylbutane
(C) n-pentane (D)
2, 2-dimethylpropane
(a) \[A>B>C>D~\] (b)\[B>C>D>A\] (c)\[~D\text{
}>\text{ }C\text{ }>\text{ }B\text{ }>\text{ }A\] (d) \[C\text{
}>\text{ }B\text{ }>\text{ }D\text{ }>\text{ }A\]
View Answer play_arrow
-
question_answer41)
Arrange the halogens \[{{F}_{2}},C{{l}_{2}},B{{r}_{2}},{{I}_{2}}\]in
order of their increasing reactivity with alkanes :
(a) \[{{I}_{2}}<B{{r}_{2}}<C{{l}_{2}}<{{F}_{2}}\] (b)
\[B{{r}_{2}}<C{{l}_{2}}<{{F}_{2}}<{{I}_{2}}\] (c) \[{{F}_{2}}<C{{l}_{2}}<B{{r}_{2}}<{{I}_{2}}\] (d)
\[B{{r}_{2}}<{{I}_{2}}<C{{l}_{2}}<{{F}_{2}}\]
View Answer play_arrow
-
question_answer42)
The increasing order of
reduction of alkyl halides with zinc and dilute \[HCl\] is :
(a) \[R-Cl<R-I<R-Br\] (b)
\[B{{r}_{2}}<C{{l}_{2}}<{{F}_{2}}<{{I}_{2}}\]
(c)\[{{F}_{2}}<C{{l}_{2}}<B{{r}_{2}}<{{I}_{2}}\] (d)
\[B{{r}_{2}}<{{I}_{2}}<C{{l}_{2}}<{{F}_{2}}\]
View Answer play_arrow
-
question_answer43)
The correct IUPAC name of the
following alkane is:
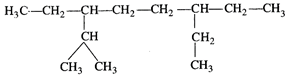 (a) 3, 6-diethyl-2-methyloctane (b)
5-isopropyl-3-ethyloctane
(c) 3-ethyl-5-isopropyloctane (d)
3-isopropyl-6-ethyloctane
(a) 3, 6-diethyl-2-methyloctane (b)
5-isopropyl-3-ethyloctane
(c) 3-ethyl-5-isopropyloctane (d)
3-isopropyl-6-ethyloctane
View Answer play_arrow
-
question_answer44)
The addition of \[HBr\]to
1-butene gives a mixture of products A, B and C.
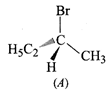
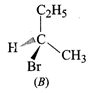 \[\underset{(C)}{\mathop{C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-Br}}\,\]
The mixture consists of:
(a) A and B as major and C as
minor products (b) B as major, A and C as minor products
(c) B as minor, A and C as major
products (d) A and B as minor and C as major products
\[\underset{(C)}{\mathop{C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-Br}}\,\]
The mixture consists of:
(a) A and B as major and C as
minor products (b) B as major, A and C as minor products
(c) B as minor, A and C as major
products (d) A and B as minor and C as major products
View Answer play_arrow
-
question_answer45)
Which of the following will not
show geometrical isomerism?
(a) 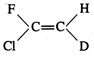 (b)
(b) 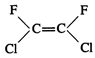 (c)
(c) 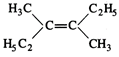 (d)
(d) 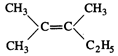
View Answer play_arrow
-
question_answer46)
Arrange the following hydrogen
halides in order of their decreasing reactivity with propene:
(a) \[HCl>HBr>HI~~~~~\] (b)
\[HBr>HI>HCl\] (c)\[HI>HBr>HCl\] (d) \[HCl>HI>HBr\]
View Answer play_arrow
-
question_answer47)
Arrange the following carbanions
in order of their decreasing stability :
(A) \[{{H}_{3}}C-C\equiv
{{C}^{-}}\] (B) \[H-C\equiv {{C}^{-}}\] (C) \[{{H}_{3}}C-CH_{2}^{-}\]
(a) \[A\text{ }>B>C~\] (b)\[B>A>C\] (c)
\[C\text{ }>\text{ }B\text{ }>\text{ }A\] (d) \[C>A>B\]
View Answer play_arrow
-
question_answer48)
Arrange the following alkyl
halides in decreasing order of the rate of \[\beta \]-elimination reaction with
alcoholic KOH.
(A) \[{{H}_{3}}C-C\equiv
{{C}^{-}}\] (B) \[H-C\equiv {{C}^{-}}\] (C) \[{{H}_{3}}C-CH_{2}^{-}\]
(a) \[A>B>C\] (b)
\[C>B>A\] (c) \[B>C>A~~~~~~~~~~\] (d) \[A>C>B\]
View Answer play_arrow
-
question_answer49)
Which of the following reactions
of methane is incomplete combustion?
(a) \[2C{{H}_{4}}+{{O}_{2}}\xrightarrow{Cu/523K/100atm.}2C{{H}_{3}}OH\] (b)
\[C{{H}_{4}}+{{O}_{2}}\xrightarrow{M{{o}_{2}}{{O}_{3}}}HCHO+{{H}_{2}}O\]
(c) \[C{{H}_{4}}+{{O}_{2}}\xrightarrow{{}}C(s)+2{{H}_{2}}O(l)\] (d)
\[C{{H}_{4}}+2{{O}_{2}}\xrightarrow[{}]{{}}C{{O}_{2}}(g)+2{{H}_{2}}O(l)\]
View Answer play_arrow
-
question_answer50)
Some oxidation reactions of
methane are given below. Which of them is/are controlled oxidation reactions?
(a) \[C{{H}_{4}}(g)+2{{O}_{2}}(g)\xrightarrow{{}}C{{O}_{2}}(g)+2{{H}_{2}}O(l)\] (b)
\[C{{H}_{4}}+{{O}_{2}}(g)\xrightarrow[{}]{{}}C(s)+2{{H}_{2}}O(l)\]
(c) \[C{{H}_{4}}(g)+{{O}_{2}}(g)\xrightarrow{M{{o}_{2}}{{O}_{3}}}HCHO+{{H}_{2}}O\] (d)
\[2C{{H}_{4}}(g)+{{O}_{2}}(g)\xrightarrow{Cu/523k/100atm.}2C{{H}_{3}}OH\]
View Answer play_arrow
-
question_answer51)
Which of the following alkenes
on ozonolysis give a mixture of ketones only?
(a) \[C{{H}_{3}}-CH=CH-C{{H}_{3}}\] (b)
\[C{{H}_{3}}-\underset{\underset{\text{C}{{\text{H}}_{\text{3}}}}{\mathop{|}}\,}{\mathop{CH}}\,-CH=C{{H}_{3}}\]
(c) 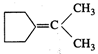 (d)
(d)
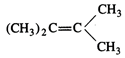
View Answer play_arrow
-
question_answer52)
Which are the correct IUPAC
names of the following compound?
\[{{H}_{3}}C-C{{H}_{2}}C{{H}_{2}}\overset{\overset{\text{HC(C}{{\text{H}}_{\text{3}}}{{\text{)}}_{\text{2}}}}{\mathop{|}}\,}{\mathop{C}}\,H\underset{\underset{{{\text{H}}_{\text{3}}}\text{C-CH-C}{{\text{H}}_{\text{2}}}\text{C}{{\text{H}}_{\text{3}}}}{\mathop{|}}\,}{\mathop{C}}\,{{H}_{2}}C{{H}_{2}}C{{H}_{2}}-C{{H}_{3}}\]
(a) 5-Butyl-4-isopropyldecane (b)
5-Ethyl-4-propyldecane
(c) 5-sec-Butyl-4-iso-propyldecane (d)
4-(1-methylethyl)-5\[-(\text{ }1\text{ }methylpropyl)\]decane
View Answer play_arrow
-
question_answer53)
Which are the correct IUPAC
names of the following compound?
\[{{H}_{3}}C-C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}-\underset{\underset{\underset{\underset{{{H}_{3}}C-\underset{\underset{C{{H}_{3}}}{\mathop{|}}\,}{\mathop{C}}\,-C{{H}_{3}}}{\mathop{|}}\,}{\mathop{\text{C}{{\text{H}}_{\text{2}}}}}\,}{\mathop{|}}\,}{\mathop{CH}}\,-C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}-C{{H}_{3}}\]
(a)
5-(2?, 2?-Dimethylpropyl)-decane (b) 4-Butyl-2, 2-dimethylnonane
(c) 2,2-Dimethyl-4-pentyloctane (d)
5-neo-Pentyldecane
View Answer play_arrow
-
question_answer54)
For an electrophilic
substitution reaction, the presence of a halogen atom in the benzene ring
......... .
(a) deactivates the ring by
inductive effect
(b) deactivates the ring by
resonance
(c) increases the charge density
at ortho and para positions relative to meta position by resonance
(d) directs the incoming
electrophile to meta position by increasing the charge density relative to
ortho and para positions
View Answer play_arrow
-
question_answer55)
In an electrophilic substitution
reaction of nitrobenzene, the presence of nitro group..........
(a) deactivates the ring by
inductive effect
(b) activates the ring by
inductive effect
(c) decreases the charge density
at ortho and para positions of the ring relative to meta position by resonance
(d) increases the charge density
at meta position relative to the ortho and para positions of the ring by
resonance
View Answer play_arrow
-
question_answer56)
Which of the following are
correct?
(a)\[C{{H}_{3}}-O-CH_{2}^{\oplus
}\] is more stable than \[C{{H}_{3}}-CH_{2}^{\oplus
}\]
(b) \[{{(C{{H}_{3}})}_{2}}C{{H}^{\oplus
}}\]is less stable than \[C{{H}_{3}}-C{{H}_{2}}-CH_{2}^{\oplus }\]
(c) \[C{{H}_{2}}=CH-CH_{2}^{\oplus
}\]is more stable than \[C{{H}_{3}}C{{H}_{2}}CH_{2}^{\oplus }\]
(d) \[C{{H}_{2}}=C{{H}^{\oplus
}}\]is more stable than \[C{{H}_{3}}-CH_{2}^{\oplus }\]
View Answer play_arrow
-
question_answer57)
Four structures are given in
options (a) to (d). Examine them and select the aromatic structures :
(a) 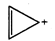 (b)
(b)
 (c)
(c) 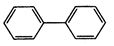 (d)
(d) 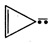
View Answer play_arrow
-
question_answer58)
The molecules having dipole
moment are..........
(a) 2, 2-Dimethylpropane (b)
trans-Pent-2-ene
(c) cis-Hex-3-ene (d)
2, 2, 3, 3-Tetramethylbutane
View Answer play_arrow
-
question_answer59)
Why do alkenes prefer to undergo
electrophilic addition reaction while arenes prefer electrophilic substitution reactions?
Explain.
View Answer play_arrow
-
question_answer60)
Alkynes on reduction with sodium
in liquid ammonia for mirons alkenes. Will the butene thus formed on reduction
of 2-butyne show the geometrical isomerism?
View Answer play_arrow
-
question_answer61)
Rotation around carbon-carbon
single bond of ethane is not completely free. Justify the statement.
View Answer play_arrow
-
question_answer62)
Draw Newman and Sawhorse
projections for the eclipsed and staggered conformations of ethane. Which of
these conformations is more stable and why?
View Answer play_arrow
-
question_answer63)
The intermediate carbocation
formed in the reactions of \[HI,\text{ }HBr\]and \[HCl\] with propene is the
same and the bond energy of \[HCl,HBr\]and \[HI\] is\[430.5\text{
}kJmo{{l}^{-1}}\], \[363.7\text{ }kJmo{{l}^{-1}}\]and\[296.8\text{
}kJmo{{l}^{-1}}\]respectively. What will be the order of reactivity of these
halogen acids?
View Answer play_arrow
-
question_answer64)
What will be the product
obtained as a result of the following reaction and why?
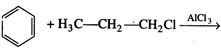
View Answer play_arrow
-
question_answer65)
How will you convert benzene
into :
(i) \[p-nitrobromobenzene\]
(ii) \[m-nitrobromobenzene?\]
View Answer play_arrow
-
question_answer66)
Arrange the following set of
compounds in the order of their decreasing relative reactivity with an
electrophile.
Give reason.
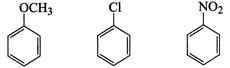
View Answer play_arrow
-
question_answer67)
Despite their \[-I\] effect, halogens
are \[o-\]and \[p\]-directing in haloarenes. Explain.
View Answer play_arrow
-
question_answer68)
Why does presence of a nitro
group make the benzene ring less reactive in comparison to the unsubstituted
benzene ring? Explain.
View Answer play_arrow
-
question_answer69)
Suggest a route for the
preparation of nitrobenzene starting from acetylene.
View Answer play_arrow
-
question_answer70)
Predict the major product(s) of
the following reactions and explain their formation.
\[{{H}_{3}}C-CH=C{{H}_{2}}\xrightarrow[HBr]{{{(Ph-COO)}_{2}}}\]
\[{{H}_{3}}C-CH=C{{H}_{2}}\xrightarrow{HBr}\]
View Answer play_arrow
-
question_answer71)
Nucleophiles and electrophiles
are reaction intermediate shaving electron rich and electron deficient centres respectively.
Hence, they tend to attack electron deficient and electron rich centres
respectively. Classify the following species as electrophiles and nucleophiles.
(i) \[{{H}_{3}}C{{O}^{-}}\] (ii)\[{{H}_{3}}C-\overset{\overset{\text{O}}{\mathop{||}}\,}{\mathop{C}}\,-{{O}^{-}}\]
(iii) \[\overset{\centerdot }{\mathop{Cl}}\,\] (iv)
\[C{{l}_{2}}C_{\centerdot }^{\centerdot }\]
(v) \[{{({{H}_{3}}C)}_{3}}{{C}^{+}}\] (vi)
\[B{{r}^{-}}\] (vii) \[{{H}_{3}}COH\] (viii) \[R-NH-R\]
View Answer play_arrow
-
question_answer72)
The relative reactivity of \[1{}^\circ
,\text{ }2{}^\circ ,\text{ }3{}^\circ \]hydrogen's towards chlorination is 1:
3.8: 5. Calculate the percentages of all mono chlorinated products obtained
from 2-methylbutane.
View Answer play_arrow
-
question_answer73)
Write the structures and names
of products obtained in the reactions of sodium with a mixture of 1-iodo-2-methylpropane
and 2-iodopropane.
View Answer play_arrow
-
question_answer74)
Write hydrocarbon radicals that
can be formed as intermediates during mono chlorination of 2-methyl-propane?
Which of them is more stable? Give reasons.
View Answer play_arrow
-
question_answer75)
An alkane \[{{C}_{8}}{{H}_{18}}\]
is obtained as the only product on subjecting a primary alkyl halide to Wurtz
reaction. On monobromination this alkane yields a single isomer of a tertiary
bromide. Write the structure of alkane and the tertiary bromide.
View Answer play_arrow
-
question_answer76)
The ring systems having
following characteristics are aromatic.
(i) Planar ring containing
conjugated \[\pi \]-bonds.
(ii) Complete delocalization of
the \[\pi \]-electrons in ring system, i.e., each atom in the ring has
unhybridised p-orbital and
(iii) Presence of (4n + 2)\[\pi
\]-electrons in the ring where n is an integer (n = 0, 1, 2,......) (Huckel?s
rule).
Using this information classify
the following compounds as aromatic/non aromatic.
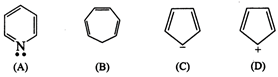
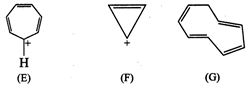
View Answer play_arrow
-
question_answer77)
Which of the following compounds
are aromatic according to Huckel?s rule?
(a) 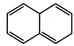 (b)
(b)
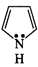 (c)
(c) 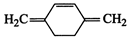 (d)
(d)
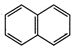 (e)
(e) 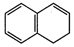 (f)
(f)
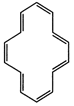
View Answer play_arrow
-
question_answer78)
Suggest a route to prepare ethyl
hydrogen sulphate\[(C{{H}_{3}}-C{{H}_{2}}-OS{{O}_{2}}-OH)\]starting from ethanol\[({{C}_{2}}{{H}_{5}}OH)\].
View Answer play_arrow
-
question_answer79)
Match the reagent from Column I which
on reaction with\[C{{H}_{3}}CH=C{{H}_{2}}\]gives some product given in Column
II as per the codes given below :
|
Column I
|
Column II
|
|
(i) \[{{O}_{3}}/Zn+{{H}_{2}}O\]
|
(a) Acetic acid and \[C{{O}_{2}}\]
|
|
(ii) \[KMn{{O}_{4}}/{{H}^{+}}\]
|
(b) Propan-1-ol
|
|
(iii) \[KMn{{O}_{4}}/O{{H}^{-}}\]
|
(c) Propan-2-ol
|
|
(iv) \[{{H}_{2}}O/{{H}^{+}}\]
|
(d) Acetaldehyde and formaldehyde
|
|
(v) \[{{B}_{2}}{{H}_{6}}/NaOH\]and \[{{H}_{2}}{{O}_{2}}\]
|
(e) Propane-1,2-diol
|
View Answer play_arrow
-
question_answer80)
Match the hydrocarbons in Column
I with the boiling points given in Column II.
|
Column I
|
Column II
|
|
(i) \[n-Pentane\]
|
(a)\[282.5K\]
|
|
(ii) \[iso-\text{Pentane}\]
|
(b) \[309K\]
|
|
(iii) \[neo-\text{Pentane}\]
|
(c) \[301K\]
|
View Answer play_arrow
-
question_answer81)
Match the following reactants in
Column I with the corresponding reaction products in Column II.
|
Column I
|
Column II
|
|
(i) Benzene \[+C{{l}_{2}}\xrightarrow{AlC{{l}_{3}}}\]
|
(a) Benzoic acid
|
|
(ii) Benzene \[+C{{H}_{3}}Cl\xrightarrow{AlC{{l}_{3}}}\]
|
(b) Methyl phenyl ketone
|
|
(iii) Benzene\[+C{{H}_{3}}COCl\xrightarrow{AlC{{l}_{3}}}\]
|
(c) Toluene
|
|
(iv) Toluene \[+\xrightarrow{KMn{{O}_{4}}/NaOH}\]
|
(d) Chlorobenzene
|
|
|
(e) Benzene hexachloride
|
View Answer play_arrow
-
question_answer82)
Match the reactions given in
Column I with the reaction types in Column II.
|
Column I
|
Column II
|
|
(i) \[{{H}_{2}}C=C{{H}_{2}}+{{H}_{2}}O\xrightarrow{{{H}^{+}}}\]\[C{{H}_{3}}C{{H}_{2}}OH\]
|
(a) Hydrogenation
|
|
(ii) \[{{H}_{2}}C=C{{H}_{2}}+{{H}_{2}}\xrightarrow{Pd}\]\[C{{H}_{3}}-C{{H}_{3}}\]
|
(b) \[\text{Halogenation}\]
|
|
(iii) \[{{H}_{2}}C=C{{H}_{2}}+C{{l}_{2}}\xrightarrow{{}}\]\[ClC{{H}_{2}}C{{H}_{2}}Cl\]
|
(c) \[\text{Polymerisation}\]
|
|
(iv) \[3HC\equiv CH\xrightarrow[\Delta ]{Cu\,tube}{{C}_{6}}{{H}_{6}}\]
|
(d) \[Hydration\]
|
|
|
(e) \[Condensation\]
|
View Answer play_arrow
-
question_answer83)
In the following questions a
statement of Assertion (A) followed by a statement of Reason (R) is given.
Choose the correct option out of the choices given below:
(a) If both A and R
are correct and R is the correct explanation of the A.
(b) If both A and R
are correct but R is not the correct explanation of the A.
(c) If A is correct but R
is incorrect.
(d) If A is incorrect but
R is correct.
(A) The compound
cyclooctene has the following structural formula :
 It is cyclic and has conjugated \[8\pi
\]-electron system but it is not an aromatic compound.
(R)\[(4n+2)\pi \]-
electrons rule does not hold good and ring is not planar.
It is cyclic and has conjugated \[8\pi
\]-electron system but it is not an aromatic compound.
(R)\[(4n+2)\pi \]-
electrons rule does not hold good and ring is not planar.
View Answer play_arrow
-
question_answer84)
(A) Toluene on
Friedel-Crafts methylation gives o- and p-xylene.
(R) \[C{{H}_{3}}\] ?group
bonded to benzene ring increases electron density at o- and p-positions.
View Answer play_arrow
-
question_answer85)
(A) Nitration of benzene
with nitric acid requires the use of concentrated sulphuric acid.
(R) The mixture of
concentrated sulphuric acid and concentrated nitric acid produces the
electrophile, \[NO_{2}^{+}\].
View Answer play_arrow
-
question_answer86)
(A) Among isomeric
pentanes, 2,2-dimethylpropane has lowest boiling point.
(R) Branching does not
affect the boiling point.
View Answer play_arrow
-
question_answer87)
An alkyl halide \[{{C}_{5}}{{H}_{11}}Br(A)\]Br
(A) reacts with ethanolic KOH to give an alkene ?B? which reacts with \[B{{r}_{2}}\]
to give a compound 'C?, which on dehydrobromination gives an alkyne 'D?. On
treatment with sodium metal in liquid ammonia one mole of 'D? gives one mole of
the sodium salt of 'D? and half a mole of hydrogen gas. Complete hydrogenation
of 'D' yields a straight chain alkane. Identify A, B, C and D. Give the
reactions involved.
View Answer play_arrow
-
question_answer88)
An unsaturated hydrocarbon 'A'
adds two molecules of \[{{H}_{2}}\]and on reductive ozonolysis gives butane-1,4-dial,
ethanol and propanone. Give the structure of 'A', write its IUPAC name and
explain the reactions involved.
View Answer play_arrow
-
question_answer89)
In the presence of peroxide addition of \[HBr\]to
propene takes place according to anti Markownikoffs rule but peroxide effect is
not seen in the case of \[HCl\] and \[HI\]. Explain.
View Answer play_arrow
 (f) \[C{{H}_{3}}{{(C{{H}_{2}})}_{4}}\underset{\underset{\text{C}{{\text{H}}_{\text{2}}}\text{-CH(C}{{\text{H}}_{\text{3}}}{{\text{)}}_{\text{2}}}}{\mathop{|}}\,}{\mathop{C}}\,H{{(C{{H}_{2}})}_{3}}C{{H}_{3}}\]
(g)\[{{H}_{3}}C-CH=CH-C{{H}_{2}}-CH=CH\] \[-\underset{\underset{{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}}}{\mathop{\text{
}\!\!|\!\!\text{ }}}\,}{\mathop{CH}}\,-C{{H}_{2}}-CH=C{{H}_{2}}\]
(f) \[C{{H}_{3}}{{(C{{H}_{2}})}_{4}}\underset{\underset{\text{C}{{\text{H}}_{\text{2}}}\text{-CH(C}{{\text{H}}_{\text{3}}}{{\text{)}}_{\text{2}}}}{\mathop{|}}\,}{\mathop{C}}\,H{{(C{{H}_{2}})}_{3}}C{{H}_{3}}\]
(g)\[{{H}_{3}}C-CH=CH-C{{H}_{2}}-CH=CH\] \[-\underset{\underset{{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{5}}}}{\mathop{\text{
}\!\!|\!\!\text{ }}}\,}{\mathop{CH}}\,-C{{H}_{2}}-CH=C{{H}_{2}}\]
 (b)
(b)  (c)
(c)  (d)
(d) 
![]()
