 \[\underset{(C)}{\mathop{C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-Br}}\,\]
The mixture consists of:
(a) A and B as major and C as
minor products (b) B as major, A and C as minor products
(c) B as minor, A and C as major
products (d) A and B as minor and C as major products
\[\underset{(C)}{\mathop{C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-Br}}\,\]
The mixture consists of:
(a) A and B as major and C as
minor products (b) B as major, A and C as minor products
(c) B as minor, A and C as major
products (d) A and B as minor and C as major products
Answer:
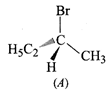
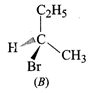
(a) \[C{{H}_{3}}-\underset{1-Butene}{\mathop{C{{H}_{2}}-CH}}\,=C{{H}_{2}}+HBr\xrightarrow{Mark.addition}\]\[C{{H}_{3}}-C{{H}_{2}}-\underset{I(major)}{\mathop{\underset{\begin{smallmatrix}
| \\
Br
\end{smallmatrix}}{\mathop{\overset{*}{\mathop{CH}}\,}}\,}}\,-C{{H}_{3}}+\underset{C(minor)}{\mathop{C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}Br}}\,\]
Since \[I\] contains a chiral
carbon, it exist in two enantiomers(A and B) which are mirror images of each
other.
You need to login to perform this action.
You will be redirected in
3 sec
