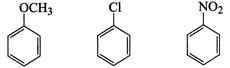
Answer:
Relative reactivity of the given
compounds are :
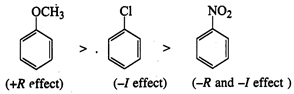 Greater is the \[(+R)\] effect
of the group, more is the reactivity of the corresponding compound.
Or
The reactivity of arenes towards
electrophiles depends upon their relative electron density in the benzene ring.
Higher the electron density (due to strong electron donating groups),more
reactive is the arene.
Greater is the \[(+R)\] effect
of the group, more is the reactivity of the corresponding compound.
Or
The reactivity of arenes towards
electrophiles depends upon their relative electron density in the benzene ring.
Higher the electron density (due to strong electron donating groups),more
reactive is the arene.
You need to login to perform this action.
You will be redirected in
3 sec
