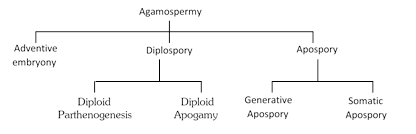
 (i) Adventive embryony : Formation of embryo directly from the diploid sporophytic cells (nucellus or integument) of ovule is called adventive embryony. Such embryos are formed without involving meiosis and sexual fusion, e.g., Citrus, Opuntia, etc. In Citrus, a seed may possess upto 40 embryos (one normal and rest adventive).
(ii) Diplospory : In this case, the archesporium differentiates but megaspore mother cell directly gives rise to an unreduced (i.e., without meiosis) embryo sac. It may produce two types of embryos :
(a) Diploid parthenogenesis : Embryo develops from unfertilized diploid egg.
(b) Diploid apogamy : Embryo develops from any diploid cell of embryo sac except egg.
(iii) Apospory : It is the formation of complete embryo sac from the sporophytic cell without meiosis so that the gametophyte remains diploid. Apospory may be of two types :
(a) Somatic apospory : Embryo sac is formed from somatic cell.
(b) Generative apospory : Embryo sac is formed from archesporium without meiosis.
(2) Vegetative propagation: Regeneration or Formation of a new individual from any vegetative part of the body is called vegetative reproduction or vegetative propagation. The lower plants reproduce vegetatively through budding, fission, fragmentation, gemmae, resting buds, spores, etc. Among flowering plants, every part of the body such as roots, stem, leaves and buds take part in vegetative propagation. It is very common mode of reproduction and it may be natural vegetative propagation or artificial vegetative propagation.
(i) Natural methods of vegetative propagation : In natural vegetative propagation, a portion gets deattached from the body of mother plant and it grows into a new individual plant under suitable conditions. Different plant parts are variously modified for vegetative propagation. Some of these are given below.
(a) Vegetative propagation by stems: The modified stems like bulbs, runners, rhizomes, corms, tubers, offsets, etc., help the plant to multiply under favourable conditions.
(i) Adventive embryony : Formation of embryo directly from the diploid sporophytic cells (nucellus or integument) of ovule is called adventive embryony. Such embryos are formed without involving meiosis and sexual fusion, e.g., Citrus, Opuntia, etc. In Citrus, a seed may possess upto 40 embryos (one normal and rest adventive).
(ii) Diplospory : In this case, the archesporium differentiates but megaspore mother cell directly gives rise to an unreduced (i.e., without meiosis) embryo sac. It may produce two types of embryos :
(a) Diploid parthenogenesis : Embryo develops from unfertilized diploid egg.
(b) Diploid apogamy : Embryo develops from any diploid cell of embryo sac except egg.
(iii) Apospory : It is the formation of complete embryo sac from the sporophytic cell without meiosis so that the gametophyte remains diploid. Apospory may be of two types :
(a) Somatic apospory : Embryo sac is formed from somatic cell.
(b) Generative apospory : Embryo sac is formed from archesporium without meiosis.
(2) Vegetative propagation: Regeneration or Formation of a new individual from any vegetative part of the body is called vegetative reproduction or vegetative propagation. The lower plants reproduce vegetatively through budding, fission, fragmentation, gemmae, resting buds, spores, etc. Among flowering plants, every part of the body such as roots, stem, leaves and buds take part in vegetative propagation. It is very common mode of reproduction and it may be natural vegetative propagation or artificial vegetative propagation.
(i) Natural methods of vegetative propagation : In natural vegetative propagation, a portion gets deattached from the body of mother plant and it grows into a new individual plant under suitable conditions. Different plant parts are variously modified for vegetative propagation. Some of these are given below.
(a) Vegetative propagation by stems: The modified stems like bulbs, runners, rhizomes, corms, tubers, offsets, etc., help the plant to multiply under favourable conditions.
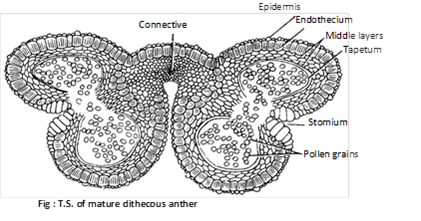 (1) Structure of anther: The fertile portion of stamens is called anther. Each anther is usually made up of two lobes connected by a connective. In turn each anther lobe contains two pollen chambers placed longitudinally. Each pollen chamber represents a microsporangium and is filled with a large number of pollen grains or microspores.
A typical anther consist of four microsporangia (tetrasporangiate) and such anthers is called dithecous e.g. mostly plants. In members of Malvaceae anthers are reniform or kidney shaped and consist of two microspoangia (bisporangiate), such anthers is called monothecous. In the smallest parasitic angiosperm, Arceuthobium minutissimum, anthers consist of only one microsporangium (monosporangiate).
The pollen sacs are surrounded by following 4 layers :
(i) Epidermis : This is the outermost single layered and protective. In Arceuthobium, cells of epidermis develops a fibrous thickening and the epidermis is designated as exothecium.
(ii) Endothecium : Inner to epidermis, there is a single layer of radially elongated cells. Cells of endothecium develop fibrous thickening (made up of cellulose with a little pectin and lignin) which help in the dehiscence of anther. In between these cells, a few cells without thickening are also present. These thick walled cells collectively form the stomium.
(iii) Middle layer : Three to four layers of thin walled cells situated just below the endothecium are known as middle layers. Cells of this layer are ephemeral and degenerate to provide nourishment to growing microspore mother cells.
(iv) Tapetum : This is the innermost layer of the wall. The cells are multinucleate (undergo endopolyploidy) and polyploid. Tapetal cells are nutritive.
In these cells the Ubisch bodies are present which help in the ornamentation of microspore walls. A compound sporopollenin is secreted in the exine of microspore wall. According to Periasamy and Swamy (1966), developmentally the tapetum has dual nature.
The tapetum is of two types :
(a) Amoeboid or Periplasmodial tapetum : In young condition cell wall of tapetal cells breaks, so protoplast of these cells become free between microspore mother cell and form mass of tapetal periplasmodium. e.g. Alisma, Typha, Tradescantia.
(b) Secretory or Glandular tapetum : This is the most common type of tapetum which remains insitu as such throughout. The tapetal cells secretes nourishment that passes into sporogenous cells. This tapetum attains its maximum development at the stage of pollen tetrads and then degenerates.
(2) Development of anther and formation of microspores (Pollen grains): The young anther consists of homogenous mass of paranchymatous cells surrounded by epidermis. It soon becomes four lobed. In each of the four lobes, some of the hypodermal cells begin to act as archesporial initials. Each archesporial initial divides into an outer more...
(1) Structure of anther: The fertile portion of stamens is called anther. Each anther is usually made up of two lobes connected by a connective. In turn each anther lobe contains two pollen chambers placed longitudinally. Each pollen chamber represents a microsporangium and is filled with a large number of pollen grains or microspores.
A typical anther consist of four microsporangia (tetrasporangiate) and such anthers is called dithecous e.g. mostly plants. In members of Malvaceae anthers are reniform or kidney shaped and consist of two microspoangia (bisporangiate), such anthers is called monothecous. In the smallest parasitic angiosperm, Arceuthobium minutissimum, anthers consist of only one microsporangium (monosporangiate).
The pollen sacs are surrounded by following 4 layers :
(i) Epidermis : This is the outermost single layered and protective. In Arceuthobium, cells of epidermis develops a fibrous thickening and the epidermis is designated as exothecium.
(ii) Endothecium : Inner to epidermis, there is a single layer of radially elongated cells. Cells of endothecium develop fibrous thickening (made up of cellulose with a little pectin and lignin) which help in the dehiscence of anther. In between these cells, a few cells without thickening are also present. These thick walled cells collectively form the stomium.
(iii) Middle layer : Three to four layers of thin walled cells situated just below the endothecium are known as middle layers. Cells of this layer are ephemeral and degenerate to provide nourishment to growing microspore mother cells.
(iv) Tapetum : This is the innermost layer of the wall. The cells are multinucleate (undergo endopolyploidy) and polyploid. Tapetal cells are nutritive.
In these cells the Ubisch bodies are present which help in the ornamentation of microspore walls. A compound sporopollenin is secreted in the exine of microspore wall. According to Periasamy and Swamy (1966), developmentally the tapetum has dual nature.
The tapetum is of two types :
(a) Amoeboid or Periplasmodial tapetum : In young condition cell wall of tapetal cells breaks, so protoplast of these cells become free between microspore mother cell and form mass of tapetal periplasmodium. e.g. Alisma, Typha, Tradescantia.
(b) Secretory or Glandular tapetum : This is the most common type of tapetum which remains insitu as such throughout. The tapetal cells secretes nourishment that passes into sporogenous cells. This tapetum attains its maximum development at the stage of pollen tetrads and then degenerates.
(2) Development of anther and formation of microspores (Pollen grains): The young anther consists of homogenous mass of paranchymatous cells surrounded by epidermis. It soon becomes four lobed. In each of the four lobes, some of the hypodermal cells begin to act as archesporial initials. Each archesporial initial divides into an outer more... 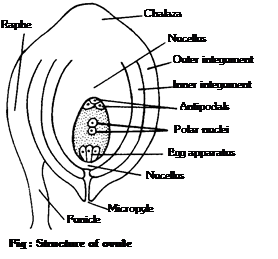 (1) Structure of ovule: Ovule is considered to be an integumented megasporangium. The ovule consists of the stalk and the body. The stalk is called funicle. One end of the funicle is attached to placenta and the other end to the body of the ovule. The point of attachment of funicle with the body is called hilum. Sometimes funicle gets fused with the body of the ovule one side and forms a ridge known as raphe. The body of the ovule shows two ends: the basal end, often called the chalazal end and the upper end is called micropylar end. The main body of the ovule is covered with one or two envelopes called integuments. These leave an opening at the top of the ovule called micropyle. The integuments enclose a large parenchymatous tissue known as nucellus.
The residual part of nucellus in the mature seed is called perisperm. In the centre of the nucellus is situated a female gametophyte known as embryo sac.
Following are the conditions seen in ovule in relation to integuments:
(i) Unitegmic : Ovule with a single integument, e.g., sympetalous or gamopetalous dicotyledons.
(ii) Bitegmic: Ovule with two integuments as in polypetalous (Archichlamydeae) dicotyledons and monocotyledons.
(iii) Aril : This is a collar-like outgrowth from the base of the ovule and forms third integument. Aril is found in litchi, nutmeg, etc.
(iv) Caruncle : It is formed as an outgrowth of the outer integument in the micropylar region. Caruncle is common in the ovules of Euphorbiaceae. e.g., Castor (Ricinus).
(v) Ategmic : In some parasites like Loranthus, Viscum, Santalum etc., there is no integument. Such an ovule is called ategmic.
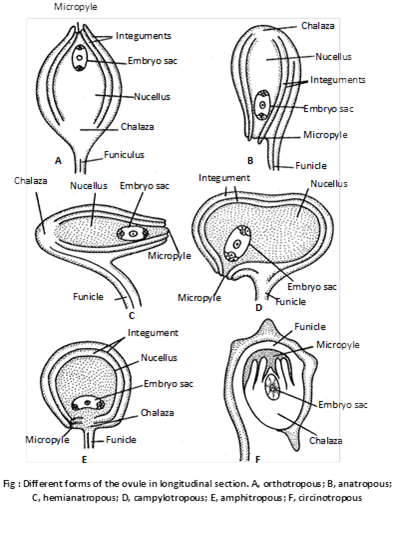
(2) Kinds of ovules: Depending upon the shape and orientation, the ovules of angiosperms are classified into following types :
(i) Orthotropous or Atropus : The micropyle, chalaza and funicle are in straight line. This is most primitive type of ovules. e.g. Betel, Piper, Polygonum.
(ii) Anatropous : The body of the ovule is completely inverted (turn at 180o angle ) so that micropyle and hilum come to lie very close to each other. e.g. 82% of angiosperm families.
(iii) Hemianatropous : Ovule turns at 90o angle upon the funicle or body of ovule is at right angle to the funicle e.g. Ranunculus.
(iv) Campylotropous : Ovule is circled more or less at right angle to funicle. Micropylar end is bent down slightly. e.g. in members of Leguminaceae and Cruciferae.
(1) Structure of ovule: Ovule is considered to be an integumented megasporangium. The ovule consists of the stalk and the body. The stalk is called funicle. One end of the funicle is attached to placenta and the other end to the body of the ovule. The point of attachment of funicle with the body is called hilum. Sometimes funicle gets fused with the body of the ovule one side and forms a ridge known as raphe. The body of the ovule shows two ends: the basal end, often called the chalazal end and the upper end is called micropylar end. The main body of the ovule is covered with one or two envelopes called integuments. These leave an opening at the top of the ovule called micropyle. The integuments enclose a large parenchymatous tissue known as nucellus.
The residual part of nucellus in the mature seed is called perisperm. In the centre of the nucellus is situated a female gametophyte known as embryo sac.
Following are the conditions seen in ovule in relation to integuments:
(i) Unitegmic : Ovule with a single integument, e.g., sympetalous or gamopetalous dicotyledons.
(ii) Bitegmic: Ovule with two integuments as in polypetalous (Archichlamydeae) dicotyledons and monocotyledons.
(iii) Aril : This is a collar-like outgrowth from the base of the ovule and forms third integument. Aril is found in litchi, nutmeg, etc.
(iv) Caruncle : It is formed as an outgrowth of the outer integument in the micropylar region. Caruncle is common in the ovules of Euphorbiaceae. e.g., Castor (Ricinus).
(v) Ategmic : In some parasites like Loranthus, Viscum, Santalum etc., there is no integument. Such an ovule is called ategmic.
(2) Kinds of ovules: Depending upon the shape and orientation, the ovules of angiosperms are classified into following types :
(i) Orthotropous or Atropus : The micropyle, chalaza and funicle are in straight line. This is most primitive type of ovules. e.g. Betel, Piper, Polygonum.
(ii) Anatropous : The body of the ovule is completely inverted (turn at 180o angle ) so that micropyle and hilum come to lie very close to each other. e.g. 82% of angiosperm families.
(iii) Hemianatropous : Ovule turns at 90o angle upon the funicle or body of ovule is at right angle to the funicle e.g. Ranunculus.
(iv) Campylotropous : Ovule is circled more or less at right angle to funicle. Micropylar end is bent down slightly. e.g. in members of Leguminaceae and Cruciferae.
 more...
more... 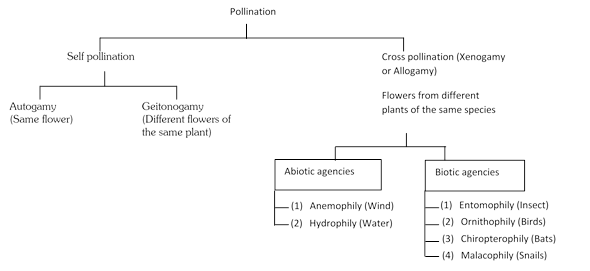 (1) Self-pollination: This process involves the transfer of pollen grains from the anthers to the stigma of the same flower or of another flower borne by the same plant. It is of two types:
(i) Autogamy : It is a kind of pollination in which the pollen from the anthers of a flower are transferred to the stigma of the same flower.
(ii) Geitonogamy : It is an kind of pollination in which the pollen from the anthers of one flower are transferred to the stigma of another flower borne on the same plant. It usually occurs in plants which show monoecious condition (unisexual, male and female flowers are borne on the same plant). Geitonogamy involves two flowers but these belong to the same parent plant.
Merits
(1) Self-pollination: This process involves the transfer of pollen grains from the anthers to the stigma of the same flower or of another flower borne by the same plant. It is of two types:
(i) Autogamy : It is a kind of pollination in which the pollen from the anthers of a flower are transferred to the stigma of the same flower.
(ii) Geitonogamy : It is an kind of pollination in which the pollen from the anthers of one flower are transferred to the stigma of another flower borne on the same plant. It usually occurs in plants which show monoecious condition (unisexual, male and female flowers are borne on the same plant). Geitonogamy involves two flowers but these belong to the same parent plant.
Merits
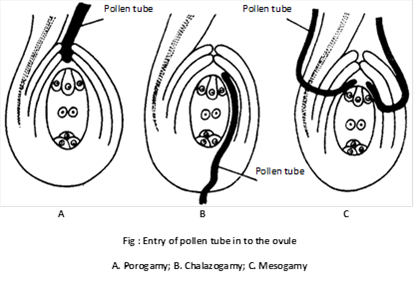 (2) Entry of pollen tube into ovule: After reaching ovary, the pollen tube enters the ovule. Pollen tube may enter the ovule by any one of the following routes:
(i) Porogamy : When the pollen tube enters the ovule through micropyle, it is called porogamy. It is the most common type. e.g. Lily.
(ii) Chalazogamy : The entry of pollen tube into the ovule from chalazal region is known as chalazogamy. Chalazogamy is less common. e.g. Casuarina, Juglans, Betula, etc. It was first observed by Treub (1981) in Casuarina.
(iii) Mesogamy : The pollen tube enters the ovule through its middle part i.e. through integument (e.g. Cucurbita, Populus) or through funicle (e.g. Pistacia).
(3) Entry of pollen tube into embryo sac: The pollen tube enters the embryo sac only from the micropylar end irrespective of its mode of entry into the ovule. The pollen tube either passes between a synergid and the egg cell or enters into one of the synergids through filiform apparatus. The synergids direct the growth of pollen tube by secreting some chemical substances (chemotropic secretion). The tip of pollen tube enters into one synergid. The penetrated synergid starts degenerating. After penetration, the tip of pollen tube enlarge and ruptures releasing most of its contents including the two male gametes and the vegetative nucleus into the synergid.
(4) Double fertilization: The nuclei of both the male gametes are released in the embryo sac. One male gamete fuses with the egg to form the diploid zygote. The process is called syngamy or generative fertilization. This syngamy was discovered by Strasburger (1884). The diploid zygote finally develops into embryo. The other male gamete fuses with the two polar nuclei (or secondary nucleus) to form the triploid primary endosperm nucleus. The process is called triple fusion or vegetative fertilization. These two acts of fertilizations constitute the process of double fertilization. The process was discovered more...
(2) Entry of pollen tube into ovule: After reaching ovary, the pollen tube enters the ovule. Pollen tube may enter the ovule by any one of the following routes:
(i) Porogamy : When the pollen tube enters the ovule through micropyle, it is called porogamy. It is the most common type. e.g. Lily.
(ii) Chalazogamy : The entry of pollen tube into the ovule from chalazal region is known as chalazogamy. Chalazogamy is less common. e.g. Casuarina, Juglans, Betula, etc. It was first observed by Treub (1981) in Casuarina.
(iii) Mesogamy : The pollen tube enters the ovule through its middle part i.e. through integument (e.g. Cucurbita, Populus) or through funicle (e.g. Pistacia).
(3) Entry of pollen tube into embryo sac: The pollen tube enters the embryo sac only from the micropylar end irrespective of its mode of entry into the ovule. The pollen tube either passes between a synergid and the egg cell or enters into one of the synergids through filiform apparatus. The synergids direct the growth of pollen tube by secreting some chemical substances (chemotropic secretion). The tip of pollen tube enters into one synergid. The penetrated synergid starts degenerating. After penetration, the tip of pollen tube enlarge and ruptures releasing most of its contents including the two male gametes and the vegetative nucleus into the synergid.
(4) Double fertilization: The nuclei of both the male gametes are released in the embryo sac. One male gamete fuses with the egg to form the diploid zygote. The process is called syngamy or generative fertilization. This syngamy was discovered by Strasburger (1884). The diploid zygote finally develops into embryo. The other male gamete fuses with the two polar nuclei (or secondary nucleus) to form the triploid primary endosperm nucleus. The process is called triple fusion or vegetative fertilization. These two acts of fertilizations constitute the process of double fertilization. The process was discovered more... 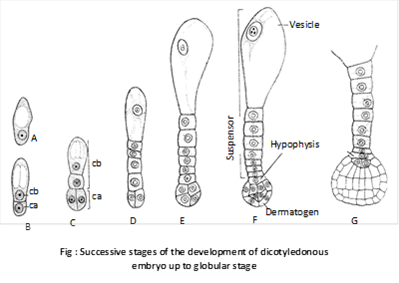
 The two apical cells first divide by longitudinal division (at right angle to first one) and then by transverse and periclinal division. So sixteen celled globular embryo is produced. Due to differentiation of cotyledons globular embryo becomes heart shaped.
Mature embryo in dicots consists of two lateral cotyledons, terminal plumule or stem tip and radicle or root tip.
(ii) In monocotyledons: The normal type of monocot embryo development has been studied in Sagittaria sagittaefolia. The early development of dicot and monocot embryos is similar upto globular stage. Later on differentiation starts. Suspensor is single celled and vascular. There is only one terminal cotyledon called scutellum (shield shaped). In grasses the second cotyledon is reduced called epiblast.
The two apical cells first divide by longitudinal division (at right angle to first one) and then by transverse and periclinal division. So sixteen celled globular embryo is produced. Due to differentiation of cotyledons globular embryo becomes heart shaped.
Mature embryo in dicots consists of two lateral cotyledons, terminal plumule or stem tip and radicle or root tip.
(ii) In monocotyledons: The normal type of monocot embryo development has been studied in Sagittaria sagittaefolia. The early development of dicot and monocot embryos is similar upto globular stage. Later on differentiation starts. Suspensor is single celled and vascular. There is only one terminal cotyledon called scutellum (shield shaped). In grasses the second cotyledon is reduced called epiblast.
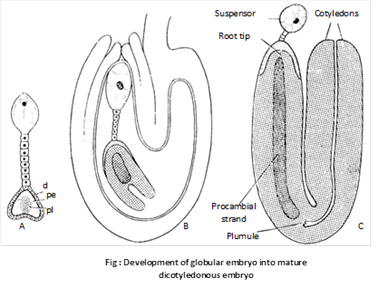
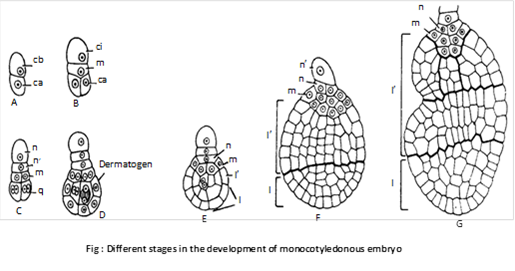 The basal cell (cb) divides by a transverse wall into two cells – ci and m. The cell ci divides once again to form n and n’ cells. Of these n’ is the outermost which develops into suspensor. The cell n forms parts of root cap the cell m contributes to the remaining part of root cap and a part of the radicle.
The terminal cell (ca) divides by two vertical walls, at right angles to one another. This results in the formation of a quadrant (q). Cells of the quadrant divide periclinally differentiating into the peripheral cells and the inner group of cells. The repeated divisions in both peripheral and central group of cells results in the formation of two regions –l and l’. Region l produces the lower part of cotyledon while upper part of cotyledon, hypocotyl and plumule are formed by l’ region.
(2) Polyembryony: Occurrence of more than two embryo in the seed is known as polyembryony. It was discovered by A.V. Leeuwenhock (1719) in Citrus. It may be :
(i) Cleavage polyembryony more...
The basal cell (cb) divides by a transverse wall into two cells – ci and m. The cell ci divides once again to form n and n’ cells. Of these n’ is the outermost which develops into suspensor. The cell n forms parts of root cap the cell m contributes to the remaining part of root cap and a part of the radicle.
The terminal cell (ca) divides by two vertical walls, at right angles to one another. This results in the formation of a quadrant (q). Cells of the quadrant divide periclinally differentiating into the peripheral cells and the inner group of cells. The repeated divisions in both peripheral and central group of cells results in the formation of two regions –l and l’. Region l produces the lower part of cotyledon while upper part of cotyledon, hypocotyl and plumule are formed by l’ region.
(2) Polyembryony: Occurrence of more than two embryo in the seed is known as polyembryony. It was discovered by A.V. Leeuwenhock (1719) in Citrus. It may be :
(i) Cleavage polyembryony more... 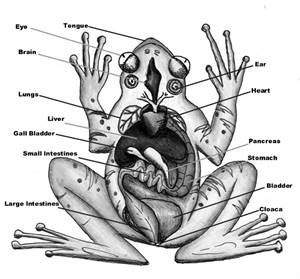 Morphology of Frogs
Though larvae have tails, adult frogs are tailless. An adult frog has a stout body which is differentiated into head and trunk. Other external features are a pair of nostrils, protruding eyes, a membranous tympanum (ear), slippery/warty moist skin and webbed limbs.
Frogs
Frogs generally have a slippery moist and highly permeable skin through which they absorb water and respire. Thus, the moist skin acts as a respiratory organ in frogs. Also, the skin is glandular in nature which produces mucus and toxic substances to warn them of their predators. The color of the skin can vary from brown and green to vivid colors as per secretions.
The locomotion of frogs takes place with the help of their forelimbs and hind limbs. Frogs are unisexual i.e., they show sexual dimorphism. A male frog is distinguished from a female frog by the presence of vocal sacs and a copulatory pad on forelimbs. A female frog lacks these body features.
Anatomy
The body plan of frogs consists of well-developed structures which help them in their physiological activities. The body cavity accommodates all the organ systems such as digestive, respiratory, circulatory, excretory, nervous and reproductive systems whose functions are almost similar to human body systems.
Digestive system:
The alimentary canal together with the accessory organs makes up the digestive system of the frog. Since frogs are carnivorous they have short intestine. The alimentary canal begins at the mouth (buccal or oral cavity), passes through the pharynx, esophagus or food pipe, stomach, small intestines, large intestines, rectum and finally ending at the cloaca. The food particles get digested gradually as they travel through various compartments of the alimentary canal.
more...
Morphology of Frogs
Though larvae have tails, adult frogs are tailless. An adult frog has a stout body which is differentiated into head and trunk. Other external features are a pair of nostrils, protruding eyes, a membranous tympanum (ear), slippery/warty moist skin and webbed limbs.
Frogs
Frogs generally have a slippery moist and highly permeable skin through which they absorb water and respire. Thus, the moist skin acts as a respiratory organ in frogs. Also, the skin is glandular in nature which produces mucus and toxic substances to warn them of their predators. The color of the skin can vary from brown and green to vivid colors as per secretions.
The locomotion of frogs takes place with the help of their forelimbs and hind limbs. Frogs are unisexual i.e., they show sexual dimorphism. A male frog is distinguished from a female frog by the presence of vocal sacs and a copulatory pad on forelimbs. A female frog lacks these body features.
Anatomy
The body plan of frogs consists of well-developed structures which help them in their physiological activities. The body cavity accommodates all the organ systems such as digestive, respiratory, circulatory, excretory, nervous and reproductive systems whose functions are almost similar to human body systems.
Digestive system:
The alimentary canal together with the accessory organs makes up the digestive system of the frog. Since frogs are carnivorous they have short intestine. The alimentary canal begins at the mouth (buccal or oral cavity), passes through the pharynx, esophagus or food pipe, stomach, small intestines, large intestines, rectum and finally ending at the cloaca. The food particles get digested gradually as they travel through various compartments of the alimentary canal.
more... You need to login to perform this action.
You will be redirected in
3 sec
