The velocity v of a particle at time t is given by \[v=at+\frac{b}{t+c},\]where a, b and c are constants. The dimensions of a, b and c are respectively:
A)
\[{{[LT]}^{2}},[L]\]and [T]
done
clear
B)
\[[{{L}^{2}}],[T]\]and\[[L{{T}^{2}}]\]
done
clear
C)
\[[L{{T}^{2}}],[LT]\] and [L]
done
clear
D)
[L], [LT] and\[[{{T}^{2}}]\]
done
clear
View Answer play_arrow
If force (F), velocity (v) and time (T) are taken as fundamental units, then the dimensions of mass are
A)
\[[Fv{{T}^{-1}}]\]
done
clear
B)
\[[Fv{{T}^{-2}}]\]
done
clear
C)
\[[F{{v}^{-1}}{{T}^{-1}}]\]
done
clear
D)
\[[F{{v}^{-1}}T]\]
done
clear
View Answer play_arrow
Two balls are dropped from heights h and 2h respectively from the earth surface. The ratio of time of these balls to reach the earth is
A)
\[1:\sqrt{2}\]
done
clear
B)
\[\sqrt{2}:1\]
done
clear
C)
2 : 1
done
clear
D)
1 : 4
done
clear
View Answer play_arrow
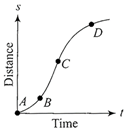
A particle shows distance-time curve as given in this figure. The maximum instantaneous velocity of the particle is around the point
A)
B
done
clear
B)
C
done
clear
C)
D
done
clear
D)
A
done
clear
View Answer play_arrow
The displacement \['x'\] (in meter) of a particle of mass \['m'\] (in kg) moving in one dimension under the action of a force is released to time \['t'\] (in sec) by \[t=\sqrt{x}+3.\] The displacement of the particle when its velocity is zero will be
A)
2 m
done
clear
B)
4 m
done
clear
C)
zero
done
clear
D)
6 m
done
clear
View Answer play_arrow
A projectile is fired at an angle of \[{{45}^{o}}\]with the horizontal. Elevation angle of the projectile at its highest point as seen from the point of projection is
A)
\[{{60}^{o}}\]
done
clear
B)
\[{{\tan }^{-1}}\left( \frac{1}{2} \right)\]
done
clear
C)
\[{{\tan }^{-1}}\left( \frac{\sqrt{3}}{2} \right)\]
done
clear
D)
\[{{45}^{o}}\]
done
clear
View Answer play_arrow
The velocity of a projectile at the initial point A is \[(2\hat{i}+3\hat{j})\,m/s.\] Its velocity (in m/s) at point B is
A)
\[-2\hat{i}-3\hat{j}\]
done
clear
B)
\[-2\hat{i}+3\hat{j}\]
done
clear
C)
\[2\hat{i}-3\hat{j}\]
done
clear
D)
\[2\hat{i}+3\hat{j}\]
done
clear
View Answer play_arrow
A block B is pushed momentarily along a horizontal surface with an initial velocity v. If \[\mu \]is the coefficient of sliding friction between B and the surface, block B will come to rest after a time:
A)
\[\frac{v}{g\mu }\]
done
clear
B)
\[\frac{g\mu }{v}\]
done
clear
C)
\[\frac{g}{v}\]
done
clear
D)
\[\frac{v}{g}\]
done
clear
View Answer play_arrow
A car of mass 1000 kg negotiates a banked curve of radius 90 m on a frictionless road. If the banking angle is\[45{}^\circ ,\] the speed of the car is:
A)
\[~10\,m{{s}^{-1}}\]
done
clear
B)
\[~20\,m{{s}^{-1}}\]
done
clear
C)
\[~30\,m{{s}^{-1}}\]
done
clear
D)
\[~5\,m{{s}^{-1}}\]
done
clear
View Answer play_arrow
Two stones of masses m and 1m are whirled in horizontal circles, the heavier one in a radius \[\frac{r}{2}\] and the lighter one in radius r. The tangential speed of lighter stone is n times that of the value of heavier stone when they experience same centripetal forces. The value of n is:
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
A force \[\vec{F}=(5\hat{i}+4\hat{j})N\]acts on a body and produces a displacement \[\vec{F}=(6\hat{i}-5\hat{j}+3\hat{k})m.\] The work done will be
A)
10 J
done
clear
B)
20 J
done
clear
C)
30 J
done
clear
D)
40 J
done
clear
View Answer play_arrow
A ball moving with velocity \[2\,m{{s}^{-1}}\]collides head on with another stationary ball of double the mass. If the coefficient of restitution is 0.5, then their velocities (in\[\,m{{s}^{-1}}\]) after collision will be
A)
0, 1
done
clear
B)
1, 1
done
clear
C)
1, 0.5
done
clear
D)
0, 2
done
clear
View Answer play_arrow
Two similar springs P and Q have spring constants \[{{K}_{P}}\]and \[{{K}_{Q}}\]such that\[{{K}_{P}}>{{K}_{Q}}\]. They are stretched first by the same amount (case a), then by the same force (case b). The work done by the springs \[{{W}_{P}}\]and \[{{W}_{Q}}\]are related as, in case and case [b], respectively.
A)
\[{{W}_{p}}={{W}_{Q}};{{W}_{p}}>{{W}_{Q}}\]
done
clear
B)
\[{{W}_{p}}={{W}_{Q}};{{W}_{p}}={{W}_{Q}}\]
done
clear
C)
\[{{W}_{p}}={{W}_{Q}};{{W}_{Q}}>{{W}_{p}}\]
done
clear
D)
\[{{W}_{p}}<{{W}_{Q}};{{W}_{Q}}<{{W}_{p}}\]
done
clear
View Answer play_arrow
A circular disk of moment of inertia \[{{I}_{t}}\] is rotating in a horizontal plane about its symmetry axis, with a constant angular speed\[{{\omega }_{i}}\]. Another disk of moment of inertia \[{{I}_{b}}\] is dropped coaxially onto the rotating disk, initially the second disk has zero angular speed. Eventually both the disks rotate with a constant angular speed \[{{\omega }_{f}}.\]The energy lost by the initially rotating disc due to friction is:
A)
\[\frac{1}{2}\frac{I_{b}^{2}}{({{I}_{t}}+{{I}_{b}})}\omega _{i}^{2}\]
done
clear
B)
\[\frac{1}{2}\frac{I_{t}^{2}}{({{I}_{t}}+{{I}_{b}})}\omega _{i}^{2}\]
done
clear
C)
\[\frac{1}{2}\frac{{{I}_{b}}-{{I}_{t}}}{({{I}_{t}}+{{I}_{b}})}\omega _{i}^{2}\]
done
clear
D)
\[\frac{1}{2}\frac{{{I}_{b}}{{I}_{t}}}{({{I}_{t}}+{{I}_{b}})}\omega _{i}^{2}\]
done
clear
View Answer play_arrow
A body of mass m is placed on the earth's surface. It is taken from the earth's surface to a height \[h=3R.\]The change in gravitational potential energy of the body is
A)
\[\frac{2}{3}mgR\]
done
clear
B)
\[\frac{3}{4}mgR\]
done
clear
C)
\[\frac{mgR}{2}\]
done
clear
D)
\[\frac{mgR}{4}\]
done
clear
View Answer play_arrow
The cylindrical tube of a spray pump has radius R, one end of which has n fine holes, each of radius r. If the speed of the liquid in the tube is V, the speed of the ejection of the liquid through the holes is:
A)
\[\frac{{{V}^{2}}R}{nr}\]
done
clear
B)
\[\frac{V{{R}^{2}}}{{{n}^{2}}{{r}^{2}}}\]
done
clear
C)
\[\frac{V{{R}^{2}}}{n{{r}^{2}}}\]
done
clear
D)
\[\frac{V{{R}^{2}}}{{{n}^{3}}{{r}^{2}}}\]
done
clear
View Answer play_arrow
Mercury boils at\[367{}^\circ C.\]However, mercury thermometers are made such that they can measure temperature up to\[500{}^\circ C.\] This is done by
A)
maintaining vacuum above mercury column in the stem of the thermometer
done
clear
B)
filling nitrogen gas at high pressure above the mercury column
done
clear
C)
filling nitrogen gas at low pressure above the mercury level
done
clear
D)
filling oxygen gas at high pressure above the mercury column
done
clear
View Answer play_arrow
If 300 ml of a gas at\[27{}^\circ \]is cooled to\[7{}^\circ \]at constant pressure, then its final volume will be
A)
540 ml
done
clear
B)
350 ml
done
clear
C)
280 ml
done
clear
D)
135 ml
done
clear
View Answer play_arrow
The particle executing simple harmonic motion has a kinetic energy \[{{K}_{0}}{{\cos }^{2}}\omega t.\]The maximum values of the potential energy and the total energy are respectively:
A)
0 and\[2{{K}_{0}}\]
done
clear
B)
\[\frac{{{K}_{0}}}{2}\]and \[{{K}_{0}}\]
done
clear
C)
\[{{K}_{0}}\]and \[2{{K}_{0}}\]
done
clear
D)
\[{{K}_{0}}\]and \[{{K}_{0}}\]
done
clear
View Answer play_arrow
A wave travelling in positive X-direction with A = 0.2 m has a velocity of 360 m/sec. If \[\lambda =60\,m,\] then correct expression for the wave is
A)
\[y=0.2\sin \left[ 2\pi \left( 6t+\frac{x}{60} \right) \right]\]
done
clear
B)
\[y=0.2\sin \left[ \pi \left( 6t+\frac{x}{60} \right) \right]\]
done
clear
C)
\[y=0.2\sin \left[ 2\pi \left( 6t-\frac{x}{60} \right) \right]\]
done
clear
D)
\[y=0.2\sin \left[ \pi \left( 6t-\frac{x}{60} \right) \right]\]
done
clear
View Answer play_arrow
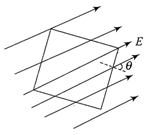
A square surface of side L metre in the plane of the paper is placed in a uniform electric field E (volt/m) acting along the same place at an angle \[\theta \] with the horizontal side of the square as shown in the figure. The electric flux linked to the surface in unit of V-m is
A)
\[E{{L}^{2}}\]
done
clear
B)
\[E{{L}^{2}}\cos \theta \]
done
clear
C)
\[E{{L}^{2}}\sin \theta \]
done
clear
D)
0
done
clear
View Answer play_arrow
A capacitor of capacity\[{{C}_{1}}\] is charged up to V volt and then connected to an uncharged capacitor of capacity\[{{C}_{2}}.\]Then final potential difference across each will be
A)
\[\frac{{{C}_{2}}V}{{{C}_{1}}+{{C}_{2}}}\]
done
clear
B)
\[\left( 1+\frac{{{C}_{2}}}{{{C}_{1}}} \right)V\]
done
clear
C)
\[\frac{{{C}_{1}}V}{{{C}_{1}}+{{C}_{2}}}\]
done
clear
D)
\[\left( 1-\frac{{{C}_{2}}}{{{C}_{1}}} \right)V\]
done
clear
View Answer play_arrow
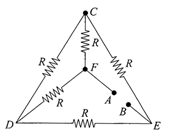
Five equal resistances each of resistance R are connected as shown in the figure. A battery of V volts is connected between A and B. The current flowing in AFCEB will be
A)
\[\frac{3V}{R}\]
done
clear
B)
\[\frac{V}{R}\]
done
clear
C)
\[\frac{V}{2R}\]
done
clear
D)
\[\frac{2V}{R}\]
done
clear
View Answer play_arrow
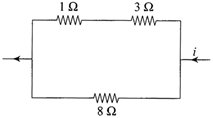
Power dissipated across the \[8\,\Omega \] resistor in the circuit shown here is 2 W. The power dissipated in watt units across the \[3\,\Omega \] resistor is
A)
2.0
done
clear
B)
1.0
done
clear
C)
0.5
done
clear
D)
3.0
done
clear
View Answer play_arrow
When a charged particle moving with velocity \[\vec{V}\]is subjected to a magnetic field of induction \[\vec{B}\]the force on it is non -zero. This implies that:
A)
angle between \[\vec{V}\]and\[\vec{B}\] is necessarily\[90{}^\circ \]
done
clear
B)
angle between\[\vec{V}\]and\[\vec{B}\]can have any value other than \[90{}^\circ \]
done
clear
C)
angle between \[\vec{V}\]and \[\vec{B}\]can have any value other than zero and\[180{}^\circ \]
done
clear
D)
angle between \[\vec{V}\]and\[\vec{B}\]is either zero or \[180{}^\circ \]
done
clear
View Answer play_arrow
A coil in the shape of an equilateral triangle of side I is suspended between the pole pieces of a permanent magnet such that\[\vec{B}\]is in plane of the coil. If due to a current i in the triangle a torque \[\tau \]acts on it, then the side I of the triangle is
A)
\[\frac{2}{\sqrt{3}}{{\left( \frac{\tau }{Bi} \right)}^{1/2}}\]
done
clear
B)
\[\frac{2}{\sqrt{3}}\left( \frac{\tau }{Bi} \right)\]
done
clear
C)
\[2{{\left( \frac{\tau }{\sqrt{3}Bi} \right)}^{1/2}}\]
done
clear
D)
\[\frac{1}{\sqrt{3}}\frac{\tau }{Bi}\]
done
clear
View Answer play_arrow
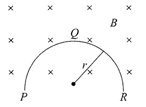
A thin semicircular conducting ring (PQR) of radius r is falling with its plane vertical in a horizontal magnetic field B, as shown in the figure. The potential difference developed across the ring when its speed is v is
A)
Zero
done
clear
B)
\[Bv\pi {{r}^{2}}/2\]and P is at higher potential
done
clear
C)
\[\pi rBv\]and R is at higher potential
done
clear
D)
2rBv and R is at higher potential
done
clear
View Answer play_arrow
A transformer is used to light a 100 W and 110 V lamp from a 220 V mains. If the main current is 0.5 A, the Efficiency of the transformer is approximately:
A)
30%
done
clear
B)
50%
done
clear
C)
90%
done
clear
D)
10%
done
clear
View Answer play_arrow
The electric field part of an electromagnetic wave in a medium is represented by\[{{E}_{x}}=0;\]\[{{E}_{y}}=2.5\,\frac{N}{C}\cos \left[ \left( 2\pi \times {{10}^{6}}\frac{rad}{m} \right)t-\left( \pi \times {{10}^{-2}}\frac{rad}{s} \right)x \right]\]\[{{E}_{z}}=0\]. The wave is
A)
moving along y-direction with frequency \[2\pi \times {{10}^{6}}Hz\] and wavelength 200 m.
done
clear
B)
moving along\[x-\]direction with frequency \[{{10}^{6}}Hz\]and wavelength 100 m
done
clear
C)
moving along \[x-\]direction with frequency \[{{10}^{6}}Hz\]and wavelength 200 m
done
clear
D)
moving along\[x-\]direction with frequency \[{{10}^{6}}Hz\]and wavelength 200 m
done
clear
View Answer play_arrow
Ray optics is valid when characteristic dimensions are
A)
Of the same order as the wavelength of light
done
clear
B)
Much smaller than the wavelength of light
done
clear
C)
Of the order of one millimetre
done
clear
D)
Much larger than the wavelength of light
done
clear
View Answer play_arrow
The dimensions of universal gravitational constant are:
A)
\[[{{M}^{-1}}{{L}^{3}}{{T}^{-2}}]\]
done
clear
B)
\[[M{{L}^{2}}{{T}^{-1}}]\]
done
clear
C)
\[[{{M}^{-2}}{{L}^{3}}{{T}^{-2}}]\]
done
clear
D)
\[[{{M}^{-2}}{{L}^{2}}{{T}^{-1}}]\]
done
clear
View Answer play_arrow
The acceleration due to gravity on the planet A is 9 times the acceleration due to gravity on planet B. A man jumps to a height of 2 m on the surface of A. What is the height of jump by the same person on the planet B? [2003]
A)
18 m
done
clear
B)
6 m
done
clear
C)
\[\frac{2}{3}\,m\]
done
clear
D)
\[\frac{2}{9}\,m\]
done
clear
View Answer play_arrow
Two bodies A (of mass 1 kg) and B (of mass 3 kg) are dropped from heights of 16 m and 25 m, respectively. The ratio of the time taken by them to reach the ground is:
A)
5/4
done
clear
B)
12/5
done
clear
C)
5/12
done
clear
D)
4/5
done
clear
View Answer play_arrow
A particle of mass m is projected with velocity v making an angle of \[45{}^\circ \] with the horizontal. When the particle lands on the level ground the magnitude of the change in its momentum will be:
A)
\[2\,mv\]
done
clear
B)
\[mv/\sqrt{2}\]
done
clear
C)
\[mv\sqrt{2}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
A tube of length L is filled completely with an incompressible liquid of mass M and closed at both the ends. The tube is then rotated in a horizontal plane about one of its ends with a uniform angular velocity \[\omega .\]The force exerted by the liquid at the other end is:
A)
\[\frac{ML{{\omega }^{2}}}{2}\]
done
clear
B)
\[\frac{M{{L}^{2}}\omega }{2}\]
done
clear
C)
\[ML{{\omega }^{2}}\]
done
clear
D)
\[\frac{M{{L}^{2}}{{\omega }^{2}}}{2}\]
done
clear
View Answer play_arrow
A body of mass M hits normally a rigid wall with velocity v and bounces back with the same velocity. The impulse experienced by the body is:
A)
1.5 Mv
done
clear
B)
2 Mv
done
clear
C)
zero
done
clear
D)
Mv
done
clear
View Answer play_arrow
A stone is tied to a string of length \[l\] and is whirled in a vertical circle with the other end of the string as the centre. At a certain instant of time, the stone is at its lowest position and has a speed \[{{u}_{1}}.\]The magnitude of the change in velocity as it reaches a position where the string is horizontal (g being acceleration due to gravity) is
A)
\[\sqrt{2({{u}^{2}}-gl)}\]
done
clear
B)
\[\sqrt{{{u}^{2}}-gl}\]
done
clear
C)
\[u-\sqrt{{{u}^{2}}-2gl}\]
done
clear
D)
\[\sqrt{2gl}\]
done
clear
View Answer play_arrow
A body of mass 1 kg is thrown upwards with a velocity \[20\,m{{s}^{-1}}.\]It momentarily comes to rest after attaining a height of 18 m. How much energy is lost due to air friction? \[(g=10\,m{{s}^{-2}})\]
A)
20 J
done
clear
B)
30 J
done
clear
C)
40 J
done
clear
D)
10 J
done
clear
View Answer play_arrow
A car of mass m starts from rest and accelerates so that the instantaneous power delivered to the car has a constant magnitude\[{{P}_{0}}\]. The instantaneous velocity of this car is proportional to
A)
\[{{t}^{2}}{{P}_{0}}\]
done
clear
B)
\[{{t}^{1/2}}\]
done
clear
C)
\[{{t}^{-1/2}}\]
done
clear
D)
\[t/\sqrt{m}\]
done
clear
View Answer play_arrow
A uniform rod AB of length \[l\] and mass m is free to rotate about point A. The rod is released from rest in the horizontal position. Given that the moment of inertia of the rod about A is\[\frac{m{{l}^{2}}}{3}.\] the Initial angular acceleration of the rod will be:
A)
\[\frac{2g}{3l}\]
done
clear
B)
\[mg\frac{l}{2}\]
done
clear
C)
\[\frac{3}{2}gl\]
done
clear
D)
\[\frac{3g}{2l}\]
done
clear
View Answer play_arrow
Two bodies of mass 1 kg and 3 kg have position vectors \[\hat{i}+2\hat{j}+\hat{k}\]and \[-3\hat{i}-2\hat{j}+\hat{k},\]respectively. The centre of mass of this system has a position vector.
A)
\[-2\hat{i}+2\hat{k}\]
done
clear
B)
\[-2\hat{i}-\hat{j}+\hat{k}\]
done
clear
C)
\[2\hat{i}-\hat{j}-2\hat{k}\]
done
clear
D)
\[-\hat{i}+\hat{j}+\hat{k}\]
done
clear
View Answer play_arrow
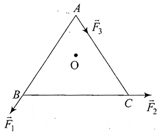
ABC is an equilateral triangle with O as its centre. \[{{\vec{F}}_{1}},{{\vec{F}}_{2}}\]and\[{{\vec{F}}_{3}}\]represent three forces acting along the sides AB, BC and AC respectively. If the total torque about O is zero then the magnitude of \[{{\vec{F}}_{3}}\]is
A)
\[2({{F}_{1}}+{{F}_{2}})\]
done
clear
B)
\[{{F}_{1}}+{{F}_{2}}\]
done
clear
C)
\[{{F}_{1}}-{{F}_{2}}\]
done
clear
D)
\[\frac{{{F}_{1}}+{{F}_{2}}}{2}\]
done
clear
View Answer play_arrow
The acceleration due to gravity is g at a point distant r from the centre of earth of radius R. If r < R, then
A)
\[g\propto r\]
done
clear
B)
\[g\propto {{r}^{2}}\]
done
clear
C)
\[g\propto {{r}^{-1}}\]
done
clear
D)
\[g\propto {{r}^{-2}}\]
done
clear
View Answer play_arrow
There is a hole in the bottom of tank having water. If total pressure at bottom is 3 atm \[(1\,atm\,={{10}^{5}}N/{{m}^{2}}),\]then the velocity of water flowing from hole is
A)
\[\sqrt{400}\,m/s\]
done
clear
B)
\[\sqrt{600}\,m/s\]
done
clear
C)
\[\sqrt{60}\,m/s\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
The Young's modulus of steel is twice that of brass. Two wires of the same length and of the same area of cross section, one of steel and another of brass are suspended from the same roof. If we want the lower ends of the wires to be at the same level, then the weights added to the steel and brass wires must be in the ratio of
A)
1 : 1
done
clear
B)
1 : 2
done
clear
C)
2 : 1
done
clear
D)
4 : 1
done
clear
View Answer play_arrow
An element X has the following isotopic com- position\[:\]\[{{\,}^{200}}X:90%{{\,}^{199}}X:8.0%{{\,}^{202}}X:2.0%\]. The weighted average atomic mass of the naturally occurring element X is closest to
A)
201 amu
done
clear
B)
202 amu
done
clear
C)
199 amu
done
clear
D)
200 amu
done
clear
View Answer play_arrow
The energy of second Bohr's orbit in H-atom is \[-328\,kJ\,mo{{l}^{-1}},\] hence the energy of fourth Bohr's orbit would be
A)
\[-82\,kJ\,mo{{l}^{-1}}\]
done
clear
B)
\[-41\,kJ\,mo{{l}^{-1}}\]
done
clear
C)
\[-1312\,kJ\,mo{{l}^{-1}}\]
done
clear
D)
\[-164\,kJ\,mo{{l}^{-}}\]
done
clear
View Answer play_arrow
Which of the following statements is not correct for sigma and pi-bonds formed between two carbon atoms?
A)
Sigma-bond determines the direction between carbon atoms but a pi-bond has no primary effect in this regard
done
clear
B)
Sigma-bond is stronger than a pi-bond
done
clear
C)
Bond energies of sigma- and pi-bonds are of the order of 264 kJ/mol and 347 kJ/mol, respectively
done
clear
D)
Free rotation of atoms about a sigma-bond is 11 lowed but not in case of a pi-bond
done
clear
View Answer play_arrow
For real gives, van der Waals' equation is written as\[\left( p+\frac{a{{n}^{2}}}{{{V}^{2}}} \right)(V-nb)=nRT\], where \[a\] and \[b\] are van der Waals' constants. Two sets of gases are:\[(I)\,{{O}_{2}},C{{O}_{2}},{{H}_{2}}\] and \[He\,\,(II)C{{H}_{4}},{{O}_{2}}\]and\[{{H}_{2}}\].The gases given in set I in increasing order of A and gases given in set II in decreasing order of a are arranged below. Select the correct order from the following:
A)
\[(I)\,He<{{H}_{2}}<C{{O}_{2}}<{{O}_{2}},\]\[(II)\,C{{H}_{4}}>{{H}_{2}}>{{O}_{2}}\]
done
clear
B)
\[(I)\,{{O}_{2}}<He<{{H}_{2}}<C{{O}_{2}},\]\[(II)\,{{H}_{2}}>{{O}_{2}}>C{{H}_{4}}\]
done
clear
C)
\[(I)\,{{H}_{2}}<He<{{O}_{2}}<C{{O}_{2}},\]\[(II)C{{H}_{4}}>{{O}_{2}}>{{H}_{2}}\]
done
clear
D)
\[(I)\,{{H}_{2}}<{{O}_{2}}<He<C{{O}_{2}},\]\[(II)\,{{O}_{2}}>C{{H}_{4}}>{{H}_{2}}\]
done
clear
View Answer play_arrow
Standard enthalpy and standard entropy change for the oxidation of \[N{{H}_{3}}\] at 298 K are -3 82.64 kJ \[mo{{l}^{-1}}\]and \[-145.6\,J\,mo{{l}^{-1}}\] respectively. Standard free energy change for the same reaction at 298 K is
A)
\[-221.1\,kJ\,mo{{l}^{-1}}\]
done
clear
B)
\[-339.3\text{ }kJ\text{ }mo{{l}^{-1}}\]
done
clear
C)
\[-439.3\text{ }kJ\,mo{{l}^{-1}}\]
done
clear
D)
\[-523.2\text{ }kJ\text{ }mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
Consider the following reactions:
(i)\[{{H}^{+}}(aq)+O{{H}^{-}}(aq)\to {{H}_{2}}O(l);\]
\[\Delta H =-{{X}_{1}}\,kJ\,mo{{l}^{-1}}\]
(ii) \[{{H}_{2}}(g)+\frac{1}{2}{{O}_{2}}(g)\to {{H}_{2}}O(l);\]
\[\Delta H =-{{X}_{2}}\,kJ\,mo{{l}^{-1}}\]
(iii) \[C{{O}_{2}}(g)+{{H}_{2}}(g)\to CO(g)+{{H}_{2}}O(l);\]
\[\Delta H =-{{X}_{3}}kJ\,mo{{l}^{-1}}\]
(iv) \[{{C}_{2}}{{H}_{2}}(g)+\frac{5}{2}{{O}_{2}}(g)\to 2C{{O}_{2}}g+{{H}_{2}}O(l);\]
\[\Delta H =+{{X}_{4}}\,kJ\,mo{{l}^{-1}}\]
Enthalpy of formation of \[{{H}_{2}}O(l)\] is
A)
\[-{{x}_{2}}\,\,kJ\,mo{{l}^{-1}}\]
done
clear
B)
\[+{{x}_{3}}\,\,kJ\,mo{{l}^{-1}}\]
done
clear
C)
\[-{{x}_{4}}\,\,kJ\,mo{{l}^{-1}}\]
done
clear
D)
\[-{{x}_{1}}\,\,kJ\,mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
The value of equilibrium constant of the reaction, \[HI(g)\rightleftharpoons \frac{1}{2}{{H}_{2}}(g)+\frac{1}{2}{{I}_{2}}(g)\]is 8.0. The equilibrium constant of the reaction\[{{H}_{2}}(g)+{{I}_{2}}(g)\rightleftharpoons 2HI(g)\]will be
A)
\[\frac{1}{64}\]
done
clear
B)
16
done
clear
C)
\[\frac{1}{8}\]
done
clear
D)
\[\frac{1}{16}\]
done
clear
View Answer play_arrow
In the equilibrium constant for \[{{N}_{2}}(g)+{{O}_{2}}(g)\rightleftharpoons 2NO(g)\]is K, the equilibrium constant for \[\frac{1}{2}{{N}_{2}}(g)+\frac{1}{2}{{O}_{2}}(g)\rightleftharpoons NO(g)\]will be:
A)
K
done
clear
B)
\[{{K}^{2}}\]
done
clear
C)
\[{{K}^{1/2}}\]
done
clear
D)
\[\frac{1}{2}K\]
done
clear
View Answer play_arrow
Which has the highest pH?
A)
\[C{{H}_{3}}COOK\]
done
clear
B)
\[N{{a}_{2}}C{{O}_{3}}\]
done
clear
C)
\[N{{H}_{4}}Cl\]
done
clear
D)
\[NaN{{O}_{3}}\]
done
clear
View Answer play_arrow
The dissociation constant for acetic acid and HCN at \[25{}^\circ C\]are \[1.5\times {{10}^{-5}}\]and \[4.5\times {{10}^{-10}}\] respectively. The equilibrium constant for the equilibrium\[C{{N}^{-}}+C{{H}_{3}}COOH\rightleftharpoons HCN+C{{H}_{3}}CO{{O}^{-}}\] would be:
A)
\[3.0\times {{10}^{4}}\]
done
clear
B)
\[3.0\times {{10}^{-5}}\]
done
clear
C)
\[3.0\times {{10}^{5}}\]
done
clear
D)
\[3.0\times {{10}^{-4}}\]
done
clear
View Answer play_arrow
What is the pH of the resulting solution when equal volumes of \[0.1\,M\,NaOH\]and 0.01 MHCl are mixed?
A)
7.0
done
clear
B)
1.04
done
clear
C)
12.65
done
clear
D)
2.0
done
clear
View Answer play_arrow
Oxidation numbers of \[\text{P}\]in \[PO_{4}^{3-}\]of \[\text{S}\] in \[SO_{4}^{2-}\]and that of Cr in \[C{{r}_{2}}O_{7}^{2-}\]are respectively:
A)
\[-3,+6\]and +6
done
clear
B)
\[+5,+3\]and +6
done
clear
C)
\[+3,+6\]and +5
done
clear
D)
+5, +6 and +6
done
clear
View Answer play_arrow
When a substance A reacts with water it produces a combustible gas B and a solution of substance C in water. When another substance D reacts with this solution of C, it also produces the same gas B on warming but D can produce gas B on reaction with dilute sulphuric acid at room temperature. A imparts a deep golden yellow colour to a smokeless flame of Bunsen burner. A, B, C and D respectively are
A)
\[K,{{H}_{2}},KOH,Al\]
done
clear
B)
\[Na,{{H}_{2}},NaOH,Zn\]
done
clear
C)
\[Ca,{{H}_{2}},Ca{{(OH)}_{2}},Sn\]
done
clear
D)
\[Ca{{C}_{2}},{{C}_{2}}{{H}_{2}},Ca{{(OH)}_{2}},Fe\]
done
clear
View Answer play_arrow
The correct increasing covalent character is
A)
\[NaCl<LiCl<BeC{{l}_{2}}\]
done
clear
B)
\[BeC{{l}_{2}}<NaCl<LiCl\]
done
clear
C)
\[BeC{{l}_{2}}<LiCl<NaCl\]
done
clear
D)
\[LiCl<NaCl<BeC{{l}_{2}}\]
done
clear
View Answer play_arrow
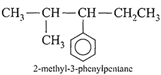
Which nomenclature is not according to IUPAC system?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
If there is no rotation of plane polarized light by a compound in a specific solvent, thought to be chiral, it means that:
A)
it is certainly meso
done
clear
B)
it is racemic mixture
done
clear
C)
it is certainly not chiral
done
clear
D)
no such compound
done
clear
View Answer play_arrow
. A(Predominantly) is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
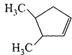
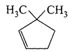
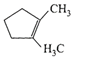
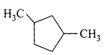
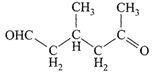
A single compound of the structure
is obtainable from ozonolysis of which of the following cyclic compounds?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
If 'a' stands for the edge length of the cubic systems : simple cubic, body-centred and face- centred, then the ratio of radii of the spheres in these systems will be respectively
A)
\[\frac{1}{2}a:\sqrt{3}a:\frac{1}{\sqrt{2}}a\]
done
clear
B)
\[\frac{1}{2}a:\frac{\sqrt{3}}{2}a:\frac{\sqrt{2}}{2}a\]
done
clear
C)
\[\frac{1}{2}a:\sqrt{3}a:\sqrt{2}a\]
done
clear
D)
\[\frac{1}{2}a:\frac{\sqrt{3}}{4}a:\frac{1}{2\sqrt{2}}a\]
done
clear
View Answer play_arrow
A 0.0020 m aqueous solution in an ionic com- pound \[Co{{(N{{H}_{3}})}_{5}}(N{{O}_{2}})Cl\]freezes at\[-0.00732{{\,}^{o}}C.\] Number of moles of ions which 1 mol of ionic compound produces on being dissolved in water will be \[({{K}_{f}}=+1.86{{\,}^{o}}C/m)\]
A)
1
done
clear
B)
4
done
clear
C)
3
done
clear
D)
2
done
clear
View Answer play_arrow
What is the fraction of the solute in a 1.00 m aqueous solution?
A)
0.0354
done
clear
B)
0.0177
done
clear
C)
0.177
done
clear
D)
1.770
done
clear
View Answer play_arrow
4.5 g of Al (atomic mass 27 amu) is deposited at cathode from \[A{{l}^{3+}}\]solution by a certain quantity of charge. The volume of \[{{H}_{2}}\]produced at STP from \[{{H}^{+}}\]ions in solution by the same quantity of charge will be
A)
11.2 L
done
clear
B)
44.8 L
done
clear
C)
5.6 L
done
clear
D)
22.4 L
done
clear
View Answer play_arrow
Standard electrode potential for\[S{{n}^{4+}}/S{{n}^{2+}}\]couple is +0.15 V and that for the \[C{{r}^{3+}}/Cr\]couple is -0.74 V. These two couples in their standard state are connected to make a cell. The cell potential will be
A)
+1.83 V
done
clear
B)
+1.19 V
done
clear
C)
+0.18 V
done
clear
D)
+0.89 V
done
clear
View Answer play_arrow
A device that converts energy of combustion of fuels like hydrogen and methane, directly into electrical energy is known as
A)
dynamo
done
clear
B)
Ni-Cd cell
done
clear
C)
fuel cell
done
clear
D)
electrolytic cell
done
clear
View Answer play_arrow
The activation energy for a simple chemical reaction \[A\to B\]is\[{{E}_{a}}\]in forward direction. The activation energy for reverse reaction
A)
is always double of\[{{E}_{a}}\]
done
clear
B)
is negative of\[{{E}_{a}}\]
done
clear
C)
is always less than\[{{E}_{a}}\]
done
clear
D)
can be less than or more than \[{{E}_{a}}\]
done
clear
View Answer play_arrow
For the reaction \[\underset{(g)}{\mathop{{{N}_{2}}{{O}_{5}}}}\,\xrightarrow{{}}\underset{(g)}{\mathop{2N{{O}_{2}}}}\,+\underset{(g)}{\mathop{\frac{1}{2}{{O}_{2}}}}\,,\] the rate of disappearance of \[{{N}_{2}}{{O}_{5}}\]is\[6.25\times {{10}^{-3}}\,mol\,{{L}^{-1}}{{s}^{-1}}\]. The rate of formation of \[N{{O}_{2}}\]and \[{{O}_{2}}\] will be respectively.
A)
\[6.25\times {{10}^{-3}}\,mol\,{{L}^{-1}}{{s}^{-1}}\]and \[6.25\times {{10}^{-3}}\,mol\,{{L}^{-1}}{{s}^{-1}}\]
done
clear
B)
\[1.25\times {{10}^{-2}}mol\,{{L}^{-1}}{{s}^{-1}}\]and \[3.125\times {{10}^{-3}}\]\[mol{{L}^{-1}}{{s}^{-1}}\]
done
clear
C)
\[6.25\times {{10}^{-3}}mol{{L}^{-1}}{{s}^{-1}}\]and \[3.125\times {{10}^{-3}}\]\[mol\,{{L}^{-1}}{{s}^{-1}}\]
done
clear
D)
\[1.25\times {{10}^{-2}}mol{{L}^{-1}}{{s}^{-1}}\]and \[6.25\times {{10}^{-3}}mol\,{{L}^{-1}}{{s}^{-1}}\]
done
clear
View Answer play_arrow
The rate constant of the reaction\[A\to B\]is \[0.6\times {{10}^{-3}}\]mole per second. If the concentration of A is 5 M, then concentration of B after 20 minutes is:
A)
0.36 M
done
clear
B)
0.72 M
done
clear
C)
1.08 M
done
clear
D)
3.60 M
done
clear
View Answer play_arrow
Which of the following pairs of metals is purified by van Arkel method?
A)
Zr and Ti
done
clear
B)
Ga and in
done
clear
C)
Ni and Fe
done
clear
D)
Ag and Au
done
clear
View Answer play_arrow
\[BC{{l}_{3}}\]is a planar molecule whereas\[NC{{l}_{3}}\]is pyramidal because
A)
\[BC{{l}_{3}}\] is a planar molecule whereas \[NC{{l}_{3}}\] has a lone pair of electrons
done
clear
B)
\[B-Cl\]bond is more polar than\[N-Cl\]bond
done
clear
C)
nitrogen atom is smaller than boron atom
done
clear
D)
N-Cl bond is more covalent than \[B-Cl\] bond
done
clear
View Answer play_arrow
When \[{{H}_{2}}S\]is passed through acidified \[KMn{{O}_{4}},\]we get
A)
\[\,{{K}_{2}}S{{O}_{3}}\]
done
clear
B)
\[Mn{{O}_{2}}\]
done
clear
C)
\[KHS{{O}_{3}}\]
done
clear
D)
Sulphur
done
clear
View Answer play_arrow
The number of moles of\[KMn{{O}_{4}}\]reduced by one mole of KI in alkaline is:
A)
one
done
clear
B)
two
done
clear
C)
five
done
clear
D)
one-fifth
done
clear
View Answer play_arrow
Number of moles of \[MnO_{4}^{-}\]required to oxidize one mole of ferrous oxalate completely in acidic medium will be:
A)
0.6 moles
done
clear
B)
0.4 moles
done
clear
C)
7.5 moles
done
clear
D)
0.2 moles
done
clear
View Answer play_arrow
In hydrogen atom, energy of first excited state is -3.4 eV. Then, KJE of the same orbit of hydrogen atom is
A)
+3.4 eV
done
clear
B)
+6.8 eV
done
clear
C)
-13.6 eV
done
clear
D)
+13.6 eV
done
clear
View Answer play_arrow
Which of the following is not permissible arrangement of electrons in an atom?
A)
\[n=3,l=2,m=-2,s=-1/2\]
done
clear
B)
\[n=4,l=0,m=0,s=-1/2\]
done
clear
C)
\[n=5,l=3,m=0,s=+1/2\]
done
clear
D)
\[n=3,l=2,m=-3,s=-1/2\]
done
clear
View Answer play_arrow
Which of the following lanthanoids ions is diamagnetic? (At. nos.: Ce = 58, Sm = 62, Eu = 63, Yb = 70)
A)
\[S{{m}^{2+}}\]
done
clear
B)
\[E{{u}^{2+}}\]
done
clear
C)
\[C{{e}^{2+}}\]
done
clear
D)
\[Y{{b}^{2+}}\]
done
clear
View Answer play_arrow
In which of the following\[p\pi -d\pi \]bonding is observed?
A)
\[NO_{3}^{-}\]
done
clear
B)
\[SO_{3}^{2-}\]
done
clear
C)
\[BO_{3}^{3-}\]
done
clear
D)
\[CO_{3}^{2-}\]
done
clear
View Answer play_arrow
In which of the following molecules are all the bonds not equal?
A)
\[~Al{{F}_{3}}\]
done
clear
B)
\[~N{{F}_{3}}\]
done
clear
C)
\[~Cl{{F}_{3}}\]
done
clear
D)
\[~B{{F}_{3}}\]
done
clear
View Answer play_arrow
Four diatomic species are listed below in different sequences. Which of these represents the correct order of their increasing bond order?
A)
\[NO<C_{2}^{2-}<O_{2}^{-}<He_{2}^{+}\]
done
clear
B)
\[C_{2}^{2-}<He_{2}^{+}<NO<O_{2}^{-}\]
done
clear
C)
\[He_{2}^{+}<O_{2}^{-}<NO<C_{2}^{2-}\]
done
clear
D)
\[O_{2}^{-}<NO<C_{2}^{2-}<He_{2}^{+}\]
done
clear
View Answer play_arrow
Amongst the elements with following electronic configurations, which one of them may have the highest ionization energy?
A)
\[Ne[3{{s}^{2}}3{{p}^{1}}]\]
done
clear
B)
\[Ne[3{{s}^{2}}3{{p}^{3}}]\]
done
clear
C)
\[Ne[3{{s}^{2}}3{{p}^{2}}]\]
done
clear
D)
\[Ar[3{{d}^{10}}4{{s}^{2}}4{{p}^{3}}]\]
done
clear
View Answer play_arrow
In which of the following pairs, both the species are not isostructural?
A)
\[N{{H}_{3}},P{{H}_{3}}\]
done
clear
B)
\[Xe{{F}_{4}},Xe{{O}_{4}}\]
done
clear
C)
\[SiC{{l}_{4}},PC{{l}_{4}}\]
done
clear
D)
Diamond, silicon carbide
done
clear
View Answer play_arrow
Three moles of an ideal gas expanded spontaneously into vacuum. The work done will be
A)
9 joules
done
clear
B)
3 joules
done
clear
C)
Zero
done
clear
D)
Infinite
done
clear
View Answer play_arrow
Enthalpy of\[C{{H}_{4}}+\frac{1}{2}{{O}_{2}}\xrightarrow{{}}C{{H}_{3}}OH\]is negative. If enthalpy of combustion of \[C{{H}_{4}}\]and \[C{{H}_{3}}OH\]are\[x\]and y respectively, then which relation is correct?
A)
\[x>y\]
done
clear
B)
\[x<y\]
done
clear
C)
\[x=y\]
done
clear
D)
\[x\ge y\]
done
clear
View Answer play_arrow
A reaction occurs spontaneously if:
A)
\[T\Delta S=\Delta H\] and both \[\Delta H\]and \[\Delta S\]are positive
done
clear
B)
\[T\Delta S>\Delta H\]and both\[\Delta H\]and \[\Delta S\]are positive
done
clear
C)
\[T\Delta S>\Delta H\]and both \[\Delta H\]and\[\Delta S\]are positive
done
clear
D)
\[T\Delta S>\Delta H\] and \[\Delta H\]is positive and\[\Delta S\]is negative
done
clear
View Answer play_arrow
Which of the following are not state functions?
(I)\[q+w\] (II) q (III)\[w\] (IV)\[H-TS\]
A)
(II), (III) and (IV)
done
clear
B)
(I), (II) and (III)
done
clear
C)
(II) and (III)
done
clear
D)
(I) and (IV)
done
clear
View Answer play_arrow
In which of the following reactions, standard reaction entropy change\[(\Delta {{S}^{o}})\] is positive and standard Gibb's energy change \[(\Delta {{G}^{o}})\] decreases sharply with increasing temperature?
A)
\[Mg(s)+\frac{1}{2}{{O}_{2}}(g)\xrightarrow{{}}MgO(s)\]
done
clear
B)
\[\frac{1}{2}C(graphite)+\frac{1}{2}{{O}_{2}}(g)\xrightarrow{{}}\frac{1}{2}C{{O}_{2}}(g)\]
done
clear
C)
\[C(graphite)+\frac{1}{2}{{O}_{2}}(g)\xrightarrow{{}}CO(g)\]
done
clear
D)
\[CO(g)+\frac{1}{2}{{O}_{2}}(g)\xrightarrow{{}}C{{O}_{2}}(g)\]
done
clear
View Answer play_arrow
Which one of the following statements about viruses is correct?
A)
Viruses possess their own metabolic system
done
clear
B)
All viruses contain both RNA and DNA
done
clear
C)
Viruses are obligate parasites
done
clear
D)
Nucleic acid of viruses is known as capsid
done
clear
View Answer play_arrow
Lichens are well-known combination of an alga and a fungus where fungus has:
A)
An epiphytic relationship with the alga
done
clear
B)
A parasitic relationship with the alga
done
clear
C)
A symbiotic relationship with the alga
done
clear
D)
A saprophytic relationship with the alga
done
clear
View Answer play_arrow
The causative agent of mad-cow disease is a
A)
Bacterium
done
clear
B)
Prion
done
clear
C)
Worm
done
clear
D)
Virus
done
clear
View Answer play_arrow
In the light of recent classification of living organisms into three domains of life (bacteria, archaea and eukarya), which one of the following statements is true about archaea?
A)
Archaea completely differ from both prokaryotes and eukaryotes
done
clear
B)
Archaea completely differ from prokaryotes
done
clear
C)
Archaea resemble eukarya in all respects
done
clear
D)
Archaea have some novel features that are absent in other prokaryotes and eukaryotes
done
clear
View Answer play_arrow
Which one pair of examples will correctly represent the grouping Spermatophyta according to one of the schemes of classifying plants?
A)
Acacia, Sugarcane
done
clear
B)
Pinus, Cycas
done
clear
C)
Rhizopus, Triticum
done
clear
D)
Ginkgo, Pisum
done
clear
View Answer play_arrow
Flagellated male gametes are present in all the three of which one of the following sets?
A)
Riccia, Dryopferis and Cycas
done
clear
B)
Anthoceros, Funaria and Spirogyra
done
clear
C)
Zygnema, Saproiegnia and Hydrilla
done
clear
D)
Fucus, Marsilea and Calotropis
done
clear
View Answer play_arrow
In which one of the following sets of animals do all the four give birth to young ones?
A)
Lion, Bat, Whale, Ostrich
done
clear
B)
Platypus, Penguin, Bat, Hippopotamus
done
clear
C)
Shrew, Bat, Cat, Kiwi
done
clear
D)
Kangaroo, Hedgehog, Dolphin, Loris
done
clear
View Answer play_arrow
Which one of the following is the true description about an animal concerned?
A)
Rat-Left kidney is slightly higher in position than the right one
done
clear
B)
Cockroach- 10 pairs of spiracles (2 pairs on thorax and 8 pairs on abdomen)
done
clear
C)
Earthworm-The alimentary canal consists of a sequence of pharynx, oesopha- gus, stomach, gizzard and intestine
done
clear
D)
Frog-Body divisible into three regions- head, neck and trunk
done
clear
View Answer play_arrow
Which one of the following statements about all the four of Spongilla, Leech, Dolphin and Penguin is correct?
A)
Penguin is homoiothermic while the remaining three are poikilothermic
done
clear
B)
Leech is fresh water form while all others are marine
done
clear
C)
Spongilla has special collared cells called choanocytes, not found in the remaining three
done
clear
D)
All are bilaterally symmetrical
done
clear
View Answer play_arrow
In which one of the following the genus name its two characters and its phylum are not correctly matched, whereas the remaining threes correct
A)
Genus name Two characters Phylum Aslericis [a] Spiny skinned Echinoder- mate [b] Water vascular system
done
clear
B)
Genus name Two characters Phylum Sycon [a] Pore bearing Porifera [b] Canal system
done
clear
C)
Genus name Two characters Phylum Periplaneta [a] Jointed appendages Arthropoda [b] Chitinous exoskeleton
done
clear
D)
Genus name Two characters Phylum Pila [a] Body segmented Mollusca [b] Mouth with Radula
done
clear
View Answer play_arrow
Which of the following is a flowering plant with nodules containing filamentous nitrogen-fixing microorganism?
A)
Cicer arietimnn
done
clear
B)
Casuarina equisetifolia
done
clear
C)
Crotalaria juncea
done
clear
D)
Cycas revolute
done
clear
View Answer play_arrow
When the margins of sepals or petals overlap one another without any particular direction, the condition is termed as:
A)
Vexillary
done
clear
B)
Imbricate
done
clear
C)
Twisted
done
clear
D)
Valvate
done
clear
View Answer play_arrow
Anatomically fairly old dicotyledonous root is distinguished from the dicotyledonous stem by:
A)
Position of protoxylem
done
clear
B)
Absence of secondary xylem
done
clear
C)
Absence of secondary phloem
done
clear
D)
Presence of cortex
done
clear
View Answer play_arrow
The common bottle cork is a product of
A)
Xylem
done
clear
B)
Vascular Cambium
done
clear
C)
Dermatogen
done
clear
D)
Phellogen
done
clear
View Answer play_arrow
Transmission tissue is characteristic feature of
A)
Wet stigma
done
clear
B)
Hollow style
done
clear
C)
Solid style
done
clear
D)
Dry stigma
done
clear
View Answer play_arrow
Choose the correctly matched pair:
A)
Tendon-Specialized connective tissue
done
clear
B)
Adipose tissue-Dense connective tissue
done
clear
C)
Areolar tissue-Loose connective tissue
done
clear
D)
Cartilage-Loose connective tissue
done
clear
View Answer play_arrow
Cellulose is the major component of cell walls of:
A)
Pseudomonas
done
clear
B)
Saccharomyces
done
clear
C)
Pythium
done
clear
D)
Xanthomonas
done
clear
View Answer play_arrow
Which one of the following has its own DNA?
A)
Mitochondria
done
clear
B)
Dictyosome
done
clear
C)
Lysosome
done
clear
D)
Peroxisome
done
clear
View Answer play_arrow
DNA is not present in:
A)
Mitochondria
done
clear
B)
Chloroplast
done
clear
C)
Ribosomes
done
clear
D)
Nucleus
done
clear
View Answer play_arrow
The catalytic efficiency of two different enzymes can be compared by the:
A)
\[{{K}_{m}}\]value
done
clear
B)
pH optimum value
done
clear
C)
Molecular size of the enzyme
done
clear
D)
Formation of the product
done
clear
View Answer play_arrow
The essential chemical components of many coenzymes are:
A)
Proteins
done
clear
B)
Nucleic acids
done
clear
C)
Carbohydrates
done
clear
D)
Vitamins
done
clear
View Answer play_arrow
During which phase(s) of cell cycle, amount of DNA in a cell remains at 4 C level if the initial amount is denoted as 2 C?
A)
\[{{G}_{0}}\]and\[{{G}_{1}}\]
done
clear
B)
\[{{G}_{1}}\]and S
done
clear
C)
Only\[{{G}_{2}}\]
done
clear
D)
\[{{G}_{2}}\]and M
done
clear
View Answer play_arrow
Which one of the following pairs is wrongly matched?
A)
Textile-amylase
done
clear
B)
Detergents-lipase
done
clear
C)
Alcohol-nitrogenase
done
clear
D)
Fruit juice-pectinase
done
clear
View Answer play_arrow
Which one of the following is wrong statement?
A)
Phosphorus is a constituent of cell mem- branes, certain nucleic acids and all proteins
done
clear
B)
Nitrosomonas and Nitrobacter are chemoautotrophs
done
clear
C)
Anabaena and Nostoc are capable of fixing nitrogen in free-living state also
done
clear
D)
Root nodule forming nitrogen fixers live as aerobes under free living conditions
done
clear
View Answer play_arrow
The wavelength of light absorbed by Pr form of phytochrome is:
A)
620 nm
done
clear
B)
640 nm
done
clear
C)
680 nm
done
clear
D)
720 nm
done
clear
View Answer play_arrow
Pigment-containing membranous extensions in some cyanobacteria are:
A)
Heterocysts
done
clear
B)
Basal bodies
done
clear
C)
Pneumatophores
done
clear
D)
Chromatophores
done
clear
View Answer play_arrow
How does pruning help in making the hedge dense?
A)
It induces the differentiation of new shoots from the rootstock
done
clear
B)
It frees axillary buds from apical dominance
done
clear
C)
The apical shoot grows faster after pruning
done
clear
D)
It released wound hormones
done
clear
View Answer play_arrow
Anxiety and eating spicy food together in a otherwise normal human may lead to
A)
Diarrhoea
done
clear
B)
Vomiting
done
clear
C)
Indigestion
done
clear
D)
Jaundice
done
clear
View Answer play_arrow
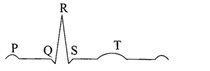
In a standard ECG which one of the following alphabets is the correct representation of the respective activity of the human heart?
A)
P-depolarisation of the atria
done
clear
B)
R-repolarisation of ventricles
done
clear
C)
S-start of systole
done
clear
D)
T-end of diastole
done
clear
View Answer play_arrow
The diagram given here is the standard ECG of a normal person. The P-wave represents the:
A)
Contraction of both the atria
done
clear
B)
Initiation of the ventricular contraction
done
clear
C)
Beginning of the systole
done
clear
D)
End of systole
done
clear
View Answer play_arrow
The maximum amount of electrolytes and water (70-80 percent) from the glomerular filtrate is reabsorbed in which part of the nephron
A)
Proximal convoluted tubule
done
clear
B)
Descending limb of loop of Henie
done
clear
C)
Ascending limb of loop of Henie
done
clear
D)
Distal convoluted tubule
done
clear
View Answer play_arrow
During the transmission of nerve impulse through a nerve fibre, the potential on the inner side of the plasma membrane has which type of electric charge?
A)
First positive, then negative and again back to positive
done
clear
B)
First negative, then positive and again back to negative
done
clear
C)
First positive, then negative and continue to be negative
done
clear
D)
First negative, then positive and continue to be positive
done
clear
View Answer play_arrow
A gymnast is able to balance his body upside down even in the total darkness because of:
A)
Organ of corti
done
clear
B)
Cochlea
done
clear
C)
Vestibular apparatus
done
clear
D)
Tectorial membrane
done
clear
View Answer play_arrow
Which part of ovary in mammals acts as an endocrine gland after ovulation?
A)
Vitelline membrane
done
clear
B)
Graafian follicle
done
clear
C)
Stroma
done
clear
D)
Germinal epithelium
done
clear
View Answer play_arrow
Injury to adrenal cortex is not likely to affect the secretion of which one of the following?
A)
Aldosterone
done
clear
B)
Both androstenedione and dehydroepi- androsterone
done
clear
C)
Adrenaline
done
clear
D)
Cortisol
done
clear
View Answer play_arrow
Select the answer which correctly matches the endocrine gland with the hormone it secretes and its function/deficiency symptom:
A)
Endocrine gland Hormone Function/deficiency symptoms Anterior pituitary Oxytocin Stimulates uterus contraction during child birth
done
clear
B)
Endocrine gland Hormone Function/deficiency symptoms Posterior pituitary Growth Hormone (GH) Oversecretion stimulates abnormal growth
done
clear
C)
Endocrine gland Hormone Function/deficiency symptoms Thyroid gland Thyroxine Lack of iodine in diet results in goitre
done
clear
D)
Endocrine gland Hormone Function/deficiency symptoms Corpus luteum Testosterone Stimulates spermatogenesis
done
clear
View Answer play_arrow
Which one of the following pairs of plant structures has haploid number of chromosomes?
A)
Nucellus and antipodal cells
done
clear
B)
Egg nucleus and secondary nucleus
done
clear
C)
Megaspore mother cell and antipodal cells
done
clear
D)
Egg cell and antipodal cells
done
clear
View Answer play_arrow
Wind pollination is common in:
A)
Orchids
done
clear
B)
Legumes
done
clear
C)
Lilies
done
clear
D)
Grasses
done
clear
View Answer play_arrow
Which one of the following statements is correct?
A)
Hard outer layer of pollen is called inline
done
clear
B)
Sporogenous tissue is haploid
done
clear
C)
Endothecjum produces the microspores
done
clear
D)
Tapetum nourishes the developing pollen
done
clear
View Answer play_arrow
Megasporangium is equivalent to:
A)
Embryo sac
done
clear
B)
Fruit
done
clear
C)
Nucellus
done
clear
D)
Ovule
done
clear
View Answer play_arrow
Which one of the following may require pollinators, but is genetically similar to autogamy?
A)
Cleistogamy
done
clear
B)
Geitonogamy
done
clear
C)
Xenogamy
done
clear
D)
Apogamy
done
clear
View Answer play_arrow
Seminal plasma in human males is rich in:
A)
fructose and calcium
done
clear
B)
glucose and calcium
done
clear
C)
DNA and testosterone
done
clear
D)
ribose and potassium
done
clear
View Answer play_arrow
Menstrual flow occurs due to lack of:
A)
Progesterone
done
clear
B)
FSH
done
clear
C)
Oxytocin
done
clear
D)
Vasopressin
done
clear
View Answer play_arrow
Which of these is not an important component of initiation of parturition in humans?
A)
Release of prolactin
done
clear
B)
Increase in estrogen and progesterone ratio
done
clear
C)
Synthesis of prostaglandins
done
clear
D)
Release of oxytocin
done
clear
View Answer play_arrow
After a mutation at a genetic locus the character of an organism changes due to the change in:
A)
DNA replication
done
clear
B)
Protein synthesis pattern
done
clear
C)
RNA transcription pattern
done
clear
D)
Protein structure
done
clear
View Answer play_arrow
Which of the following is not a hereditary dis- ease?
A)
Haemophilia
done
clear
B)
Cretinism
done
clear
C)
Cystic fibrosis
done
clear
D)
Thalassemia
done
clear
View Answer play_arrow
Test cross involves:
A)
Crossing between two genotypes with recessive trait
done
clear
B)
Crossing between two \[{{F}_{1}}\]hybrids
done
clear
C)
Crossing the \[{{F}_{1}}\]hybrid with a double recessive genotype
done
clear
D)
Crossing between two genotypes with dominant trait
done
clear
View Answer play_arrow
The genetic defect-adenosine deaminase (ADA) deficiency may be cured permanently by:
A)
Enzyme replacement therapy
done
clear
B)
Periodic infusion of genetically engineered lymphocytes having functional ADA cDNA
done
clear
C)
Administering adenosine deaminase activators
done
clear
D)
Introducing bone marrow cells producing ADA into cells at early embryonic stages
done
clear
View Answer play_arrow
Which one of the following conditions correctly describes the manner of determining the sex in the given examples?
A)
Homozygous sex chromosomes (XX) produce male in Drosophila
done
clear
B)
Homozygous sex chromosomes (ZZ) determine female sex in birds
done
clear
C)
XO type of sex chromosomes determine male sex in grasshopper
done
clear
D)
XO condition in humans as found in Turner syndrome determines female sex
done
clear
View Answer play_arrow
A human female with Turner's syndrome:
A)
has 45 chromosomes with XO
done
clear
B)
has one additional X chromosome.
done
clear
C)
exhibits male characters
done
clear
D)
is able to produce children with normal husband.
done
clear
View Answer play_arrow
What does "lac" refer to in what we call the lac operon?
A)
Lactose
done
clear
B)
Lactase
done
clear
C)
Lac insect
done
clear
D)
The number 1,00,000
done
clear
View Answer play_arrow
E. coli cells with a mutated z gene of the lac operon cannot grow in medium containing only lactose as the source of energy because
A)
They cannot synthesize functional beta galactosidase
done
clear
B)
They cannot transport lactose from the medium into the cell
done
clear
C)
The lac operon is constitutively active in these cells
done
clear
D)
In the presence of glucose, E. coli cells do not utilize lactose
done
clear
View Answer play_arrow
Which one of the following pairs of nitrogenous bases of nucleic acids is wrongly matched with the category mentioned against it?
A)
Guanine, Adenine - Purines
done
clear
B)
Adenine, Thymine - Purines
done
clear
C)
Thymine, Uracil - Pyrimidines
done
clear
D)
Uracil, Cytosine - Pyrimidines
done
clear
View Answer play_arrow
Satellite DNA is useful tool in;
A)
organ transplantation
done
clear
B)
sex determination
done
clear
C)
forensic science
done
clear
D)
genetic engineering
done
clear
View Answer play_arrow
Commonly used vectors for human genome sequencing are:
A)
T-DNA
done
clear
B)
BAC and YAC
done
clear
C)
Expression Vectors
done
clear
D)
T/A Cloning Vectors
done
clear
View Answer play_arrow
The DNA molecules to which the gene of interest is integrated for cloning is called:
A)
Carrier
done
clear
B)
Transformer
done
clear
C)
Vector
done
clear
D)
Template
done
clear
View Answer play_arrow
Random genetic drift in a population probably results from:
A)
Highly genetically variable individuals
done
clear
B)
Interbreeding within this population
done
clear
C)
Constant low mutation rate
done
clear
D)
Large population size
done
clear
View Answer play_arrow
There are two opposing views about origin of modern man. According to one view Homo erectus in Asia were the ancestors of modem man. A study of variation of DNA however suggested African origin of modem man. What kind of observation of DNA variation could suggest this?
A)
Greater variation in Asia than in Africa
done
clear
B)
Similar variation in Africa and Asia
done
clear
C)
Variation only in Asia and no variation in Africa
done
clear
D)
Greater variation in Africa than in Asia
done
clear
View Answer play_arrow
What is common between parrot, platypus and kangaroo?
A)
Oviparity
done
clear
B)
Homoiothermy
done
clear
C)
Toothless jaws
done
clear
D)
Functional post-anal tail
done
clear
View Answer play_arrow
Variation in gene frequencies within populations can occur by chance rather than by natural selection. This is referred to as:
A)
genetic flow
done
clear
B)
genetic drift
done
clear
C)
random mating
done
clear
D)
genetic load
done
clear
View Answer play_arrow
The wings of a bird and the wings of an insect are:
A)
homologous structures and represent convergent evolution
done
clear
B)
homologous structures and represent divergent evolution
done
clear
C)
analogous structures and represent convergent evolution
done
clear
D)
phylogenetic structures and represent divergent evolution
done
clear
View Answer play_arrow
Ultrasound of how much frequency is beamed into human body for sonography?
A)
45-70 MHz
done
clear
B)
30-45 MHz
done
clear
C)
15-30 MHz
done
clear
D)
1-15 MHz
done
clear
View Answer play_arrow
Which one of the following statement is correct?
A)
Malignant tumours may exhibit metastasis
done
clear
B)
Patients who have undergone surgery are given cannabinoids to relieve pain.
done
clear
C)
Benign tumours show the property of metastasis
done
clear
D)
Heroin accelerates body functions.
done
clear
View Answer play_arrow
Which one of the following acts as a physi- ological barrier to the entry of microorganisms in human body?
A)
Skin
done
clear
B)
Epithelium of urogenital tract
done
clear
C)
Tears
done
clear
D)
Monocytes
done
clear
View Answer play_arrow
Grafted kidney may be rejected in a patient due to
A)
Innate immune response
done
clear
B)
Humeral immune response
done
clear
C)
Cell-mediated immune response
done
clear
D)
Passive immune response
done
clear
View Answer play_arrow
Before the European invader which vegetable was absent in India:
A)
Potato and tomato
done
clear
B)
Simla mirch and brinjal
done
clear
C)
Maize and chichinda
done
clear
D)
Bitter gourd
done
clear
View Answer play_arrow
The technique of obtaining large number of Plantlets by tissue culture method is called
A)
Organ culture
done
clear
B)
Micropropagation
done
clear
C)
Macropropagation
done
clear
D)
Plantlet culture
done
clear
View Answer play_arrow
Curing of tea leaves is brought about by the activity of
A)
Bacteria
done
clear
B)
Mycorrhiza
done
clear
C)
Viruses
done
clear
D)
Fungi
done
clear
View Answer play_arrow
Somaclones are obtained by:
A)
Genetic engineering
done
clear
B)
Tissue culture
done
clear
C)
Plant breeding
done
clear
D)
Irradiation genetic engineering
done
clear
View Answer play_arrow
Two microbes found to be very useful in genetic engineering are
A)
Escherichia coli and Agrobacterium tumefaciens
done
clear
B)
Vibrio cholerae and a tailed bacteriophage
done
clear
C)
Diplococcus sp. and Pseudomonas sp
done
clear
D)
Crown gall bacterium and Caenorhabditis elegans
done
clear
View Answer play_arrow
Which one of the following helps in absorption of phosphorus from soil by plants?
A)
Anabaena
done
clear
B)
Glomus
done
clear
C)
Rhizobium
done
clear
D)
prankia
done
clear
View Answer play_arrow
During sewage treatment, biogases are produced which include:
A)
methane, hydrogen sulphide, carbon dioxide
done
clear
B)
methane, oxygen, hydrogen sulphide
done
clear
C)
hydrogen sulphide, methane, sulphur dioxide
done
clear
D)
hydrogen sulphide, nitrogen, methane
done
clear
View Answer play_arrow
Polyethylene glycol method is used for-
A)
Energy production from sewage
done
clear
B)
Gene transfer without a vector
done
clear
C)
Biodiesel production
done
clear
D)
Seedless fruit production
done
clear
View Answer play_arrow
For transformation, micro-particles coated with DNA to be bombarded with gene are made up of
A)
Silicon or platinum
done
clear
B)
Gold or tungsten
done
clear
C)
Silver or platinum
done
clear
D)
Platinum or zinc
done
clear
View Answer play_arrow
The name of Norman Borlaug is associated with
A)
Green Revolution
done
clear
B)
White Revolution
done
clear
C)
Yellow Revolution
done
clear
D)
Blue Revolution
done
clear
View Answer play_arrow
Some of the characteristics of Bt cotton are
A)
Long fibre and resistance to aphids
done
clear
B)
Medium yield, long fibre and resistance to beetle pests
done
clear
C)
High yield and production of toxic protein crystals which kill dipteran pests
done
clear
D)
High yield and resistance to bollworms
done
clear
View Answer play_arrow
Which of the following Bt crops is being grown in India by the farmers?
A)
Maize
done
clear
B)
Cotton
done
clear
C)
Brinjal
done
clear
D)
Soybean
done
clear
View Answer play_arrow
The crops engineered for glyphosate are resistant/tolerant to;
A)
Herbicides
done
clear
B)
Fungi
done
clear
C)
Bacteria
done
clear
D)
insects
done
clear
View Answer play_arrow
In a population, unrestricted reproductive capacity is called as:
A)
Biotic potential
done
clear
B)
Fertility
done
clear
C)
Carrying capacity
done
clear
D)
Birthrate
done
clear
View Answer play_arrow
The formula for exponential population growth is
A)
\[dt/dN=rN\]
done
clear
B)
\[dN/rN=dt\]
done
clear
C)
\[rN/dN=dt\]
done
clear
D)
\[dN/dt=rN\]
done
clear
View Answer play_arrow
What is true about the isolated small tribal populations?
A)
Wrestlers who develop strong body muscles in their life time pass their character on to their progeny
done
clear
B)
There is no change in population size as they have a large gene pool
done
clear
C)
There is a decline in population as boys marry girls only from their own tribe
done
clear
D)
Hereditary diseases like colour blindness do not spread in the isolated population
done
clear
View Answer play_arrow
Consider the following four conditions (a-d) and select the correct pair of them as adaptation to environment in desert lizards. The conditions:
a. burrowing in soil to escape high temperature b. losing heat rapidly from the body during high temperature c. bask in sun when temperature is low d. insulating body due to thick fatty dermis
A)
a, b
done
clear
B)
c, d
done
clear
C)
a, c
done
clear
D)
b, d
done
clear
View Answer play_arrow
If by radiation all nitrogenase enzymes are inactivated, then there will be no:
A)
Fixation of atmospheric nitrogen
done
clear
B)
Conversion from nitrate to nitrite in legumes
done
clear
C)
Conversion from ammonium to nitrate in soil
done
clear
D)
Fixation of nitrogen in legumes
done
clear
View Answer play_arrow
About 70% of total global carbon is found in:
A)
Oceans
done
clear
B)
Forests
done
clear
C)
Grasslands
done
clear
D)
Agroecosystems
done
clear
View Answer play_arrow
Which one of the following is not a functional unit of an ecosystem?
A)
Productivity
done
clear
B)
Stratification
done
clear
C)
Energy flow
done
clear
D)
Decomposition
done
clear
View Answer play_arrow
In which of the following pairs is the specific characteristic of a soil not correctly mathed:
A)
Terra rossa - Most suitable for roses
done
clear
B)
Chemozems - Richest soil in the world
done
clear
C)
Black soil - Rich in calcium carbonate
done
clear
D)
Laterite - Contains aluminium compound
done
clear
View Answer play_arrow
Which one of the following is the correctly matched pair of an endangered animal and a National Park?
A)
Lion - Corbett National Park
done
clear
B)
Rhinoceros - Kaziranga National Park
done
clear
C)
Wild Ass - Dudhwa National Park
done
clear
D)
Great Indian - Keoladeo National Park Bustard
done
clear
View Answer play_arrow
A collection of plants and seeds having diverse alleles of all the genes of a crop is called:
A)
Genome
done
clear
B)
Herbarium
done
clear
C)
Germplasm
done
clear
D)
Gene library
done
clear
View Answer play_arrow
Identify the correctly matched pair
A)
Kyoto protocol - Climatic change
done
clear
B)
Montreal Protocol - Global warming
done
clear
C)
Basal Convention - Biodiversity conservation
done
clear
D)
Ramsar Convention - Ground water pollution
done
clear
View Answer play_arrow
Stirred-tank bioreactors have been designed for
A)
Addition of preservatives to the product
done
clear
B)
Purification of the product
done
clear
C)
Ensuring anaerobic conditions in the culture vessel
done
clear
D)
Availability of oxygen throughout the process
done
clear
View Answer play_arrow

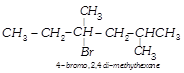
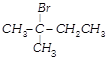
 is obtainable from ozonolysis of which of the following cyclic compounds?
is obtainable from ozonolysis of which of the following cyclic compounds?