-
question_answer1)
The limiting molar conductivities \[{{\wedge }^{0}}\]for NaCl, KBr and KCl are 126, 152 and 150 \[S\ c{{m}^{2}}mo{{l}^{-1}}\] respectively. The \[{{\wedge }^{0}}\] for NaBr is [AIEEE 2004]
A)
\[278\ S\ c{{m}^{2}}mo{{l}^{-1}}\] done
clear
B)
\[176\ S\ c{{m}^{2}}mo{{l}^{-1}}\] done
clear
C)
\[128\ S\ c{{m}^{2}}mo{{l}^{-1}}\] done
clear
D)
\[302\ S\ c{{m}^{2}}mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer2)
On the basis of the electrochemical theory of aqueous corrosion, the reaction occurring at the cathode is [MP PET 1994; UPSEAT 2001]
A)
\[{{O}_{2\,(g)}}+4H_{(aq)}^{+}+4{{e}^{-}}\to 2{{H}_{2}}{{O}_{(l)}}\] done
clear
B)
\[F{{e}_{(s)}}\to Fe_{(aq)}^{2+}+2{{e}^{-}}\] done
clear
C)
\[Fe_{(aq)}^{2+}\to Fe_{(aq)}^{3+}+{{e}^{-}}\] done
clear
D)
\[{{H}_{2(g)}}+2OH_{(aq)}^{-}\to 2{{H}_{2}}{{O}_{(l)}}+2{{e}^{-}}\] done
clear
View Solution play_arrow
-
question_answer3)
The reaction \[\tfrac{1}{2}{{H}_{2}}(g)+AgCl(s)\,\to \,{{H}^{+}}(aq)+C{{l}^{-}}(aq)+Ag(s)\] occurs in the galvanic cell [IIT 1985; AMU 2002; KCET 2003]
A)
\[Ag/AgCl(s)\,KCl\,(\text{soln})\,\,||\,\,AgN{{O}_{3}}\,(\text{soln})\,/Ag\] done
clear
B)
\[Pt/{{H}_{2}}(g)\,HCl\,(\text{soln})\,\,||\,\,AgN{{O}_{3}}\,(\text{soln})\,/Ag\] done
clear
C)
\[Pt/{{H}_{2}}(g)\,HCl\,(\text{soln})\,\,||\,\,AgCl\,(s)\,/Ag\] done
clear
D)
\[Pt/{{H}_{2}}(g)\,KCl\,(\text{soln})\,\,||\,\,AgCl\,(s)\,/Ag\] done
clear
View Solution play_arrow
-
question_answer4)
The standard reduction potential \[NaCl\]for the half reactions are as \[Zn=Z{{n}^{2+}}+2{{e}^{-}};\,\,{{E}^{o}}=+0.76\,V\] \[Fe=F{{e}^{2+}}+2{{e}^{-}};\,\,{{E}^{o}}=+0.41\,V\] The EMF for cell reaction \[F{{e}^{2+}}+Zn\,\to \,Z{{n}^{2+}}+Fe\] is [IIT 1988; CBSE PMT 1993, 96; BHU 1995, 2000; CPMT 2000; KCET 2000; AIIMS 2001; Orissa JEE 2002]
A)
\[-\,0.35\,V\] done
clear
B)
\[+\,0.35\,V\] done
clear
C)
\[+\,1.17\,V\] done
clear
D)
\[-\,1.17\,V\] done
clear
View Solution play_arrow
-
question_answer5)
The number of electrons to balance the following equation \[NO_{3}^{-}+4{{H}^{+}}+{{e}^{-}}\,\to \,2{{H}_{2}}O+NO\] is [IIT Screening 1991]
A)
5 done
clear
B)
4 done
clear
C)
3 done
clear
D)
2 done
clear
View Solution play_arrow
-
question_answer6)
The standard \[EMF\] for the given cell reaction \[Zn+C{{u}^{2+}}=Cu+Z{{n}^{2+}}\] is \[1.10V\] at \[{{25}^{o}}C\]. The \[EMF\] for the cell reaction, when \[0.1M\,C{{u}^{2+}}\] and \[0.1M\,Z{{n}^{2+}}\] solutions are used, at \[{{25}^{o}}C\] is [MNR 1994; AMU 1999; UPSEAT 2002]
A)
\[1.10\,V\] done
clear
B)
\[0.110\,V\] done
clear
C)
\[-1.10\,V\] done
clear
D)
\[-\,0.110\,V\] done
clear
View Solution play_arrow
-
question_answer7)
A gas \[X\] at \[1\,atm\] is bubbled through a solution containing a mixture of \[1\,M\,{{Y}^{-}}\] and \[1\,M\,{{Z}^{-}}\] at \[{{25}^{o}}C\]. If the reduction potential of \[Z>Y>X\], then [IIT 1999]
A)
\[Y\] will oxidize \[X\] and not \[Z\] done
clear
B)
\[Y\] will oxidize \[Z\] and not \[X\] done
clear
C)
\[Y\] will oxidize both \[X\] and \[Z\] done
clear
D)
\[Y\] will reduce both \[X\] and \[Z\] done
clear
View Solution play_arrow
-
question_answer8)
The oxidation potential of a hydrogen electrode at pH = 10 and \[p{{H}_{1}}=1\] [JIPMER 2000]
A)
0.059 V done
clear
B)
0.59 V done
clear
C)
0.00 V done
clear
D)
0.51 V done
clear
View Solution play_arrow
-
question_answer9)
The decomposition of hydrogen peroxide is an example of [Roorkee 2000]
A)
Exothermic reaction done
clear
B)
Endothermic reaction done
clear
C)
Negative catalysis done
clear
D)
Auto-oxidation done
clear
View Solution play_arrow
-
question_answer10)
Aluminium oxide may be electrolysed at 1000°C to furnish aluminium metal (At. Mass = 27 amu; 1 Faraday = 96,500 Coulombs). The cathode reaction is \[A{{l}^{3}}+3{{e}^{-}}\to Al{}^\circ \] To prepare 5.12kg of aluminium metal by this method would require [AIEEE 2005]
A)
\[5.49\times {{10}^{7}}C\] of electricity done
clear
B)
\[1.83\times {{10}^{7}}C\] of electricity done
clear
C)
\[5.49\times {{10}^{4}}C\] of electricity done
clear
D)
\[5.49\times {{10}^{1}}C\] of electricity done
clear
View Solution play_arrow
-
question_answer11)
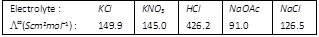
Calculate \[\Lambda _{HOAc}^{\infty }\] using appropriate molar conductances of the electrolytes listed above at infinite dilution in \[{{H}_{2}}O\] at 25°C [AIEEE 2005]
A)
517.2 done
clear
B)
552.7 done
clear
C)
390.7 done
clear
D)
217.5 done
clear
View Solution play_arrow
-
question_answer12)
The mass of carbon anode consumed (giving only carbondioxide) in the production of 270kg of aluminium metal from bauxite by the Hall process is [CBSE PMT 2005]
A)
180kg done
clear
B)
270kg done
clear
C)
540kg done
clear
D)
90kg done
clear
View Solution play_arrow
-
question_answer13)
4.5g of aluminium (at mass 27amu) is deposited at cathode from \[A{{l}^{3+}}\] solution by a certain quantity of electric charge. The volume of hydrogen produced at STP from \[{{H}^{+}}\] ions in solution by the same quantity of electric charge will be [CBSE PMT 2005]
A)
22.4 L done
clear
B)
44.8 L done
clear
C)
5.6 L done
clear
D)
11.2 L done
clear
View Solution play_arrow
-
question_answer14)
What amount of Cl2 gas liberated at anode, if 1 amp. current is passed for 30 min. from NaCl solution. [BHU 2005]
A)
0.66 moles done
clear
B)
0.33 moles done
clear
C)
0.66 g done
clear
D)
0.33 g done
clear
View Solution play_arrow
