question_answer 1) A particle of mass \[m\] moving with velocity \[v\] collides with a stationary particle of mass\[2m\]. The speed of the system, will be:
A)
\[3v\]
done
clear
B)
\[\frac{v}{2}\]
done
clear
C)
\[v/3\]
done
clear
D)
\[2v\]
done
clear
View Answer play_arrow
question_answer 2) The dimensions of gravitational constant are :
A)
\[M{{L}^{3}}{{T}^{-2}}\]
done
clear
B)
\[{{M}^{-1}}{{L}^{2}}{{T}^{-2}}\]
done
clear
C)
\[M{{L}^{-2}}{{T}^{2}}\]
done
clear
D)
\[{{M}^{-1}}{{L}^{3}}{{T}^{-2}}\]
done
clear
View Answer play_arrow
question_answer 3) A stone is thrown with an initial speed of \[4.9\,\,m/s\] from a bridge in vertically upward direction. It falls down in water after\[2\sec \]. The height of bridge is:
A)
\[24.7\,\,m\]
done
clear
B)
\[19.8\,\,m\]
done
clear
C)
\[9.8\,\,m\]
done
clear
D)
\[4.9\,\,m\]
done
clear
View Answer play_arrow
question_answer 4) A ball of mass \[10\,\,g\] moving with acceleration of \[20m/{{s}^{2}}\] is hit by a force which acts on it for 0.1 sec. The impulsive force is:
A)
\[1.2\,\,Ns\]
done
clear
B)
\[0.3\,\,Ns\]
done
clear
C)
\[0.1\,\,Ns\]
done
clear
D)
\[0.5\,\,Ns\]
done
clear
View Answer play_arrow
question_answer 5) The earth of mass \[=6\times {{10}^{24}}kg\] revolves around the sun with an angular velocity of \[2\times {{10}^{-7}}\,\,rad/s\] in a circular orbit of radius\[1.5\times {{10}^{-8}}\,\,km\]. The force exerted by the sun on earth is:
A)
\[6\times {{10}^{19}}N\]
done
clear
B)
\[18\times {{10}^{25}}N\]
done
clear
C)
\[36\times {{10}^{21}}N\]
done
clear
D)
\[27\times {{10}^{39}}N\]
done
clear
View Answer play_arrow
question_answer 6) A particle moves from position\[{{\overset{\to }{\mathop{r}}\,}_{1}}=3\mathbf{\hat{i}}+2\widehat{\mathbf{j}}-6\widehat{\mathbf{k}}\]to position\[\overset{\to }{\mathop{{{r}_{2}}}}\,=14\widehat{\mathbf{i}}+13\widehat{\mathbf{j}}+9\widehat{\mathbf{k}}\]under action of force\[\overset{\to }{\mathop{\mathbf{F}}}\,=4\widehat{\mathbf{i}}+\widehat{\mathbf{j}}+3\widehat{\mathbf{k}}\,\,N\]. The work done will be:
A)
\[100\,\,\text{J}\]
done
clear
B)
\[50\,\,\text{J}\]
done
clear
C)
\[200\,\,\text{J}\]
done
clear
D)
\[75\,\,\text{J}\]
done
clear
View Answer play_arrow
question_answer 7) A cylinder contains \[10\,\,kg\] of gas at pressure of\[107N/{{m}^{2}}\]. The quantity of gas taken out of the cylinder, if final pressure is\[2.5\times {{10}^{6}}N/{{m}^{2}}\], will be (temperature of gas is constant):
A)
\[15.2\,\,kg\]
done
clear
B)
\[3.7\,\,kg\]
done
clear
C)
\[zero\]
done
clear
D)
\[7.5\,\,kg\]
done
clear
View Answer play_arrow
question_answer 8) In junction diode, the holes are because of:
A)
missing electrons
done
clear
B)
extra electrons
done
clear
C)
protons
done
clear
D)
neutrons
done
clear
View Answer play_arrow
question_answer 9) The work done in pulling up a block of wood weighing \[2\,\,kN\] for a length of \[10\,\,m\] on a smooth plane inclined at an angle of \[{{15}^{o}}\] with the horizontal is:
A)
\[9.82\,\,kJ\]
done
clear
B)
\[89\,\,kJ\]
done
clear
C)
\[4.35\,\,kJ\]
done
clear
D)
\[5.17\,\,kJ\]
done
clear
View Answer play_arrow
question_answer 10) If the heat of \[110\,\,J\] is added to a gaseous system, whose internal energy is\[40\,\,J\], then the amount of external work done is:
A)
\[180\,\,\text{J}\]
done
clear
B)
\[70\,\,\text{J}\]
done
clear
C)
\[110\,\,\text{J}\]
done
clear
D)
\[30\,\,\text{J}\]
done
clear
View Answer play_arrow
question_answer 11) A resonance air column of length \[40\,\,cm\] resonates with a tuning fork of frequency\[450\,\,Hz\]. Ignoring end correction, the velocity of sound in air will be:
A)
\[1020\,\,m/s\]
done
clear
B)
\[920\,\,m/s\]
done
clear
C)
\[820\,\,m/s\]
done
clear
D)
\[720\,\,m/s\]
done
clear
View Answer play_arrow
question_answer 12) If vibration of string are to be two triose, then tension in the string must be made:
A)
eight times
done
clear
B)
four times
done
clear
C)
twice
done
clear
D)
half
done
clear
View Answer play_arrow
question_answer 13) A siren emitting sound of frequency 500 Hz is going away from a stationary listener with a speed of\[50\,\,m/s\], the frequency of sound to be heard directly from the siren is:
A)
\[286.5\,\,Hz\]
done
clear
B)
\[481\,\,Hz\]
done
clear
C)
\[434.2\,\,Hz\]
done
clear
D)
\[580\,\,Hz\]
done
clear
View Answer play_arrow
question_answer 14) The half-life of a radioactive substance is 3.6 days. How much of \[20\,\,mg\] of this radioactive substance will remain after 36 days:
A)
\[0.0019\,\,mg\]
done
clear
B)
\[1.019\,\,mg\]
done
clear
C)
\[1.109\,\,mg\]
done
clear
D)
\[0.019\,\,mg\]
done
clear
View Answer play_arrow
question_answer 15) The kinetic energy of an electron is\[5\,\,eV\]. Calculate the de-Broglie wavelength associated with it (\[h=6.6\times {{10}^{-34}}\,\,Js\],\[{{m}_{e}}=9.1\times {{10}^{-31}}kg\]):
A)
\[5.47\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[10.9\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[2.7\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 16) A charged particle is suspended in equilibrium in a uniform vertical electric field of intensity\[20000\,\,V/m\]. If mass of the particle is \[9.6\times {{10}^{-16}}kg\], the charge on it and excess number of electrons on the particle are respectively\[(g=10m/s)\]:
A)
\[4.8\times {{10}^{-19}}C,\,\,3\]
done
clear
B)
\[5.8\times {{10}^{-19}}C,\,\,4\]
done
clear
C)
\[3.8\times {{10}^{-19}}C,\,\,2\]
done
clear
D)
\[2.8\times {{10}^{-19}}C,\,\,1\]
done
clear
View Answer play_arrow
question_answer 17) The energy in \[MeV\] is released due to transformation of \[1\,\,kg\] mass completely into energy\[(c=3\times {{10}^{8}}m/s)\]:
A)
\[7.625\times 10\,\,MeV\]
done
clear
B)
\[10.5\times {{10}^{29}}\,\,MeV\]
done
clear
C)
\[2.8\times {{10}^{-28}}\,\,MeV\]
done
clear
D)
\[5.625\times {{10}^{29}}\,\,MeV\]
done
clear
View Answer play_arrow
question_answer 18) \[20\,\,kV\] potential is applied across \[X-\]ray tube, the minimum wavelength of \[X-\]ray emitted will be:
A)
\[0.62\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[0.37\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[1.62\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[1.31\,\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
question_answer 19) In the \[CB\] mode of a transistor, when the collector voltage is changed by\[0.5\,\,volt\]. The collector current changes by\[0.05\,\,mA\]. The output resistance will be:
A)
\[10\,\,k\Omega \]
done
clear
B)
\[20\,\,k\Omega \]
done
clear
C)
\[5\,\,k\Omega \]
done
clear
D)
\[2.5\,\,k\Omega \]
done
clear
View Answer play_arrow
question_answer 20) A transmitting station transmits radio waves of wavelength\[360\,\,m\]. Calculate the inductance of coil required with a condenser of capacity \[1.20\mu F\] in the resonant circuit to receive them:
A)
\[3.07\times {{10}^{-8}}H\]
done
clear
B)
\[2.07\times {{10}^{-8}}H\]
done
clear
C)
\[4.07\times {{10}^{-8}}H\]
done
clear
D)
\[6.07\times {{10}^{-8}}H\]
done
clear
View Answer play_arrow
question_answer 21) A circular coil of mean radius of\[7\,\,cm\] and having 4000 turns is rotated at the rate of 1800 revolutions per minute in the earths magnetic field (\[B=0.5\]gauss), the emf induced in coil will be:
A)
\[1.158\,\,V\]
done
clear
B)
\[0.58\,\,V\]
done
clear
C)
\[0.29\,\,V\]
done
clear
D)
\[5.8\,\,V\]
done
clear
View Answer play_arrow
question_answer 22) The peak value of alternating current is \[5\sqrt{2}\] ampere. The mean square value of current will be:
A)
\[5\,\,A\]
done
clear
B)
\[2.5\,\,A\]
done
clear
C)
\[5\sqrt{2}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 23) A lamp in which \[10\,\,amp\] current can flow at\[15\,\,V\], is connected with an alternating source of potential\[220\,\,V\]. The frequency of source is\[50Hz\]. The impedance of choke coil required to light the bulb is:
A)
\[0.07\,\,H\]
done
clear
B)
\[0.14\,\,H\]
done
clear
C)
\[0.28\,\,H\]
done
clear
D)
\[1.07\,\,H\]
done
clear
View Answer play_arrow
question_answer 24) If the current \[30\,\,A\] flowing in the primary coil is made zero is\[0.1\,\,\sec \]. The emf induced in the secondary coil is\[1.5\,\,volt\]. The mutual inductance between the coil is:
A)
\[0.05H\]
done
clear
B)
\[1.05H\]
done
clear
C)
\[0.1H\]
done
clear
D)
\[0.2H\]
done
clear
View Answer play_arrow
question_answer 25) Two equal resistances \[R\] are joined with voltage source \[V\] in (i) series (h) parallel, the ratio of electrical power consumed in two cases will be:
A)
\[1:4\]
done
clear
B)
\[4:1\]
done
clear
C)
\[2:1\]
done
clear
D)
\[2:1\]
done
clear
View Answer play_arrow
question_answer 26) A proton enters a magnetic field of intensity \[1.5\,\,wb/{{m}^{2}}\] with a velocity \[2\times 10\,\,m/s\] in a direction at an angle \[{{30}^{o}}\] with the field. The force on the proton will be (charge on proton is\[1.6\times {{10}^{-19}}C\]):
A)
\[2.4\times {{10}^{-12}}N\]
done
clear
B)
\[4.8\times {{10}^{-12}}N\]
done
clear
C)
\[1.2\times {{10}^{-12}}N\]
done
clear
D)
\[7.2\times {{10}^{-12}}N\]
done
clear
View Answer play_arrow
question_answer 27) 20 ampere current is flowing in a long straight wire. The intensity of magnetic field at a distance \[10\,\,cm\] from the wire will be:
A)
\[2\times {{10}^{-5}}wb/{{m}^{2}}\]
done
clear
B)
\[9\times {{10}^{-5}}wb/{{m}^{2}}\]
done
clear
C)
\[8\times {{10}^{-5}}wb/{{m}^{2}}\]
done
clear
D)
\[6\times {{10}^{-5}}wb/{{m}^{2}}\]
done
clear
View Answer play_arrow
question_answer 28) The resistance of a galvanometer is\[50\Omega \]. When \[0.01\,\,A\] current flows in it, full scale deflection is obtained in galvanometer, the resistance of shunt connected to convert galvanometer into an ammeter of range\[5\,\,A\], will be:
A)
\[0.1\Omega \]
done
clear
B)
\[0.2\Omega \]
done
clear
C)
\[0.3\Omega \]
done
clear
D)
\[0.4\Omega \]
done
clear
View Answer play_arrow
question_answer 29) When a resistance of \[\text{a}\] \[\Omega \] is connected at the ends of a battery, its potential difference decreases from \[40\,\,V\] to\[30\,\,V\]. The internal resistance of the battery is:
A)
\[3\,\,\Omega \]
done
clear
B)
\[6\,\,\Omega \]
done
clear
C)
\[1.5\,\,\Omega \]
done
clear
D)
\[4\,\,\Omega \]
done
clear
View Answer play_arrow
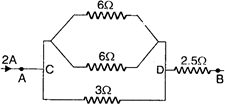
question_answer 30)
The equivalent resistance and potential difference between \[A\] and \[B\] for the circuit is respectively:
A)
\[4\,\,\Omega ,\,\,8\,\,V\]
done
clear
B)
\[8\,\,\Omega ,\,\,4\,\,V\]
done
clear
C)
\[2\,\,\Omega ,\,\,2\,\,V\]
done
clear
D)
\[16\,\,\Omega ,\,\,8\,\,V\]
done
clear
View Answer play_arrow
question_answer 31) A wire of resistance \[R\] is divided in equal parts, then these parts are joined in parallel, the equivalent resistance of the combination will be:
A)
\[nR\]
done
clear
B)
\[\frac{R}{n}\]
done
clear
C)
\[\frac{n}{R}\]
done
clear
D)
\[\frac{R}{{{n}^{2}}}\]
done
clear
View Answer play_arrow
question_answer 32) The capacity of an air condenser is\[2.0\mu F\]. If a medium is placed between its plates. The capacity becomes\[12\mu F\]. The dielectric constant of the medium will be:
A)
5
done
clear
B)
4
done
clear
C)
3
done
clear
D)
6
done
clear
View Answer play_arrow
question_answer 33) Taking the earth to be a spherical conductor of diameter\[12.8\times {{10}^{3}}km\]. Its capacity will be:
A)
\[711\,\,\mu F\]
done
clear
B)
\[611\,\,\mu F\]
done
clear
C)
\[811\,\,\mu F\]
done
clear
D)
\[511\,\,\mu F\]
done
clear
View Answer play_arrow
question_answer 34) The ratio of electrostatic and gravitational forces acting between electron and proton separated by a distance\[5\times {{10}^{-11}}m\], will be: (Charge on electron\[=1.6\times {{10}^{-19}}C\], mass of electron\[=9.1\times {{10}^{-31}}kg\], mass of proton\[=1.6\times {{10}^{-27}}\,\,kg\],\[G=6.7\times {{10}^{-11}}N{{m}^{2}}/k{{g}^{2}}\])
A)
\[2.36\times {{10}^{39}}\]
done
clear
B)
\[2.36\times {{10}^{40}}\]
done
clear
C)
\[2.34\times {{10}^{41}}\]
done
clear
D)
\[2.34\times {{10}^{42}}\]
done
clear
View Answer play_arrow
question_answer 35) A student can distinctly see the object upto a distance\[15\,\,cm\]. He wants to see the black board at a distance of\[3\,\,m\]. Focal length and power of lens used respectively will be:
A)
\[-4.8\,\,cm,\,\,-3.3\,\,D\]
done
clear
B)
\[-5.8\,\,cm,\,\,-4.3\,\,D\]
done
clear
C)
\[-7.5\,\,cm,\,\,-6.3\,\,D\]
done
clear
D)
\[-15.8\,\,cm,\,\,-6.33\,\,D\]
done
clear
View Answer play_arrow
question_answer 36) A this glass prism \[(\mu =1.5)\] in the position of minimum deviation deviates the monochromatic light ray by\[{{10}^{o}}\], the refracting angle of prism is:
A)
\[{{20}^{o}}\]
done
clear
B)
\[{{10}^{o}}\]
done
clear
C)
\[{{30}^{o}}\]
done
clear
D)
\[{{45}^{o}}\]
done
clear
View Answer play_arrow
question_answer 37) Two thin lenses of powers \[12\,\,D\] and \[-2\,\,D\] respectively are placed in contact, the power, focal length and nature respectively will be:
A)
\[8\,\,D,\,\,0.8\,\,m,\]convex
done
clear
B)
\[14\,\,D,\,\,0.5\,\,m,\]convex
done
clear
C)
\[5\,\,D,\,\,0.2\,\,m,\]convex
done
clear
D)
\[10\,\,D,\,\,0.1\,\,m,\]convex
done
clear
View Answer play_arrow
question_answer 38) Amount of work which can be obtained from \[200\,\,cal\] heat, will be:
A)
\[280\,\,\text{J}\]
done
clear
B)
\[800\,\,\text{J}\]
done
clear
C)
\[420\,\,\text{J}\]
done
clear
D)
\[840\,\,\text{J}\]
done
clear
View Answer play_arrow
question_answer 39) The spring extends by \[x\] on loading, then energy stored by the spring is: (if \[T\] is the tension in spring and \[k\] is spring constant)
A)
\[\frac{{{T}^{2}}}{2k}\]
done
clear
B)
\[\frac{{{T}^{2}}}{2{{k}^{2}}}\]
done
clear
C)
\[\frac{2k}{{{T}^{2}}}\]
done
clear
D)
\[\frac{2{{T}^{2}}}{k}\]
done
clear
View Answer play_arrow
question_answer 40) The kinetic energy of one molecule of a gas at normal temperature and pressure will be \[(k=8.31\,\,\text{J}\]/mole\[k\]):
A)
\[1.7\times {{10}^{3}}\text{J}\]
done
clear
B)
\[10.2\times {{10}^{3}}\text{J}\]
done
clear
C)
\[3.4\times {{10}^{3}}\text{J}\]
done
clear
D)
\[6.8\times {{10}^{3}}\text{J}\]
done
clear
View Answer play_arrow
question_answer 41) The frequency of a light ray is\[6\times {{10}^{14}}Hz\]. Its frequency when it propagates in a medium of refractive index \[1.5\] will be:
A)
\[1.67\times {{10}^{14}}Hz\]
done
clear
B)
\[9\times {{10}^{4}}Hz\]
done
clear
C)
\[6\times {{10}^{14}}Hz\]
done
clear
D)
\[4\times {{10}^{14}}Hz\]
done
clear
View Answer play_arrow
question_answer 42) Shorter wavelength is of:
A)
infrared rays
done
clear
B)
\[\gamma -\]rays
done
clear
C)
visible light
done
clear
D)
\[X-\]rays
done
clear
View Answer play_arrow
question_answer 43) A light of wavelength \[5890\overset{\text{o}}{\mathop{\text{A}}}\,\] falls normally on a thin air film. The minimum thickness of the film such that the film appears dark in reflected light:
A)
\[2.945\times {{10}^{-7}}m\]
done
clear
B)
\[3.945\times {{10}^{-7}}m\]
done
clear
C)
\[4.945\times {{10}^{-7}}m\]
done
clear
D)
\[1.945\times {{10}^{-7}}m\]
done
clear
View Answer play_arrow
question_answer 44) A Carnots engine works between temperature \[{{72}^{o}}C\] and\[{{27}^{o}}C\], the efficiency of the engine is:
A)
\[100%\]
done
clear
B)
\[90%\]
done
clear
C)
\[35%\]
done
clear
D)
\[70%\]
done
clear
View Answer play_arrow
question_answer 45) de-Broglie wavelength of a body of mass \[1\,\,kg\] moving with velocity of \[2000\,\,m/s\] is:
A)
\[3.32\times {{10}^{-27}}\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[1.5\times {{10}^{-27}}\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[0.55\times {{10}^{-22}}\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 46) A wave is represented by the equation \[y=a\sin \,\,(0.01x-2t)\] where \[a\] and \[x\] are in \[cm\], velocity of propagation is:
A)
\[200\,\,cm/s\]
done
clear
B)
\[100\,\,cm/s\]
done
clear
C)
\[50\,\,cm/s\]
done
clear
D)
\[10\,\,cm/s\]
done
clear
View Answer play_arrow
question_answer 47) A mass m attached to a spring oscillate every\[2\sec \]. If the mass increases by\[2\,\,kg\], then time period increases by\[1\sec \]. The initial mass of the body is:
A)
\[12.6\,\,kg\]
done
clear
B)
\[9.6\,\,kg\]
done
clear
C)
\[3.2\,\,kg\]
done
clear
D)
\[1.6\,\,kg\]
done
clear
View Answer play_arrow
question_answer 48) The isothermal bulk modulus of a gas at atmospheric pressure is:
A)
\[2.026\times {{10}^{5}}N/{{m}^{2}}\]
done
clear
B)
\[1.013\times {{10}^{5}}N/{{m}^{2}}\]
done
clear
C)
\[1mm\]of\[Hg\]
done
clear
D)
\[13.6\,\,mm\]of\[Hg\]
done
clear
View Answer play_arrow
question_answer 49) If a body of mass \[3\,\,kg\] is dropped from the top of a tower of height \[25\,\,m\], then kinetic energy after \[3\sec \] will be:
A)
\[557\,\,\text{J}\]
done
clear
B)
\[246\,\,\text{J}\]
done
clear
C)
\[1048\,\,\text{J}\]
done
clear
D)
\[1297\,\,\text{J}\]
done
clear
View Answer play_arrow
question_answer 50) A \[500\,\,kg\] can takes a turn of radius \[50\,\,m\] with velocity of\[36\,\,km/hr\]. The centripetal force is:
A)
1200 N
done
clear
B)
1000 N
done
clear
C)
750 N
done
clear
D)
250 N
done
clear
View Answer play_arrow
question_answer 51) The significant figures in \[1.0024,\,\,1.240\] and \[0.0020\]are respectively:
A)
\[5,\,\,4,\,\,2\]
done
clear
B)
\[3,\,\,3,\,\,1\]
done
clear
C)
\[5,\,\,3,\,\,4\]
done
clear
D)
\[5,\,\,3,\,\,2\]
done
clear
View Answer play_arrow
question_answer 52) Bauxite is an ore of:
A)
boron
done
clear
B)
barium
done
clear
C)
aluminium
done
clear
D)
bismuth
done
clear
View Answer play_arrow
question_answer 53) golf-ball weigh\[40.0\,\,g\]. If it is moving with a velocity of \[20.0\,\,m{{s}^{-1}}\], its de-Broglie wavelength is:
A)
\[1.66\times {{10}^{-34}}\,\,nm\]
done
clear
B)
\[8.28\times {{10}^{-32}}\,\,nm\]
done
clear
C)
\[8.28\times {{10}^{-25}}\,\,nm\]
done
clear
D)
\[1.66\times {{10}^{-24}}\,\,nm\]
done
clear
View Answer play_arrow
question_answer 54) Which of the following elements, is a component of a compound essential for photosynthesis?
A)
Iron
done
clear
B)
Calcium
done
clear
C)
Barium
done
clear
D)
Magnesium
done
clear
View Answer play_arrow
question_answer 55) Which of the following, is a mordant dye?
A)
Aniline black
done
clear
B)
Congo-red
done
clear
C)
Alizarin
done
clear
D)
Indigo
done
clear
View Answer play_arrow
question_answer 56) Which is the shortest wavelength line in the Lyman series of the hydrogen spectrum? \[(R=1.097\times {{10}^{-2}}n{{m}^{-1}})\]
A)
\[94.21nm\]
done
clear
B)
\[91.16nm\]
done
clear
C)
\[911.6nm\]
done
clear
D)
\[933.6nm\]
done
clear
View Answer play_arrow
question_answer 57) Which of the following order of ionisation energy is correct?
A)
\[B<Be<N<C\]
done
clear
B)
\[Be>B>C>N\]
done
clear
C)
\[B<Be<C<N\]
done
clear
D)
\[Be<B<C<N\]
done
clear
View Answer play_arrow
question_answer 58) Which of the following turns potassium dichromate paper green?
A)
\[N{{O}_{2}}\]
done
clear
B)
\[HCl\]
done
clear
C)
\[N{{H}_{3}}\]
done
clear
D)
\[S{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 59) The correct order of stability of carbocations, is:
A)
\[1{}^\circ <2{}^\circ <3{}^\circ <\overset{+}{\mathop{C}}\,{{H}_{3}}\]
done
clear
B)
\[\overset{+}{\mathop{C}}\,{{H}_{3}}<1{}^\circ <2{}^\circ <3{}^\circ \]
done
clear
C)
\[3{}^\circ <2{}^\circ <1{}^\circ <\overset{+}{\mathop{C}}\,{{H}_{3}}\]
done
clear
D)
\[2{}^\circ <3{}^\circ <1{}^\circ <\overset{+}{\mathop{C}}\,{{H}_{3}}\]
done
clear
View Answer play_arrow
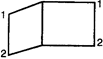
question_answer 60)
The \[IUPAC\] name of the following compound is:
A)
bicycio \[[2\,\,2.0]\] octane
done
clear
B)
bicycio\[[0.2.2]\] hexane
done
clear
C)
bicycio \[[2.1.1]\] hexane
done
clear
D)
bicycio \[[2.2.0]\] hexane
done
clear
View Answer play_arrow
question_answer 61) The following compound cannot exhibit: \[C{{H}_{3}}CH=CH-CH(OH)\cdot COOH\]
A)
geometrical isomerism
done
clear
B)
optical isomerism
done
clear
C)
metamerism
done
clear
D)
position isomerism
done
clear
View Answer play_arrow
question_answer 62) Volume of \[C{{O}_{2}}\] obtained from complete decomposition of \[20\,\,g\,\,CaC{{O}_{3}}\] at STP is:
A)
\[2.24\,\,litre\]
done
clear
B)
\[4.48\,\,litre\]
done
clear
C)
\[20\,\,litre\]
done
clear
D)
\[22.4\,\,litre\]
done
clear
View Answer play_arrow
question_answer 63) The shape of an orbital is determined by :
A)
\[n\]
done
clear
B)
\[l\]
done
clear
C)
\[m\]
done
clear
D)
\[s\]
done
clear
View Answer play_arrow
question_answer 64) Oxidation number \[Fe\] in \[{{K}_{3}}[Fe{{(CN)}_{6}}]\] is:
A)
\[+1\]
done
clear
B)
\[+2\]
done
clear
C)
\[+3\]
done
clear
D)
\[+4\]
done
clear
View Answer play_arrow
question_answer 65) The enthalpies of all elements in their standard states are:
A)
zero
done
clear
B)
\[>0\]
done
clear
C)
\[<0\]
done
clear
D)
unity
done
clear
View Answer play_arrow
question_answer 66) Which of the following, is not a unit of pressure?
A)
Atmosphere
done
clear
B)
Torr
done
clear
C)
Pascal
done
clear
D)
Newton
done
clear
View Answer play_arrow
question_answer 67) The \[Ka\] of an acid is\[3.2\times {{10}^{-5}}\]. The degree of dissociation of the acid at concentration of \[0.2\,\,M\]is:
A)
\[6.0\times {{10}^{-2}}\]
done
clear
B)
\[1.26\times {{10}^{-2}}\]
done
clear
C)
\[40\times {{10}^{-4}}\]
done
clear
D)
\[0.04\times {{10}^{-3}}\]
done
clear
View Answer play_arrow
question_answer 68) Which of the following ions, has smallest size?
A)
\[C{{a}^{2+}}\]
done
clear
B)
\[C{{l}^{-}}\]
done
clear
C)
\[{{S}^{2-}}\]
done
clear
D)
\[{{K}^{+}}\]
done
clear
View Answer play_arrow
question_answer 69) Which of the following acids is monobasic?
A)
\[{{H}_{3}}P{{O}_{3}}\]
done
clear
B)
\[{{H}_{3}}P{{O}_{2}}\]
done
clear
C)
\[{{H}_{2}}S{{O}_{3}}\]
done
clear
D)
\[{{H}_{3}}P{{O}_{4}}\]
done
clear
View Answer play_arrow
question_answer 70) Which of the following is paramagnetic?
A)
\[CO\]
done
clear
B)
\[{{N}_{2}}\]
done
clear
C)
\[{{O}_{2}}\]
done
clear
D)
\[{{H}_{2}}{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 71) Yellow phosphorus is kept in:
A)
water
done
clear
B)
kerosene
done
clear
C)
ether
done
clear
D)
alcohol
done
clear
View Answer play_arrow
question_answer 72) The oxide that gives \[{{H}_{2}}{{O}_{2}}\] on treatment with dilute acid is:
A)
\[Pb{{O}_{2}}\]
done
clear
B)
\[Mn{{O}_{2}}\]
done
clear
C)
\[N{{a}_{2}}{{O}_{2}}\]
done
clear
D)
\[Ti{{O}_{2}}\]
done
clear
View Answer play_arrow
question_answer 73) Which of the following dissolves in hot \[conc.\,\,NaOH\] solution?
A)
\[Fe\]
done
clear
B)
\[Zn\]
done
clear
C)
\[Cu\]
done
clear
D)
\[Ag\]
done
clear
View Answer play_arrow
question_answer 74) \[C{{H}_{3}}CHC{{l}_{2}}\], on hydrolysis, will give:
A)
\[C{{H}_{3}}CHO\]
done
clear
B)
\[C{{H}_{3}}COOH\]
done
clear
C)
\[CHC{{l}_{3}}\]
done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}OH\]
done
clear
View Answer play_arrow
question_answer 75) The compound which reacts faster with Lucas reagent is:
A)
butan-1-ol
done
clear
B)
butan-2-ol
done
clear
C)
2-methyl-propan-l-ol
done
clear
D)
2-methyl propan-2-ol
done
clear
View Answer play_arrow
question_answer 76) What is obtained, when potassium succinate solution is electrolysed?
A)
Ethane
done
clear
B)
Ethene
done
clear
C)
Ethyne
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 77) Which of the following has the maximum acidic strength?
A)
\[p-\]nitrophenol
done
clear
B)
\[p-\]nitrobenzoic acid
done
clear
C)
\[o-\]nitrobenzoic acid
done
clear
D)
\[m-\]nitrobenzoic acid
done
clear
View Answer play_arrow
question_answer 78) Which of the following statement is not correct?
A)
Adsorption energy for chemical adsorption is generally greater than that of physical adsorption
done
clear
B)
Physical adsorption is due to van der Waal forces
done
clear
C)
Physical adsorption decreases at high temperature and low pressure
done
clear
D)
Physical adsorption is irreversible
done
clear
View Answer play_arrow
question_answer 79) An element \[Y\] emits one \[\alpha -\]and two \[\beta -\]particles to give \[X\].\[X\] and \[Y\] are:
A)
isotones
done
clear
B)
isobars
done
clear
C)
isotopes
done
clear
D)
iso electronics
done
clear
View Answer play_arrow
question_answer 80) For a reaction, the half-life is independent of initial concentration. What is the order of that reaction?
A)
Zero
done
clear
B)
One
done
clear
C)
Two
done
clear
D)
Three
done
clear
View Answer play_arrow
question_answer 81) Addition of a non-volatile solute causes lowering in vapour pressure of a solvent from\[0.8\,\,atm.\] to\[0.2\,\,atm\]. What is the mole fraction of solvent?
A)
\[0.25\]
done
clear
B)
\[0.75\]
done
clear
C)
\[0.50\]
done
clear
D)
Cannot be predicted
done
clear
View Answer play_arrow
question_answer 82) Which of the following has \[s{{p}^{3}}-\]hybridisation?
A)
\[BC{{l}_{3}}\]
done
clear
B)
\[PC{{l}_{3}}\]
done
clear
C)
\[BeC{{l}_{2}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 83) The \[pH\] of \[{{10}^{-8}}\,\,M\,\,NaOH\] is:
A)
\[8.0\]
done
clear
B)
\[6.0\]
done
clear
C)
\[6.96\]
done
clear
D)
\[7.04\]
done
clear
View Answer play_arrow
question_answer 84) For the reaction,\[2N{{O}_{2}}2NO+{{O}_{2}}\], the value of\[{{K}_{p}}\]is equal to\[K\]. What will the value of \[{{K}_{p}}\] for reaction, \[NO+\frac{1}{2}{{O}_{2}}N{{O}_{2}}\]
A)
\[\sqrt{K}\]
done
clear
B)
\[\frac{1}{K}\]
done
clear
C)
\[\sqrt{\frac{1}{K}}\]
done
clear
D)
\[\frac{1}{{{K}^{2}}}\]
done
clear
View Answer play_arrow
question_answer 85) Two metals \[A\] and \[B\] have \[E_{red}^{\text{o}}\] value equal to \[0.36\,\,V\] and\[1.23\,\,V\]. What will be the correct cell equation?
A)
\[A+{{B}^{2}}\xrightarrow{{}}{{A}^{2}}+B\]
done
clear
B)
\[{{A}^{2+}}+B\xrightarrow{{}}A+{{B}^{2+}}\]
done
clear
C)
Both [a] and [b]
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 86) Lunar caustic is:
A)
\[AgCl\]
done
clear
B)
\[AgN{{O}_{3}}\]
done
clear
C)
\[NaOH\]
done
clear
D)
\[KN{{O}_{3}}\]
done
clear
View Answer play_arrow
question_answer 87) Which of the following forms molecular solid?
A)
\[S{{O}_{2}}\]
done
clear
B)
\[Si{{O}_{2}}\]
done
clear
C)
\[SiC\]
done
clear
D)
Diamond
done
clear
View Answer play_arrow
question_answer 88) \[C{{H}_{3}}COOH\xrightarrow{P/B{{r}_{2}}}C{{H}_{2}}BrCOOH\] This reaction is called:
A)
Schotton-Bauman reaction
done
clear
B)
Finkelstin reaction
done
clear
C)
Hell-Volhard-Zelinsky reaction
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 89) What is \[X\] in the following sequence? \[Ar{{N}_{2}}Cl\xrightarrow[KCN]{CuCN}Z\xrightarrow{H.OH}Y\]\[\xrightarrow[\Delta ]{\text{soda-lime}}X\]
A)
Benzoic acid
done
clear
B)
Sodium benzoate
done
clear
C)
Benzaldehyde
done
clear
D)
Benzene
done
clear
View Answer play_arrow
question_answer 90) Solvent loving colloids are known as:
A)
hydrophobic
done
clear
B)
lyophilic
done
clear
C)
hydrophillic
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 91) If \[\Delta H>0\] and\[\Delta S>0\], in which of the following cases, can a reaction be spontaneous?
A)
\[\Delta H>T.\Delta S\]
done
clear
B)
\[\Delta H=T.\Delta S\]
done
clear
C)
\[\Delta H<T.\Delta S\]
done
clear
D)
The reaction will be non-spontaneous
done
clear
View Answer play_arrow
question_answer 92) Tin stone \[(Sn{{O}_{2}})\] is concentrated by:
A)
electromagnetic separation
done
clear
B)
gravity separation
done
clear
C)
roasting
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 93) Which type of isomerism is shown by\[CO{{(N{{H}_{3}})}_{6}}]C{{l}_{3}}\]?
A)
Coordination
done
clear
B)
Hydration
done
clear
C)
lonisation
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 94) When chlorobenzene is heated with chloral in presence of the little\[conc.{{H}_{2}}S{{O}_{4}}\], the product obtained is:
A)
DDT
done
clear
B)
BHC
done
clear
C)
lindane
done
clear
D)
melathion
done
clear
View Answer play_arrow
question_answer 95) Arrhenius equation can be represented as:
A)
\[k=A.{{e}^{-{{E}_{a}}/RT}}\]
done
clear
B)
\[\log \frac{{{k}_{2}}}{{{k}_{1}}}=\frac{{{E}_{a}}}{2.303R}\left[ \frac{{{T}_{2}}-{{T}_{1}}}{{{T}_{1}}{{T}_{2}}} \right]\]
done
clear
C)
\[\log k=\log A-\frac{{{E}_{a}}}{2.303RT}\]
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 96) Aureomycin is an:
A)
antibiotic
done
clear
B)
antiseptic
done
clear
C)
analeptic
done
clear
D)
carcinogenic agent
done
clear
View Answer play_arrow
question_answer 97) DNA is a polymer of:
A)
nucleosides
done
clear
B)
nucleotides
done
clear
C)
purine and pyrimidine bases
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 98) Dacron is polymer of:
A)
glycol and formaldehyde
done
clear
B)
glycol and phenol
done
clear
C)
glycol and phthalic acid
done
clear
D)
glycol and terephthalic acid
done
clear
View Answer play_arrow
question_answer 99) Which cell convert electrical energy into chemical energy?
A)
Voltaic cell
done
clear
B)
Electrolytic cell
done
clear
C)
Galvanic cell
done
clear
D)
Electrochemical cell
done
clear
View Answer play_arrow
question_answer 100) Molarity of pure water is:
A)
\[38.6\,\,M\]
done
clear
B)
\[12.4\,\,M\]
done
clear
C)
\[55.5\,\,M\]
done
clear
D)
cannot be determined
done
clear
View Answer play_arrow
question_answer 101) Mannitol is reserve food in:
A)
Rhodophyceae
done
clear
B)
Chlorophyceae
done
clear
C)
Phaeophyceae
done
clear
D)
Xanthophyceae
done
clear
View Answer play_arrow
question_answer 102) Plants growing on sand and gravel are called:
A)
eremophytes
done
clear
B)
psammophytes
done
clear
C)
psilophytes
done
clear
D)
oxylophytes
done
clear
View Answer play_arrow
question_answer 103) The inflorescence in Ocimum is:
A)
cyathium
done
clear
B)
verticillaster
done
clear
C)
hypanthodium
done
clear
D)
raceme
done
clear
View Answer play_arrow
question_answer 104) If the pollen tube enters the ovule through funiculus, the phenomenon is known as:
A)
porogamy
done
clear
B)
chalazogamy
done
clear
C)
mesogamy
done
clear
D)
heterogamy
done
clear
View Answer play_arrow
question_answer 105) The first phylogenetic system of plant classification was proposed by:
A)
Engler
done
clear
B)
Engler and Pranti
done
clear
C)
Eichler
done
clear
D)
Wetlstein
done
clear
View Answer play_arrow
question_answer 106) Cellulose fibre is obtained from Gossypium:
A)
stem surface
done
clear
B)
seed hair
done
clear
C)
leaf surface
done
clear
D)
root hair
done
clear
View Answer play_arrow
question_answer 107) In split genes the coding sequences are called:
A)
introns
done
clear
B)
exons
done
clear
C)
cistrons
done
clear
D)
operons
done
clear
View Answer play_arrow
question_answer 108) The real forces responsible for the movement of water from one cell to another cell is mainly:
A)
osmotic pressure (OP)
done
clear
B)
turgor pressure (TP)
done
clear
C)
diffusion pressure deficit (DPD)
done
clear
D)
imbibition
done
clear
View Answer play_arrow
question_answer 109) Which of the following is the most resistant material in pollen grain?
A)
sporopollenin
done
clear
B)
intine
done
clear
C)
exine
done
clear
D)
certicle
done
clear
View Answer play_arrow
question_answer 110) The amphibians of plant kingdom are:
A)
multicellular non motile algae
done
clear
B)
bryophytes with simple internal organisation
done
clear
C)
unicellular motile algae
done
clear
D)
pteridophytes with complex internal organisation
done
clear
View Answer play_arrow
question_answer 111) The root system growing near the base of the radicle in monocots is:
A)
haptera
done
clear
B)
anchoring roots
done
clear
C)
clinging roots
done
clear
D)
seminal roots
done
clear
View Answer play_arrow
question_answer 112) The point at which polytene chromosome appear, to be attached together is called
A)
centriole
done
clear
B)
centromere
done
clear
C)
chromomere
done
clear
D)
chromo centre
done
clear
View Answer play_arrow
question_answer 113) Black rust of wheat is a fungal disease caused by:
A)
Melampsora lini
done
clear
B)
Claviceps purpurea
done
clear
C)
Albugo Candida
done
clear
D)
Puccinia graminis tritici
done
clear
View Answer play_arrow
question_answer 114) Which of the following statement ii correct about DDT?
A)
It is a biomagnifying biodegradable pollutant
done
clear
B)
It is non biomagnifying biodegradable pollutant
done
clear
C)
It is biomanifying non-biodegradable pollutant
done
clear
D)
It is not a pollutant
done
clear
View Answer play_arrow
question_answer 115) In Chlorophyceae the mode of sexual reproduction is:
A)
anisogamy
done
clear
B)
oogamy
done
clear
C)
isogamy
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 116) Restriction enzymes are used in genetic engineering, because they:
A)
can degrade harmful proteins
done
clear
B)
can join different DNA flagments
done
clear
C)
can cut DNA at specific base sequence
done
clear
D)
are nucleases that cut DNA at variable sites
done
clear
View Answer play_arrow
question_answer 117) In which genera endospores are formed for reproduction?
A)
Mono coccus and Clostridium
done
clear
B)
Bacillus and Clostridium
done
clear
C)
Mucor and Bacillus
done
clear
D)
None of the above
done
clear
View Answer play_arrow
question_answer 118) Agricultural chemicals include:
A)
growth regulators
done
clear
B)
fertilizers
done
clear
C)
pesticides
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 119) In-1 condition represent :
A)
tetrasomy
done
clear
B)
trisomy
done
clear
C)
monosomy
done
clear
D)
multisomy
done
clear
View Answer play_arrow
question_answer 120) \[CO\] is harmful to human being because:
A)
it decreases \[C{{O}_{2}}\] concentration
done
clear
B)
it competes \[{{O}_{2}}\] to combine with haemoglobin
done
clear
C)
it is carcinogenic
done
clear
D)
it depletes \[{{O}_{3}}\] layer
done
clear
View Answer play_arrow
question_answer 121) Which of the following is a day neutral plant?
A)
Helianthus annuus
done
clear
B)
Euphorbia pulcherrima
done
clear
C)
Avena sativa
done
clear
D)
Beta vulgaris
done
clear
View Answer play_arrow
question_answer 122) Which of the following is an ant gibberellin?
A)
Auxin
done
clear
B)
ABA
done
clear
C)
Ethylene
done
clear
D)
Cytokinin
done
clear
View Answer play_arrow
question_answer 123) Heterospory is the production of:
A)
sexual and asexual spores
done
clear
B)
large and small spores
done
clear
C)
haploid and diploid spores
done
clear
D)
diploid and tetraploid spores
done
clear
View Answer play_arrow
question_answer 124) Algal zone is present in:
A)
normal root of Cycas
done
clear
B)
coralloid root of Cycas
done
clear
C)
normal root of Pinus
done
clear
D)
stem of Cycas
done
clear
View Answer play_arrow
question_answer 125) Double fertilization means:
A)
fusion of nucleus of male gamete with the egg nucleus
done
clear
B)
fusion of two polar nuclei
done
clear
C)
fusion of sperm nucleus with secondary nucleus
done
clear
D)
fusion of one sperm nucleus with nucleus and fusion of other sperm nucleus with secondary nucleus
done
clear
View Answer play_arrow
question_answer 126) \[\underset{+}{\overset{_{\uparrow }}{\mathop{O}}}\,{{K}_{(5)}}{{C}_{(5)}}{{A}_{5}}{{G}_{(2)}}\] is die floral formula of:
A)
Solanaceae
done
clear
B)
Asteraceae
done
clear
C)
Malvaceae
done
clear
D)
Cruciferae
done
clear
View Answer play_arrow
question_answer 127) The resolving power of a compound microscope is about:
A)
\[0.275\mu \]
done
clear
B)
\[0.5\mu \]
done
clear
C)
\[0.75\mu \]
done
clear
D)
\[1.5\mu \]
done
clear
View Answer play_arrow
question_answer 128) The magnitude of root pressure ranges from:
A)
\[2-5\,\,atm\]
done
clear
B)
\[1-5\,\,atm\]
done
clear
C)
\[0.1-0.2\,\,atm\]
done
clear
D)
\[4-6\,\,atm\]
done
clear
View Answer play_arrow
question_answer 129) Which of the following element plays an important role in nitrogen fixation?
A)
Zinc
done
clear
B)
Molybdenum
done
clear
C)
Manganese
done
clear
D)
Copper
done
clear
View Answer play_arrow
question_answer 130) The terminal electron acceptor during electron transport system is:
A)
oxygen
done
clear
B)
cyto\[{{a}_{3}}\]
done
clear
C)
cyto\[c\]
done
clear
D)
plasto quinone
done
clear
View Answer play_arrow
question_answer 131) The Orobanche plant is:
A)
partial stem parasite
done
clear
B)
total root parasite
done
clear
C)
symbiont
done
clear
D)
total stem parasite
done
clear
View Answer play_arrow
question_answer 132) Glycolysis occurs in:
A)
cytosol
done
clear
B)
mitochondria
done
clear
C)
golgi complex
done
clear
D)
ribosomes
done
clear
View Answer play_arrow
question_answer 133) Closure of lid of pitcher in pitcher plant is:
A)
tropic movement
done
clear
B)
paratonic movement
done
clear
C)
turgor movement
done
clear
D)
autonomous movement
done
clear
View Answer play_arrow
question_answer 134) Reindeer moss is common name for :
A)
Funaria hygrometrica
done
clear
B)
Marchantia sp
done
clear
C)
Cladonia rangifera
done
clear
D)
Polytrichum sp
done
clear
View Answer play_arrow
question_answer 135) Microorganism which act as a vehicle for foreign gene transfer in higher plants:
A)
Agrobacterium
done
clear
B)
E. coli
done
clear
C)
T.M.V.
done
clear
D)
Bacillus anthracis
done
clear
View Answer play_arrow
question_answer 136) DNA polymerase enzyme is required for synthesis of:
A)
DNA from RNA
done
clear
B)
RNA from DNA
done
clear
C)
DNA from DNA
done
clear
D)
RNA from RNA
done
clear
View Answer play_arrow
question_answer 137) In terrestrial ecosystem such as forest, maximum energy is found in which trophic level?
A)
\[{{T}_{1}}\]
done
clear
B)
\[{{T}_{2}}\]
done
clear
C)
\[{{T}_{3}}\]
done
clear
D)
\[{{T}_{4}}\]
done
clear
View Answer play_arrow
question_answer 138) The part of earth in which life exist is known as?
A)
Lithosphere
done
clear
B)
Biosphere
done
clear
C)
Atmosphere
done
clear
D)
Hydrosphere
done
clear
View Answer play_arrow
question_answer 139) Contour farming is usually employed in:
A)
hilly areas
done
clear
B)
sandy areas
done
clear
C)
sea beaches
done
clear
D)
all of these
done
clear
View Answer play_arrow
question_answer 140) Periderm includes:
A)
cork cambium only
done
clear
B)
cork cambium and cork
done
clear
C)
cork and secondary cortex
done
clear
D)
cork cambium, cork and secondary cortex
done
clear
View Answer play_arrow
question_answer 141) In some plants sleep movement of leaves is due to:
A)
excess of photosynthesis
done
clear
B)
osmotic changes at base of leaf
done
clear
C)
excess of respiration
done
clear
D)
excess of transpiration
done
clear
View Answer play_arrow
question_answer 142) Which stain shows negative bacteria during bacterial staining?
A)
White
done
clear
B)
Red
done
clear
C)
Black
done
clear
D)
Purple
done
clear
View Answer play_arrow
question_answer 143) Yeast belongs to which class?
A)
Basidiomycetes
done
clear
B)
Ascomycetes
done
clear
C)
Eumycetes
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 144) In Spirogyra sometimes a ladder like structure is present due to:
A)
vegetative reproduction
done
clear
B)
asexual reproduction
done
clear
C)
lateral conjugation
done
clear
D)
scalariform conjugation
done
clear
View Answer play_arrow
question_answer 145) In Hatch and Slack type plants\[C{{O}_{2}}\]acceptor is:
A)
malate
done
clear
B)
pyruvate
done
clear
C)
phosphoenol pyruvate
done
clear
D)
aspartate
done
clear
View Answer play_arrow
question_answer 146) In CAM plants:
A)
stomata open in day and close at night
done
clear
B)
stomata remain open in day and night both
done
clear
C)
stomata remain closed in day and night both
done
clear
D)
stomata open at night and close in day
done
clear
View Answer play_arrow
question_answer 147) Which type of DNA is found in bacteria?
A)
Helical DNA
done
clear
B)
Membrane bound DNA
done
clear
C)
Straight DNA
done
clear
D)
Circular free DNA
done
clear
View Answer play_arrow
question_answer 148) Which is social foresting species?
A)
Leucaena leucocephala
done
clear
B)
Tectona grandis
done
clear
C)
Mangifera indica
done
clear
D)
Rosa indica
done
clear
View Answer play_arrow
question_answer 149) If there are 4 cells in a anther what will be the number of pollen grains:
A)
4
done
clear
B)
9
done
clear
C)
12
done
clear
D)
16
done
clear
View Answer play_arrow
question_answer 150) In Ficus inflorescence is:
A)
hypanthodium
done
clear
B)
cymose
done
clear
C)
raceme
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 151) ADH controls water permeability of:
A)
collecting tube
done
clear
B)
proximal convoluted tubules
done
clear
C)
distal convoluted tubules
done
clear
D)
all of the above
done
clear
View Answer play_arrow
question_answer 152) At high altitude, RBCs of human blood will:
A)
increase in number
done
clear
B)
decrease in number
done
clear
C)
decrease in size
done
clear
D)
increase in size
done
clear
View Answer play_arrow
question_answer 153) A person who is trisomic for twenty first pair of chromosome suffers from?
A)
Klinefelters syndrome
done
clear
B)
Downs syndrome
done
clear
C)
Turners syndrome
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 154) Enzymes are absent in:
A)
algae
done
clear
B)
plants
done
clear
C)
virus
done
clear
D)
bacteria
done
clear
View Answer play_arrow
question_answer 155) The basic unit of chitin is:
A)
N-acetyl glucosamine
done
clear
B)
glucose
done
clear
C)
galactose
done
clear
D)
fructose
done
clear
View Answer play_arrow
question_answer 156) Striped muscles are:
A)
anucleate
done
clear
B)
binucleate
done
clear
C)
uninucleate
done
clear
D)
syncytial
done
clear
View Answer play_arrow
question_answer 157) Ventricles are related to:
A)
heart only
done
clear
B)
brain only
done
clear
C)
both \[a\] and \[b\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 158) Incomplete division of egg during cleavage is known as:
A)
meroblastic
done
clear
B)
holoblastic
done
clear
C)
meridional
done
clear
D)
spiral
done
clear
View Answer play_arrow
question_answer 159) In which blood group antigens are absent?
A)
O
done
clear
B)
B
done
clear
C)
AB
done
clear
D)
A
done
clear
View Answer play_arrow
question_answer 160) Which one of the following exhibit concentric tube within tube plan?
A)
Cnidaria
done
clear
B)
Annelida
done
clear
C)
Platyhelminthes
done
clear
D)
Nematoda
done
clear
View Answer play_arrow
question_answer 161) Correct sequence of stages in the evolution of the modern man, (Homo sapiens), is:
A)
Neanderthal man, Austmlopithecus, Cro-magnon man, Homo erectus, Modern man
done
clear
B)
Australopithecus, Homo erectus, Neanderthal man, Cro-magnon man, Modem man
done
clear
C)
Homo erectus, Australopithecus Neanderthal man, Cro-magnon man, Modem man
done
clear
D)
Australopithecus, Neanderthal man, Cro-magnon man, Homo erectus Modem man
done
clear
View Answer play_arrow
question_answer 162) WBC accumulate at site of wound by :
A)
hypertension
done
clear
B)
arteriosclerosis
done
clear
C)
haemopoiesis
done
clear
D)
diapedesis
done
clear
View Answer play_arrow
question_answer 163) Each active sites in enzyme is bounded by how many amino acids?
A)
Twenty
done
clear
B)
Infinite
done
clear
C)
Three
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 164) Arachidonic acid is:
A)
non-essential fatty acid
done
clear
B)
essential fatty acid
done
clear
C)
poly unsaturated fatty acid
done
clear
D)
both \[b\] and \[c\]
done
clear
View Answer play_arrow
question_answer 165) The immediate cause of induction of ovulation in female is the large plasma surge of
A)
progesterone
done
clear
B)
estriadiol
done
clear
C)
LH
done
clear
D)
FSH
done
clear
View Answer play_arrow
question_answer 166) Which type of enzyme is present in lysosome?
A)
ATPase
done
clear
B)
Hydrolytic
done
clear
C)
Lyase
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 167) The true stomach in ruminants where most of digestion takes place is:
A)
rumen
done
clear
B)
omasum
done
clear
C)
reticulum
done
clear
D)
abomasum
done
clear
View Answer play_arrow
question_answer 168) The effect of intraspecific competition on niches is to make them:
A)
smaller
done
clear
B)
larger
done
clear
C)
change location
done
clear
D)
more triangular
done
clear
View Answer play_arrow
question_answer 169) The larvicidial fish used in biocontrol of mosquitoes:
A)
Gambusia
done
clear
B)
Hilsa
done
clear
C)
Scalophagns
done
clear
D)
Gold fish
done
clear
View Answer play_arrow
question_answer 170) The joint of radio-ulna with the upper arm is:
A)
hinge joint
done
clear
B)
pivot joint
done
clear
C)
socket joint
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 171) Which of the following amino acids is present in omithine cycle?
A)
Valine and cysteine
done
clear
B)
Arginine and citrulline
done
clear
C)
Glycine and methionine
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 172) During the process of blood coagulation vitamin K helps in:
A)
the formation of prothrombin
done
clear
B)
the formation of thromboplastin
done
clear
C)
the conversion of fibrinogen into fibrin
done
clear
D)
the conversion of prothrombin into thrombin
done
clear
View Answer play_arrow
question_answer 173) Third cleavage of frogs development is:
A)
equatorial
done
clear
B)
vertical
done
clear
C)
latitudinal
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 174) Which of the following is both exocrine and endocrine gland?
A)
Liver
done
clear
B)
Pancreas
done
clear
C)
Thyroid
done
clear
D)
Adrenal
done
clear
View Answer play_arrow
question_answer 175) Which of the following is the part of mid brain of rabbit?
A)
Diencephalon
done
clear
B)
Cerebrum
done
clear
C)
Corpora quadrigemina
done
clear
D)
None of these
done
clear
View Answer play_arrow
question_answer 176) Anus is absent in:
A)
Unio
done
clear
B)
Pheretima
done
clear
C)
Periplaneta
done
clear
D)
Fasciola
done
clear
View Answer play_arrow
question_answer 177) Spiral valve is present in:
A)
right auricle
done
clear
B)
sinus venosus
done
clear
C)
right ventricle
done
clear
D)
truncus arteriosus
done
clear
View Answer play_arrow
question_answer 178) Which of the following is colourless Hydra?
A)
Hydra fusca
done
clear
B)
Hydra viridis
done
clear
C)
Hydra oligactis
done
clear
D)
Hydra vidgaris
done
clear
View Answer play_arrow
question_answer 179) Phospholipids are:
A)
amphipathic
done
clear
B)
amphibolie
done
clear
C)
hydrophobic
done
clear
D)
none of these
done
clear
View Answer play_arrow
question_answer 180) Which of the following assists in the locomotion of the organism stated?
A)
Epithelium of Pheretima
done
clear
B)
Trichocysts of Paramecium
done
clear
C)
Pedicellaria of Star fish
done
clear
D)
Posterior sucker of Hirudinaria
done
clear
View Answer play_arrow
question_answer 181) Dinosaurs become extinct in:
A)
Permian age
done
clear
B)
Triassic period
done
clear
C)
Jurassic period
done
clear
D)
Cretaceous period
done
clear
View Answer play_arrow
question_answer 182) Blood calcium level can be increased by the administration of:
A)
glucagon
done
clear
B)
parathormone
done
clear
C)
thyroxine
done
clear
D)
calcitonin
done
clear
View Answer play_arrow
question_answer 183) The wish bone or merry thought bone of bird is:
A)
sternum
done
clear
B)
scapula
done
clear
C)
coracoid
done
clear
D)
clavicle
done
clear
View Answer play_arrow
question_answer 184) Regarding blood circulation, it may be said that in Pheretima the dorsal vessel is a:
A)
collecting vessel in first two segments and distributing vessel in the other
done
clear
B)
distributing vessel in first five segments and collecting vessel in other
done
clear
C)
collecting vessel in first thirteen segments and distributing vessel in intestinal region
done
clear
D)
distributing vessel in first thirteen segments and collecting vessel in intestinal region
done
clear
View Answer play_arrow
question_answer 185) The first heart sound is produced when:
A)
diastole begins
done
clear
B)
semilunar valve close quickly
done
clear
C)
inter ventricular pressure decreases
done
clear
D)
bicuspid and tricuspid valve close quickly
done
clear
View Answer play_arrow
question_answer 186) The epithelial lining in the alveoli of the frogs facing lung cavity is:
A)
squamous ciliated
done
clear
B)
columnar ciliated
done
clear
C)
columnar non ciliated
done
clear
D)
squamous non ciliated
done
clear
View Answer play_arrow
question_answer 187) The infective stage of Plasmodhim is:
A)
sporozoite
done
clear
B)
trophozoite
done
clear
C)
merozoite
done
clear
D)
schizont
done
clear
View Answer play_arrow
question_answer 188) A man of blood group A, marries a woman of blood group B, both of them are heterozygous for blood group, chances of their first child having blood group AB will be:
A)
25%
done
clear
B)
50%
done
clear
C)
75%
done
clear
D)
100%
done
clear
View Answer play_arrow
question_answer 189) The scientist, who described the birds as glorified reptiles, was:
A)
Romer
done
clear
B)
Huxley
done
clear
C)
Mendel
done
clear
D)
Robert Hook
done
clear
View Answer play_arrow
question_answer 190) Which one of the following is vestigial organ of human?
A)
Hair
done
clear
B)
Intestine
done
clear
C)
Wisdom teeth
done
clear
D)
Muscle of glottis
done
clear
View Answer play_arrow
question_answer 191) Organ of Corti is found in:
A)
heart
done
clear
B)
kidneys
done
clear
C)
inner ear
done
clear
D)
nasal chamber
done
clear
View Answer play_arrow
question_answer 192) Which of the following is a transparent tissue?
A)
Tendon
done
clear
B)
Hyaline cartilage
done
clear
C)
Fibrous cartilage
done
clear
D)
All of these
done
clear
View Answer play_arrow
question_answer 193) In frog, the vertebra with an anterior convex surface (i.e., double convexties) is:
A)
atlas
done
clear
B)
urostyle
done
clear
C)
8th vertebra
done
clear
D)
9th vertebra
done
clear
View Answer play_arrow
question_answer 194) Peyers patches produce:
A)
mucus
done
clear
B)
trypsin
done
clear
C)
lymphocytes
done
clear
D)
enterokinase
done
clear
View Answer play_arrow
question_answer 195) The vital capacity of lungs is equal to:
A)
\[4000\,\,ml\]
done
clear
B)
\[5000\,\,ml\]
done
clear
C)
\[6000\,\,ml\]
done
clear
D)
\[7000\,\,ml\]
done
clear
View Answer play_arrow
question_answer 196) What is left when bath sponges dries up?
A)
Spicules
done
clear
B)
Spongin fibres
done
clear
C)
Tentacles
done
clear
D)
Holdfast
done
clear
View Answer play_arrow
question_answer 197) RBCs are nucleated in:
A)
man
done
clear
B)
rat
done
clear
C)
frog
done
clear
D)
rabbit
done
clear
View Answer play_arrow
question_answer 198) Which of the following pairs is correctly matched?
A)
Central dogma - Codon
done
clear
B)
Okazaki fragments - Splicing
done
clear
C)
RNA polymerase - RNA primer
done
clear
D)
Restriction enzymes - Genetic engineering
done
clear
View Answer play_arrow
question_answer 199) A nucleoside is:
A)
base + sugar
done
clear
B)
base + phosphate
done
clear
C)
sugar + phosphate
done
clear
D)
base + sugar + phosphate
done
clear
View Answer play_arrow
question_answer 200) Stanley Miller performed an experiment to prove the origin of life. They took gases \[N{{H}_{3}}\] and \[{{H}_{2}}\] alongwith:
A)
\[{{N}_{2}}\]and\[{{H}_{2}}O\]
done
clear
B)
\[{{H}_{2}}O\]and\[C{{H}_{4}}\]
done
clear
C)
\[C{{H}_{4}}\]and\[{{N}_{2}}\]
done
clear
D)
\[C{{O}_{2}}\]and\[N{{H}_{3}}\]
done
clear
View Answer play_arrow