The dominant mechanism for motion of charge carriers in forward and reverse biased silicon p-n junction are
A)
diffusion in forward biased, drift in reverse bias
done
clear
B)
drift in forward biased, drift in reverse biased
done
clear
C)
diffusion in both forward and reverse bias
done
clear
D)
drift in both forward and reverse bias
done
clear
View Answer play_arrow
If binding energy of electron in a hydrogen atom is 13.6 eV, then energy required to remove the electron from the first excited state of \[L{{i}^{+\,+}}\] is
A)
13.6 eV
done
clear
B)
3.4 eV
done
clear
C)
30.6 eV
done
clear
D)
122.4 eV
done
clear
View Answer play_arrow
Magnetic field does not cause deflection in
A)
\[\alpha -rays\]
done
clear
B)
\[\beta -rays\]
done
clear
C)
H-atom
done
clear
D)
None of these
done
clear
View Answer play_arrow
For a telescope, larger the diameter of the objective lens,
A)
greater is the magnifying power
done
clear
B)
greater is the resolving power
done
clear
C)
smaller is the magnifying power
done
clear
D)
smaller is the resolving power
done
clear
View Answer play_arrow
An X-rays tube operate at 10 kV. The ratio of X-rays wavelength to that of de-Broglie is
A)
\[1 : 10\]
done
clear
B)
\[10: 1\]
done
clear
C)
\[1 : 100\]
done
clear
D)
\[100 : 1\]
done
clear
View Answer play_arrow
During melting of an ice slab at 273 K at atmospheric pressure
A)
positive work is done by ice water system on the atmosphere
done
clear
B)
positive work is done on ice water system by the atmosphere
done
clear
C)
internal energy of ice water system decreases
done
clear
D)
None of the above
done
clear
View Answer play_arrow
The ratio of \[{{C}_{P}}\,\,and\,\,{{C}_{V}}\] depends on temperature T according to relation
A)
\[\gamma \propto T\]
done
clear
B)
\[\gamma \propto T\]
done
clear
C)
\[\gamma \propto \frac{1}{T}\]
done
clear
D)
\[\gamma \propto \sqrt{T}\]
done
clear
View Answer play_arrow
A body executing SHM with an amplitude A. At what displacement from the mean position is potential energy of the body is one-fourth of its total energy
A)
\[\frac{A}{4}\]
done
clear
B)
\[\frac{A}{2}\]
done
clear
C)
\[\frac{3A}{4}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
A capillary tube of radius R is immersed in water and water rises in it to a height H. Mass of water in capillary tube is M. If the radius of tube is doubled, mass of water that will rise in capillary will be
A)
M
done
clear
B)
\[\frac{M}{2}\]
done
clear
C)
2 M
done
clear
D)
4 M
done
clear
View Answer play_arrow
Two bodies of mass 1 kg and 3 kg have position vector\[\hat{i} + 2\hat{j} + \hat{k}\,\,and\,\,-3\hat{i} - 2\hat{j} + \hat{k}\], respectively. The centre of mass of this system has a position vector
A)
\[2\hat{i}\,-\hat{j}\,\,-2\hat{k}\]
done
clear
B)
\[-2\hat{i}\,-\hat{j}\,\,+\,\,\hat{k}\]
done
clear
C)
\[-\hat{i}\,+\hat{j}\,\,+\,\,\hat{k}\]
done
clear
D)
\[-\,2\hat{i}\,+2\,\,\hat{k}\]
done
clear
View Answer play_arrow
A thin circular ring of mass M and radius R is rotating in a horizontal plane about an axis vertical to its plane with a constant angular velocity co. If two objects each of mass m be attached gently to opposite ends of a diameter of the ring, the ring will then rotate with an angular velocity
A)
\[\frac{\omega M}{M+m}\]
done
clear
B)
\[\frac{\omega (M-2m)}{M+2\,m}\]
done
clear
C)
\[\frac{\omega M}{M+2\,m}\]
done
clear
D)
\[\frac{\omega (M+2m)}{M}\]
done
clear
View Answer play_arrow
If a faulty thermometer has its fixed points marked as \[5{}^\circ and 95{}^\circ \]. If the temperature measured by the faulty thermometer is \[59{}^\circ \], then the correct temperature of the body on Celsius scale is
A)
\[30{}^\circ C\]
done
clear
B)
\[60{}^\circ C\]
done
clear
C)
\[120{}^\circ C\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
An open organ pipe produces fundamental note. All of a sudden, one of its ends is close [d] If again fundamental note is emitted, the frequency of note will be
A)
half
done
clear
B)
double
done
clear
C)
same
done
clear
D)
None of these
done
clear
View Answer play_arrow
Two spheres of the same material have radii 1 m and 4 m and temperatures 4000 K and 2000 K respectively. The ratio of energy radiated per second by the first sphere to that by the second is
A)
\[1 :1\]
done
clear
B)
\[16:1\]
done
clear
C)
\[4:1\]
done
clear
D)
\[1:9\]
done
clear
View Answer play_arrow
Two wires of the same length and same material but radii in the ratio of \[1:2\] are stretched by unequal forces to produce equal elongation. The ratio of forces is
A)
\[1:4\]
done
clear
B)
\[1:2\]
done
clear
C)
\[2:1\]
done
clear
D)
\[1:\sqrt{2}\]
done
clear
View Answer play_arrow
Which of the following is not the cause of low conductivity of electrolytes?
A)
high resistance offered by the solution to the motion of ions
done
clear
B)
ionisation of salt
done
clear
C)
low number of density of charge carriers?
done
clear
D)
low drift velocity of ions
done
clear
View Answer play_arrow
A ball is released from the top of a tower and exactly 1s later another ball is released. The distance between two balls 2 s after the release of second ball is\[(g =9.8 m/{{s}^{2}})\].
A)
14.5 m
done
clear
B)
24.5 m
done
clear
C)
34.5 m
done
clear
D)
None of these
done
clear
View Answer play_arrow
Pick out which one of these is for AND, NAND and NOT gates, respectively I.
II.
III.
IV.
A)
III, II and I
done
clear
B)
III, II and IV
done
clear
C)
II, III and IV
done
clear
D)
II, IV and III
done
clear
View Answer play_arrow
The equivalent resistance between points A and D is (given wire used is idle)
A)
zero
done
clear
B)
R
done
clear
C)
2 R
done
clear
D)
None of these
done
clear
View Answer play_arrow
The electrostatic potential V at a distance r from the centre of a charged metallic sphere of radius JR for \[\operatorname{r} < R\] will vary according to relation
A)
\[V\propto \frac{1}{r}\]
done
clear
B)
\[V=r\]
done
clear
C)
\[V\propto \frac{1}{{{r}^{2}}}\]
done
clear
D)
\[V\propto {{r}^{0}}\]
done
clear
View Answer play_arrow
Three capacitors of equal capacity C are connected as shown in figure, the equivalent capacitance of the combination between A and B is C
A)
C
done
clear
B)
2 C
done
clear
C)
\[\frac{C}{2}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
P, Q and R are three points in a uniform electric field. The electric potential is
A)
minimum at R
done
clear
B)
minimum at O
done
clear
C)
minimum at P
done
clear
D)
Same at all three points
done
clear
View Answer play_arrow
A motor pumps water continuously through a hose. Water leaves the hose with velocity v and m is the mass flowing per unit length of water jet, then the rate at which kinetic energy is imparted to water is
A)
\[\frac{1}{2}m{{v}^{2}}\]
done
clear
B)
\[\frac{1}{2}{{m}^{2}}v\]
done
clear
C)
\[\frac{1}{2}m{{v}^{3}}\]
done
clear
D)
\[m{{v}^{3}}\]
done
clear
View Answer play_arrow
Two bodies are thrown with same initial velocity at angles \[\theta \] and \[\left( 90{}^\circ -\theta \right)\] with horizontal. The ratio of their horizontal ranges are
A)
1
done
clear
B)
\[\frac{1}{2}\]
done
clear
C)
\[ta{{n}^{2}}\theta \]
done
clear
D)
None of these
done
clear
View Answer play_arrow
A 10 m long wire of resistance \[20\,\Omega \] is connected in series with a battery of emf 3 V (negligible internal resistance) and a resistance of \[10\,\Omega \]., then the potential gradient along the wire is
A)
0.1
done
clear
B)
0.2
done
clear
C)
0.4
done
clear
D)
None of these
done
clear
View Answer play_arrow
If the cold junction of a thermocouple is lowered then the neutral temperature
A)
decreases
done
clear
B)
increases
done
clear
C)
remains the same
done
clear
D)
approaches inversion temperature
done
clear
View Answer play_arrow
Two square metal plates of same thickness and material are connected in series as shown in figure. The length of B is twice the length of A. If resistance of A is represented by \[{{R}_{A}}\] and resistance of B is denoted by \[{{R}_{B}}\] then \[\frac{{{R}_{A}}}{{{R}_{B}}}\] is
A)
1
done
clear
B)
2
done
clear
C)
\[\frac{1}{2}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
A magnet of magnetic moment M is rotated through \[360{}^\circ \] in a magnetic field H, the work done will be
A)
0
done
clear
B)
2 MH
done
clear
C)
- MH
done
clear
D)
-2 MH
done
clear
View Answer play_arrow
Earth's magnetic field always has a horizontal component except at
A)
magnetic pole
done
clear
B)
equator
done
clear
C)
\[\operatorname{at} altitude of 60{}^\circ \]
done
clear
D)
\[\operatorname{at} altitude of 45{}^\circ \]
done
clear
View Answer play_arrow
What is self-inductance of a coil which produces 5 V, when current in it changes from 3 A to 2 A in one millisecond.
A)
5 mH
done
clear
B)
5 H
done
clear
C)
50 H
done
clear
D)
- 5 mH
done
clear
View Answer play_arrow
A voltmeter has a resistance of G Ohm and range of V volt. The value of resistance used in series to convert it into a voltmeter of range nV volt is
A)
nG
done
clear
B)
\[\frac{G}{(n-1)}\]
done
clear
C)
\[\left( n-1 \right)G\]
done
clear
D)
\[\frac{G}{n}\]
done
clear
View Answer play_arrow
A particle of mass \[{{m}_{1}}\] is moving with a velocity \[{{v}_{1}}\] and another particle of mass \[{{m}_{2}}\] moving with a velocity\[{{v}_{2}}\]. Both of them have same momentum but different kinetic energy \[{{E}_{1}}\,\,and\,\,{{E}_{2}}\] respectively. If \[{{m}_{1}}\,\,>\,\,{{m}_{2}}\], then
A)
\[{{E}_{1}}>{{E}_{2}}\]
done
clear
B)
\[{{E}_{1}}={{E}_{2}}\]
done
clear
C)
\[\frac{{{E}_{1}}}{{{E}_{2}}}=\frac{{{m}_{1}}}{{{m}_{2}}}\]
done
clear
D)
\[{{E}_{1}}<{{E}_{2}}\]
done
clear
View Answer play_arrow
A 5 m aluminum wire \[\left( y=7\times 1{{0}^{10}}N/{{m}^{2}} \right)\] of diameter 3 mm supports a 40 kg mass. In order to have same elongation in a copper wire \[\left( Y=12\times 1{{0}^{10}}N/{{m}^{2}} \right)\] of same length under same weight, the diameter should be, in mm
A)
2.0
done
clear
B)
2.3
done
clear
C)
1.75
done
clear
D)
5.0
done
clear
View Answer play_arrow
A transverse wave is represented by the equation\[\operatorname{y}={{y}_{0}}\,\,sin\,\frac{2\pi }{\lambda }\,(vt-x)\]. For what value of is maximum particle velocity equal to two times of the wave velocity
A)
\[\lambda =2\pi \,{{y}_{0}}\]
done
clear
B)
\[\lambda =\pi \,{{y}_{0}}\]
done
clear
C)
\[\lambda =\frac{\pi \,{{y}_{0}}}{3}\]
done
clear
D)
\[\lambda =\frac{\pi \,{{y}_{0}}}{2}\]
done
clear
View Answer play_arrow
If angle between two vectors X and Y is \[120{}^\circ \], then its resultant Z will be
A)
\[\left| Z \right|=\left| X\,-Y \right|\]
done
clear
B)
\[\left| Z \right|<\left| X\,-Y \right|\]
done
clear
C)
\[\left| Z \right|>\left| X\,\,-\,\,Y \right|\]
done
clear
D)
\[\left| Z \right|=\left| X+Y \right|\]
done
clear
View Answer play_arrow
If length of rod X is \[\left( 2.25 \pm 0.02 \right)\]cm and that of Y is \[\left( 5.19 \pm 0.02 \right)\] cm, then rod Y is longer then rod X by
A)
\[\left( 2.94 \pm 0.02 \right)\]
done
clear
B)
\[\left( 2.94 \pm 0.00 \right)\]
done
clear
C)
\[\left( 2.94 \pm 0.04 \right)\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
If a radioactive nucleus decays by two different processes, the half-life for first process is \[{{t}_{1}}\] and for second process it is \[{{t}_{2}}\], then the effective half-life t of the nucleus is given by
A)
\[t={{t}_{1}}+{{t}_{2}}\]
done
clear
B)
\[\sqrt{t}=\sqrt{{{t}_{1}}}+\sqrt{{{t}_{2}}}\]
done
clear
C)
\[{{t}^{-1}}=t_{1}^{-1}+t_{2}^{-1}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
A TV tower has a height of 80m. If average density of population is 3000 persons per \[k{{m}^{2}}\], then the population covered by TV tower is
A)
40 lakh
done
clear
B)
80 lakh
done
clear
C)
96 lakh
done
clear
D)
192 lakh
done
clear
View Answer play_arrow
What will be the time constant for the given circuit?
A)
RC
done
clear
B)
2.5 RC
done
clear
C)
1.2 RC
done
clear
D)
6 RC
done
clear
View Answer play_arrow
Two particles 1 and 2 of masses \[{{M}_{1}}\,\,and\,\,{{M}_{2}}\] respectively enter in a uniform magnetic field as shown in figure. If \[{{v}_{1}}\,\,and\,\,{{v}_{2}}\] are their respective velocities then (assume both particles have same charge)
A)
\[{{M}_{1}}{{v}_{1}}<{{M}_{2}}{{v}_{2}}\]
done
clear
B)
\[{{M}_{1}}{{v}_{1}}>{{M}_{2}}{{v}_{2}}\]
done
clear
C)
\[{{M}_{1}}{{v}_{1}}={{M}_{2}}{{v}_{2}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
The effective resistance of 'n' number of resistors when connected in parallel is A ohm. If one of the resistor is removed then the effective resistance becomes B ohm, then the resistance of resistor which is removed is
A)
\[\frac{AB}{B-A}\]
done
clear
B)
\[\frac{AB}{A-B}\]
done
clear
C)
\[\frac{A+B}{AB}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
If the density of water at the surface of the lake is D. The bulk modulus of water is B, then the density of lake water at a depth where the pressure is n p? \[[p :atomospheric pressure]\]
A)
\[\frac{DB}{B+(n-1)p}\]
done
clear
B)
\[\frac{DB}{B-(n-1)p}\]
done
clear
C)
\[\frac{DB}{B+(n\,+1)p}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
If the flux linked with a circuit is given by\[\phi ={{t}^{3}}+3t-t\]. The graph between time (x- axis) and induced emf (y - axis) will be a
A)
straight line through the origin
done
clear
B)
straight line with positive intercept
done
clear
C)
parabola through origin
done
clear
D)
parabola not through origin
done
clear
View Answer play_arrow
Relative permittivity and permeability of a material are \[{{\varepsilon }_{r}}\,\,and\,\,{{\mu }_{r}}\] respectively. Which of the following values of these quantities are allowed for a diamagnetic material?
A)
\[{{\varepsilon }_{r}} = 0.5, \,{{\mu }_{r}} = 1.5\]
done
clear
B)
\[{{\varepsilon }_{r}} = 1.5, \,{{\mu }_{r}} = 0.5\]
done
clear
C)
\[{{\varepsilon }_{r}} = 0.5, \,{{\mu }_{r}} = 0.5\]
done
clear
D)
\[{{\varepsilon }_{r}} = 1.5, \,{{\mu }_{r}} = 1.5\]
done
clear
View Answer play_arrow
A length of wire carries a steady current. It is bent first to form a circular of one turn. The same length is now bent more sharply to give a double loop of smaller radius. The magnetic field at centre caused by the same current is
A)
double of its first value
done
clear
B)
quarter of its first value
done
clear
C)
four times of its first value
done
clear
D)
same as the first value
done
clear
View Answer play_arrow
Today artificial sweeteners and other sugar substitutes are found in a variety of food and beverages marketed as 'sugar-free'. Which of the following is most powerful artificial sweetener?
A)
Saccharin
done
clear
B)
Aspartame
done
clear
C)
Alitame
done
clear
D)
Sucralose
done
clear
View Answer play_arrow
A greenhouse gas is a gas in an atmosphere that absorbs and emits radiation within the thermal infrared range. This process is the fundamental cause of greenhouse effect. Among the following which one is not a greenhouse gas?
A)
\[C{{O}_{2}}\]
done
clear
B)
\[C{{H}_{4}}\]
done
clear
C)
\[{{O}_{2}}\]
done
clear
D)
Vapour of water
done
clear
View Answer play_arrow
Nyctalopia also called 'Night Blindness' is a condition making it difficult or impossible to see in relative low light. It is symptom of several eye diseases. It may exist from birth, or be caused by injury or malnutrition. It is caused in human due to deficiency of vitamin
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
View Answer play_arrow
Those polymers which get decomposed by the process of biodegradation are known as biodegradable polymer. Among the following, the biodegradable polymer is
A)
cellulose
done
clear
B)
polyethene
done
clear
C)
PVC
done
clear
D)
nylon 6
done
clear
View Answer play_arrow
The shapes of \[NO_{3}^{-}\] and \[NO_{3}^{-\,\,-}\] are
A)
triangular planar
done
clear
B)
tetrahedral and triangular planar respectively
done
clear
C)
tetrahedral
done
clear
D)
angular and triangular planar respectively
done
clear
View Answer play_arrow
Compare the acidic strength of reaction product, B with respect to phenol, A and choose the correct order. Chemical reaction is
A)
\[A > B\]
done
clear
B)
\[A < B\]
done
clear
C)
\[A = B\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
What will be correct stereochemistry of product obtained on reaction ofbut-2-ene with\[OS{{O}_{4}}\]?
A)
done
clear
B)
done
clear
C)
done
clear
D)
None of these
done
clear
View Answer play_arrow
Product obtained on reaction 2-methyl cyclohexene with mCPBA followed by hydrolysis is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following is the product of Hofmann elimination of?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following will give positive carbylamine test?
A)
N, N-diethyl aniline
done
clear
B)
N methyl propyl amine
done
clear
C)
N, N-diethyl butyl amine
done
clear
D)
2, 4-dimethyl aniline
done
clear
View Answer play_arrow
IUPAC name of A is
A)
3 methyl 2 chloro butanoic acid
done
clear
B)
2 methyl 3 chloro butanoic acid
done
clear
C)
3 chloro 2 methyl butanoic acid
done
clear
D)
2 chloro 3 carboxy, 2, 3-dimethyl propane
done
clear
View Answer play_arrow
Choose the process involved in the separation of ortho and paro nitrophenol and the reason behind respectively.
A)
steam distillation, metallic bonding
done
clear
B)
sublimation, Hydrogen bonding
done
clear
C)
steam distillation, van der Waal?s forces
done
clear
D)
steam distillation, Hydrogen bonding
done
clear
View Answer play_arrow
\[CH\equiv CH\,\,\xrightarrow{{{O}_{3}}\,/\,NaOH}\,\,X\,\xrightarrow{Zn\,/\,C{{H}_{3}}COOH}\,Y\],Y is
A)
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}OH\]
done
clear
C)
\[{{\operatorname{CH}}_{3}}COOH\]
done
clear
D)
\[{{\operatorname{CH}}_{3}}OH\]
done
clear
View Answer play_arrow
Which one of the following is correct order?
A)
\[~-OH >-OR\left( order of -l effect \right)\]
done
clear
B)
\[-C{{H}_{3}} > C{{O}_{3}} > CC{{l}_{3}} \left( order of +l power \right)\]
done
clear
C)
\[-F> -OH > -N{{H}_{2}} \left( order of -l effect \right)\]
done
clear
D)
\[-NO - S{{O}_{2}}R - S{{O}_{3}}H - N{{O}_{2}} \left( set of\,\,meta\,\,directors \right)\]
done
clear
View Answer play_arrow
Vitamin \[\operatorname{B}-12\] also called coalmen is a water soluble vitamin with a peyroline the normal functioning of the brain and nervous system and for the formation of blood. Deficiency of vitamin B-12 causes
A)
scurvey
done
clear
B)
bery-bery
done
clear
C)
night blindness
done
clear
D)
None of these
done
clear
View Answer play_arrow
Bond order of CO is equal to bond order of
A)
\[B{{e}_{2}}\]
done
clear
B)
\[O_{2}^{+}\]
done
clear
C)
\[N_{2}^{+}\]
done
clear
D)
\[N_{2}^{{}}\]
done
clear
View Answer play_arrow
Effective magnetic moment of \[C{{e}^{3+}}\] ion is
A)
zero
done
clear
B)
1.83
done
clear
C)
2.83
done
clear
D)
1.73
done
clear
View Answer play_arrow
Which of the following has highest 2 nd ionisation energy?
A)
0 6
done
clear
B)
N
done
clear
C)
C
done
clear
D)
B
done
clear
View Answer play_arrow
Which of the following is a lanthanide?
A)
Curium
done
clear
B)
Californium
done
clear
C)
Erbium
done
clear
D)
Amercium
done
clear
View Answer play_arrow
Magnetic moment of \[{{\operatorname{K}}_{4}} \left[ Fe {{\left( CN \right)}_{5}} \left( {{H}_{2}}O \right) \right]\] is
A)
0
done
clear
B)
4.92
done
clear
C)
3.87
done
clear
D)
2.83
done
clear
View Answer play_arrow
IUPAC name of \[\left[ Cr{{({{H}_{2}}O)}_{4}}C{{l}_{2}} \right]\] is
A)
tetraaquadichlorochromate
done
clear
B)
tetraaquadichlorochromium (III) chloride
done
clear
C)
tetra aquadichlorochromium (II) chloride
done
clear
D)
dichlorotetraaquachromium (III) chloride
done
clear
View Answer play_arrow
Which of the following electronic configuration is correct for chromium?
A)
\[[Ar]\,3{{d}^{4}}4{{s}^{2}}\]
done
clear
B)
\[[Ar]\,3{{d}^{5}}4{{s}^{1}}\]
done
clear
C)
\[[Ar]\,3{{d}^{4}}4{{s}^{1}}\]
done
clear
D)
\[[Ar]\,3{{d}^{5}}4{{s}^{2}}\]
done
clear
View Answer play_arrow
The magnetic quantum number of 29 electron of copper is
A)
+1
done
clear
B)
- 3
done
clear
C)
-1
done
clear
D)
- 2
done
clear
View Answer play_arrow
Poorest reducing agent among following is
A)
atomic hydrogen
done
clear
B)
nascent hydrogen
done
clear
C)
dihydrogen
done
clear
D)
All have same reducing strength
done
clear
View Answer play_arrow
Which of the following elements do not impart colour to flame? Ca, Mg, Li, Na, K, Rb
A)
Ca and Mg
done
clear
B)
Li and Na
done
clear
C)
K and Rb
done
clear
D)
Na and K
done
clear
View Answer play_arrow
Which of the following code is correct regarding allotrops of carbon? I. Carbon exist in more than one allotropic form II. Diamond is semiconductor while graphite is conductor III. Diamond has layered structure while graphite has tetrahedral structure IV. Diamond is non-conductor of electricity
A)
I and II are correct
done
clear
B)
II and III are incorrect
done
clear
C)
I and IV are incorrect
done
clear
D)
All are correct
done
clear
View Answer play_arrow
Bases which keep \[B{{H}_{2}}\] in monomeric form is
A)
\[N{{N}_{3}}\]
done
clear
B)
THF
done
clear
C)
DIGLYME
done
clear
D)
All of these
done
clear
View Answer play_arrow
Which of the following shows correct Lewis acidic strength order of trihalides of boron?
A)
\[BC{{l}_{3}}>BB{{r}_{3}}>B{{F}_{3}}>B{{l}_{3}}\]
done
clear
B)
\[B{{F}_{3}}<BB{{r}_{3}}<BC{{l}_{3}}<B{{l}_{3}}\]
done
clear
C)
\[B{{F}_{3}}<BC{{l}_{3}}<BB{{r}_{3}}<B{{l}_{3}}\]
done
clear
D)
\[B{{F}_{3}}>BC{{l}_{3}}>BB{{r}_{3}}>B{{l}_{3}}\]
done
clear
View Answer play_arrow
Which of the following is correct when water and ice are at equilibrium at temperature \[0{}^\circ C\] is?
A)
\[{{G}_{ice}}>{{G}_{{{H}_{2}}O}}\]
done
clear
B)
\[{{G}_{ice}}<{{G}_{{{H}_{2}}O}}\]
done
clear
C)
\[{{G}_{ice}}={{G}_{{{H}_{2}}O}}=0\]
done
clear
D)
\[{{G}_{\,ice}}={{G}_{\,{{H}_{2}}O}}\ne 0\]
done
clear
View Answer play_arrow
Which of the following reaction will not be affected by change in pressure?
A)
\[{{H}_{2}}+{{I}_{2}}\rightleftharpoons 2HI\]
done
clear
B)
\[{{N}_{2}}+3{{H}_{2}}\rightleftharpoons 2N{{H}_{3}}\]
done
clear
C)
\[PC{{l}_{5}}\rightleftharpoons PC{{l}_{3}}+C{{l}_{2}}\]
done
clear
D)
\[\operatorname{CaO}+C{{O}_{2}} \xrightarrow{{}}\,\,CaC{{O}_{3}}\]
done
clear
View Answer play_arrow
Among the following which one is not a redox reaction?
A)
\[{{\operatorname{CaCO}}_{3}}\, \xrightarrow{{}} CaO+C{{O}_{2}}\]
done
clear
B)
\[Na+{{H}_{2}}O\xrightarrow{{}}\,NaOH+\frac{1}{2}{{H}_{2}}\]
done
clear
C)
\[\operatorname{MnC}{{l}_{3}} \xrightarrow{{}} MnC{{l}_{2}}\,+\,\frac{1}{2}\,C{{l}_{2}}\]
done
clear
D)
\[{{O}_{2}}+2{{H}_{2}}\xrightarrow{{}}2{{H}_{2}}O\]
done
clear
View Answer play_arrow
How much time is required to do the electroplating of Ag layer on a coffee tray \[\left( 30\,\,cm \times 15\,cm \right)\] to a thickness of 1 mm using a constant current of 1.0 A. Given that density of Ag is\[10.5 g/c{{m}^{3}}\].
A)
7720 sq
done
clear
B)
120 min
done
clear
C)
772 s
done
clear
D)
77.2 s
done
clear
View Answer play_arrow
Which of the following order is correct regarding rate of diffusion of gases?
A)
\[PC{{l}_{3}}>S{{O}_{3}}>S{{O}_{2}}>C{{O}_{2}}\]
done
clear
B)
\[C{{O}_{2}}>S{{O}_{2}} > PC{{l}_{3}} >S{{O}_{3}}\]
done
clear
C)
\[S{{O}_{2}}>S{{O}_{3}}>PC{{l}_{3}}>C{{O}_{2}}\]
done
clear
D)
\[C{{O}_{2}}>S{{O}_{2}}>S{{O}_{3}}>PC{{l}_{3}}\]
done
clear
View Answer play_arrow
Packing fraction of simple cubic crystal lattice is
A)
\[38\,%\]
done
clear
B)
\[74\,%\]
done
clear
C)
\[68\,%\]
done
clear
D)
\[52.4\,%\]
done
clear
View Answer play_arrow
For antifluorite structure coordination number of cations and anions are respectively
A)
6 and 6
done
clear
B)
4 and 8
done
clear
C)
8 and 4
done
clear
D)
4 and 4
done
clear
View Answer play_arrow
In the reaction, \[{{N}_{2}}{{O}_{4}}\rightleftharpoons 2N{{O}_{2}}\], a is that part of \[{{N}_{2}}{{O}_{4}}\] which dissociates, then the number of moles at equilibrium will be
A)
\[{{\left( 1-\alpha \right)}^{2}}\]
done
clear
B)
\[3\alpha \]
done
clear
C)
\[\alpha \]
done
clear
D)
\[1+\alpha \]
done
clear
View Answer play_arrow
\[\Delta E{}^\circ \] of combustion of isobutylene is \[-x\,kJ mol{{\,}^{-}}^{1}\]. The value of \[\Delta H{}^\circ \] is
A)
\[=\Delta \,E{}^\circ \]
done
clear
B)
\[>\Delta \,E{}^\circ \]
done
clear
C)
\[=zero\]
done
clear
D)
\[<\Delta \,E{}^\circ \]
done
clear
View Answer play_arrow
A plot of log \[\frac{x}{m}\] versus log p for adsorption of a gas on a solid gives a straight line with slope equal to m
A)
\[\frac{1}{n}\]
done
clear
B)
log K
done
clear
C)
- log K
done
clear
D)
n
done
clear
View Answer play_arrow
The half-life time of a first order reaction is \[1.26 \times 1{{0}^{14}}s\], then rate constant of this reaction will be
A)
\[5.5\times 1{{0}^{-10}}{{s}^{-\,1}}\]
done
clear
B)
\[5.5\times 1{{0}^{-14}}{{s}^{-\,1}}\]
done
clear
C)
\[6.6\times 1{{0}^{-14}}{{s}^{-\,1}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
Which of the following forms cationic micelles above certain concentration?
A)
Urea
done
clear
B)
Sodium dodecyl sulphate
done
clear
C)
Sodium acetate
done
clear
D)
Cetyltrimethylammonium bromide
done
clear
View Answer play_arrow
0.5 g of a metal on oxidation give 0.79 g of its oxide. The equivalent weight of the metal is
A)
10
done
clear
B)
14
done
clear
C)
20
done
clear
D)
40
done
clear
View Answer play_arrow
What will be the value of relative lowering of vapour pressure when 3 g urea is dissolved in 45 g. of water?
A)
0.05
done
clear
B)
0.04
done
clear
C)
0.02
done
clear
D)
0.01
done
clear
View Answer play_arrow
Which of the following will not show tautomerism?
A)
cyclohexen-1-ol
done
clear
B)
pentanone
done
clear
C)
acetone
done
clear
D)
2-methyl pentan-1-ol
done
clear
View Answer play_arrow
Rate constant of any reaction with \[20\,%\] complete in 10 min is
A)
0.223
done
clear
B)
0.0223
done
clear
C)
0.0322
done
clear
D)
0.322
done
clear
View Answer play_arrow
Which of the following contain N?N bond? \[{{N}_{2}}{{O}_{4}},\,\,{{N}_{2}}{{O}_{5}},\,\,{{N}_{2}}{{O}_{3}}\]
A)
\[{{N}_{2}}{{O}_{4}}, {{N}_{2}}{{O}_{3}} and {{N}_{2}}{{O}_{5}}\]
done
clear
B)
\[{{N}_{2}}{{O}_{5}} and {{N}_{2}}{{O}_{4}}\]
done
clear
C)
\[{{N}_{2}}{{O}_{3}} and {{N}_{2}}{{O}_{5}}\]
done
clear
D)
\[{{N}_{2}}{{O}_{3}} and {{N}_{2}}{{O}_{4}}\]
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Viral diseases 1. Taeniasus and ascariasis B. Bacterial diseases 2. Malaria C. Protozoan diseases 3. Rabies and polio D. Helminthic diseases 4. Cholera and TB 5. Syphilis
Codes
A)
A-5, B-1, C-4, D-2
done
clear
B)
A-3, B-4, C-2, D-1
done
clear
C)
A-1, B-2, C-5, D-3
done
clear
D)
A-4, B-3, C-1, D-2
done
clear
View Answer play_arrow
Match the following columns.
Column I (Stop codon) Column II (Also known as) A. UAG 1. Opal B. UAA 2. Ochre C. AGA 3. Amber 4. Umber
Codes
A)
A-3, B-1, C-2
done
clear
B)
A-4, B-1, C-3
done
clear
C)
A-3, B-4, C-2
done
clear
D)
A-3, B-2, C-1
done
clear
View Answer play_arrow
Which one is obtained by acetylation of morphine?
A)
Charas
done
clear
B)
Cocaine
done
clear
C)
Smack
done
clear
D)
Crack
done
clear
View Answer play_arrow
Accordance to the human genome project, select the incorrect statement.
A)
The goal of HGP was to identity or determine 3 billion chemical base pair
done
clear
B)
It was launched in 1980 2 completed in 1990
done
clear
C)
Two major approaches involved i.e., ESTs (Expressed Sequence Tags) and sequence annotation
done
clear
D)
SNPs or single nucleotide polymorphism occur in humans.
done
clear
View Answer play_arrow
Cambium is the example of which of the following types apical of meristem?
A)
Apical meristem
done
clear
B)
Lateral meristem
done
clear
C)
Marginal meristem
done
clear
D)
Intercalary meristem
done
clear
View Answer play_arrow
Which of the following are true for serum sickness? I. It is an genetic disorder. II. It is a reaction that is similar to an allergy. III. Corticosteroid is generally first line of management/treatment for serum sickness. IV. Sulfa drugs cannot involve in its etiology.
A)
II and III
done
clear
B)
I and IV
done
clear
C)
I and III
done
clear
D)
II and IV
done
clear
View Answer play_arrow
Identify the markings of below ecological pyramid from following options, if 1-Herbivores; 2-Secondary predator; 3-Decay derives; 4-Primary predators; 5-Autotrophs
A)
A-2, B-1, C-3, D-4, E-5
done
clear
B)
A-4, B-1, C-3, D-5, E-2
done
clear
C)
A-3, B-5, C-2, D-1, E-4
done
clear
D)
A-5, B-3, C-2, D-1, E-4
done
clear
View Answer play_arrow
Which of the following statements is not correct for interferons?
A)
They are mainly glycopretein in nature
done
clear
B)
They are produced by plasma B-cells only
done
clear
C)
They form the body's second line of defence
done
clear
D)
They provide a temporary protection against microbes
done
clear
View Answer play_arrow
In reference to the mRNA, select the incorrect option.
A)
mRNA is linear
done
clear
B)
It carries coded information
done
clear
C)
It does not require any processing
done
clear
D)
Nitrogen bases are unmodified
done
clear
View Answer play_arrow
Which one of the statements given below is not correct?
A)
Insectivorous plants are autotrophic and heterotrophic as they capture insects to overcome their \[{{N}_{3}}\] deficiency
done
clear
B)
Sulphur is main constituent of chlorophyll
done
clear
C)
The most important diseases due to copper deficiency are exanthema and reclamation
done
clear
D)
Nif gene is present in Rhizobium
done
clear
View Answer play_arrow
Which of the following are the causes of insulin resistance? I. Polycystic ovary disease II. Hemochromatosis III. Lipodystrophy IV. Mutations of melanocortin receptors
A)
I and IV
done
clear
B)
I, II, III and IV
done
clear
C)
II and III
done
clear
D)
II and IV
done
clear
View Answer play_arrow
Which part of male reproductive system is the site of sperm maturation and storage?
A)
Vas deferens
done
clear
B)
Testes
done
clear
C)
Epididymis
done
clear
D)
Seminal vesicle
done
clear
View Answer play_arrow
Which of the followings is not correctly matched?
A)
Cancer of muscle tissue - Myoma
done
clear
B)
Cancer of adipose tissue - Lipomas
done
clear
C)
Blood cancer - Leukaemia
done
clear
D)
Cancer of glial cells of CNS - Melanomas
done
clear
View Answer play_arrow
Which of the following statement(s) is/are correct for RAPD? I. It stands of for Random Altered Polygenic DNA. II. It stands for Random Amplified Polymorphic DNA. III. It is used to study phylogeny of diverse plant and animal species. IV. It is very easy to distinguish whether a DNA segment is amplified from heterozygous or homozygous.
A)
I and IV
done
clear
B)
II and III
done
clear
C)
II and IV
done
clear
D)
I, II, III and IV
done
clear
View Answer play_arrow
Which one of the following is not correct?
A)
The minerals, particularly \[C{{a}^{2+}},\,\,{{K}^{+}}\,and\,\,N{{a}^{+}}\] maintain the permeability of cytomembranes
done
clear
B)
Minerals like Cu, As, impart toxic effect on the proplasm under specific condition
done
clear
C)
Several elements particularly Fe, Ca, Mg, Zn, Cu, Cl act as metallic catalyst in biochemical reactions
done
clear
D)
Mg, K and Ca are essential microelements
done
clear
View Answer play_arrow
Identify the marking in below graphs.
A)
A- Allopatric, B- Sympatric, C- Parapatric
done
clear
B)
A- Sympatric, B- Parapatric, C- Allopatric
done
clear
C)
A- Parapatric, B- Allopatric, C- Sympatric
done
clear
D)
A- Allopatric, B- Parapatric, C- Sympatric
done
clear
View Answer play_arrow
Which of the following is/are true for Leber's hereditary Optic Neuropathy (LHON)
I. It is mitochondrial myopathy
II. It is characterised by short stature
III. It is characterised by acromegaly
IV. It is characterised by visual lose
A)
All are false
done
clear
B)
II and III
done
clear
C)
I and IV
done
clear
D)
II and IV
done
clear
View Answer play_arrow
\[{{C}_{4}}-plants\] differ from \[{{C}_{3}}-plants\] in respect to
A)
Number of \[C{{O}_{2}}\] molecules used
done
clear
B)
Substrate, which accept the \[C{{O}_{2}}\] molecules
done
clear
C)
Number of ATP formed
done
clear
D)
Number of \[{{O}_{2}}\] formed
done
clear
View Answer play_arrow
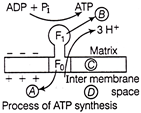
In the process of ATP synthesis, oligomycin and DCCD act at which place in figure
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A. Macrophytes 1. Rooted plants in shallow water B. Phagotrophe 2. Animals ingest food C. Abiotic components 3. Temperature D. Hydrophytes 4. Plants of dry areas 5. Plants of aquatic nasitants
Codes
A)
A-5, B-4, C-3, D-2
done
clear
B)
A-1, B-2, C-3, D-4
done
clear
C)
A-1, B-2, C-3, D-5
done
clear
D)
A-4, B-3, C-2, D-1
done
clear
View Answer play_arrow
In the modern era, due to excess stress and faulty life styles, mental problems are on increasing trend. Arrange the given component in correct sequence of cycle. 1. Avoid treatment 2. Silence and hiding 3. Stigma 4. Symptoms of mental illness 5. Discrimination
A)
A-3, B-1, C-2, D-4, E-5
done
clear
B)
A-5, B-4, C-3, D-1, E-2
done
clear
C)
A-4, B-3, C-5, D-2, E-1
done
clear
D)
A-1, B-4, C-5, D-2, E-3
done
clear
View Answer play_arrow
Assertion [A] The leaves of cauliflower become flaccid and gray in molybdenum deficiency. Reason [R] Cauliflower plant is affected by whiptail disease in molybdenum deficiency.
A)
Both A and R are true and R is correct explanation of A
done
clear
B)
Both A and R are true, but R is not the correct explanation of A
done
clear
C)
A is true but R is false
done
clear
D)
A is false but R is true
done
clear
View Answer play_arrow
Identify the incorrect combination from given options.
A)
Periyar - Elephant
done
clear
B)
Sunderban - Tiger
done
clear
C)
Dachigam National Park - Snow leopard,
done
clear
D)
Rann of Kutch - Wild ass
done
clear
View Answer play_arrow
Match the following columns
Column I (Mode of preservation of fossils) Column II (Comment) A. Adpression 1. Chdcalcified remains of plant tissue B. Fusain 2. Most commonly found plant fossils C. Petrifactions 3. Provide fine details D. Moulds and costs 4. Pith casts is it subset
Codes
A)
A-2, B-3, C-1, D-4
done
clear
B)
A-1, B-4, C-3, D-2
done
clear
C)
A-4, B-2, C-1, D-3
done
clear
D)
A-2, B-1, C-3, D-4
done
clear
View Answer play_arrow
Preparation of Fallopian tube for conveying developing embryo to uterus is aided by which of the following homona
A)
human placental lacto gen
done
clear
B)
oxytocin
done
clear
C)
progesterone
done
clear
D)
prolactin
done
clear
View Answer play_arrow
Which of the following are desirable properties for an ideal DNA markers? I. Highly polymorphic nature II. Codominant inheritance III. Frequent occurrence in genome IV. High reproducibility
A)
I and IV
done
clear
B)
II and III
done
clear
C)
I, II, III and IV
done
clear
D)
I and III
done
clear
View Answer play_arrow
Crown gall disease is caused which of the following pathogen.
A)
virus
done
clear
B)
bacteria
done
clear
C)
fungi
done
clear
D)
protozoans
done
clear
View Answer play_arrow
Which of the following is not an autotrophic microbe that fix atmospheric nitrogen?
A)
Anabaena
done
clear
B)
Rhizobium
done
clear
C)
Nostoc
done
clear
D)
Oscillatoria
done
clear
View Answer play_arrow
Which of the following is/are the first most common sign of pregnancy? I. Amenorrhea II. Quickening III. Ballottement of foetus IV. Foetal movement perceived by examiner
A)
I, II, III and IV
done
clear
B)
I and IV
done
clear
C)
Only I
done
clear
D)
II and III
done
clear
View Answer play_arrow
Choose the incorrect statement.
A)
Sieve tube elements resemble RBCs in being without nucleus in the mature state
done
clear
B)
Root hairs are found in zone of maturation
done
clear
C)
The common bottle cork is a product of xylem
done
clear
D)
Water containing cavities in vascular bundles are found in maize
done
clear
View Answer play_arrow
If your Renal Plasma Flow is 650 mL/min and Glomerular Filtration Rate is 125 mL/min. Calculate your Filtration Fraction.
A)
Given inform at ions are not complete
done
clear
B)
0.19
done
clear
C)
0.5
done
clear
D)
5.5
done
clear
View Answer play_arrow
In summer vacation you have visited to your friend's residence. He informed you that recently he has donated his kidney to his father. When you enquired further, you have been told that your friend had opted for laproscopic way of kidney donation, instead of open way of kidney donation. What might be the correct reason for it?
I. Your friend's age is below is 18 year.
II. It is less painful method
III. When laproscopic method is followed, other kidney does a quick hypertrophy.
IV. Incision is not visible (cosmetic reason).
A)
II and IV
done
clear
B)
All are true
done
clear
C)
All are false
done
clear
D)
I and II
done
clear
View Answer play_arrow
Below pie diagram shows the global biodiversity and proportionate numbers of vertebrates. Identify the markings if I. Fishes II. Amphibians III. Mammals IV. Birds V. Reptiles
A)
A-II, B-I, C-IV, D-V, E-III
done
clear
B)
A-V, B-III, C-I, D-IV, E-II
done
clear
C)
A-III, B-IV, C-V, D-II, E-I
done
clear
D)
A-I, B-IV, C-III, D-II, E-V
done
clear
View Answer play_arrow
A man has six daughters, what is the probability of next child being a son?
A)
\[10\,%\]
done
clear
B)
\[50\,%\]
done
clear
C)
\[100\,%\]
done
clear
D)
\[75\,%\]
done
clear
View Answer play_arrow
Which of the following gland do not secrete proteinaceous hormones?
A)
Pancreas
done
clear
B)
Thyroid gland
done
clear
C)
Parathyroid gland
done
clear
D)
Gonads
done
clear
View Answer play_arrow
In the given below fluid mosaic model of cell membrane, identify the markings I. Hydrophobic tails II. Cholesterol III. Integral protein IV. Hydrophilic heads
A)
A-IV, B-I, C-III, D-II
done
clear
B)
A-I, B-III, C-II, D-IV
done
clear
C)
A-III, B-II, C-IV, D-I
done
clear
D)
A-IV, B-I, C-III, D-II
done
clear
View Answer play_arrow
The aleurone layer in maize grain is specially rich in which of the following molecules
A)
starch
done
clear
B)
proteins
done
clear
C)
lipids
done
clear
D)
cellulose
done
clear
View Answer play_arrow
Study the following chain Sun \[\Downarrow \] Given 1000 units energy to phytoplanktons \[\Downarrow \] Phytoplanktons are consumed by hebivorous zooplankton, and in turn received A unit of energy, \[\Downarrow \] Carnivorous zooplanktons eat the herbivorous zooplankton and get B unit of energy. \[\Downarrow \] Carnivorous fish takes carnivorous zooplanktons and received C unit of energy. \[\Downarrow \] In last, Tunas find their turn and engulf carnivore fishes to get D unit of energy. Sun \[\Downarrow \] Given 1000 units energy to phytoplanktons \[\Downarrow \] Phytoplanktons are consumed by hebivorous zooplankton, and in turn received A unit of energy, \[\Downarrow \] Carnivorous zooplanktons eat the herbivorous zooplankton and get B unit of energy. \[\Downarrow \] Carnivorous fish takes carnivorous zooplanktons and received C unit of energy. \[\Downarrow \] In last, Tunas find their turn and engulf carnivore fishes to get D unit of energy. Identify A, B, C and D If I. 0.1 unit II. 10 unit III. 100 unit IV. 1 unit
A)
A-II, B-III, C-IV, D-I
done
clear
B)
A-IV, B-I, C-III, D-II
done
clear
C)
A-III, B-II, C-IV, D-I
done
clear
D)
Given information is not sufficient to answer
done
clear
View Answer play_arrow
Intertribal hybrids were first obtained by which of the following scientist
A)
Gibbs and Hofmann (1966)
done
clear
B)
Gibbs and Hofmann (1982)
done
clear
C)
Gibbs and Hofmann (1976)
done
clear
D)
Gibbs and Hofmann (1953)
done
clear
View Answer play_arrow
Label the given graph.
A)
(i)-Log phase (ii)-Exponential phase (iii)-Stationary phase
done
clear
B)
(i)-Exponential phase (ii)-Log phase (iii)-Stationary phase
done
clear
C)
(i)-Stationary phase (ii)-Log phase (iii)-Exponential phase
done
clear
D)
(i)-Log phase (ii)-Stationary phase (iii)-Exponential phase
done
clear
View Answer play_arrow
Identify the incorrect statement(s) from following options I. Glycolate is formed, in illuminated chloroplast of \[{{C}_{3}}\] plants. II. For every 2 glycolate molecules that leaves the chloroplast, one glycerate, derived from glycolate III. One of the 4 carbons from the two glycolates is lost as\[C{{O}_{2}}\]. IV. As 3-phosphoglycerate, the 3 remaining carbons can reenter the Calvin cycle.
A)
II and IV
done
clear
B)
I and III
done
clear
C)
None of these statement is false
done
clear
D)
I and II
done
clear
View Answer play_arrow
Choose the incorrect pair from the following options.
A)
Ethylene - Gas hormone
done
clear
B)
Abscisic acid - Flowering hormone
done
clear
C)
Auxin - To grow
done
clear
D)
Gibberellins - Gibbered fujikuroi
done
clear
View Answer play_arrow
Which of the following option is true regarding Flavr Savr?
A)
It is toxic insecticidal protein
done
clear
B)
It is slow ripening tomato
done
clear
C)
It is transgenic tomato
done
clear
D)
Its a pesticide variety
done
clear
View Answer play_arrow
The formation of acetyl coenzyme from pyruvic acid is the result of its
A)
reduction
done
clear
B)
dehydration
done
clear
C)
dephosphorylation
done
clear
D)
oxidative decarboxylation
done
clear
View Answer play_arrow
Assertion [A] Linnaeus system of animal classification is essentially an artificial system, yet it has become a natural system. Reason [R] Similarities forming the basis in Linnaeus systems are indicative of genetic relationship.
A)
Both A and R are true and R is the correct explanation of A
done
clear
B)
Both A and R are true, but R can?t explain A
done
clear
C)
Only A is the true and R is false
done
clear
D)
A is false and R is true
done
clear
View Answer play_arrow
Your one of the relatives has just married. She called you and requested to suggest the most appropriate method of contraception. Which of the following contraception would be your first suggested choice?
A)
Oral contraceptive pills
done
clear
B)
IUDS (Intrauterine devices)
done
clear
C)
Tubectomy
done
clear
D)
I-pill
done
clear
View Answer play_arrow
Taxonomic hierarchy refers to one of the following statement.
A)
nomenclature of plants and animal species by highly qualified group of individual
done
clear
B)
classification of all the species based on the phylogenetic analysis
done
clear
C)
classification of species on the basis of level in which they are present in food chain
done
clear
D)
stepwise arrangement of all categories for classification of plants and animals
done
clear
View Answer play_arrow
Spironolactone acts at which part of the nephron.
A)
Its site of action depends upon it concentration
done
clear
B)
It is still the matter of research
done
clear
C)
It acts on collecting tubules
done
clear
D)
It acts on PCT
done
clear
View Answer play_arrow
In a given case if \[C{{O}_{2}}\] liberated during respiration is more than the volume of \[{{O}_{2}}\] used, the substance will be
A)
fats
done
clear
B)
organic acid
done
clear
C)
protein
done
clear
D)
carbohydrate
done
clear
View Answer play_arrow
In the following processes of synthesis of norepinephrine identify the markings correctly. 1. \[A\xrightarrow{D}Dopa\] 2. \[\operatorname{Dopa} \xrightarrow{B} Dopamine\] 3. Transport of dopamine into the vesicles 4. \[Dopamine\text{ }\xrightarrow{C}\text{ }Norepinephrine\] If I. Tyrosine II. Hydroxylation III. Decarboxylation IV. Phenylacanin
A)
A-IV, B-II, C-III, D-III
done
clear
B)
A-I, B-II, C-III, D-III
done
clear
C)
A-I, B-III, C-II, D-II
done
clear
D)
A-IV, B-III, C-II, D-II
done
clear
View Answer play_arrow
Choose the correct statement in reference to primary myofilament.
A)
They are more than secondary myofilament
done
clear
B)
Primary myofilament have smooth surface
done
clear
C)
Primary my filament consists of two proteins
done
clear
D)
They slide during muscle contraction
done
clear
View Answer play_arrow
In a type of apomixis known as adventive embryony, embryos develop directly from the
A)
endosperm
done
clear
B)
nucellus or integuments
done
clear
C)
synergids
done
clear
D)
anti podals
done
clear
View Answer play_arrow
Which of the following is true for the arrangement of the nuclei in a Polygonum type of embryo sac in the angiospermic plants?
A)
\[2+4+2\]
done
clear
B)
\[2+3+3\]
done
clear
C)
\[3+2+3\]
done
clear
D)
\[3+3+2\]
done
clear
View Answer play_arrow
Which of the following is agranulocytic type of white blood cells?
A)
Lymphocyte
done
clear
B)
Neutrophil
done
clear
C)
Basophil
done
clear
D)
Eosinophil
done
clear
View Answer play_arrow
Which of the following option is correct for the life span of WBC?
A)
10 months
done
clear
B)
10 weeks
done
clear
C)
Less than 10 days
done
clear
D)
Less than 10 hours
done
clear
View Answer play_arrow
Consider the following statements. I. \[{{C}_{4}}\] species have greater rate of \[C{{O}_{2}}\] assimilation than \[{{C}_{3}}\] species. II. \[C{{O}_{2}}\] compensation point is very high in \[{{C}_{4}}\] plants. III. The process of photorespiration involves the chloroplasts, peroxysomes and mitochondria. Which of the statements given above are correct?
A)
I and II
done
clear
B)
I and III
done
clear
C)
II and III
done
clear
D)
I, II and III
done
clear
View Answer play_arrow
Which of the following EEG waves of Brain occur mainly in children and in adults during stress?
A)
Alpha waves
done
clear
B)
Beta waves
done
clear
C)
Delta waves
done
clear
D)
Theta waves
done
clear
View Answer play_arrow
Gossamer silk is produced by which of the following organism?
A)
Silk moth
done
clear
B)
Scorpion
done
clear
C)
Tusser moth
done
clear
D)
Spider
done
clear
View Answer play_arrow
Which of the following theories explains the structure of protoplasm?
A)
Surface-tension theory
done
clear
B)
Colloidal theory
done
clear
C)
Sol-gel theory
done
clear
D)
Viscosity theory
done
clear
View Answer play_arrow
Which of the following is a connecting link as well as a living fossil?
A)
Latimeria
done
clear
B)
Neopilina
done
clear
C)
Euglena
done
clear
D)
Archaeopteryx
done
clear
View Answer play_arrow
What is true about the first living organisms?
A)
They originated in an oxidising atmosphere.
done
clear
B)
They were aerobic
done
clear
C)
Made their own food
done
clear
D)
Obtained energy by fermentation of organic molecules
done
clear
View Answer play_arrow
Correct sequence of stage in evolution of Modern man. I. Australopithecus II. Homoerectus III. Neanderthal man IV. Cro-magnon man V. Modern man
A)
V, III, I, IV, II
done
clear
B)
I, II, III, IV, V
done
clear
C)
III, II, I, V, IV
done
clear
D)
II, IV, V, I, III
done
clear
View Answer play_arrow
Vermis is found in which of the following brain structure.
A)
cerebrum
done
clear
B)
medulla oblongata
done
clear
C)
cerebellum
done
clear
D)
brain stem
done
clear
View Answer play_arrow
Mental retardation is commonly associated with genetic diseas as, but it is not related with one of the genetic defect.
A)
trisomy 21
done
clear
B)
phenylalanine hydroxylase
done
clear
C)
myelinisation of neuron
done
clear
D)
Y-chromosome
done
clear
View Answer play_arrow
Which of the following is principal cation of blood plasma?
A)
\[M{{g}^{+\,2}}\]
done
clear
B)
\[{{K}^{+\,}}\]
done
clear
C)
\[N{{a}^{+\,2}}\]
done
clear
D)
\[C{{a}^{+\,2}}\]
done
clear
View Answer play_arrow
The genes, which remain confined to differential region of Y-chromosome are
A)
holandric genes
done
clear
B)
autosomal genes
done
clear
C)
completely sex-linked genes
done
clear
D)
mutant genes
done
clear
View Answer play_arrow
Which of the following statement is not correct?
A)
Pineal gland secretes serotonin and melatonin
done
clear
B)
Adrenaline is an amine
done
clear
C)
Kidney do not secrete steroid hormones
done
clear
D)
Oxytocin is secreted by posterior lobe of pituitary
done
clear
View Answer play_arrow
Which among the following is a function of restriction enzyme in nature?
A)
Plasmids
done
clear
B)
Destroy foreign DNA
done
clear
C)
Destroy phage DNA
done
clear
D)
Splice up DNA of the cells
done
clear
View Answer play_arrow
Choose the incorrect statement.
A)
RNA is a genetic material
done
clear
B)
DNA replication is semiconservative
done
clear
C)
Ribonucleoside \[\to \] Nitrogenous base \[\to \] Ribonucleotide
done
clear
D)
Adenine and guanine are purines.
done
clear
View Answer play_arrow
Which transgenic food crop helps in solving the problem of night blindness?
A)
Transgenic maize
done
clear
B)
Golden rice
done
clear
C)
Bt soyabean
done
clear
D)
Bt brinjal
done
clear
View Answer play_arrow
Down's syndrome is due to
A)
non-disjunction of chromosome
done
clear
B)
duplication
done
clear
C)
crossing over
done
clear
D)
linkage
done
clear
View Answer play_arrow
Which of the following is incorrect? I. Stabilising selection - Favours average size individuals. II. Disruptive selection - Favours exotic species III. Directional selection - Favours both small and large sized individuals IV. Industrial melanism - Supports evolution by use and disuse
A)
All except II
done
clear
B)
All except I
done
clear
C)
All of these
done
clear
D)
None of these
done
clear
View Answer play_arrow
Why the tropical deciduous trees shed their leaves?
A)
To protect itself from heat and water
done
clear
B)
To enhance rate of respiration and rate of absorption
done
clear
C)
To reduce the rate of transpiration
done
clear
D)
To save energy
done
clear
View Answer play_arrow
GMFs have proven their importance as
A)
they cause less damage to environment
done
clear
B)
they allow the increase of crop quantity
done
clear
C)
they improve profitability
done
clear
D)
they improve convince for customers.
done
clear
View Answer play_arrow
Which is not X-linked?
A)
Duchenne muscular dystrophy
done
clear
B)
Protanopia
done
clear
C)
GbPD deficiency
done
clear
D)
Congenital adrenal hyperplasia
done
clear
View Answer play_arrow
The cell is fully turgid when one of the following condition fulfilled.
A)
Diffusion pressure deficit = Turgor pressure
done
clear
B)
Diffusion pressure deficit = Suction pressure
done
clear
C)
Osmotic pressure = Diffusion pressure deficit
done
clear
D)
Diffusion pressure deficit = Zero
done
clear
View Answer play_arrow
Which of the following statement(s) is/are true for vccine?
A)
collection of attenuated disease causing bacteria and virus
done
clear
B)
collection of microorganism
done
clear
C)
collection of killed a disease causing bacteria and virus
done
clear
D)
Both [a] and [c]
done
clear
View Answer play_arrow
Which of the following is the golden age of reptiles?
A)
Mesozoic era
done
clear
B)
Archaeozoic era
done
clear
C)
Coenozoic era
done
clear
D)
Paraepzpicera
done
clear
View Answer play_arrow
In our nature, cleistogamous type of flowers are
A)
animal pollinated
done
clear
B)
insect-pollinated
done
clear
C)
bird pollinated
done
clear
D)
self-pollinated
done
clear
View Answer play_arrow
Which of the following is not an autosomal recessive trait?
A)
Phenylketonuria
done
clear
B)
Cystic fibrosis
done
clear
C)
Albinism
done
clear
D)
Polydactyl
done
clear
View Answer play_arrow
Blunt ends are produced by which of the following restriction enzyme?
A)
Alu I
done
clear
B)
Pst I
done
clear
C)
Sma I
done
clear
D)
Hind III
done
clear
View Answer play_arrow
Bolistic technique is characterisation of
A)
germplasm conservation
done
clear
B)
conjugation
done
clear
C)
tissue culture
done
clear
D)
gene transfer process
done
clear
View Answer play_arrow
Auxin (IAA) was isolated by Thiamann in 1935 from
A)
bryophytes
done
clear
B)
algae
done
clear
C)
fungi
done
clear
D)
pteridophytes
done
clear
View Answer play_arrow
Under given suitable conditions, the diffusion pressure deficit will be more than osmotic pressure.
A)
When osmotic pressure is equal to turgor pressure
done
clear
B)
When osmotic pressure is less than turgor pressure
done
clear
C)
When osmotic pressure is greater than turgor pressure
done
clear
D)
When turgor pressure is negative
done
clear
View Answer play_arrow
Viriods differ from viruses in having
A)
naked DNA molecule only
done
clear
B)
naked DNA packed with viral genome
done
clear
C)
satellite RNA packed with viral genome only
done
clear
D)
naked RNA molecule only
done
clear
View Answer play_arrow
The joint between atlas and axis is called
A)
angular joint
done
clear
B)
pivot joint
done
clear
C)
saddle joint
done
clear
D)
hinge joint
done
clear
View Answer play_arrow
Which one of the following is Permanent cure of Adenosine Deaminase (ADA) deficiency is
A)
enzyme replacement therapy
done
clear
B)
introducing isolated gene from marrow cells producing ADA into the cells at early embryonic stages
done
clear
C)
injecting the functional ADA, CDNA into the patient
done
clear
D)
administering adenosine deaminase through injections
done
clear
View Answer play_arrow
Which of the following is correct pairing of site of action and substrate of rennin?
A)
Mouth-starch
done
clear
B)
Stomach-fat
done
clear
C)
Stomach-caseinogen
done
clear
D)
Small intestine-fat
done
clear
View Answer play_arrow
Which of the following stabilises microtubules and interferes with mitosis?
A)
Colchicine
done
clear
B)
Tubulin
done
clear
C)
Taxol
done
clear
D)
Vincristine
done
clear
View Answer play_arrow
Which of the following is an example of gene silencing?
A)
Bt brinjal
done
clear
B)
Bt cotton
done
clear
C)
Golden rice
done
clear
D)
Flavr savr tomato
done
clear
View Answer play_arrow

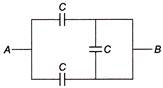







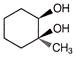
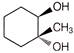
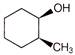




 Components are
Components are 
 If
If 
