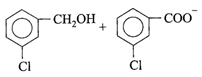
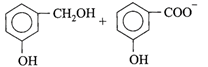
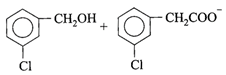
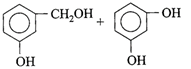
Candela is the unit of
A)
Electric intensity
done
clear
B)
Luminous intensity
done
clear
C)
Sound intensity
done
clear
D)
None of these
done
clear
View Answer play_arrow
The displacement of a particle, moving in a straight line, is given by \[s=2{{t}^{2}}+2t+4\] where 5 is in metres and t in seconds. The acceleration of the particle is
A)
\[2\text{ }m/{{s}^{2}}\]
done
clear
B)
\[~4\text{ }m/{{s}^{2}}\]
done
clear
C)
\[~6\text{ }m/{{s}^{2}}\]
done
clear
D)
\[~8\text{ }m/{{s}^{2}}\]
done
clear
View Answer play_arrow
A car runs at a constant speed on a circular track of radius 100 m, taking 62.8 s for every circular loop. The average velocity and average speed for each circular loop respectively is:
A)
0, 0
done
clear
B)
0, 10 m/s
done
clear
C)
10 m/s, 10 m/s
done
clear
D)
10 m/s, 0
done
clear
View Answer play_arrow
A man weighing 80 kg is standing in a trolley weighing 320 kg. The trolley is resting on frictionless horizontal rails. If the man starts walking on the trolley with a speed of 1 m/s, then after 4 sec his displacement relative to the ground will be
A)
5 m
done
clear
B)
4.8 m
done
clear
C)
3.2 m
done
clear
D)
3.0 m
done
clear
View Answer play_arrow
A mass of 0.5 kg moving with a speed of 1.5 m/s on a horizontal smooth surface collides with a nearly weightless spring of force constant \[k=50\,n/m.\]The maximum compression of the spring would be:
A)
0.15 m
done
clear
B)
0.12 m
done
clear
C)
1.5 m
done
clear
D)
0.5 m
done
clear
View Answer play_arrow
A wheel of radius 1 m rolls forward half a revolution on a horizontal ground. The magnitude of the displacement of the point of the wheel initially in contact with the ground is
A)
\[\pi \]
done
clear
B)
\[2\pi \]
done
clear
C)
\[\sqrt{2}\pi \]
done
clear
D)
\[\sqrt{{{\pi }^{2}}+4}\]
done
clear
View Answer play_arrow
Consider a system of two particles having masses \[{{m}_{1}}\]and\[{{m}_{2}}.\]If the particle of mass\[{{m}_{1}}\]is pushed towards the mass centre of particles through a distance d, by what distance would the particle of mass \[{{m}_{2}}\]move so as to keep the mass centre of particles at the original position?
A)
\[\frac{{{m}_{1}}}{{{m}_{1}}+{{m}_{2}}}d\]
done
clear
B)
\[\frac{{{m}_{1}}}{{{m}_{2}}}d\]
done
clear
C)
d
done
clear
D)
\[\frac{{{m}_{2}}}{{{m}_{1}}}d\]
done
clear
View Answer play_arrow
The acceleration due to gravity on the planet A is 9 times the acceleration due to gravity on planet B. A man jumps to a height of 2 m on the surface of A. What is the height of jump by the same person on the planet B?
A)
\[\frac{2}{3}m\]
done
clear
B)
\[\frac{2}{9}m\]
done
clear
C)
18 m
done
clear
D)
6 m
done
clear
View Answer play_arrow
For a satellite moving in an orbit around the earth, the ratio of kinetic energy to potential energy is
A)
2
done
clear
B)
\[\frac{1}{2}\]
done
clear
C)
\[\frac{1}{\sqrt{2}}\]
done
clear
D)
\[\sqrt{2}\]
done
clear
View Answer play_arrow
Copper of fixed volume V is drawn into wire of length \[l.\] When this wire is subjected to a constant force F, the extension produced in the wire is \[\Delta l.\]Which of the following graphs is a straight line?
A)
\[\Delta l\] versus\[\frac{1}{l}\]
done
clear
B)
\[\Delta l\]versus \[{{l}^{2}}\]
done
clear
C)
\[\Delta l\]versus\[\frac{1}{{{l}^{2}}}\]
done
clear
D)
\[\Delta l\]versus \[l\]
done
clear
View Answer play_arrow
A wind with speed 40 m/s blows parallel to the roof of a house. The area of the roof is \[250\text{ }{{m}^{2}}.\] Assuming that the pressure inside the house is atmospheric pressure, the force exerted by the wind on the roof and the direction of the force will be\[({{P}_{air}}=1.2\,kg/{{m}^{3}})\]
A)
\[4.8\times {{10}^{5}}N,\] downwards
done
clear
B)
\[4.8\times {{10}^{5}}N,\]upwards
done
clear
C)
\[2.4\times {{10}^{5}}N,\]upwards
done
clear
D)
\[2.4\times {{10}^{5}}N,\] downwards
done
clear
View Answer play_arrow
Which of the following is the example of ideal black body?
A)
Kajal
done
clear
B)
Blackboard
done
clear
C)
A pin hole in a box
done
clear
D)
None of these
done
clear
View Answer play_arrow
We consider the radiation emitted by the human body. Which of the following statements is true?
A)
The radiation is emitted only during the day
done
clear
B)
The radiation is emitted during the summers and absorbed during the winters
done
clear
C)
The radiation emitted lies in the ultraviolet region and hence is not visible
done
clear
D)
The radiation emitted is in the infrared region
done
clear
View Answer play_arrow
Efficiency of a Carnot engine is 50% when temperature of outlet is 500 K-. In order to increase efficiency up to 60% keeping temperature of intake the same what is temperature of outlet
A)
200 K
done
clear
B)
400 K
done
clear
C)
600 K
done
clear
D)
800 K
done
clear
View Answer play_arrow
The equation of state for 5 g of oxygen at a pressure P and temperature T, when occupying a volume V, will be:
A)
\[PV=(5/32)RT\]
done
clear
B)
\[PV=5RT\]
done
clear
C)
\[PV=(5/2)RT\]
done
clear
D)
\[PV=(5/16)RT\]
done
clear
View Answer play_arrow
There is a body having mass m and performing SHM with amplitude a. There is a restoring force \[F=-Kx,\]where\[x\] is the displacement. The total energy of body depends upon
A)
\[K,x\]
done
clear
B)
\[K,a\]
done
clear
C)
\[K,a,x\]
done
clear
D)
\[K,a,v\]
done
clear
View Answer play_arrow
The time period of a mass suspended from a spring is T. If the spring is cut into four equal parts and the same mass is suspended from one of the parts, then the new time period will be
A)
T
done
clear
B)
\[\frac{T}{2}\]
done
clear
C)
\[2T\]
done
clear
D)
\[\frac{T}{4}\]
done
clear
View Answer play_arrow
The time of reverberation of a room A is one second. What will be the time (in seconds) of reverberation of a room, having all the dimensions double of those of room A?
A)
2
done
clear
B)
4
done
clear
C)
\[\frac{1}{2}\]
done
clear
D)
1
done
clear
View Answer play_arrow
The driver of a car travelling with speed \[30\,m{{s}^{-1}}\] towards a hill sounds a horn of frequency 600 Hz. If the velocity of sound in air is\[330\,m{{s}^{-1}},\]the frequency of reflected sound as heard by driver is
A)
550 Hz
done
clear
B)
555.5 Hz
done
clear
C)
720 HZ
done
clear
D)
500 Hz
done
clear
View Answer play_arrow
The mean free path of electrons in a metal is \[4\times {{10}^{-8}}m.\]The electric field which can give on an average 2 eV energy to an electron in the metal will be in unit of \[V{{m}^{-1}}\]
A)
\[8\times {{10}^{-7}}\]
done
clear
B)
\[5\times {{10}^{-11}}\]
done
clear
C)
\[8\times {{10}^{-11}}\]
done
clear
D)
\[5\times {{10}^{7}}\]
done
clear
View Answer play_arrow
The electric potential V at any point \[(x,y,z),\]all in metres in space is given by \[V=4{{x}^{2}}\]volt. The electric field at the point (1,0,2) in volt/metre is
A)
8 along positive X-axis
done
clear
B)
16 along negative Y-axis
done
clear
C)
16 along positive X-axis
done
clear
D)
8 along, negative X-axis
done
clear
View Answer play_arrow
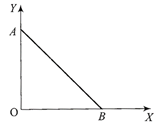
As per this diagram a point charge + q is placed at the origin O. Work done in taking another point charge -Q from the point A [co-ordinates (0, a)] to another point B [co-ordinates (a, 0)] along the straight path AB is
A)
Zero
done
clear
B)
\[\left( \frac{-qQ}{4\pi {{\varepsilon }_{0}}}\frac{1}{{{a}^{2}}} \right)\sqrt{2}\,a\]
done
clear
C)
\[\left( \frac{qQ}{4\pi {{\varepsilon }_{0}}}\frac{1}{{{a}^{2}}} \right)\frac{a}{\sqrt{2}}\]
done
clear
D)
\[\left( \frac{qQ}{4\pi {{\varepsilon }_{0}}}\frac{1}{{{a}^{2}}} \right)\sqrt{2}\]
done
clear
View Answer play_arrow
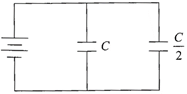
Two condensers, one of capacity C and the other of capacity C/2, are connected to a F volt battery, as shown.
The work done in charging fully both the condensers is
A)
\[2C{{V}^{2}}\]
done
clear
B)
\[\frac{1}{4}C{{V}^{2}}\]
done
clear
C)
\[\frac{3}{4}C{{V}^{2}}\]
done
clear
D)
\[\frac{1}{2}C{{V}^{2}}\]
done
clear
View Answer play_arrow
A 6 V battery is connected to the terminals of a three metre long wire of uniform thickness and resistance of \[100\Omega .\]The difference of potential, between two points on the wire separated by a distance or 50 cm, will be
A)
2 V
done
clear
B)
3 V
done
clear
C)
1 V
done
clear
D)
1.5 V
done
clear
View Answer play_arrow
In a Wheatstone's bridge all the four arms have equal resistance R. If the resistance of the galvanometer arm is also R, the equivalent resistance of the combination as seen by the battery is
A)
\[\frac{R}{2}\]
done
clear
B)
R
done
clear
C)
2 R
done
clear
D)
\[\frac{R}{4}\]
done
clear
View Answer play_arrow
A cell can be balanced against 110 cm and 100 cm of potentiometer wire, respectively with and without being short circuited through a resistance of \[10\,\Omega .\] Its internal resistance is
A)
\[1.0\Omega \]
done
clear
B)
\[0.5\,\Omega \]
done
clear
C)
\[2.0\,\Omega \]
done
clear
D)
zero
done
clear
View Answer play_arrow
The rate of increase of thermo-emf with temperature at the neutral temperature of a thermo- couple
A)
is zero
done
clear
B)
depends upon the choice of the two materials of the thermocouple
done
clear
C)
is negative
done
clear
D)
is positive
done
clear
View Answer play_arrow
An electric current passes through a long straight wire. At a distance 5 cm from the wire the magnetic field is B. The field at 20 cm from the wire would be
A)
\[\frac{B}{6}\]
done
clear
B)
\[\frac{B}{4}\]
done
clear
C)
\[\frac{B}{3}\]
done
clear
D)
\[\frac{B}{2}\]
done
clear
View Answer play_arrow
A galvanometer of \[50\,\Omega \]resistance has 25 divisions. A current of \[4\times {{10}^{-4}}\] A gives a deflection of one division. To convert this galvanometer into a voltmeter having a range of 25 V, it should be connected with a resistance of
A)
\[2500\,\Omega \] as a shunt
done
clear
B)
\[245\,\Omega \] as a shunt
done
clear
C)
\[2550\,\Omega \]in series
done
clear
D)
\[2450\,\Omega \] in series
done
clear
View Answer play_arrow
Nickel shows ferromagnetic property at room temperature. If the temperature is increased beyond Curie temperature, then it will show
A)
paramagnetism
done
clear
B)
anti-ferromagnetism
done
clear
C)
no magnetic property
done
clear
D)
diamagnetism
done
clear
View Answer play_arrow
A compass needle which is allowed to move in a horizontal plane is taken to a geomagnetic pole. It
A)
will stay in east-west direction only
done
clear
B)
will become rigid showing no movement
done
clear
C)
will stay in any position
done
clear
D)
will stay in north-south direction only
done
clear
View Answer play_arrow
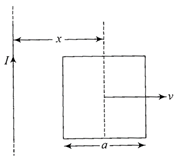
A conducting square frame of side a and a long straight wire carrying current / are located in the same plane as shown in the figure. The frame moves to the right with a constant velocity V. The emf induced in the frame will be proportional to
A)
\[\frac{1}{{{x}^{2}}}\]
done
clear
B)
\[\frac{1}{{{(2x-a)}^{2}}}\]
done
clear
C)
\[\frac{1}{{{(2x+a)}^{2}}}\]
done
clear
D)
\[\frac{1}{(2x-a)(2x+a)}\]
done
clear
View Answer play_arrow
A conducting circular loop is placed in a uniform magnetic field 0.04 T with its plane perpendicular to the magnetic field. The radius of the loop starts shrinking at \[2mm{{s}^{-1}}.\] The induced emf in the loop when the radius is 2 cm is
A)
\[3.2\,\pi \mu V\]
done
clear
B)
\[4.8\,\pi \mu V\]
done
clear
C)
\[0.8\,\pi \mu V\]
done
clear
D)
\[1.6\,\pi \mu V\]
done
clear
View Answer play_arrow
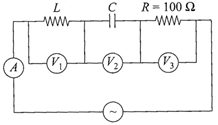
In the given circuit the reading of voltmeter \[{{V}_{1}}\]and \[{{V}_{2}}\]are 300 V each. The reading to the voltmeter \[{{V}_{3}}\]and ammeter A are respectively
A)
150 V, 2.2 A
done
clear
B)
200 V, 2.2 A
done
clear
C)
220 V, 2.0 A
done
clear
D)
100 V, 2.0 A
done
clear
View Answer play_arrow
The instantaneous values of alternating current and voltages in a circuit are given as\[i=\frac{1}{\sqrt{2}}\sin (100\pi t)\]ampere\[e=\frac{1}{\sqrt{2}}\sin (100\pi t+\pi /3)\,\text{volt}\]. The average power in watts consumed in the circuit is
A)
\[\frac{1}{4}\]
done
clear
B)
\[\frac{\sqrt{3}}{4}\]
done
clear
C)
\[\frac{1}{2}\]
done
clear
D)
\[\frac{1}{8}\]
done
clear
View Answer play_arrow
Which of the following statement is false for the properties of electromagnetic waves?
A)
Both electric and magnetic field vectors attain the maxima and minima at the same place and the same time
done
clear
B)
The energy in electromagnetic wave is divided equally between electric and magnetic vectors
done
clear
C)
Both electric and magnetic field vectors are parallel to each other and perpendicular to the direction of propagation of wave
done
clear
D)
These waves do not require any material medium for propagation
done
clear
View Answer play_arrow
A radiation of energy 'E' falls normally on a perfectly reflecting surface. The momentum transferred to the surface is (C = velocity of light)
A)
\[\frac{E}{C}\]
done
clear
B)
\[\frac{2E}{C}\]
done
clear
C)
\[\frac{2E}{{{C}^{2}}}\]
done
clear
D)
\[\frac{E}{{{C}^{2}}}\]
done
clear
View Answer play_arrow
The chromatic aberration in lenses is due to
A)
Dissimilarity of main axis of rays
done
clear
B)
Dissimilarity of radii of curvature
done
clear
C)
Variation of focal length of lenses with wavelength
done
clear
D)
None of these
done
clear
View Answer play_arrow
The angular resolution of a 10 cm diameter telescope at a wavelength of \[5000\text{ }\overset{\text{o}}{\mathop{\text{A}}}\,\]is of the order
A)
\[{{10}^{6}}rad\]
done
clear
B)
\[{{10}^{-2}}rad\]
done
clear
C)
\[{{10}^{-4}}rad\]
done
clear
D)
\[{{10}^{-6}}rad\]
done
clear
View Answer play_arrow
Red light is generally used to observe diffraction pattern from single slit. If blue light is used instead of red light, then diffraction pattern.
A)
Will be more-clear
done
clear
B)
Will contract
done
clear
C)
Will expanded
done
clear
D)
Will not be visualized
done
clear
View Answer play_arrow
In a double-slit experiment, the two slits are 1 mm apart and the screen is placed 1 m away. A monochromatic light of wavelength 500 nm is used. What will be the width of each slit for obtaining ten maxima of double slit within the central maxima of single-slit pattern?
A)
0.2 mm
done
clear
B)
0.1 mm
done
clear
C)
0.5 mm
done
clear
D)
0.02 mm
done
clear
View Answer play_arrow
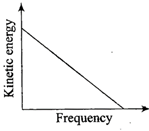
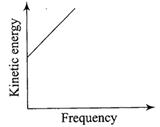
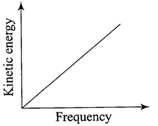
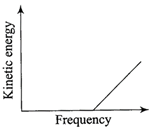
According to Einstein's photoelectric equation, the graph between the kinetic energy of photo- electrons ejected and the frequency of incident radiation is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
A 5 W source emits monochromatic light of wavelength\[5000\text{ }\overset{\text{o}}{\mathop{\text{A}}}\,\]. When placed 0.5 m away, it liberates photoelectrons from a photosensitive metallic surface. When the source is moved to a distance of 1.0 m the number of photoelectrons liberated will be reduced by a factor of
A)
4
done
clear
B)
8
done
clear
C)
16
done
clear
D)
2
done
clear
View Answer play_arrow
The energy of a hydrogen atom in the ground state is -13.6 eV. The energy of a\[H{{e}^{+}}\,ion\] in the first excited state will be
A)
-13.6 eV
done
clear
B)
-27.2 eV
done
clear
C)
-54.4 eV
done
clear
D)
-6.8 eV
done
clear
View Answer play_arrow
The transition from the state n = 3 to n = 1 in a hydrogen-like atom results in ultraviolet radiation. Infrared radiation will be obtained in the transition from
A)
\[2\to 1\]
done
clear
B)
\[3\to 2\]
done
clear
C)
\[4\to 2\]
done
clear
D)
\[4\to 3\]
done
clear
View Answer play_arrow
Percentage of Se in peroxidase anhydrase enzyme is 0.5% by weight (at. wt. = 78.4), then minimum molecular weight of peroxidase anhydrase enzyme is:
A)
\[1.568\times {{10}^{3}}\]
done
clear
B)
15.68
done
clear
C)
\[1.568\times {{10}^{4}}\]
done
clear
D)
\[2.136\times {{10}^{4}}\]
done
clear
View Answer play_arrow
The value of Planck's constant is \[6.63\times {{10}^{-34}}\,Js.\] The velocity of light is \[3.0\times {{10}^{8}}m{{s}^{-1}}.\] Which value is closest to the wavelength in nanometer of a quantum of light with frequency of\[8\times {{10}^{15}}{{s}^{-1}}?\]
A)
\[2\times {{10}^{-25}}nm\,\]
done
clear
B)
\[5\times {{10}^{-18}}\,nm\]
done
clear
C)
4 nm
done
clear
D)
\[3\times {{10}^{-7}}\,nm\]
done
clear
View Answer play_arrow
The species \[Ar,{{K}^{+}}\]and \[C{{a}^{2+}}\]contain the same number of electrons. In which order do their radii increase?
A)
\[C{{a}^{2+}}<{{K}^{+}}<Ar\]
done
clear
B)
\[{{K}^{+}}<Ar<C{{a}^{2+}}\]
done
clear
C)
\[Ar<{{K}^{+}}<C{{a}^{2+}}\]
done
clear
D)
\[C{{a}^{2+}}<Ar<{{K}^{+}}\]
done
clear
View Answer play_arrow
In\[X-H---Y,\]both X and Y are electronegative elements
A)
Electron density on X will increase and on H will decrease
done
clear
B)
In both electron density will decrease
done
clear
C)
In both electron density will increase
done
clear
D)
Electron density will decrease on X and will increase on H
done
clear
View Answer play_arrow
By what factor does the average velocity of a gaseous molecule increase when the temperature (in Kelvin) is doubled?
A)
1.4
done
clear
B)
2.0
done
clear
C)
2.8
done
clear
D)
4.0
done
clear
View Answer play_arrow
In a closed insulated container a liquid is stirred with a paddle to increase the temperature, which of the following is true?
A)
\[\Delta U+W\ne 0,q=0\]
done
clear
B)
\[\Delta U+W=q\ne 0\]
done
clear
C)
\[\Delta U+Q,W=q\ne 0\]
done
clear
D)
\[W=0,\Delta U=q\ne 0\]
done
clear
View Answer play_arrow
For which reaction\[{{K}_{p}}\ne {{K}_{c}}?\]
A)
\[2N{{O}_{2}}(g)\rightleftharpoons {{N}_{2}}(g)+{{O}_{2}}(g)\]
done
clear
B)
\[S{{O}_{2}}(g)+N{{O}_{2}}(g)\rightleftharpoons S{{O}_{3}}(g)+NO(g)\]
done
clear
C)
\[{{I}_{2}}(g)+{{H}_{2}}(g)\rightleftharpoons 2HI(g)\]
done
clear
D)
\[2C(s)+{{O}_{2}}(g)\xrightarrow{{}}2CO(g)\]
done
clear
View Answer play_arrow
In \[H{{S}^{-}},{{I}^{-}},R-N{{H}_{2}},N{{H}_{3}}\]order of proton accepting tendency will be
A)
\[{{I}^{-}}>N{{H}_{3}}>RN{{H}_{2}}>H{{S}^{-}}\]
done
clear
B)
\[N{{H}_{3}}>RN{{H}_{2}}>H{{S}^{-}}>{{I}^{-}}\]
done
clear
C)
\[RN{{H}_{2}}>N{{H}_{3}}>H{{S}^{-}}>{{I}^{-}}\]
done
clear
D)
\[H{{S}^{-}}>RN{{H}_{2}}>N{{H}_{3}}>{{I}^{-}}\]
done
clear
View Answer play_arrow
Oxidation no. of P in \[{{H}_{4}}{{P}_{2}}{{O}_{5}},\text{ }{{H}_{4}}{{P}_{2}}{{O}_{6}},\]and \[{{H}_{4}}{{P}_{2}}{{O}_{7}}\]are respectively
A)
+3, +4, +5
done
clear
B)
+4, +3, +5
done
clear
C)
+3, +5, +4
done
clear
D)
+5, +3, +4
done
clear
View Answer play_arrow
Which statement about zeolite is false?
A)
They are used as cation exchanger
done
clear
B)
They have open structure which enables them to take up small molecules
done
clear
C)
Zeolites are alumino silicates having three dimensional networks
done
clear
D)
Some of the\[SiO_{4}^{4-}\]units are replaced by \[AlO_{4}^{5-}\] and\[AlO_{6}^{9-}\]ions in zeolites
done
clear
View Answer play_arrow
Match List I (substance) with List II (processes) employed in the manufacture of the substance and select the correct option.
List I (Substances) List II (Processes) [a] Sulphuric acid (i) Haber's process [b] Steel (ii) Bessemer's process [c] Sodium hydroxide (iii) Lebanc process [d] Ammonia (iv) Contact process
A)
(A-i), (B-iv), (C-ii), (D-iii)
done
clear
B)
(A-i), (B-ii), (C-iii), (D-iv)
done
clear
C)
(A-iv), (B-iii), (C-ii), (D-i)
done
clear
D)
(A-iv), (B-ii), (C-iii), (D-i)
done
clear
View Answer play_arrow
\[{{H}_{2}}O\]is dipolar, whereas \[Be{{F}_{2}}\]is not. It is because
A)
the electronegativity of F is greater than O
done
clear
B)
\[{{H}_{2}}O\]involves H-bonding, whereas\[Be{{F}_{2}}\]is discrete molecule
done
clear
C)
\[{{H}_{2}}O\]is linear, and\[Be{{F}_{2}}\]is angular
done
clear
D)
\[{{H}_{2}}O\]is angular, and\[Be{{F}_{2}}\]is linear
done
clear
View Answer play_arrow
\[A{{l}_{2}}{{O}_{3}}\]can be converted to anhydrous \[AlC{{l}_{3}}\]by heating
A)
a mixture of\[A{{l}_{2}}{{O}_{3}}\]and carbon in dry \[C{{l}_{2}}\]gas
done
clear
B)
\[A{{l}_{2}}{{O}_{3}}\]with \[C{{l}_{2}}\] gas
done
clear
C)
\[A{{l}_{2}}{{O}_{3}}\] and HCl gas
done
clear
D)
\[A{{l}_{2}}{{O}_{3}}\] with NaCl in solid state
done
clear
View Answer play_arrow
Name the type of the structure of silicate in which one oxygen atom of \[{{[Si{{O}_{4}}]}^{4-}}\]is shared
A)
Three dimensional
done
clear
B)
Linear chain silicate
done
clear
C)
Sheet silicate
done
clear
D)
Pyrosilicate
done
clear
View Answer play_arrow
In Kjeldahl's method, nitrogen present is estimated as:
A)
\[{{N}_{2}}\]
done
clear
B)
\[~N{{H}_{3}}\]
done
clear
C)
\[~N{{O}_{2}}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
The Lassaigne's extract is boiled with conc. \[HN{{O}_{3}}\]while testing for halogens. By doing so it:
A)
increases the concentration of\[NO_{3}^{-}\]ions
done
clear
B)
decomposes \[N{{a}_{2}}S\]and NaCN, if formed
done
clear
C)
helps in the precipitation of AgCl
done
clear
D)
increases the solubility product of AgCl
done
clear
View Answer play_arrow
The stability of carbanions in the following:
Is in the order of:
A)
(2) > (3) > (4) > (1)
done
clear
B)
(4) > (2) > (3) > (1)
done
clear
C)
(1) > (3) > (2) > (4)
done
clear
D)
(1) > (2) > (3) > (4)
done
clear
View Answer play_arrow
The IUPAC name of the compound \[C{{H}_{3}}CH=CHC\text{ }\equiv \text{ }CH\]
A)
pent-4-yn-2-ene
done
clear
B)
pent-3-en-l-yne
done
clear
C)
pent-2-en-4-yne
done
clear
D)
pentl-lyn-3-ene
done
clear
View Answer play_arrow
Which of the following acids does not exhibit optical isomerism?
A)
Lactic acid
done
clear
B)
Tartaric acid
done
clear
C)
Maleicacid
done
clear
D)
done
clear
View Answer play_arrow
Two possible stereostructures of\[C{{H}_{3}}CHOH.COOH\]Which are optically active, are called:
A)
Enantiomers
done
clear
B)
Mesomers
done
clear
C)
Diastereomers
done
clear
D)
Atropisomers
done
clear
View Answer play_arrow
Benzene reacts with \[C{{H}_{2}}Cl\]in the presence of anhydrous \[AlC{{l}_{3}}\]to form
A)
xylene
done
clear
B)
toluene
done
clear
C)
chlorobenzene
done
clear
D)
benzylchloride
done
clear
View Answer play_arrow
Which of the following compounds will not undergo Friedel-Crafts reaction easily?
A)
Xylene
done
clear
B)
Nitrobenzene
done
clear
C)
Toluene
done
clear
D)
Cumene
done
clear
View Answer play_arrow
The greenhouse effect is because of the
A)
presence of gases, with in general are strong infrared absorbers, in the atmosphere
done
clear
B)
presence of\[C{{O}_{2}}\]only in the atmosphere
done
clear
C)
presence of \[{{O}_{3}}\] and \[C{{H}_{4}}\]in the atmosphere
done
clear
D)
\[{{N}_{2}}O\]and chlorofluorocarbons in the at- mosphere
done
clear
View Answer play_arrow
Which of the following is not a common component of photochemical smog?
A)
Ozone
done
clear
B)
Acrolein
done
clear
C)
Peroxyacetyl nitrate
done
clear
D)
Chlorofluorocarbons
done
clear
View Answer play_arrow
Number of neutrons in a parent nucleus X which gives\[_{7}{{N}^{14}}\]after two successive\[\beta -\]emissions would be
A)
6
done
clear
B)
7
done
clear
C)
8
done
clear
D)
9
done
clear
View Answer play_arrow
The half-life of a substance in a certain enzyme- catalysed reaction is 13 8 s. The time required for the concentration of the substance to fall from \[1.28\text{ }mg\text{ }{{L}^{-1}}\]to \[0.04\text{ }mg\text{ }{{L}^{-1}}\] is
A)
414 s
done
clear
B)
552 s
done
clear
C)
690 s
done
clear
D)
276 s
done
clear
View Answer play_arrow
Percentage of free space in a body-centred cubic unit cell is
A)
32%
done
clear
B)
34%
done
clear
C)
28%
done
clear
D)
30%
done
clear
View Answer play_arrow
A given metal crystallises out with a cubic structure having edge length of 361 pm. If there are four metal atoms in one unit cell, what is the radius of one atom?
A)
80 pm
done
clear
B)
108 pm
done
clear
C)
40 pm
done
clear
D)
127 pm
done
clear
View Answer play_arrow
The vapour pressure of two liquids P and Q are 80 ton- and 60 ton- respectively. The total vpour pressure obtained by mixing 3 mole of P and 2 mole of Q would be
A)
68 torr
done
clear
B)
20 torr
done
clear
C)
140 torr
done
clear
D)
72 torr
done
clear
View Answer play_arrow
\[{{p}_{A}}\]and\[{{p}_{B}}\]are the vapour pressure of pure liquid components, A and B, respectively of an ideal binary solution. If \[{{x}_{A}}\]represents the mole fraction of component A, the total pressure of the solution will be
A)
\[{{p}_{B}}+{{x}_{A}}({{p}_{B}}-{{p}_{A}})\]
done
clear
B)
\[{{p}_{A}}+{{x}_{A}}({{p}_{A}}-{{p}_{B}})\]
done
clear
C)
\[{{p}_{A}}+{{x}_{A}}({{p}_{B}}-{{p}_{A}})\]
done
clear
D)
\[{{p}_{B}}+{{x}_{A}}({{p}_{A}}-{{p}_{B}})\]
done
clear
View Answer play_arrow
Standard reduction potentials of the half reactions are given below:
\[{{F}_{2}}(g)+2{{e}^{-}}\xrightarrow{{}}2{{F}^{-}}(aq);{{E}^{o}}=+2.85\,V\] \[C{{l}_{2}}(g)+2{{e}^{-}}\xrightarrow{{}}2C{{l}^{-}}(aq);{{E}^{o}}=+1.36\,V\] \[B{{r}_{2}}(i)+2{{e}^{-}}\xrightarrow{{}}2B{{r}^{-}}(aq);{{E}^{o}}=+1.06\,V\] \[{{I}_{2}}(S)+2{{e}^{-}}\xrightarrow{{}}2{{I}^{-}}(aq);{{E}^{o}}=+0.53\,V\]
The strongest oxidizing and reducing agents respectively are
A)
\[{{F}_{2}}\]and\[{{I}^{-}}\]
done
clear
B)
\[B{{r}_{2}}\]and \[C{{l}^{-}}\]
done
clear
C)
\[C{{l}_{2}}\]and\[B{{r}^{-}}\]
done
clear
D)
\[C{{l}_{2}}\]and\[{{I}_{2}}\]
done
clear
View Answer play_arrow
The rate of reaction between two reactants A and B decreases by a factor of 4 if the concentration of reactant B is doubled. The order of this reaction with respect to reactant B is
A)
-1
done
clear
B)
-2
done
clear
C)
1
done
clear
D)
2
done
clear
View Answer play_arrow
Which property of colloids is not dependent on the, 2015 charge on colloidal particles?
A)
Tyndal effect
done
clear
B)
Coagulation
done
clear
C)
Electrophoresis
done
clear
D)
Electro-osmosis
done
clear
View Answer play_arrow
Which of the following statements about the advantage of roasting of sulphide ore before reduction is not true?
A)
\[\Delta G_{f}^{o}\]of the sulphide is greater than \[C{{S}_{2}}\]and\[{{H}_{2}}S\]
done
clear
B)
\[\Delta G_{f}^{o}\]is negative for roasting of sulphide ore to oxide
done
clear
C)
Roasting of the sulphide to oxide is ther-modynamically feasible
done
clear
D)
Carbon and hydrogen are suitable reducing agents for metal sulphides
done
clear
View Answer play_arrow
Nitrogen dioxide and sulphur dioxide have some properties in common. Which property is shown by one of these compounds, but not by the other?
A)
Is soluble in water
done
clear
B)
Is used as a food preservative
done
clear
C)
Forms 'acid-rain'.
done
clear
D)
Is a reducing agent
done
clear
View Answer play_arrow
S-S bond is not present in
A)
\[{{S}_{2}}O_{7}^{2-}\]
done
clear
B)
\[{{S}_{4}}O_{6}^{2-}\]
done
clear
C)
\[{{S}_{2}}O_{4}^{2-}\]
done
clear
D)
\[{{S}_{2}}O_{3}^{2-}\]
done
clear
View Answer play_arrow
Which of the following has greatest reducing power?
A)
HI
done
clear
B)
HBr
done
clear
C)
HCl
done
clear
D)
HF
done
clear
View Answer play_arrow
Which of the following gases is/are called rare gas?
A)
Ne
done
clear
B)
He
done
clear
C)
Kr
done
clear
D)
All of these
done
clear
View Answer play_arrow
Reason of lanthanide contraction is
A)
negligible screening effect of f-orbitals
done
clear
B)
increasing nuclear charge
done
clear
C)
decreasing nuclear charge
done
clear
D)
decreasing screening effect
done
clear
View Answer play_arrow
\[[Cr{{({{H}_{2}}O)}_{6}}]C{{l}_{3}}\] (at. no. of Cr = 24) has a magnetic moment of 3.83 BM. The correct distribution of 3d-electrons in the chromium of the complex:
A)
\[3d_{xy}^{1},3d_{yz}^{1},3d_{xz}^{1}\]
done
clear
B)
\[3d_{xy}^{1},3d_{yz}^{1},3d_{{{z}^{2}}}^{1}\]
done
clear
C)
\[(3d_{{{x}^{2}}-{{y}^{2}}}^{1}),3d_{{{z}^{2}}}^{1},3d_{xz}^{1}\]
done
clear
D)
\[3d_{xy}^{1},(3d_{{{x}^{2}}-{{y}^{2}}}^{1}),3d_{yz}^{1}\]
done
clear
View Answer play_arrow
In an \[S{{N}_{1}}\]reaction on chiral centers, there is
A)
100% retention
done
clear
B)
100% inversion
done
clear
C)
100% racemization
done
clear
D)
inversion more than retention leading to partial racemization
done
clear
View Answer play_arrow
Ethylene oxide when, treated with Grignard reagent yields:
A)
cyclopropyl alcohol
done
clear
B)
primary alcohol
done
clear
C)
secondary alcohol
done
clear
D)
tertiary alcohol
done
clear
View Answer play_arrow
Predict the products in the given reaction.
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Nitrobenzene on reaction with conc. \[HN{{O}_{3}}/{{H}_{2}}S{{O}_{4}}\]at \[80-100{{\,}^{o}}C\]forms which one of the following products?
A)
1, 3-Dinitrobenzene
done
clear
B)
1, 4-Dinitrobenzene
done
clear
C)
1, 2, 4-Trinitrobenzene
done
clear
D)
1, 2-Dinitrobenzene
done
clear
View Answer play_arrow
In DNA, the complimentary bases are:
A)
adenine and thymine; guanine and uracil
done
clear
B)
adenine and guanine; thymine and cytosine
done
clear
C)
uracil and adenine; cytosine and guanine
done
clear
D)
adenine and thymine; guanine and cytosine
done
clear
View Answer play_arrow
Organisms that obtain energy by the oxidation of reduced inorganic compounds are called:
A)
Photo autotrophs
done
clear
B)
Chemo autotrophs
done
clear
C)
Saprozoic
done
clear
D)
Coproheterotrophs
done
clear
View Answer play_arrow
Which statement is correct for bacterial transduction?
A)
Transfer of some genes from one bacteria to another bacteria through virus
done
clear
B)
Transfer of some genes from one bacteria to another bacteria by conjugation
done
clear
C)
Bacteria obtained its DNA directly
done
clear
D)
Bacteria obtained DNA from other external source
done
clear
View Answer play_arrow
Which of the following propagates through leaf tip?
A)
Sprout-leaf plant
done
clear
B)
Marchantia
done
clear
C)
Moss
done
clear
D)
Walking fern
done
clear
View Answer play_arrow
In Arthropoda, head and thorax are often fused to form cephalothorax, but in which one of the following classes is the body divided into head, thorax and abdomen?
A)
Myriapoda
done
clear
B)
Crustacea
done
clear
C)
Arachnida and Crustacea
done
clear
D)
Insecta
done
clear
View Answer play_arrow
An ovule which becomes curved so that the nucellus and embryo sac lie at right angles to the funicle is:
A)
Campylotropous
done
clear
B)
Anatropous
done
clear
C)
Orthotropous
done
clear
D)
Hemitropous
done
clear
View Answer play_arrow
Passage cells are thin-walled cells found in:
A)
Central region of style through which the pollen tube grows towards the ovary
done
clear
B)
Endodermis of roots facilitating rapid transport of water from cortex to pericycle
done
clear
C)
Phloem elements that serve as entry points for substances for transport to other plant parts
done
clear
D)
Testa of seeds to enable emergence of growing embryonic axis during seed germination.
done
clear
View Answer play_arrow
In which one of the following preparations are you likely to come across cell junctions most frequently?
A)
Hyaline cartilage
done
clear
B)
Ciliated epithelium
done
clear
C)
Thrombocytes
done
clear
D)
Tendon
done
clear
View Answer play_arrow
A major breakthrough in the studies of cells came with the development of electron micro- scope. This is because:
A)
The resolution power of the electron microscope is much higher than that of the light microscope
done
clear
B)
The resolving power of the electron microscope is 200-350 nm as compared to 0.1-0.2 nm for the light microscope
done
clear
C)
Electron beam can pass through thick materials, whereas light microscopy requires thin sections
done
clear
D)
The electron microscope is more powerful than the light microscope as it uses a beam of electrons which has wavelength much longer than that of photons
done
clear
View Answer play_arrow
Carrier ions like\[N{{a}^{+}}\]facilitate the absorption of substances like
A)
amino acids and glucose
done
clear
B)
glucose and fatty acids
done
clear
C)
fatty acids and glycerol
done
clear
D)
fructose and some amino acids
done
clear
View Answer play_arrow
In the somatic cell cycle:
A)
DNA replication takes place in S-phase
done
clear
B)
A short interphase is followed by a long mitotic phase
done
clear
C)
\[{{G}_{2}}\]phase follows mitotic phase
done
clear
D)
In\[{{G}_{1}}\]phase DNA content is double the amount of DNA present in the original cell
done
clear
View Answer play_arrow
The translocation of organic solutes in sieve tube members is supported by:
A)
Root pressure and transpiration pull
done
clear
B)
P-proteins
done
clear
C)
Mass flow involving a carrier and ATP
done
clear
D)
Cytoplasmic streaming
done
clear
View Answer play_arrow
Which of the following criteria does not pertain to facilitated transport?
A)
Requirement of special membrane proteins
done
clear
B)
High selectivity
done
clear
C)
Transport saturation
done
clear
D)
Uphill transport
done
clear
View Answer play_arrow
A free living nitrogen-fixing cyanobacterium which can also form symbiotic association with the water fern Azolla is:
A)
Chlorella
done
clear
B)
Nostoc
done
clear
C)
Anabaena
done
clear
D)
Tolypothrix
done
clear
View Answer play_arrow
Which one of the following elements is plants is not remobillised?
A)
Sulphur
done
clear
B)
Phosphorus
done
clear
C)
Calcium
done
clear
D)
Potassium
done
clear
View Answer play_arrow
As compared to a C3-plant, how many additional molecules of ATP are needed for net production of one molecule of hexose sugar by C4- plants:
A)
Zero
done
clear
B)
Six
done
clear
C)
Two
done
clear
D)
Twelve
done
clear
View Answer play_arrow
Importance of day length in flowering of plants was first shown in:
A)
Cotton
done
clear
B)
Petunia
done
clear
C)
Lemna
done
clear
D)
Tobacco
done
clear
View Answer play_arrow
During which stage in the complete oxidation of glucose are the greatest number of ATP molecules formed from ADP.
A)
Glycolysis
done
clear
B)
Krebs cycle
done
clear
C)
Electron transport chain
done
clear
D)
Conversion of pyruvic acid to acetyl CoA
done
clear
View Answer play_arrow
The energy-releasing metabolic process in which substrate is oxidised without an external electron acceptor is called:
A)
Glycolysis
done
clear
B)
Fermentation
done
clear
C)
Aerobic respiration
done
clear
D)
Photorespiration
done
clear
View Answer play_arrow
Hormone responsible for senescence:
A)
ABA
done
clear
B)
Auxin
done
clear
C)
GA
done
clear
D)
Cytokinin
done
clear
View Answer play_arrow
Treatment of seed at low temperature under moist conditions to break its dormancy is called:
A)
Sclaritication
done
clear
B)
Vernalization
done
clear
C)
Chelation
done
clear
D)
Stratification
done
clear
View Answer play_arrow
Epithelial cells of the intestine involved in food absorption have on their surface:
A)
Zymogen granules
done
clear
B)
Pinocytic vesicles
done
clear
C)
Phagocytic vesicles
done
clear
D)
Microvilli
done
clear
View Answer play_arrow
What will happen if the secretion of parietal cells of gastric glands is blocked with an inhibitor?
A)
In the absence of HCI secretion, inactive pepsinogen is not converted into the active enzyme pepsin
done
clear
B)
Enterokinase will not be released from the duodenal mucosa and so trypsinogen is not converted to trypsin
done
clear
C)
Gastric juice will be deficient in chymosin
done
clear
D)
Gastric juice will be deficient in pepsinogen
done
clear
View Answer play_arrow
The majority of carbon dioxide produced by our body cells is transported to the lungs:
A)
Dissolved in the blood
done
clear
B)
As bicarbonates
done
clear
C)
As carbonates
done
clear
D)
Attached to hemoglobin
done
clear
View Answer play_arrow
Approximately seventy percent of carbon dioxide absorbed by the blood will be transported to the lungs:
A)
as bicarbonate ions
done
clear
B)
in the form of dissolved gas molecules
done
clear
C)
by binding to R.B.C.
done
clear
D)
as carbaminohaemoglobin
done
clear
View Answer play_arrow
The most active phagocytic white blood cells are:
A)
eosinophils and lymphocytes
done
clear
B)
neutrophils and monocytes
done
clear
C)
neutrophils and eosinophils
done
clear
D)
lymphocytes and macrophages
done
clear
View Answer play_arrow
Which one of the following plasma proteins is involved in the coagulation of blood?
A)
Fibrinogen
done
clear
B)
an albumin
done
clear
C)
serum amylase
done
clear
D)
a globulin
done
clear
View Answer play_arrow
A person is undergoing prolonged fasting. His urine will be found to contain abnormal quantities of:
A)
Fats
done
clear
B)
Amino acids
done
clear
C)
Ketones
done
clear
D)
Glucose
done
clear
View Answer play_arrow
Uricotelic mode of passing out nitrogenous wastes is found in:
A)
Insects and Amphibians
done
clear
B)
Reptiles and Birds
done
clear
C)
Birds and Annelids
done
clear
D)
Amphibians and Reptiles
done
clear
View Answer play_arrow
In human body, which one of the following is anatomically correct?
A)
Cranial nerve - 10 pairs
done
clear
B)
Floating ribs - 2 pairs
done
clear
C)
Collar bones - 3 pairs
done
clear
D)
Salivary glands - 1 pair
done
clear
View Answer play_arrow
The H-zone in the skeletal muscle fibre is due to:
A)
the absence of myofibrils in the central portion of A-band
done
clear
B)
the central gap between myosin filaments in the A-band
done
clear
C)
the central gap between actin filaments extending through myosin filaments in the A band
done
clear
D)
extension of myosin filaments in the central portion of the A-band.
done
clear
View Answer play_arrow
Which one of the following is an example of negative feedback loop in humans?
A)
Secretion of sweat glands and constriction of skin blood vessels when it is too hot
done
clear
B)
Constriction of skin blood vessels and contraction of skeletal muscles when it is too cold
done
clear
C)
Secretion of tears after falling of sand particles into the eye
done
clear
D)
Salivation of mouth at the sight of delicious food
done
clear
View Answer play_arrow
Which one of the following pairs correctly matches a hormone with a disease resulting from its deficiency?
A)
Relaxin - Gigantism
done
clear
B)
Prolactin - Cretinism
done
clear
C)
Parathyroid hormone - Tetany
done
clear
D)
Insulin - Diabetes insipidus
done
clear
View Answer play_arrow
Meiosis takes place in:
A)
Meiocyte
done
clear
B)
Conidia
done
clear
C)
Gemmule
done
clear
D)
Megaspore
done
clear
View Answer play_arrow
Why is vivipary an undesirable character for annual crop plants?
A)
It reduces the vigour of the plant
done
clear
B)
The seeds exhibit long dormancy
done
clear
C)
It adversely affects the fertility of the plant
done
clear
D)
The seeds cannot be stored under normal conditions for the next season
done
clear
View Answer play_arrow
In humans, at the end of the first meiotic division, the male germ cells differentiate into the:
A)
spermatids
done
clear
B)
spermatogonia
done
clear
C)
primary spermatocytes
done
clear
D)
secondary spermatocytes
done
clear
View Answer play_arrow
Tubectomy is the method of sterilization in which:
A)
small part of the fallopian tube is removed or tied up
done
clear
B)
ovaries are removed surgically
done
clear
C)
small part of vas deferens is removed or tied up
done
clear
D)
uterus is removed surgically
done
clear
View Answer play_arrow
The genes controlling the seven pea characters studied by Mendel are now known to be located on how many different chromosomes:
A)
Seven
done
clear
B)
Six
done
clear
C)
Five
done
clear
D)
Four
done
clear
View Answer play_arrow
Genetic map is one that:
A)
Establishes sites of the genes on a chromo- some
done
clear
B)
Establishes the various stages in gene evolution
done
clear
C)
Shows the stages during the cell division
done
clear
D)
Shows the distribution of various species in a region
done
clear
View Answer play_arrow
Convergent evolution is illustrated by:
A)
Rat and dog
done
clear
B)
Bacterium and protozoan
done
clear
C)
Starfish and cuttlefish
done
clear
D)
Dogfish and whale
done
clear
View Answer play_arrow
AIDS is caused by HIV that principally infects
A)
Activator B cells
done
clear
B)
T4 lymphocytes
done
clear
C)
Cytotoxic T cells
done
clear
D)
All lymphocytes
done
clear
View Answer play_arrow
Crop plants grown in monoculture are
A)
Low in yield
done
clear
B)
Free from intraspecific competition
done
clear
C)
Characterised by poor root system
done
clear
D)
Highly prone to pests
done
clear
View Answer play_arrow
Monascus purpureus is a yeast used commercially in the production of:
A)
Citric acid
done
clear
B)
Blood cholesterol lowering statins
done
clear
C)
Ethanol
done
clear
D)
Streptokinase for removing clots from the blood vessels
done
clear
View Answer play_arrow
The colonies of recombinant bacteria appear white in contrast to blue colonies of non- recombinant bacteria because of:
A)
Non-recombinant bacteria containing beta-galactosidase
done
clear
B)
Insertional inactivation ofalpha-galacto- sidase in non-recombinant bacteria
done
clear
C)
Insertional inactivation ofalpha-galacto- sidase in recombinant bacteria
done
clear
D)
Inactivation of glycosidase enzyme in recombinant bacteria
done
clear
View Answer play_arrow
The first human hormone produced by recombinant DNA technology is:
A)
Insulin
done
clear
B)
Estrogen
done
clear
C)
Thyroxin
done
clear
D)
Progesterone
done
clear
View Answer play_arrow
What is a keystone species?
A)
A common species that has plenty of biomass, yet has a fairly low impact on the community's organization
done
clear
B)
A rare species that has minimal impact on the biomass and on other species in the community
done
clear
C)
A dominant species that constitutes a large proportion of the biomass and which affects many other species
done
clear
D)
A species which makes up only a small proportion of the total biomass of a community, yet has a huge impact on the community's organization and survival
done
clear
View Answer play_arrow
Dough kept overnight in warm weather becomes soft and spongy because of:
A)
Fermentation
done
clear
B)
Cohesion
done
clear
C)
Osmosis
done
clear
D)
Absorption of carbon dioxide from atmosphere
done
clear
View Answer play_arrow
Gray spots of oat are caused by deficiency of:
A)
Cu
done
clear
B)
Zn
done
clear
C)
Mn
done
clear
D)
Fe
done
clear
View Answer play_arrow
PGA as the first \[C{{O}_{2}}\]fixation product was dis-covered in photosynthesis of:
A)
Bryophyte
done
clear
B)
Gymnosperm
done
clear
C)
Angiosperm
done
clear
D)
Algae
done
clear
View Answer play_arrow
Chromatophores take part in:
A)
Respiration
done
clear
B)
Photosynthesis
done
clear
C)
Growth
done
clear
D)
Movement
done
clear
View Answer play_arrow
Phototropic curvature is the result of uneven distribution of:
A)
Gibberellin
done
clear
B)
Phytochrome
done
clear
C)
Cytokinins
done
clear
D)
Auxin
done
clear
View Answer play_arrow
The purplish red pigment rhodopsin contained in the rods type of photo receptor cells of the human eye is a derivative of:
A)
Vitamin A
done
clear
B)
Vitamin B 1
done
clear
C)
Vitamin C
done
clear
D)
Vitamin D
done
clear
View Answer play_arrow
Which one of the following is the correct statement for respiration in humans?
A)
Workers in grinding and stone-breaking industries may suffer, from lung fibrosis
done
clear
B)
About 90% of carbon dioxide \[(C{{O}_{2}})\] is carried by haemoglobin as carbamino- haemoglobin
done
clear
C)
Cigarette smoking may lead to inflammation of bronchi
done
clear
D)
Neural signals from pneumotoxic centre in pons region of brain can increase the duration of inspiration
done
clear
View Answer play_arrow
You are required to draw blood from a patient and to keep it in a test tube for analysis of blood corpuscles and plasma. You are also provided with the following four types of test tubes. Which of them will you not use for the purpose?
A)
Chilled test tube
done
clear
B)
Test tube containing heparin
done
clear
C)
Test tube containing sodium oxalate
done
clear
D)
Test tube containing calcium bicarbonate
done
clear
View Answer play_arrow
Which one of the following characteristics is common both in humans and adult frogs?
A)
Internal fertilization
done
clear
B)
Nucleated RBCs
done
clear
C)
Ureotelic mode of excretion
done
clear
D)
Four-chambered heart
done
clear
View Answer play_arrow
Select the correct statement regarding the specific disorder of muscular or skeletal system
A)
Myasthenia gravis - autoimmune disorder which inhibits sliding of myosin filaments
done
clear
B)
Gout - inflammation of joints due to extra deposition of calcium
done
clear
C)
Muscular dystrophy - age related shorting of muscles
done
clear
D)
Osteoporosis - decrease in bone mass and higher chances of fractures with advancing age
done
clear
View Answer play_arrow
In which animal nerve cell is present but brain is absent?
A)
Sponge
done
clear
B)
Earthworm
done
clear
C)
Cockroach
done
clear
D)
Hydra
done
clear
View Answer play_arrow
Which part of human brain is concerned with the regulation of body temperature?
A)
Hypothalamus
done
clear
B)
Medulla oblongata
done
clear
C)
Cerebellum
done
clear
D)
Cerebrum
done
clear
View Answer play_arrow
Parkinson's disease (characterized by tremors and progressive rigidity of limbs) is caused by degeneration of brain neurons that are involved in movement control and make use of neurotransmitter:
A)
Norepinephrine
done
clear
B)
Acetylcholine
done
clear
C)
GABA
done
clear
D)
Dopamine
done
clear
View Answer play_arrow
A person is having problems with calcium and phosphorus metabolism in his body. Which one of the following glands may not be functioning properly?
A)
Thyroid
done
clear
B)
Parathyroid
done
clear
C)
Parotid
done
clear
D)
Pancreas
done
clear
View Answer play_arrow
In angiosperm all the four microspores of tetrad are covered by a layer which is formed by:
A)
Pectocellulose
done
clear
B)
Callose
done
clear
C)
Cellulose
done
clear
D)
Sporopollenin
done
clear
View Answer play_arrow
Male gametes in angiosperms are formed by the division of:
A)
Microspore mother cell
done
clear
B)
Microspore
done
clear
C)
Generative cell
done
clear
D)
Vegetative cell
done
clear
View Answer play_arrow
Which of the following is the correct matching of the events occurring during menstrual cycle?
A)
Menstruation \[\times \] Breakdown of myometrium and ovum not fertilized
done
clear
B)
Ovulation \[\times \] LH and FSH attain peak level and sharp fall in the secretion of progesterone
done
clear
C)
Proliferative phase \[\times \] Rapid regeneration of myometrium and maturation of Graa- fian follicle
done
clear
D)
Development of corpus \[\times \] Secretory phase and increased secretion of progesterone. Luteum
done
clear
View Answer play_arrow
The testes in humans are situated outside the abdominal cavity inside a pouch called scrotum. The purpose served is for:
A)
providing a secondary sexual feature for exhibiting the male sex
done
clear
B)
maintaining the scrotal temperature lower than the internal body temperature
done
clear
C)
escaping any possible compression by the visceral organs
done
clear
D)
providing more space for the growth of Epididymis
done
clear
View Answer play_arrow
Medical Termination of Pregnancy (MTP) is considered safe up to how many weeks of pregnancy?
A)
Six weeks
done
clear
B)
Eight weeks
done
clear
C)
Twelve weeks
done
clear
D)
Eighteen weeks
done
clear
View Answer play_arrow
One of the legal methods of birth control is:
A)
abortion by taking an appropriate medicine
done
clear
B)
by abstaining from coitus from day 10 to 17 of the menstrual cycle
done
clear
C)
by having coitus at the time of day break
done
clear
D)
by a premature ejaculation during coitus
done
clear
View Answer play_arrow
Forecoming generations are less adaptive than their parental generation due to:
A)
Natural selection
done
clear
B)
Mutation
done
clear
C)
Genetic drift
done
clear
D)
Adaptation
done
clear
View Answer play_arrow
Which of the following is the example of sex- linked disease?
A)
AIDS
done
clear
B)
Colour-blindness
done
clear
C)
Syphilis
done
clear
D)
Gonorrhoea
done
clear
View Answer play_arrow
m-RNA is synthesised on DNA template in which direction:
A)
\[5'\to 3'\]
done
clear
B)
\[3'\to 5'\]
done
clear
C)
Both
done
clear
D)
Any
done
clear
View Answer play_arrow
Transformation experiment was first performed on which bacteria:
A)
E. coli
done
clear
B)
Diplococcus pneumoniae
done
clear
C)
Salmonella
done
clear
D)
Pasteurella pestis
done
clear
View Answer play_arrow
Which of the following is important for speciation?
A)
Seasonal isolation
done
clear
B)
Reproductive isolation
done
clear
C)
Behavioural isolation
done
clear
D)
Tropical isolation
done
clear
View Answer play_arrow
Age of fossils in the past was generally determined by radiocarbon method and other methods involving radioactive elements found in the rocks. More precise methods, which were used recently and led to the revision of the evolutionary periods for different groups of organisms includes
A)
Study of the conditions of fossilization
done
clear
B)
Electron spin resonance (ESR) and fossil DNA
done
clear
C)
Study of carbohydrates/proteins in rocks
done
clear
D)
Study of carbohydrates/proteins in fossils
done
clear
View Answer play_arrow
Cancerous cells can easily be destroyed by radiations due to:
A)
rapid cell division
done
clear
B)
lack of nutrition
done
clear
C)
fast mutation
done
clear
D)
lack of oxygen
done
clear
View Answer play_arrow
Match the disease in Column I with the appropriate items (pathogen / prevention / treatment) in Column II.
Column I Column II [a] Amoebiasis (i) Treponemapallidum [b] Diphtheria (ii) Use only sterilized food and water [c] Cholera (iii) DPT Vaccine [d] Syphilis (iv) Use oral rehydration therapy
A)
A = (ii), B = (i), C = (iii), D = (iv)
done
clear
B)
A = (ii), B = (iii), C = (iv), D = (i)
done
clear
C)
A = (i), B = (ii), C = (iii), D = (iv)
done
clear
D)
A = (ii), B = (iv), C = (i), D = (iii)
done
clear
View Answer play_arrow
Which of the following crops have been brought to India from New World?
A)
Cashew nut, potato, rubber
done
clear
B)
Mango, tea
done
clear
C)
Tea, rubber, mango
done
clear
D)
Coffee
done
clear
View Answer play_arrow
Parthenocarpic tomato fruits can be produced by
A)
Removing androecium of flowers before pollen grains are released
done
clear
B)
Treating the plants with low concentrations of gibberellic acid and auxins
done
clear
C)
Raising the plants from vernalized seeds
done
clear
D)
Treating the plants with phenylmercuric acetate
done
clear
View Answer play_arrow
Which of the following is true pair of biofertil- izers?
A)
Azolla and BGA
done
clear
B)
Nostoc and legume
done
clear
C)
Rhizobium and grasses
done
clear
D)
Salmonella and E. coli
done
clear
View Answer play_arrow
A common biocontrol agent for the control of plant diseases is
A)
Baculovirus
done
clear
B)
Bacillusthuringiensis
done
clear
C)
Glomus
done
clear
D)
Trichoderma
done
clear
View Answer play_arrow
Introduction of food plants developed by genetic engineering is not desirable because:
A)
Economy of developing countries may suffer
done
clear
B)
These products are less tasty as compared to the already existing products
done
clear
C)
This method is costly
done
clear
D)
There is danger of coming viruses, allergens and toxins with introduced crop
done
clear
View Answer play_arrow
Which one of the following is used as vector for cloning genes into higher organisms?
A)
Baculovirus
done
clear
B)
Salmonella typhimurium
done
clear
C)
Rhizopus nigricans
done
clear
D)
Retrovirus
done
clear
View Answer play_arrow
Which of the following is expected to have the highest value (gm/m /yr) in a grassland ecosystem?
A)
Tertiary production
done
clear
B)
Gross production (GP)
done
clear
C)
Net production (NP)
done
clear
D)
Secondary production
done
clear
View Answer play_arrow
A lake near a village suffered heavy mortality of fishes within a few days. Consider the following reasons for this?
(i) Lots of urea and phosphate fertilizer were used in the crops in the vicinity (ii) The area was sprayed with DDT by an aircraft (iii) The lake water turned green and stinky (iv) Phytoplankton populations in the lake declined initially, thereby greatly reducing photosynthesis
Which two of the above were the main causes offish mortality in the lake?
A)
(i),(iii)
done
clear
B)
(i), (ii)
done
clear
C)
(ii),(iii)
done
clear
D)
(iii),(iv)
done
clear
View Answer play_arrow
Identify the possible link "A" in the following food chain: Plant®in sec t®frog®"A"®Eagle
A)
Cobra
done
clear
B)
Parrot
done
clear
C)
Rabbit
done
clear
D)
Wolf
done
clear
View Answer play_arrow
At which latitude, heat gain through insolation approximately equals heat loss through terrestrial radiation
A)
42 \[1/{{2}^{o}}\]North and South
done
clear
B)
22 \[1/{{2}^{o}}\]North and South
done
clear
C)
\[{{40}^{o}}\]North and South
done
clear
D)
\[{{66}^{o}}\]North and South
done
clear
View Answer play_arrow
Quercus species are the dominant component in:
A)
Scrub forests
done
clear
B)
Tropical rain forests
done
clear
C)
Temperate deciduous forests
done
clear
D)
Alpine forests
done
clear
View Answer play_arrow
Which of the following represent maximum number of species among global biodiversity?
A)
Algae
done
clear
B)
Lichens
done
clear
C)
Fungi
done
clear
D)
Mosses and ferns
done
clear
View Answer play_arrow
In 1984, the Bhopal gas tragedy took place because methyl isocyanate:
A)
Reacted with ammonia
done
clear
B)
Reacted with \[C{{O}_{2}}\]
done
clear
C)
Reacted with water
done
clear
D)
Reacted with DDT
done
clear
View Answer play_arrow
Montreal Protocol which calls for appropriate action to protect the ozone layer from human activities was passed in the year
A)
1986
done
clear
B)
1987
done
clear
C)
1988
done
clear
D)
1985
done
clear
View Answer play_arrow
World Summit on Sustainable Development (2002) was held in:
A)
Argentina
done
clear
B)
South Africa
done
clear
C)
Brazil
done
clear
D)
Sweden
done
clear
View Answer play_arrow
Which one of the following is commonly used in the transfer of foreign DNA into crop plants?
A)
Penicillium expansum
done
clear
B)
Trichoderma harzianum
done
clear
C)
Meloidogyne incognita
done
clear
D)
Agrobacterium tumefaciens
done
clear
View Answer play_arrow
The population of an insect species shows an explosive increase in numbers during rainy season followed by its disappearance at the end of the season. What does this show?
A)
The population of its predators increases enormously
done
clear
B)
S-shaped or sigmoid growth of this insect
done
clear
C)
The food plants mature and die at the end of the rainy season
done
clear
D)
Its population growth curve is of J-type
done
clear
View Answer play_arrow

 The work done in charging fully both the condensers is
The work done in charging fully both the condensers is