-
question_answer1)
In the given conformation \[{{C}_{2}}\] is rotated about \[{{C}_{2}}-{{C}_{3}}\] bond anticlockwise by an angle of \[120{}^\circ \] then the conformation obtained is [AIIMS 2004]
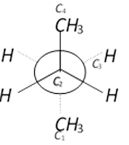
A)
Fully eclipsed conformation done
clear
B)
Partially eclipsed conformation done
clear
C)
Gauche conformation done
clear
D)
Staggered conformation done
clear
View Solution play_arrow
-
question_answer2)
The molecular formula of diphenyl methane,
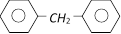
, is \[{{C}_{13}}{{H}_{12}}\] How many structural isomers are possible when one of the hydrogens is replaced by a chlorine atom [CBSE PMT 2004]
A)
8 done
clear
B)
7 done
clear
C)
6 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer3)
\[S{{N}^{1}}\] reaction is faster in [Orissa JEE 2004]
A)
\[C{{H}_{3}}C{{H}_{2}}Cl\] done
clear
B)
\[\begin{matrix} C{{H}_{3}} \\ C{{H}_{3}} \\ \end{matrix}>CH-Cl\] done
clear
C)
\[C{{H}_{3}}-\underset{C{{H}_{3}}\ }{\overset{C{{H}_{3}}\ }{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,-Cl}}}\,\] done
clear
D)
\[C{{H}_{3}}-\underset{C{{H}_{3}}}{\mathop{\underset{\underset{|}{\mathop{C}}\,{{H}_{2}}}{\mathop{\underset{|}{\mathop{C}}\,H\ }}\,}}\,-Cl\] done
clear
View Solution play_arrow
-
question_answer4)
How many enantiomer pairs are obtained by monochlorination of 2, 3-dimethylbutane [Kerala PMT 2004]
A)
Nil done
clear
B)
Four done
clear
C)
Two done
clear
D)
Three done
clear
E)
One done
clear
View Solution play_arrow
-
question_answer5)
Among the following compounds which can be dehydrated very easily is [AIEEE 2004]
A)
\[C{{H}_{3}}-C{{H}_{2}}-\underset{OH\ \ \ \ \ \ }{\overset{C{{H}_{3}}\ \ \ \ }{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,-C{{H}_{2}}}}}\,-C{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-\overset{OH}{\mathop{\overset{|}{\mathop{C}}\,H}}\,-C{{H}_{3}}\] done
clear
C)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-OH\] done
clear
D)
\[C{{H}_{3}}-C{{H}_{2}}-\underset{C{{H}_{3}}\ }{\mathop{\underset{|}{\mathop{C}}\,H-}}\,C{{H}_{2}}-C{{H}_{2}}-OH\] done
clear
View Solution play_arrow
-
question_answer6)
Which of the following statements is not characteristic of free radical chain reaction [JIPMER 1997]
A)
It gives major product derived from most stable free radical done
clear
B)
It is usually sensitive to change in solvent polarity done
clear
C)
It proceeds in three main steps like initiation, propagation and termination done
clear
D)
It may be initiated by U.V. light done
clear
View Solution play_arrow
-
question_answer7)
Most stable carbanion is [BHU 2003]
A)
\[CH_{3}^{-}\] done
clear
B)
\[C{{H}_{3}}CH_{2}^{-}\] done
clear
C)
D)
View Solution play_arrow
-
question_answer8)
Among the following the dissociation constant is highest for [AIIMS 2004]
A)
\[{{C}_{6}}{{H}_{5}}OH\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}OH\] done
clear
C)
\[C{{H}_{3}}C\equiv CH\] done
clear
D)
\[C{{H}_{3}}NH_{3}^{+}C{{l}^{-}}\] done
clear
View Solution play_arrow
-
question_answer9)
Which one of the following compounds is most acidic [CBSE PMT 2005]
A)
\[Cl-C{{H}_{2}}-C{{H}_{2}}-OH\] done
clear
B)
C)
D)
View Solution play_arrow
-
question_answer10)
Which one is electrophilic addition [AMU (Engg.) 1999]
A)
\[C{{H}_{3}}-C{{H}_{3}}+C{{l}_{2}}\to {{C}_{2}}{{H}_{5}}Cl+HCl\] done
clear
B)
\[C{{H}_{3}}CH=O+HCN\to {{(C{{H}_{3}})}_{2}}C(OH)CN\] done
clear
C)
\[B{{r}_{2}}\to B{{r}^{\bullet }}+B{{r}^{\bullet }}\] done
clear
D)
\[C{{H}_{2}}=C{{H}_{2}}+B{{r}_{2}}\to C{{H}_{2}}BrC{{H}_{2}}Br\] done
clear
View Solution play_arrow
-
question_answer11)
A compound has 3 chiral carbon atoms. The number of possible optical isomers it can have [DCE 2004]
A)
3 done
clear
B)
2 done
clear
C)
8 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer12)
How many chiral isomers can be drawn from 2-bromo, 3-chloro butane [DCE 2003]
A)
2 done
clear
B)
3 done
clear
C)
4 done
clear
D)
5 done
clear
View Solution play_arrow
-
question_answer13)
Number of isomers of \[{{C}_{4}}{{H}_{10}}\] is [CBSE PMT 1996; AFMC 1997; RPMT 2002; MP PMT 1997]
A)
2 done
clear
B)
3 done
clear
C)
4 done
clear
D)
Isomerism not exist done
clear
View Solution play_arrow
-
question_answer14)
The number of possible isomers for compound \[{{C}_{2}}{{H}_{3}}C{{l}_{2}}Br\] is [MP PMT 1999]
A)
2 done
clear
B)
3 done
clear
C)
4 done
clear
D)
5 done
clear
View Solution play_arrow
-
question_answer15)
The optically active tartaric acid is named as D-(+)- tartaric acid because it has a positive [IIT-JEE 1999]
A)
Optical rotation and is derived from D-glucose done
clear
B)
pH in organic solvent done
clear
C)
Optical rotation and is derived from D(+) glyceraldehyde done
clear
D)
Optical rotation only when substituted by deuterium done
clear
View Solution play_arrow
-
question_answer16)
Among the following compounds (I-III) the correct order of reaction with electrophilic reagent is
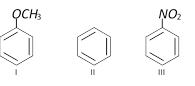
[CBSE PMT 1997]
A)
II > III > I done
clear
B)
III < I < II done
clear
C)
I > II > III done
clear
D)
I = II > III done
clear
View Solution play_arrow
-
question_answer17)
Carbocation which is most stable [BHU 2003]
A)
\[C{{H}_{3}}C{{H}_{2}}^{+}\] done
clear
B)
\[C{{H}_{3}}^{+}\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}^{+}\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}^{+}\] done
clear
View Solution play_arrow
-
question_answer18)
Tautomerism is exhibited by [CBSE PMT 1997; KCET 2002]
A)
\[{{(C{{H}_{3}})}_{3}}CNO\] done
clear
B)
\[{{(C{{H}_{3}})}_{2}}NH\] done
clear
C)
\[{{R}_{3}}CN{{O}_{2}}\] done
clear
D)
\[RC{{H}_{2}}N{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer19)
Which of the following will have geometrical isomers [MP PET 1996; MP PMT 1997; AFMC 1997]
A)
2-methylpropene done
clear
B)
2-butene done
clear
C)
1-butene done
clear
D)
Propene done
clear
View Solution play_arrow
-
question_answer20)
Examine the following three pairs of possible isomers
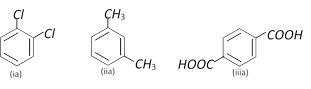
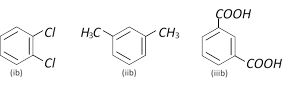
Now state whether the pairs represent identical compounds or different isomers
A)
All three pairs represent different compounds done
clear
B)
(ia) and (ib) are identical; (iia) and (iib) are identical; and (iiia) and (iiib) are identical done
clear
C)
(ia) and (ib) are isomers; (iia) and (iib) are identical; and (iiia) and (iiib) are isomers done
clear
D)
(ia) and (ib) are identical; (iia) and (iib) are identical, and (iiia) and (iiib) are isomers done
clear
View Solution play_arrow
-
question_answer21)
Tautomerism is exhibited by [IIT-JEE 1998]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer22)
The following reaction is described as [CBSE PMT 1997] \[\begin{matrix} C{{H}_{3}}{{(C{{H}_{2}})}_{5}} \\ {} \\ {{H}_{3}}C \\ \end{matrix}>\underset{H}{\mathop{\underset{|}{\mathop{C}}\,}}\,-Br\xrightarrow{OH}HO-\underset{H}{\mathop{\underset{|}{\mathop{C}}\,}}\,<\begin{matrix} {{(C{{H}_{2}})}_{5}}C{{H}_{3}} \\ {} \\ C{{H}_{3}}\,\,\,\,\,\,\,\,\,\,\, \\ \end{matrix}\]
A)
\[S{{E}^{2}}\] done
clear
B)
\[S{{N}^{1}}\] done
clear
C)
\[S{{N}^{2}}\] done
clear
D)
\[S{{N}^{0}}\] done
clear
View Solution play_arrow
-
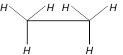
question_answer23)
Which one of the following represents eclipsed form of ethane
A)
B)
C)
D)
None of these done
clear
View Solution play_arrow
-
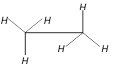
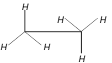
question_answer24)
Which are the staggered forms of ethane
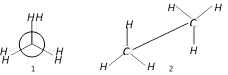
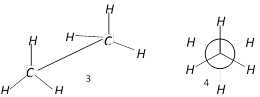
A)
1 and 4 done
clear
B)
3 and 4 done
clear
C)
2 and 4 done
clear
D)
1 and 3 done
clear
View Solution play_arrow
-
question_answer25)
Which of the following is the most stable cation [CBSE PMT 1988; MNR 1988; AIIMS 1985]
A)
\[C{{H}_{3}}C{{H}_{2}}\overset{+}{\mathop{C}}\,HC{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}-\underset{C{{H}_{3}}\,\,\,\,}{\mathop{\underset{|\,\,\,\,\,\,\,\,\,\,\,}{\mathop{\overset{+}{\mathop{C}}\,C{{H}_{3}}}}\,}}\,\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}\overset{+}{\mathop{C}}\,{{H}_{2}}\] done
clear
D)
\[CH_{3}^{+}\] done
clear
View Solution play_arrow
-
question_answer26)
In a reaction of \[{{C}_{6}}{{H}_{5}}Y,\] the major product (> 60%) is m-isomer, so the group Y is [AIIMS 1997; UPSEAT 2003]
A)
\[-COOH\] done
clear
B)
\[-N{{H}_{2}}\] done
clear
C)
\[-OH\] done
clear
D)
\[-Cl\] done
clear
View Solution play_arrow
-
question_answer27)
Dehydrohalogenation in presence of \[O{{H}^{-}}\] is correctly represented by [Orissa JEE 2004]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer28)
Number of structural isomers of \[{{C}_{4}}{{H}_{10}}O\] are [CPMT 1983, 84, 89, 91; MADT Bihar 1984; MNR 1984; MP PET 1997; Pb. PMT 1999; MH CET 2003]
A)
3 done
clear
B)
6 done
clear
C)
7 done
clear
D)
10 done
clear
View Solution play_arrow
-
question_answer29)
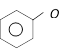
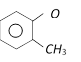
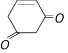
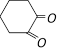
Among the following the aromatic compound is [AIIMS 2004]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer30)
Which of the following compounds are not arranged in order of decreasing reactivity towards electrophilic substitution [DCE 2003]
A)
Fluoro benzene > chloro benzene > bromo benzene done
clear
B)
Phenol > n-propyl benzene > benzoic acid done
clear
C)
Chloro toluene > para-nitro toluene > 2-chloro-4-nitro toluene done
clear
D)
Benzoic acid > phenol > n-propyl benzene done
clear
View Solution play_arrow
-
question_answer31)
Most stable carbonium ion is [Pb. CET 2004]
A)
\[{{\overset{+}{\mathop{C}}\,}_{2}}{{H}_{5}}\] done
clear
B)
\[{{(C{{H}_{3}})}_{3}}\overset{+}{\mathop{C}}\,\] done
clear
C)
\[{{({{C}_{6}}{{H}_{5}})}_{3}}\overset{+}{\mathop{C}}\,\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}\overset{+}{\mathop{C}}\,{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer32)
Which one of the following species is most stable [IIT-JEE 1995]
A)
\[p-{{O}_{2}}N-{{C}_{6}}{{H}_{4}}-\overset{+}{\mathop{C}}\,{{H}_{2}}\] done
clear
B)
\[p-C{{H}_{3}}O-{{C}_{6}}{{H}_{4}}-\overset{+}{\mathop{C}}\,{{H}_{2}}\] done
clear
C)
\[p-Cl-{{C}_{6}}{{H}_{4}}-\overset{+}{\mathop{C}}\,{{H}_{2}}\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}-\overset{+}{\mathop{C}}\,{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer33)
Which of the following gives most stable carbocation by dehydration [RPMT 2002]
A)
\[(C{{H}_{3}}){{}_{2}}CH-OH\] done
clear
B)
\[{{(C{{H}_{3}})}_{3}}C-OH\] done
clear
C)
\[C{{H}_{3}}-C{{H}_{2}}-OH\] done
clear
D)
\[C{{H}_{3}}-C{{H}_{2}}-O-C{{H}_{2}}-C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer34)
Which of the following orders regarding relative stability of free radicals is correct [UPSEAT 2004]
A)
\[3{}^\circ <2{}^\circ <1{}^\circ \] done
clear
B)
\[3{}^\circ \text{ }>\text{ }2{}^\circ \text{ }>\text{ }1{}^\circ \] done
clear
C)
\[1{}^\circ \text{ }<\text{ }2{}^\circ \text{ }>\text{ }3{}^\circ \] done
clear
D)
\[3{}^\circ \text{ }>\text{ }2{}^\circ \text{ }<\text{ }1{}^\circ \] done
clear
View Solution play_arrow
-
question_answer35)
The +I effect of alkyl groups is in the order [DCE 2002]
A)
\[2{}^\circ \text{ }>\text{ }3{}^\circ \text{ }>\text{ }1{}^\circ ~~~~~~~~~~\] done
clear
B)
\[1{}^\circ \text{ }>\text{ }2{}^\circ \text{ }>\text{ }3{}^\circ \] done
clear
C)
\[3{}^\circ \text{ }>\text{ }2{}^\circ \text{ }>\text{ }1{}^\circ ~~~~~~~~~~\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer36)
Which one has asymmetric C-atom [Roorkee 1995]
A)
\[C{{H}_{3}}-C{{H}_{2}}-\underset{Br}{\mathop{\underset{|\,\,\,\,\,}{\mathop{C{{H}_{2}}}}\,}}\,\] done
clear
B)
\[C{{H}_{3}}-\underset{Br}{\mathop{\underset{|\,\,\,}{\mathop{CH}}\,}}\,--\underset{C{{H}_{3}}}{\mathop{\underset{|\,\,\,\,\,\,\,}{\mathop{CH-}}\,}}\,C{{H}_{3}}\] done
clear
C)
\[C{{H}_{3}}-C{{H}_{2}}-\underset{Br}{\mathop{\underset{|\,\,\,}{\mathop{CH}}\,}}\,-C{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}-\underset{Br\,}{\mathop{\underset{|\,\,\,\,}{\mathop{\overset{C{{H}_{3}}}{\mathop{\overset{|\,\,\,\,}{\mathop{C-}}\,}}\,}}\,}}\,C{{H}_{2}}-C{{H}_{2}}-C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer37)
Which of the following compounds will show metamerism [KCET 1996]
A)
\[C{{H}_{3}}COO{{C}_{2}}{{H}_{5}}\] done
clear
B)
\[{{C}_{2}}{{H}_{5}}-S-{{C}_{2}}{{H}_{5}}\] done
clear
C)
\[C{{H}_{3}}-O-C{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}-O-{{C}_{2}}{{H}_{5}}\] done
clear
View Solution play_arrow
-
question_answer38)
How many carbon atoms in the molecule \[HCOO-{{(CHOH)}_{2}}-COOH\]are asymmetric [MP PET 2001]
A)
1 done
clear
B)
2 done
clear
C)
3 done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer39)
Which of the following act as nucleophiles [Roorkee 1999]
A)
\[C{{H}_{3}}N{{H}_{2}}\] done
clear
B)
\[R{{O}^{-}}\] done
clear
C)
\[AlC{{l}_{3}}\] done
clear
D)
\[C{{H}_{3}}MgBr\] done
clear
View Solution play_arrow
-
question_answer40)
Which of the following has the highest nucleophilicity [IIT-JEE Screening 2000]
A)
\[{{F}^{-}}\] done
clear
B)
\[O{{H}^{-}}\] done
clear
C)
\[CH_{3}^{-}\] done
clear
D)
\[NH_{2}^{-}\] done
clear
View Solution play_arrow
-
question_answer41)
Keto-enol tautomerism is found in [IIT-JEE 1988; MADT Bihar 1995]
A)
\[{{H}_{5}}{{C}_{6}}-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-H\] done
clear
B)
\[{{H}_{5}}{{C}_{6}}-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-{{C}_{6}}{{H}_{5}}\] done
clear
C)
\[{{H}_{5}}{{C}_{6}}-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-C{{H}_{3}}\] done
clear
D)
\[{{H}_{5}}{{C}_{6}}-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-C{{H}_{2}}-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer42)
Which of the following compounds will show geometrical isomerism [IIT-JEE 1998]
A)
2-butene done
clear
B)
Propene done
clear
C)
1-phenylpropene done
clear
D)
2-methyl-2-butene done
clear
View Solution play_arrow
-
question_answer43)
Which behaves both as a nucleophile and electrophile [IIT-JEE Screening 1991; Pb. CET 1985]
A)
\[C{{H}_{3}}N{{H}_{2}}\] done
clear
B)
\[C{{H}_{3}}Cl\] done
clear
C)
\[C{{H}_{3}}CN\] done
clear
D)
\[C{{H}_{3}}OH\] done
clear
View Solution play_arrow
-
question_answer44)
The number of optical isomers of an organic compound having n asymmetric carbon atoms will be [MP PET 1994]
A)
\[{{2}^{n+1}}\] done
clear
B)
\[{{n}^{2}}\] done
clear
C)
\[{{2}^{n}}\] done
clear
D)
\[{{2}^{n-1}}\] done
clear
View Solution play_arrow
-
question_answer45)
Total number of isomers of \[{{C}_{6}}{{H}_{14}}\] are [IIT-JEE 1987; DPMT 1983; CPMT 1991; MNR 1990; MP PET 1995; UPSEAT 2001]
A)
4 done
clear
B)
5 done
clear
C)
6 done
clear
D)
7 done
clear
View Solution play_arrow
-
question_answer46)
With a change in hybridisation of the carbon bearing the charge, the stability of a carbanion increase in the order [DCE 2003]
A)
\[sp<s{{p}^{2}}<s{{p}^{3}}\] done
clear
B)
\[sp<s{{p}^{3}}<s{{p}^{2}}\] done
clear
C)
\[s{{p}^{3}}<s{{p}^{2}}<sp\] done
clear
D)
\[s{{p}^{2}}<sp<s{{p}^{3}}\] done
clear
View Solution play_arrow
-
question_answer47)
The C - C bond length of the following molecules is in the order [IIT-JEE 1991]
A)
\[{{C}_{2}}{{H}_{6}}>{{C}_{2}}{{H}_{4}}>{{C}_{6}}{{H}_{6}}>{{C}_{2}}{{H}_{2}}\] done
clear
B)
\[{{C}_{2}}{{H}_{2}}<{{C}_{2}}{{H}_{4}}<{{C}_{6}}{{H}_{6}}<{{C}_{2}}{{H}_{6}}\] done
clear
C)
\[{{C}_{2}}{{H}_{6}}>{{C}_{2}}{{H}_{2}}>{{C}_{6}}{{H}_{6}}>{{C}_{2}}{{H}_{4}}\] done
clear
D)
\[{{C}_{2}}{{H}_{4}}>{{C}_{2}}{{H}_{6}}>{{C}_{2}}{{H}_{2}}>{{C}_{6}}{{H}_{6}}\] done
clear
View Solution play_arrow
-
question_answer48)
In the reaction \[C{{H}_{3}}CHO+HCN\to C{{H}_{3}}CH(OH)CN\]a chiral centre is produced. This product would be [CBSE PMT 1995]
A)
Laevorotatory done
clear
B)
Meso compound done
clear
C)
Dextrorotatory done
clear
D)
Racemic mixture done
clear
View Solution play_arrow
-
question_answer49)
Cyclic hydrocarbon molecule ?A? has all the carbon and hydrogen in a single plane. All the carbon-carbon bonds are of same length less than 1.54Å, but more than 1.34Å. The \[C-C\] bond angle will be [BVP 2003]
A)
\[{{109}^{o}}2{8}'\] done
clear
B)
\[{{100}^{o}}\] done
clear
C)
\[{{180}^{o}}\] done
clear
D)
\[{{120}^{o}}\] done
clear
View Solution play_arrow
-
question_answer50)
How many structural isomers are possible for a compound with molecular formula \[{{C}_{3}}{{H}_{7}}Cl\] [CBSE PMT 2001]
A)
2 done
clear
B)
5 done
clear
C)
7 done
clear
D)
9 done
clear
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear


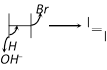 done
clear
done
clear
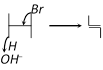 done
clear
done
clear
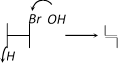 done
clear
done
clear
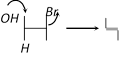 done
clear
done
clear
