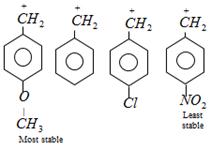
A) \[p-{{O}_{2}}N-{{C}_{6}}{{H}_{4}}-\overset{+}{\mathop{C}}\,{{H}_{2}}\]
B) \[p-C{{H}_{3}}O-{{C}_{6}}{{H}_{4}}-\overset{+}{\mathop{C}}\,{{H}_{2}}\]
C) \[p-Cl-{{C}_{6}}{{H}_{4}}-\overset{+}{\mathop{C}}\,{{H}_{2}}\]
D) \[{{C}_{6}}{{H}_{5}}-\overset{+}{\mathop{C}}\,{{H}_{2}}\]
Correct Answer: B
Solution :
You need to login to perform this action.
You will be redirected in
3 sec
