question_answer 1)
Two elements X and Y have following electronic Configurations
\[X:1{{s}^{2}}\,2{{s}^{2}}\,2{{p}^{2}}\,3{{s}^{2}}\,3{{p}^{6}}4{{s}^{2}}\]
\[Y:1{{s}^{2}}\,2{{s}^{2}}\,2{{p}^{6}}\,3{{s}^{2}}\,3{{p}^{5}}\]
The expected compound formed by combination of X and Y will be expressed as
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 2)
The compound which contains both covalent and co-ordinate bond is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 3)
Which of the following Lewis structure does not contribute in resonance?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 4)
The correct statement for the molecule \[Cs{{I}_{3}}\] is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 5)
Out of the following which has highest dipole moment?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 6)
Which of the following have distorted octahedral structure?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
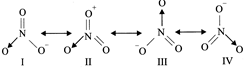
question_answer 7)
The statement true for azide ion (\[{{N}_{3}}^{\odot }\]) is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 8)
\[{{H}_{2}}O\] is dipolar, whereas \[Be{{F}_{2}}\] is not. It I because
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 9)
The electronegativity difference between N and F is greater than that between N and H, yet the dipole moment of \[N{{H}_{3}}\] (1.5 D) is larger than that of \[N{{F}_{3}}\] (0.2 D). This is because
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 10)
In which of the following molecules/ions\[B{{F}_{3}}\],\[N{{O}^{-}}_{2}\] \[N{{H}^{-}}_{2}\], and \[{{H}_{2}}O\] the central atom is \[s{{p}^{2}}_{}\] hybridized?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 11)
In which of the following pairs bond angle is\[109{}^\circ 28'\]?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 12)
Which one of the following species is diamagnetic in nature?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 13)
In which of the following ionization processes, the bond order has increased and the magnetic behavior has changed?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 14)
In which of the following pairs, the two species are not isostructural?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 15)
Which one of the following constitutes a group of the isoelectronic species?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 16)
Specify the coordination geometry around and hybridization Of N and B atoms in a 1 : 1 complex of \[B{{F}_{3}}\] and \[N{{H}_{3}}\]:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 17)
Which of the following diatomic molecules would be stabilised by the removal of an electron?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 18)
Which of the following statements is correct for CO?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 19)
\[As{{F}_{5}}\] molecule is \[s{{p}^{3}}\]d hybridised and is trigonal bipyramidal (Tbp) shape. Which J-orbital is involved in \[s{{p}^{3}}\]d hybridisation.
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 20)
Which of the following have been arranged in increasing bond order as well as bond dissociation energy?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 21)
An ionic bond is established between a positive ion \[{{A}^{+}}\] and negative ion\[{{B}^{-}}\]. How many times strength of the ionic bond is affected by doubling the charge on \[{{A}^{+}}\] and making the radius halved?
View Solution play_arrow
question_answer 22)
Find out number of \[P\]-orbitals of boron atom which participate in hybridisation in \[{{B}_{2}}{{H}_{6}}\] (number of\[p\]-orbitals taking part in hybridisation per atom of boron).
View Solution play_arrow
question_answer 23)
\[A{{X}_{4}}\] Forms square planar type structure. Distance between A and X-atoms is 4 units. What is distance between two X-atoms in trans-position?
View Solution play_arrow
question_answer 24)
At 300 K and 1.00 atm, density of gaseous HF is 3.17\[g{{L}^{-1}}\]. How many HF molecules are associated by H-bonding?
View Solution play_arrow
question_answer 25)
Based on VSEPR theory, the number of 90 degree F-Br-F angles in \[Br{{F}_{5}}\]is
View Solution play_arrow