One mole of ideal gas undergoes cyclic process shown in figure. Process 1 \[\to \] 2 is adiabatic. Heat given to gas in the process is (nearly)-
A)
-59 J
done
clear
B)
-47J
done
clear
C)
-42 J
done
clear
D)
None of these
done
clear
View Answer play_arrow
The pressure and volume of a given mass of gas at a given temperature are P and V respectively. Keeping temperature constant, the pressure is increased by 10% and then decreased by 10%. The volume now will be-
A)
less than V
done
clear
B)
more than V
done
clear
C)
equal to V
done
clear
D)
less than V for diatomic and more than V for monoatomic
done
clear
View Answer play_arrow
A wire is 4 m long and has a mass 0.2 kg. The wire is kept horizontally. A transverse pulse is generated by plucking one end of the taut (tight) wire. The pulse makes four trips back and forth along the cord in 0.8 sec. The tension is the cord will be-
A)
80 N
done
clear
B)
160 N
done
clear
C)
240 N
done
clear
D)
320 N
done
clear
View Answer play_arrow
Two tuning forks when sounded together produced 4 beats/sec. The frequency of one fork is 256. The number of beats heard increases when the fork is loaded with wax. The frequency of the other fork is-
A)
504
done
clear
B)
520
done
clear
C)
260
done
clear
D)
252
done
clear
View Answer play_arrow
The apparent frequency of the whistle of an engine changes by the ratio 5/3 as the engine passes a stationary observer. If the velocity of sound is 340 m/s, then the velocity of the engine is-
A)
340 m/s
done
clear
B)
170 m/s
done
clear
C)
85 m/s
done
clear
D)
42.5 m/s
done
clear
View Answer play_arrow
The frequency of a light ray is\[6\,\,\times \,\,{{10}^{14}}\,Hz\]. Its frequency when it propagates in a medium of refractive index 1.5, will be-
A)
\[1.67\times {{10}^{14}}Hz\]
done
clear
B)
\[\,9.10\times {{10}^{14}}\,Hz\]
done
clear
C)
\[\,6\times {{10}^{14}}Hz\]
done
clear
D)
\[\,4\times {{10}^{14}}\,Hz\]
done
clear
View Answer play_arrow
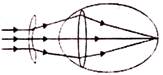
A converging beam of light forms a sharp image on a screen. A lens is placed 10 cm from the screen in the path of the beam. It was found that the screen has to be moved 8 cm further away to obtain the sharp image. Focal length of the lens is-
A)
6.43 cm
done
clear
B)
-2.5 cm
done
clear
C)
- 22.5 cm
done
clear
D)
2.66 cm
done
clear
View Answer play_arrow
The angle of prism is \[60{}^\circ \] and its refractive index is 1.5. There will be no emergent light if the angle of incidence on the first face is \[\left( Given\text{ }sin\text{ }42{}^\circ =2/3,\text{ }sin\text{ }18{}^\circ =0.309\text{ } \right)\]
A)
equal to \[30{}^\circ \]
done
clear
B)
less than \[30{}^\circ \]
done
clear
C)
more than \[30{}^\circ \]
done
clear
D)
equal to \[60{}^\circ \]
done
clear
View Answer play_arrow
The objective lens of an astronomical telescope has a focal length of 60 cm and the eyepiece has a focal length of 2 cm. How far apart should the lenses be placed in order to form a final image at infinity?
A)
62 cm
done
clear
B)
44 cm
done
clear
C)
76 cm
done
clear
D)
60 cm
done
clear
View Answer play_arrow
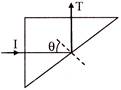
A triangular prism of glass is shown in figure, A ray incident normal to one face is totally reflected. If \[\theta \] is \[45{}^\circ \], then index of refraction of the glass is-
A)
less than 1.41
done
clear
B)
equal to 1.41
done
clear
C)
greater than 1.41
done
clear
D)
None of these
done
clear
View Answer play_arrow
The central fringe of the interference pattern produced by light of wavelength 6000 A is found to shift to the position of 4th bright fringe after a glass plate of refractive index 1.5 is introduced. The thickness of the glass plate would be-
A)
\[4.8\,\mu m\]
done
clear
B)
\[8.23\,\mu m\]
done
clear
C)
\[14.98\,\mu m\]
done
clear
D)
\[3.78\,\mu m\]
done
clear
View Answer play_arrow
Two monochromatic light waves of amplitudes A and 2A interfering at a point, have a phase difference of \[60{}^\circ \]. The intensity at that point will be proportional to-
A)
\[3{{A}^{2}}\]
done
clear
B)
\[5{{A}^{2}}\]
done
clear
C)
\[7{{A}^{2}}~\]
done
clear
D)
\[9{{A}^{2}}\]
done
clear
View Answer play_arrow
The three initial and final position of a man on the x-axis are given as:
(i) (-8m, 7m), (ii) (7m, - 3m) and (iii) (-7m, 3m) Which pair gives the negative displacement?
A)
(i)
done
clear
B)
(ii)
done
clear
C)
(iii)
done
clear
D)
(i) and (ii)
done
clear
View Answer play_arrow
The engine of a motorcycle can produce a maximum acceleration\[5\text{ }m/{{s}^{2}}\]. Its brakes can produce a maximum retardation\[10\text{ }m/{{s}^{2}}\]. What is the minimum time in which it can cover a distance of 1.5 km?
A)
30 sec
done
clear
B)
15 sec
done
clear
C)
10 sec
done
clear
D)
5 sec
done
clear
View Answer play_arrow
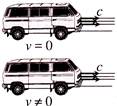
In figure, one car is at rest and velocity of light from head light is c, then velocity of light from head light for the moving car at velocity v, would be
A)
\[c+v~\]
done
clear
B)
\[c-v\]
done
clear
C)
\[c\times v\]
done
clear
D)
c
done
clear
View Answer play_arrow
A boat crosses a river from port A to port B, which are just on the opposite side. The speed of the water is \[{{V}_{W}}\] and that of boat is \[{{V}_{B}}\] relative to still water. Assume\[{{V}_{B}}=2{{V}_{W}}\]. What is the time taken by the boat, if it has to cross the river directly on the AB line?
A)
\[\frac{2D}{{{V}_{B}}\sqrt{3}}\]
done
clear
B)
\[\frac{\sqrt{3}D}{2{{V}_{B}}}\]
done
clear
C)
\[\frac{D}{{{V}_{B}}\sqrt{2}}\]
done
clear
D)
\[\frac{D\sqrt{2}}{{{V}_{B}}}\]
done
clear
View Answer play_arrow
A body is projected vertically up with a velocity v and after some time it returns to the point from which it was projected. The average velocity and average speed of the body for the total time of flight are
A)
\[\frac{\overrightarrow{v}}{2}\,\,and\,\,\frac{v}{2}\]
done
clear
B)
\[0\,\,and\,\,\frac{v}{2}\]
done
clear
C)
0 and 0
done
clear
D)
\[\,\,\,\frac{\overrightarrow{v}}{2}\,\,and\,\,0\]
done
clear
View Answer play_arrow
A circular road of radius 1000 m has banking angle\[45{}^\circ \]. The maximum safe speed of a car having mass 2000 kg will be, if the coefficient of friction between tyre and road is 0.5:
A)
172 m/s
done
clear
B)
124 m/s
done
clear
C)
99 m/s
done
clear
D)
86 m/s
done
clear
View Answer play_arrow
Force acting on a particle is\[\left( 2\widehat{i}+3\widehat{j} \right)N\]. Work one by this force is zero, when a particle is moved on the line\[3y+kx=5\]. Here value of k is-
A)
2
done
clear
B)
4
done
clear
C)
6
done
clear
D)
8
done
clear
View Answer play_arrow
A force \[\overrightarrow{F}=4\widehat{i}-5\widehat{j}+3\widehat{k}\] is acting a point? \[{{\overrightarrow{r}}_{1}}=\widehat{i}+2\widehat{j}+3\widehat{k}\]. The torque acting about a point \[{{\overrightarrow{r}}_{2}}=3\widehat{i}-2\widehat{j}-3\widehat{k}\] is-
A)
Zero
done
clear
B)
\[42\widehat{i}-30\widehat{j}+6\widehat{k}\]
done
clear
C)
\[42\widehat{i}+30\widehat{j}+6\widehat{k}\]
done
clear
D)
\[42\widehat{i}+30\widehat{j}-6\widehat{k}\]
done
clear
View Answer play_arrow
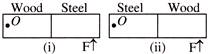
In the figure (i) half of the meter scale is made of wood while the other half, of steel. The wooden part is pivoted at O. A force F is applied at the end of steel part. In figure (ii) the steel part is pivoted at O and the same force is applied at the wooden end
A)
More acceleration will be produced in (i)
done
clear
B)
More acceleration will be produced in (ii)
done
clear
C)
Same acceleration will be produced in both conditions
done
clear
D)
Information is incomplete
done
clear
View Answer play_arrow
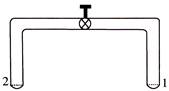
A glass tube of uniform internal radius (r) has a valve separating the two identical ends. Initially, the valve is in a tightly closed position. End 1 has a hemispherical soap bubble of radius r. End 2 has subhemispherical soap-bubble as shown in figure. Just after opening the valve.
A)
Air from end 1 flows towards end 2. No change in the volume of the soap bubbles
done
clear
B)
Air from end 1 flows towards end 2. Volume of the soap bubble at end 1 decreases
done
clear
C)
No change occurs
done
clear
D)
Air from end 2 flows towards end 1. Volume of the soap bubble at end 1 increases
done
clear
View Answer play_arrow
The displacement of a particle varies according to the relation \[x=4\,\,\,(cos\,\,\pi t\,\,+\,\,sin\,\,\pi t)\]. The amplitude of the particle is-
A)
8
done
clear
B)
-4
done
clear
C)
4
done
clear
D)
\[4\sqrt{2}\]
done
clear
View Answer play_arrow
A spring balance is attached to the ceiling of a lift. A man hangs his bag on the spring and the spring reads 49 N, when the lift is stationary. If the lift moves downward with an acceleration of \[5\text{ }m/{{s}^{2}}\], the reading of the spring balance will be:
A)
49 N
done
clear
B)
24 N
done
clear
C)
74 N
done
clear
D)
15 N
done
clear
View Answer play_arrow
The escape velocity for a body projected vertically upwards from the surface of earth is 11 km/s. If the body is projected at an angle of \[45{}^\circ \] with the vertical, the escape velocity will be:
A)
\[\frac{11}{\sqrt{2}}\,\,km/s\]
done
clear
B)
\[11\sqrt{2}\,\,km/s\]
done
clear
C)
22 km/s
done
clear
D)
11 km/s
done
clear
View Answer play_arrow
The photo sensitive surface is receiving light of wavelength \[5000\text{ }\overset{\text{o}}{\mathop{\text{A}}}\,\] at the rate of \[{{10}^{-8}}\text{ }J/s\]. The number of photons received per second is:
A)
\[2.5\times {{10}^{10}}\]
done
clear
B)
\[2.5\times {{10}^{11}}\]
done
clear
C)
\[2.5\,\,\times \,\,{{10}^{12}}\]
done
clear
D)
\[2.5\,\,\times \,\,{{10}^{9}}\]
done
clear
View Answer play_arrow
If electron, proton and He have same energy, then their de Broglie wavelength decreases in order:
A)
\[{{\lambda }_{e}}>{{\lambda }_{p}}>{{\lambda }_{He}}\]
done
clear
B)
\[{{\lambda }_{He}}>{{\lambda }_{p}}>{{\lambda }_{e}}\]
done
clear
C)
\[{{\lambda }_{He}}>{{\lambda }_{e}}>{{\lambda }_{p}}\]
done
clear
D)
\[{{\lambda }_{p}}>{{\lambda }_{e}}>{{\lambda }_{He}}\]
done
clear
View Answer play_arrow
The threshold frequency for a metallic surface corresponds to an energy of 6.2 eV, and the stopping potential for a radiation incident on this surface is 5V. The incident radiation lies in:
A)
X-ray region
done
clear
B)
ultraviolet region
done
clear
C)
infrared region
done
clear
D)
visible region
done
clear
View Answer play_arrow
When a metallic surface is illuminated with monochromatic light of wavelength\[\lambda \], stopping potential for photoelectric current is \[3{{V}_{0}}\], When the same metallic surface is illuminated with a light of wavelength\[2\lambda \], the stopping potential is\[{{V}_{0}}\]. The threshold wavelength for the surface is
A)
\[6\lambda \]
done
clear
B)
\[4\lambda \]
done
clear
C)
\[4\lambda /3\]
done
clear
D)
\[8\lambda \]
done
clear
View Answer play_arrow
A freshly prepared radioactive source of half- life 2 hours emits radiation of intensity which is 64 times the permissible safe level. The minimum time after which it would be possible to work safely with the source is:
A)
6 hours
done
clear
B)
12 hours
done
clear
C)
24 hours
done
clear
D)
128 hours
done
clear
View Answer play_arrow
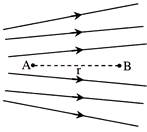
Figure shows the electric lines of force emerging from a charged body. If the electric field at A and B are \[{{E}_{A}}\,and\,\,{{E}_{B}}\] respectively and if the displacement between A and B is r then-
A)
\[{{E}_{A}}\,>\,\,{{E}_{B}}\]
done
clear
B)
\[{{E}_{A}}\,<\,\,{{E}_{B}}\]
done
clear
C)
\[{{E}_{A}}\,=\,\,\frac{{{E}_{B}}}{r}\]
done
clear
D)
\[{{E}_{A}}\,=\,\,\frac{{{E}_{B}}}{{{r}^{2}}}\]
done
clear
View Answer play_arrow
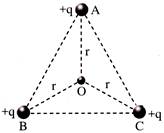
ABC is an equilateral triangle. Charges +q are placed at each comer. The electric intensity at Owill be-
A)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\,\,\frac{q}{{{r}^{2}}}\]
done
clear
B)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\,\,\frac{q}{r}\]
done
clear
C)
Zero
done
clear
D)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\,\,\frac{3q}{{{r}^{2}}}\]
done
clear
View Answer play_arrow
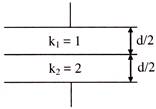
Two parallel plate of area A are separated by two different dielectrics as shown in figure. The net capacitance is-
A)
\[\frac{4{{\varepsilon }_{0}}A}{3d}\]
done
clear
B)
\[\frac{3{{\varepsilon }_{0}}A}{4d}\]
done
clear
C)
\[\frac{2{{\varepsilon }_{0}}A}{d}\]
done
clear
D)
\[\frac{{{\varepsilon }_{0}}A}{d}\]
done
clear
View Answer play_arrow
In a potentiometer experiment two cells of emf \[{{E}_{1}}and\text{ }{{E}_{2}}\] are used in series and in conjunction and the balancing length is found to be 58 cm of the wire. If the polarity of \[{{E}_{2}}\] is reversed, then the balancing length becomes 29 cm. The ratio \[{{E}_{1}}/{{E}_{2}}\] of the emfs of the two cells is-
A)
1 : 1
done
clear
B)
2 : 1
done
clear
C)
3 : 1
done
clear
D)
4 : 1
done
clear
View Answer play_arrow
If an ammeter is to be used in place of a voltmeter then we must connect with the ammeter a-
A)
Low resistance in parallel
done
clear
B)
High resistance in parallel
done
clear
C)
High resistance in series
done
clear
D)
Low resistance in series
done
clear
View Answer play_arrow
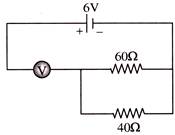
The measurement of voltmeter in the following circuit is-
A)
2.4 V
done
clear
B)
3.4 V
done
clear
C)
4.0 V
done
clear
D)
6.0 V
done
clear
View Answer play_arrow
When a magnetic field is applied in a direction perpendicular to the direction of cathode rays, then their-
A)
Energy decreases
done
clear
B)
Energy increases
done
clear
C)
Momentum increases
done
clear
D)
Momentum and energy remain unchanged
done
clear
View Answer play_arrow
If an electron is going in the direction of magnetic field \[\overrightarrow{B}\] with the velocity of \[\overrightarrow{v}\] then the force on electron is-
A)
Zero
done
clear
B)
\[e\left( \overrightarrow{v}\,.\,\overrightarrow{B} \right)\]
done
clear
C)
\[e\left( \overrightarrow{v}\,\,\times \,\,\overrightarrow{B} \right)\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
Two identical short bar magnets each having magnetic moment M are placed at a distance of 2d with their axes -L to each other in a horizontal plane. The magnetic induction at a point midway between them is-
A)
\[\frac{{{\mu }_{0}}}{4\pi }\sqrt{2}\,\frac{M}{{{d}^{3}}}\]
done
clear
B)
\[\frac{{{\mu }_{0}}}{4\pi }(\sqrt{3})\,\frac{M}{{{d}^{3}}}\]
done
clear
C)
\[\left( \frac{2{{\mu }_{0}}}{4\pi } \right)\,\frac{M}{{{d}^{3}}}\]
done
clear
D)
\[\frac{{{\mu }_{0}}}{4\pi }(\sqrt{5})\,\frac{M}{{{d}^{3}}}\]
done
clear
View Answer play_arrow
An arbitrary shaped closed coil is made of a wire of length L and a current I ampere is flowing in it. If the plane of the coil is perpendicular to magnetic filed \[\overrightarrow{B}\], the force on the coil is-
A)
Zero
done
clear
B)
IBL
done
clear
C)
2IBL
done
clear
D)
\[\frac{1}{2}\,IBL\]
done
clear
View Answer play_arrow
A solenoid has 2000 turns wound over a length of 0.30 m. Its area of cross-section is\[1.2\,\,\times \,\,{{10}^{-}}^{3}{{m}^{2}}\]. Around its central section a coil of 300 turns is wound. If an initial current of 2A in the solenoid is reversed in 0.25 sec, the emf induced in the coil is equal to-
A)
\[6\times {{10}^{-4}}\,Volt\]
done
clear
B)
\[4.8\times {{10}^{-2}}Volt\]
done
clear
C)
\[6\times {{10}^{-2}}\,Volt\]
done
clear
D)
48 kV
done
clear
View Answer play_arrow
A current \[I\,\,=\,\,10\text{ }sin\,\,(100\pi \,t)\] amp. is passed in first coil, which induces a maximum e.m.f of 57i volt in second coil. The mutual inductance between the coils is-
A)
5 mH
done
clear
B)
10 mH
done
clear
C)
15 mH
done
clear
D)
20 mH
done
clear
View Answer play_arrow
An rms voltage of 110 V is applied across a series circuit having a resistance 11 Q and impedance 22Q. The power consumed is-
A)
275 W
done
clear
B)
366 W
done
clear
C)
55 0 W
done
clear
D)
1100 W
done
clear
View Answer play_arrow
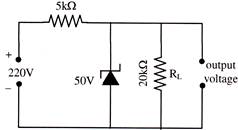
From the zener diode circuit shown in figure, the current through the zener is-
A)
34 mA
done
clear
B)
31.5 mA
done
clear
C)
36.5 mA
done
clear
D)
2.5 mA
done
clear
View Answer play_arrow
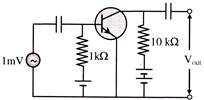
In the following common emitter configuration an NPN transistor with current gain \[\beta =100\] is used. The output voltage of the amplifier will be-
A)
10 mV
done
clear
B)
0.1 V
done
clear
C)
1.0 V
done
clear
D)
10 V
done
clear
View Answer play_arrow
1 amu is equal to-
A)
1.00758 g
done
clear
B)
0.000549 g
done
clear
C)
\[1.66\,\,\times \,\,{{10}^{-}}^{24}\]
done
clear
D)
\[6.02\,\,\times \,\,{{10}^{-}}^{23}g\]
done
clear
View Answer play_arrow
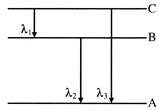
Energy levels A, B, C of a certain atom corresponds to increasing value of energy, i.e.,\[{{E}_{A}}<{{E}_{B}}<{{E}_{C}}\]. If \[{{\lambda }_{1}},\,\,{{\lambda }_{2}}\,\,and\,\,{{\lambda }_{3}}\] are the wavelengths of radiations corresponding to the transitions C to B, B to A and C to A respectively. Which of the following statement is correct-
A)
\[{{\lambda }_{3}}\,\,=\,\,\,{{\lambda }_{1}}\,+\,\,{{\lambda }_{2}}\]
done
clear
B)
\[{{\lambda }_{3}}\,=\,\,\frac{{{\lambda }_{1}}\,{{\lambda }_{2}}}{{{\lambda }_{1}}+\,{{\lambda }_{2}}}\]
done
clear
C)
\[{{\lambda }_{1}}\,+\,\,{{\lambda }_{2}}\,+\,\,{{\lambda }_{3}}\,\,=\,\,0\]
done
clear
D)
\[\lambda _{3}^{2}\,+\,\,\lambda _{1}^{2}\,\,+\,\,\lambda _{2}^{2}\,\]
done
clear
View Answer play_arrow
From the given data identify the oxide of nitrogen that will be most stable at standard state is-
\[2{{N}_{2}}{{O}_{5}}\rightleftharpoons 2{{N}_{2}}+5{{O}_{2}},\,\,{{K}_{p}}=1.2\times {{10}^{34}}\] \[2{{N}_{2}}O\rightleftharpoons 2{{N}_{2}}+{{O}_{2}},\,\,{{K}_{p}}=3.5\times {{10}^{33}}\] \[2NO\rightleftharpoons {{N}_{2}}+{{O}_{2}},\,\,{{K}_{p}}=2.2\times {{10}^{30}}\] \[2N{{O}_{2}}\rightleftharpoons {{N}_{2}}+2{{O}_{2}},\,\,{{K}_{p}}=6.71\times {{10}^{16}}\]
A)
\[N{{O}_{2}}\]
done
clear
B)
NO
done
clear
C)
\[{{N}_{2}}O\]
done
clear
D)
\[{{N}_{2}}{{O}_{5}}\]
done
clear
View Answer play_arrow
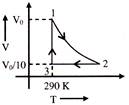
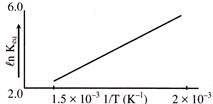
A schematic plot of \[\ell n\,\,{{K}_{eq}}\] versus inverse of temperature of a reaction is shown below
The reaction must be-
A)
One with negligible enthalpy change
done
clear
B)
Highly spontaneous at ordinary temperature
done
clear
C)
Exothermic
done
clear
D)
Endothermic
done
clear
View Answer play_arrow
Cod liver oil is-
A)
Fat dispersed in water
done
clear
B)
Water dispersed in fat
done
clear
C)
Water dispersed in oil
done
clear
D)
Fat dispersed in fat
done
clear
View Answer play_arrow
The arsenius sulphide sol has negative charge the maximum coagulating power for precipitating it is of-
A)
\[0.1\,\,\left( N \right)\,\,Zn{{\left( N{{O}_{3}} \right)}_{2}}\]
done
clear
B)
\[0.1\left( N \right)N{{a}_{3}}P{{O}_{4}}\]
done
clear
C)
\[0.1\text{ }\left( N \right)ZnS{{O}_{4}}\]
done
clear
D)
\[0.1\left( N \right)AlC{{l}_{3}}\]
done
clear
View Answer play_arrow
A buffer solution can be prepared from a mixture of-
[a] Sodium acetate and acetic acid in water [b] Sodium acetate and hydrochloric acid in water [c] Ammonia and ammonium chloride in water [d] Ammonia and sodium hydroxide in water
A)
[a], [b]
done
clear
B)
[b], [c]
done
clear
C)
[c], [d]
done
clear
D)
[a], [b], [c]
done
clear
View Answer play_arrow
The void space in fee lattice is-
A)
\[1-\frac{\pi }{6}\]
done
clear
B)
\[1-\frac{\sqrt{2}\pi }{3}\]
done
clear
C)
\[1-\frac{\sqrt{2}\pi }{6}\]
done
clear
D)
\[1-\frac{\sqrt{3}\pi }{4}\]
done
clear
View Answer play_arrow
A constant current of 1.50 amp is passed through an electrolytic cell containing 0.10 N solution of \[AgN{{O}_{3}}\] and a silver anode and platinum cathode are used. After some time, the concentration of \[AgN{{O}_{3}}\] solution may be-
A)
Equal to 0.10 M
done
clear
B)
Lessthan0.10M
done
clear
C)
Greater than 0.10M
done
clear
D)
Data are not sufficient
done
clear
View Answer play_arrow
Lead storage battery contains -
A)
Pb rod as anode
done
clear
B)
Pb rod as cathode
done
clear
C)
Pb plates coated with \[Pb{{O}_{2}}\] act as cathode
done
clear
D)
[a] & [c] both
done
clear
View Answer play_arrow
With what minimum pressure in Kpa of given volume of an ideal gas \[\left( Cp\,\,=\,\,\frac{7}{2}R \right)\] originally at 400 K and 100 kPa pressure, be adiabatically compressed in order to raise its temperature to 600 K-
A)
362.5 kpa
done
clear
B)
275 kpa
done
clear
C)
437.5 kpa
done
clear
D)
550 kpa
done
clear
View Answer play_arrow
A '15-volume' sample of an \[{{H}_{2}}S{{O}_{4}}\] solution is equivalent to-
A)
5.30 N
done
clear
B)
1.77 N
done
clear
C)
2.68 N
done
clear
D)
7.50 N
done
clear
View Answer play_arrow
When two mole of an ideal gas \[\left( Cp\,\,=\,\,\frac{5}{2}R \right)\] heated from 300 K to 600 K at constant pressure. The change in entropy of gas \[\left( \Delta S \right)\] is-
A)
\[\frac{3}{2}R\ell n2\]
done
clear
B)
\[-\frac{3}{2}R\ell n2\]
done
clear
C)
\[5\,R\ell n2\]
done
clear
D)
\[\frac{5}{2}\,R\ell n2\]
done
clear
View Answer play_arrow
pH of a 0.1 M monobasic acid is measured to be 2. Its osmotic pressure at a given temperature T K is-
A)
0.1 RT
done
clear
B)
0.11 RT
done
clear
C)
1.1 RT
done
clear
D)
0.01 RT
done
clear
View Answer play_arrow
If \[\Delta {{H}_{f}}\] of \[{{C}_{2}}{{H}_{4}}\] and \[{{C}_{2}}{{H}_{6}}\] are \[{{x}_{1}}\,and\text{ }{{x}_{2}}\] \[Kcal\text{ }mo{{l}^{-1}}\], then heat of hydrogenation of \[{{C}_{2}}{{H}_{4}}\] is-
A)
\[{{\text{x}}_{1}}+{{x}_{2}}\]
done
clear
B)
\[{{x}_{1}}-{{x}_{2}}\]
done
clear
C)
\[{{x}_{2}}-{{x}_{1}}\]
done
clear
D)
\[{{x}_{1}}+2{{x}_{2}}\]
done
clear
View Answer play_arrow
At 300 K the half life of a sample of a gaseous compound initially at 1 atm is 100 sec. When the pressure is 0.5 atm the half life is 50 sec. The order of reaction is-
A)
0
done
clear
B)
1
done
clear
C)
2
done
clear
D)
3
done
clear
View Answer play_arrow
D-glucose and D-fructose all form the same osazone derivative because all of them have:
A)
The same configuration at \[C-5\]
done
clear
B)
The same constitution at \[C-1\] and \[C-2\]
done
clear
C)
The same constitution overall
done
clear
D)
The same constitution and configuration at \[C-3,\text{ }C-4,\text{ }C-5\text{ }and\text{ }C-6\] but different constitution and configuration at \[C-1\] and \[C-2\] which becomes identical in osazone formation
done
clear
View Answer play_arrow
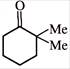
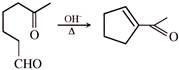
What could be the product for the following reaction?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
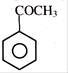
Which of the following does not react with sodium bisulphite?
(I) HCHO (II) \[C{{H}_{3}}CO{{C}_{2}}{{H}_{5}}\] (III) (IV)
A)
(III) and (IV)
done
clear
B)
(II) and (IV)
done
clear
C)
(I) only
done
clear
D)
All reacts
done
clear
View Answer play_arrow
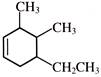
The IUPAC name of
is:
A)
2, 3-Methyl-1 -ethylcyclohex-4-ene
done
clear
B)
5-Ethyl-3, 4-dimethylcyclohex-1-ene
done
clear
C)
4-Ethyl-5, 6-dimethylcyclohex-1-ene
done
clear
D)
1-Ethyl-2, 3-dimethylcyclohex-4-ene
done
clear
View Answer play_arrow
An ice cube at \[0.00{}^\circ C\] is placed in 200 g of distilled water at\[25{}^\circ C\]. The final temperature after the ice completely melted is\[5{}^\circ C\]. What is the mass of the ice cube? \[(\Delta {{H}_{fus}},=340\,{{g}^{-1}},\,\,{{C}_{p}}=4.18\,J\,\,{{g}^{-1}}\,{}^\circ \,{{C}^{-1}})\]
A)
23.6g
done
clear
B)
36.3 g
done
clear
C)
50.0 g
done
clear
D)
800g
done
clear
View Answer play_arrow
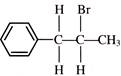
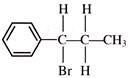
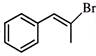
\[C{{H}_{2}}=CH-C\equiv CH\] on reaction with 1 mole of DBr gives:
A)
\[C{{H}_{2}}=CH-CBr=CHD\]
done
clear
B)
\[C{{H}_{2}}\left( Br \right)-CHDC\equiv CH\]
done
clear
C)
\[DC{{H}_{2}}-CHBrC\equiv CH\]
done
clear
D)
\[C{{H}_{2}}=CH-CD=CHBr\]
done
clear
View Answer play_arrow
The acidity order of the protons (FT) in each of the following is:
A)
(i) > (ii) > (iii)
done
clear
B)
(ii) > (iii) > (i)
done
clear
C)
(i) > (iii) > (ii)
done
clear
D)
(iii) > (i) > (ii)
done
clear
View Answer play_arrow
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
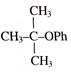
Anisole is treated with HI under two different conditions \[C+D\text{ }\xleftarrow{^{HI(g)}}~{{C}_{6}}{{H}_{5}}OC{{H}_{3}}\xrightarrow{Conc.\,HI}\,\,A\,\,+\,\,B\,\] The nature of A to D will be:
A)
A and B are \[C{{H}_{3}}I\] and \[{{C}_{6}}{{H}_{5}}OH\] while C and D are \[C{{H}_{3}}OH\] and\[{{C}_{6}}{{H}_{5}}I\].
done
clear
B)
A and B are \[C{{H}_{3}}OH\] and\[{{C}_{6}}{{H}_{5}}I\], while C and D are \[C{{H}_{3}}I\] and \[{{C}_{6}}{{H}_{5}}OH\].
done
clear
C)
Both A and B as well as both C and D are \[C{{H}_{3}}I\] and \[{{C}_{6}}{{H}_{5}}OH\]
done
clear
D)
A and B are \[C{{H}_{3}}I\] and\[{{C}_{6}}{{H}_{5}}OH\], while there is no reaction in the second case
done
clear
View Answer play_arrow
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
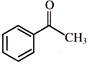
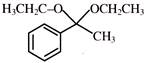
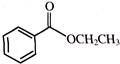
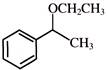
The reaction
is an example of:
A)
Oxidation
done
clear
B)
Reduction
done
clear
C)
Both
done
clear
D)
Aldol condensation
done
clear
View Answer play_arrow
Identify compound [X] in the given reaction \[Me-C\equiv C-H\xrightarrow[dil.\,{{H}_{2}}S{{O}_{4}}]{1%\,HgS{{O}_{4}}}[{{P}_{1}}]\,\xleftarrow[{{H}^{\oplus }}]{KMn{{O}_{4}}}[X]\]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The major product is :
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following statements is correct regarding the slag obtained during the extraction of a metal like copper or iron?
A)
The slag is lighter and lower melting than the metal
done
clear
B)
The slag is heavier and lower melting then the metal
done
clear
C)
The slag is lighter and higher melting than the metal
done
clear
D)
The slag is heavier and higher melting than the metal
done
clear
View Answer play_arrow
Product mixture \[\xrightarrow{{{O}_{3}}/Zn-{{H}_{2}}O}\] Final product mixture The final product mixture contains:
A)
\[C{{H}_{3}}CHO\,\,+\,\,C{{H}_{3}}COC{{H}_{3}}\]
done
clear
B)
\[C{{H}_{3}}CHO+C{{H}_{3}}C{{H}_{2}}CHO+HCHO\]
done
clear
C)
\[C{{H}_{3}}CHO\,\text{ }HCHO\]
done
clear
D)
\[C{{H}_{3}}CHO+C{{H}_{3}}COC{{H}_{3}}+HCHO\]
done
clear
View Answer play_arrow
Select incorrect statement-
A)
\[L{{i}_{2}}C{{O}_{3}}\] is only sparingly soluble in water and no \[LiHC{{O}_{3}}\] has been isolated
done
clear
B)
\[{{K}_{2}}C{{O}_{3}}\] cannot be made by solvay?s process
done
clear
C)
\[L{{i}_{2}}C{{O}_{3}}\] and \[MgC{{O}_{3}}\] both are thermally unstable
done
clear
D)
Carbonates of Ca & Mg are precipitated on adding calgon to water
done
clear
View Answer play_arrow
Select incorrect statement-
A)
Stability of superoxides of alkali metals increases with increase in size of the metal ion
done
clear
B)
Not all bicarbonates of IIA exist in solution
done
clear
C)
Increase in stability in is due to stabilisation of large anions by larger cations through lattice energy effects
done
clear
D)
The low solubility of LiF is due to its high lattice energy whereas low solubility of CsI is due to smaller hydration energy
done
clear
View Answer play_arrow
The hybridiszation present in \[I{{F}_{3}}\] is:
A)
\[\text{s}{{\text{p}}^{\text{3}}}\text{d}\]
done
clear
B)
\[s{{p}^{3}}\]
done
clear
C)
\[s{{p}^{3}}{{d}^{2}}\]
done
clear
D)
\[s{{p}^{3}}{{d}^{3}}\]
done
clear
View Answer play_arrow
Calculate the mass of anhydrous HCl in 10 ml of concentrated HCl \[\left( d=1.2\text{ }g/ml \right)\] solution having \[37%\] HCl by weight.
A)
4.44 g
done
clear
B)
4.44 mg
done
clear
C)
\[4.44\,\,\times \,\,{{10}^{-3}}\,g\]
done
clear
D)
\[0.444\text{ }\mu g\]
done
clear
View Answer play_arrow
Which of the following is not the characteristic of interhalogen compounds?
A)
They are more reactive than halogens
done
clear
B)
They are quite unstable but none of them is explosive
done
clear
C)
They are covalent in nature
done
clear
D)
They have low boiling points and are highly volatile
done
clear
View Answer play_arrow
Most favourable conditions for electrovalent bonding are-
A)
Low ionisation potential of one atom and high electron affinity of the other atom
done
clear
B)
High electron affinity and high ioinisation potential of both the atoms
done
clear
C)
Low electron affinity and low ionization potential of both the atoms
done
clear
D)
High ionisation potential of one atom and low electron affinity of the other atom
done
clear
View Answer play_arrow
The dissolution of \[Al{{\left( OH \right)}_{3}}\] by a solution of NaOH results in the formation of:
A)
\[{{\left[ Al{{\left( {{H}_{2}}O \right)}_{4}}{{\left( OH \right)}_{2}} \right]}^{+}}\]
done
clear
B)
\[\left[ Al{{\left( {{H}_{2}}O \right)}_{3}}{{\left( OH \right)}_{3}} \right]\]
done
clear
C)
\[{{\left[ Al{{\left( {{H}_{2}}O \right)}_{2}}{{\left( OH \right)}_{4}} \right]}^{-}}\]
done
clear
D)
\[\left[ Al{{\left( {{H}_{2}}O \right)}_{6}}{{\left( OH \right)}_{3}} \right]\]
done
clear
View Answer play_arrow
Methane is quite stable whereas silane is unstable? Because-
A)
C ? C bond energy is large greater than Si ? Si bond energy
done
clear
B)
Si ? H bond energy is much lower than C ? H bond energy
done
clear
C)
Si has vacant p orbitals which are more susceptible for nucleophilic attack
done
clear
D)
All of above reasons
done
clear
View Answer play_arrow
\[K{{O}_{2}}\] (potassium super oxide) is used in oxygen cylinders in space and submarines because it-
A)
Eliminates nitrogen
done
clear
B)
Eliminates moisture
done
clear
C)
Absorbs \[C{{O}_{2}}\]
done
clear
D)
Produce ozone
done
clear
View Answer play_arrow
Which of the following is correct-
A)
Solubility: \[BeS{{O}_{4}}>MgS{{O}_{4}}>CaSO4\]
done
clear
B)
Reducing character: \[Li<Na<K<Rb<Cs\]
done
clear
C)
Thermal stability: \[BeC{{O}_{3}}>MgC{{O}_{3}}>CaC{{O}_{3}}\]
done
clear
D)
\[M.P.:LiF>NaF>KF>RbF\]
done
clear
View Answer play_arrow
In the Baeyer's process-
A)
\[A{{l}_{2}}{{O}_{3}}\] goes into solution as soluble \[Al{{\left( OH \right)}_{4}}\] while other basic oxides as \[Ti{{O}_{2}}\] and \[F{{e}_{2}}{{O}_{3}}\] remain insoluble
done
clear
B)
\[A{{l}_{2}}{{O}_{3}}\] changes to AlN which in turn decomposed by \[{{H}_{2}}O\].
done
clear
C)
\[A{{l}_{2}}{{O}_{3}}\] changes to \[A{{l}_{2}}{{\left( C{{O}_{3}} \right)}_{3}}\] which changes to \[AlC{{l}_{3}}\]
done
clear
D)
Cryolite is added to lower the melting point of bauxite
done
clear
View Answer play_arrow
IUPAC name of \[\left[ Pt{{\left( N{{H}_{3}} \right)}_{3}}\left( Br \right)\left( N{{O}_{2}} \right)Cl \right]Cl\] is-
A)
Triamminechlorobromonitroplatinum (IV) chloride
done
clear
B)
Triamminebromonitrochloroplatinum (IV) chloride
done
clear
C)
Triamminebromochloronitroplatinum (IV) chloride
done
clear
D)
Triamminenitrochlorobromoplatinum (IV) chloride
done
clear
View Answer play_arrow
\[A{{g}^{+}}\] forms many complexes, some of these are \[{{\left[ Ag{{\left( N{{H}_{3}} \right)}_{2}} \right]}^{+}}\], \[{{\left[ Ag{{\left( CN \right)}_{2}} \right]}^{-}}\] and \[{{\left[ Ag{{\left( {{S}_{2}}{{O}_{3}} \right)}_{2}} \right]}^{3-}}\]-. Which of the following statement is true-
A)
In these complexes, \[A{{g}^{+}}\] is a lewis base
done
clear
B)
The hybridisation of \[A{{g}^{+}}\] is \[s{{p}^{2}}\]
done
clear
C)
The \[A{{g}^{+}}\] complexes are good reducing agents
done
clear
D)
These complexes are all linear
done
clear
View Answer play_arrow
Which of the following regarding \[{{N}_{2}}O\] is not correct?
A)
The solubility of \[{{N}_{2}}O\] in greater is cold solution as compared to a hot solution
done
clear
B)
The solubility of \[{{N}_{2}}O\] is more in hot solution as compared to a cold solution
done
clear
C)
The gas \[{{N}_{2}}O\] is known as laughing gas
done
clear
D)
Nitrous oxide mixed with oxygen is used as anaesthetic
done
clear
View Answer play_arrow
In plants, mosaic formation, leaf rolling and curling, yellowing of plant parts, vein clearing. dwarfing and stunted growth, necrosis etc. are the symptoms of
A)
bacterial diseases
done
clear
B)
mycoplasmal diseases
done
clear
C)
viral diseases
done
clear
D)
fungal diseases
done
clear
View Answer play_arrow
Cnidoblast consist of-
A)
Stinging capsule
done
clear
B)
Nematocyte
done
clear
C)
Stinging capsule or nematocyte
done
clear
D)
None of above
done
clear
View Answer play_arrow
Medusa formed by-
A)
Sexual reproduction
done
clear
B)
Asexual reproduction
done
clear
C)
Parthenogenesis
done
clear
D)
Regeneration
done
clear
View Answer play_arrow
In Aschehelminthese excretory tube removes body wastes from........ through excretory pore
A)
Blood
done
clear
B)
Tissue
done
clear
C)
Body cavity
done
clear
D)
Haemolymph
done
clear
View Answer play_arrow
Choose how many of following statement is/are not true?
[A] In cephalochordate and urochordate tail persist throughout their life [B] In echinodermata mouth is ventral and anus is dorsal [C] Medusa is free swimming while polyp is sessile [D] Planaria and fasciola have high power regeneration
A)
Two
done
clear
B)
One
done
clear
C)
Four
done
clear
D)
Three
done
clear
View Answer play_arrow
Hypostome is-
A)
Single opening of paragastric cavity of coelenterates
done
clear
B)
Single opening of gastro-vascular cavity of coelenterates
done
clear
C)
Single opening of gastro-vascular cavity of porifera
done
clear
D)
Single opening of spongocoel of porifera
done
clear
View Answer play_arrow
Poisionous snakes are-
A)
Bangarus
done
clear
B)
Naja
done
clear
C)
Vipera
done
clear
D)
All of above
done
clear
View Answer play_arrow
Name reptilian refer to-
A)
Creeping mode of locomotion
done
clear
B)
Dry and comified skin
done
clear
C)
Tympanum represents ear
done
clear
D)
All of above
done
clear
View Answer play_arrow
Hind limbs of aves are modified for-
A)
Walking
done
clear
B)
Swimming
done
clear
C)
Clasping the tree branches
done
clear
D)
All of above
done
clear
View Answer play_arrow
Claspers are-
A)
Present in pelvic fins of carchardon
done
clear
B)
Present in pelvic fins of pterophyllum
done
clear
C)
Present in pectoral fins of scoliodon
done
clear
D)
Present in pectoral fins of exocoetus
done
clear
View Answer play_arrow
Maximum mode of respiration present in-
A)
Mammals
done
clear
B)
Aves
done
clear
C)
Reptile
done
clear
D)
Amphibia
done
clear
View Answer play_arrow
Match List-I with List-II and choose the correct answer-
List-I (PGR) List-II (Application) A. Increase yield of sugarcane i. Zeatin B. Promotes lateral shoot growth ii. \[G{{A}_{3}}\] C. Sprouting of potato tuber iii. ABA D. Inhibites seed germination iv. Ethylene
A)
A-iv B-i C-ii D-iii
done
clear
B)
A-iv B-ii C-iii D-iv
done
clear
C)
A-ii B-i C-iv D-iii
done
clear
D)
A-ii B-I C-iii D-iv
done
clear
View Answer play_arrow
Which of the following chemicals are respectively antiauxin and auxin precursor?
A)
NAA, Acetyl CoA
done
clear
B)
Tryptophan, Methionin
done
clear
C)
Morphectins, Tryptophan
done
clear
D)
MH, Acetyl CoA
done
clear
View Answer play_arrow
Choose the enzyme participate in both the process of photosynthesis and cell respiration-
A)
Pyruvate dehydrogenase
done
clear
B)
ATPase
done
clear
C)
Cytochrome oxidase
done
clear
D)
PEPcase
done
clear
View Answer play_arrow
Net gain of ATP from glycolysis during aerobic respiration-
A)
2
done
clear
B)
6
done
clear
C)
10
done
clear
D)
4
done
clear
View Answer play_arrow
Choose the correct order of energy of oxidation of following respiratory substrates-
A. 1 gm of fat B. 1 gm of protein C. 1 gm of glucose D. 0.5 gm of protein & 0.5 gm of glucose
A)
\[A>B>C>D\]
done
clear
B)
\[A>B>D>C\]
done
clear
C)
\[B>A>C>D\]
done
clear
D)
\[A>D>C>B\]
done
clear
View Answer play_arrow
In \[{{C}_{4}}\] plants the photolysis of water and \[C{{O}_{2}}\] fixation into glucose occurs respectively at-
A)
Both in bundle sheeth cells
done
clear
B)
Bundle sheath cells, Mesophyll cells
done
clear
C)
Mesophyll cells, Bundle sheath cells
done
clear
D)
Both in mesophyll cells
done
clear
View Answer play_arrow
\[{{C}_{4}}\] plants are better than \[{{C}_{3}}\] plants in which of the following parameters-
[a] \[C{{O}_{2}}\] assimilation into glucose [b] Maximizing transpirational loss of water [c] Utilization of assimilatory power in \[C{{O}_{2}}\] assimilation [d] Minimizing transpirational loss
A)
A, B, C, D
done
clear
B)
A, B, C
done
clear
C)
A, D
done
clear
D)
B, D
done
clear
View Answer play_arrow
The trophic levels of Nepenthes, Utricularia, Drosera are-
A)
Primary producer
done
clear
B)
Primary consumers
done
clear
C)
Secondary consumers
done
clear
D)
Both [a] and [c]
done
clear
View Answer play_arrow
A farmer supplies Ca, Mg and Fe regularly to his crop field and yet he observed that the plant show deficiency symptoms like chlorosis and necrosis. The probable cause is-
A)
Molybdenum is deficient in plants
done
clear
B)
Mangnese may be more than critical concentration
done
clear
C)
Roots not able to properly absorb these minerals
done
clear
D)
Radial conduction of minerals should be inhibited
done
clear
View Answer play_arrow
Choose the number of incorrect statement-
[a] Cell shrink in hypotonic solutions and swells in hypertonic solution, [B] Imbibition is a special type of diffusion when water is absorbed by living cell [C] Most of water flows in the roots occurs via the symplast [D] Food translocation is mainly by symplast while water conduction is mainly by apoplast
A)
2
done
clear
B)
3
done
clear
C)
4
done
clear
D)
1
done
clear
View Answer play_arrow
In the developmental history of mammalian heart, it is observed that it passes through a two chambered fish like heart, three chambered frog like heart and finally four chambered stage. To which hypothesis can this above cited statement be approximated?
A)
Lamarck?s principle
done
clear
B)
Mendelian principle
done
clear
C)
Biogenetic law
done
clear
D)
Hardy Weinberg law
done
clear
View Answer play_arrow
Which of the following is not ?stress? for plants?
A)
Herbivores
done
clear
B)
Drought
done
clear
C)
Transpiration
done
clear
D)
Water logging
done
clear
View Answer play_arrow
Climax stage is achieved quickly in secondary succession as compared to primary succession as secondary succession-
A)
It occurs on bare area
done
clear
B)
Occurs after primary succession
done
clear
C)
Occur on area where previously build organic matter is present
done
clear
D)
Autotrophs are pioneer
done
clear
View Answer play_arrow
Select the incorrect match-
A)
Hydrilla - Rooted submerged hydrophyte
done
clear
B)
Nymphea - Non rooted floating hydrophyte
done
clear
C)
Vallisenaria - Rooted submerged hydrophyte
done
clear
D)
Typha - Rooted floating hydrophyte
done
clear
View Answer play_arrow
What will happen to an ecosystem if all herbivores are eliminated?
A)
Complete ecosystem will unstable and functioning of ecosystem damage
done
clear
B)
Population of carnivores will decrease
done
clear
C)
Rate of decomposition will decrease
done
clear
D)
No effect on stability of ecosystem as producer and decomposers can maintains the structure and function of ecosystem
done
clear
View Answer play_arrow
In Disaccharide, if the reducing group of monosaccharide are bonded than these are non- reducing sugar, which of the following is a non-reducing sugar-
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which is true regarding DNA polymerase?
A)
It is DNA dependant DNA polymerase
done
clear
B)
It uses RNA or DNA template to catalyse the polymerization of deoxynucleotide
done
clear
C)
It can start DNA replication
done
clear
D)
[a] & [b] Both
done
clear
View Answer play_arrow
Fluid nature of membrane is important in-
A)
Cell growth and division
done
clear
B)
Endocytosis
done
clear
C)
Secretion
done
clear
D)
All of the above
done
clear
View Answer play_arrow
How many of the following are not true regarding mitochondria:
[a] Matrix possess single circular DNA [b] It has protein synthesizing enzyme [c] Inner membrane form discontinous boundary [d] Mitoribosome are similar to eukaryotic ribosome
A)
Three
done
clear
B)
Four
done
clear
C)
One
done
clear
D)
Two
done
clear
View Answer play_arrow
Nucleosome contains-
A)
RNA
done
clear
B)
DNA
done
clear
C)
Non-histone protein
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Bivalent are formed in ......A........stage but they are clearly visible in ......B........stage. What are A & B respectively-
A)
Pachytene & Zygotene
done
clear
B)
Zygotene & Pachytene
done
clear
C)
Pachytene & Diplotene
done
clear
D)
Zygotene & Metaphase-I
done
clear
View Answer play_arrow
Read the following statements about cell division and select the correct ones
(i) M phase represent the phase when actual cell division occurs and I phase represents the phase between two successive M phase (ii) In the 24 hours average duration of cell cycle of a human cell, cell division proper lasts for only about an hour (iii) M phase constitutes more than \[95%\] of the duration of cell cycle
A)
(i) and (ii)
done
clear
B)
(ii) and (iii)
done
clear
C)
(i) and (iii)
done
clear
D)
(i), (ii) and (iii)
done
clear
View Answer play_arrow
What is true for 'lactational amenorrhea'?
(i) It means absence of menstruation
(ii) Ovulation does not occur during the lactational period.
(iii) Chances of contraception are almost nil upto six months following parturition.
(iv) Side-effects are almost nil
(v) Contraceptive efficiency reduces after the period of intense lactation.
(vi) It is a natural method of contraception
(vii) It increases phagocytosis of sperms
A)
(ii), (iii), (iv) (v) and (vi)
done
clear
B)
(i), (ii), (iii) and (iv)
done
clear
C)
(ii), (iii), (iv) (v) and (vii)
done
clear
D)
(i), (ii), (iii) (iv) (v) and (vi)
done
clear
View Answer play_arrow
Match the Column-I with Column-II and select the option given below-
Column-I
Column-II
M
Plum
A.
Perigynous
N
Guava
B.
Epigynous
O
Mustard
C.
Tetradynamous
P
Lady's Finger
D.
Monoadelphous
A)
M = A; N = B; O = D; P = E
done
clear
B)
M = A; N = B; O = C; P = E
done
clear
C)
M = B; N = A; O = C; P = E
done
clear
D)
M = B; N = A; O = D; P = E
done
clear
View Answer play_arrow
How many of the following show the placentation given in figure-
Onion, Lemon, wheat, cabbage Primrose, Sunflower, Gulmohar, China Rose, Tulip
A)
Six
done
clear
B)
Four
done
clear
C)
Five
done
clear
D)
Seven
done
clear
View Answer play_arrow
What can be the correct floral formula of an actinomorphic flower which is bisexual, hypogynous with five free sepal, five free petal, indefinite free stamen and marginal placentation.
A)
Data insufficient
done
clear
B)
done
clear
C)
done
clear
D)
[b] & [c] are possible
done
clear
View Answer play_arrow
Simple permanent living tissues which are made up of thin-walled similar isodiametric cells are called-
A)
parenchyma tissues
done
clear
B)
collechyma tissues
done
clear
C)
sclerenchyma tissues
done
clear
D)
meristematic tissues
done
clear
View Answer play_arrow
From evolutionary point of view tracheids and sieve cells are more primitive than vessels and sieve tube respectively. The angiosperms have which of the following attributes of xylem?
A)
vessel and sieve tube only
done
clear
B)
tracheids, vessels and sieve tube
done
clear
C)
vessel, sieve cells and sieve tube
done
clear
D)
tracheids, vessel and sieve cells
done
clear
View Answer play_arrow
In leaf, the vascular bundles are found in the-
A)
Veins
done
clear
B)
Palisade tissue
done
clear
C)
Lower epidermis
done
clear
D)
Upper epidermis
done
clear
View Answer play_arrow
Pollen grains may be viable for months in plants of
A)
Rosaceae
done
clear
B)
Leguminosae
done
clear
C)
Solanaceae
done
clear
D)
All the above
done
clear
View Answer play_arrow
The largest cell of female gametophyte is-
A)
Egg Cell
done
clear
B)
Central Cell
done
clear
C)
Antipodal Cell
done
clear
D)
Synergid Cell
done
clear
View Answer play_arrow
To the developing embryosac in megasporangium, the nutrition is provided by:
A)
Tapetum
done
clear
B)
Endosperm
done
clear
C)
Nucellus
done
clear
D)
Cotyledon
done
clear
View Answer play_arrow
A plant breeder employ technique of emasculation
A)
To cover the flower with a bag
done
clear
B)
To prevent self-pollination
done
clear
C)
To prevent stamen from being contaminated
done
clear
D)
To produce female plant
done
clear
View Answer play_arrow
Zoospores of brown algae are-
A)
Pear shaped with two equal laterally inserted flagella
done
clear
B)
Pear shaped with two equal anterior inserted flagella
done
clear
C)
Pear shaped with two unequal laterally inserted flagella
done
clear
D)
Pear shaped with two unequal anterior inserted flagella
done
clear
View Answer play_arrow
An intervening long lived dikaryophase occurs in the life cycle of-
A)
Morels, Truffles
done
clear
B)
Bread mould, Mucor
done
clear
C)
Ustilago and Puccinia
done
clear
D)
Yeast, Penicillium
done
clear
View Answer play_arrow
Select the correct options w.r.t Protonema of Mosses-
[a] Develops directly from spore [b] Creeping, green, branched and filamentous stage [c] Posses sex organs [d] Its lateral bud develops into Leafy stage [e] Helps in vegetative reproduction by fragmentation and budding
A)
b, c, d and e
done
clear
B)
a, b, d and e
done
clear
C)
a, b and d
done
clear
D)
b, c and d
done
clear
View Answer play_arrow
Choose incorrect option w.r.t. site of meiosis-
A)
Claviceps - Ascus
done
clear
B)
Ustilago - Basidium
done
clear
C)
Rhizopus - Zygospore
done
clear
D)
Colletotrichum - Oospore
done
clear
View Answer play_arrow
Which of the following can be regarded as seedless vascular plants?
A)
Angiosperms
done
clear
B)
Gymriosperms
done
clear
C)
Bryophytes
done
clear
D)
Pteridophytes
done
clear
View Answer play_arrow
Read carefully the following statement about the sexual reproduction in homosporous ferns-
I. Water is essential for fertilization. II. Male gametophyte bears antheridia, while female gametophyte bears archegonium, which produces antherozoids and egg cell, respectively. III. Antherozoids and egg cell fuses to give rise zygote. Zygote develops into young embryo. Embryo give rise to sporophyte. Which of the statements given above are correct?
A)
I and II
done
clear
B)
II and III
done
clear
C)
I and III
done
clear
D)
I, II and III
done
clear
View Answer play_arrow
Read carefully the following statements about pteridophytes.
I. They are called vascular cryptogams. II. They produce spores rather than seeds. III. They are used for medicinal purposes. IV. They are used as soil binders. V. They are frequently grown as ornaments. Which of the statements given above are correct?
A)
I, II and V
done
clear
B)
II, IV and V
done
clear
C)
II, III, IV and V
done
clear
D)
I, II, III, IV and V
done
clear
View Answer play_arrow
Select the odd one out w.r.t. category-
A)
Primata
done
clear
B)
Arthropoda
done
clear
C)
Sapindales
done
clear
D)
Poales
done
clear
View Answer play_arrow
Arrange the following taxa in increasing order of common character w.r.t. housefly-
[a] Musca [b] Insecta [c] Chordata [d] Muscidae [e] Diptera
A)
c < b < e < d < a
done
clear
B)
b < e < d < a
done
clear
C)
b > e > d > a
done
clear
D)
c > b > e > d > a
done
clear
View Answer play_arrow
What generally maintains the electrical charge across the neuronal membrane-
A)
\[N{{a}^{+}}\,-{{K}^{+}}\] pump
done
clear
B)
Action potential
done
clear
C)
Excited potential
done
clear
D)
Voltage - gated channels
done
clear
View Answer play_arrow
Which of the following statements is correct?
A)
The electrical potential difference across the resting plasma membrane is called as the resting potential
done
clear
B)
The impulse is action potential
done
clear
C)
\[N{{a}^{+}}\] ions are responsible for generating an action potential
done
clear
D)
All
done
clear
View Answer play_arrow
Hormone responsible for regulation of calcium & phosphate is secreted by-
A)
Follicular cells
done
clear
B)
Aldosterone
done
clear
C)
Oxytocin
done
clear
D)
Parathyroide
done
clear
View Answer play_arrow
Disorders caused by hypersecrtion of concerned hormone-
A)
Critinism, diabetes mellitus
done
clear
B)
Ricketes & diabetes insipidus
done
clear
C)
Gigantism & exophathalamic goitre
done
clear
D)
Addison disease & cushing disease
done
clear
View Answer play_arrow
Retinal cells involved in colour vision are-
A)
Cones
done
clear
B)
Rods
done
clear
C)
Neurons
done
clear
D)
Neuroglial cells
done
clear
View Answer play_arrow
Utriculus is the part of internal ear or membranous labyrinth which forms-
A)
Lower chamber and is concerned with maintenance of equilibrium
done
clear
B)
Lower chamber and is concerned with transmission of sound
done
clear
C)
Upper chamber and is concerned with maintenance of equilibrium
done
clear
D)
Upper chamber and is concerned with transmission of sound
done
clear
View Answer play_arrow
The figure depicts an eye defect and its correction. Select the most appropriate alternative from the following that best explain this-
A)
Myopia, concave lens of suitable focal length
done
clear
B)
Hypermetropia, concave lens of suitable focal length
done
clear
C)
Myopia, concvex lens of suitable focal length
done
clear
D)
Hypermetropia, convex lens of suitable focal length
done
clear
View Answer play_arrow
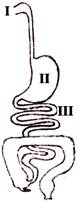
During the process of digestion, food is exposed to a range of pH. The relative pH of fluids in I, II and III regions of the alimentary canal shown in this diagram is-
A)
I < II > III
done
clear
B)
I > II > III
done
clear
C)
I < II < III
done
clear
D)
I > II < III
done
clear
View Answer play_arrow
Ependymal epithelum is made up of?
A)
Ciliated cuboidal
done
clear
B)
Ciliated squamous
done
clear
C)
Simple ciliated columnar
done
clear
D)
Stratified cuboidal
done
clear
View Answer play_arrow
Suppose evolution on earth has occurred in such a way that there are 96 amino acids instead of 20. DNA has 12 different types of bases and DNA synthesis occur in the same way as today. The minimum number of bases per DNA codon would be-
A)
12
done
clear
B)
8
done
clear
C)
2
done
clear
D)
3
done
clear
View Answer play_arrow
Select the correct choice which represents the substance which do not contain any enzyme but important in fat digestion and the organ from where it is secreted-
A)
Bile - Pancreas
done
clear
B)
Saliva - Salivary gland
done
clear
C)
Bile - Liver
done
clear
D)
Gastric juice - Stomach
done
clear
View Answer play_arrow
Which statement is false?
A)
Deficiency of vita. 'E' is too one of the cause of anaemia
done
clear
B)
The hypothalamus is a region of the hind brain important in regulating cardiovascular function, such as heart rate and blood pressure
done
clear
C)
In typical reflex arcs, impulses in sensory neurons activate motor neurons via intemeurons
done
clear
D)
Marasmus is caused by protein deficiency with low calorific value diet
done
clear
View Answer play_arrow
Heparin is produced by-
A)
Kidney cells
done
clear
B)
Blood cells
done
clear
C)
Liver cells
done
clear
D)
Bone marrow
done
clear
View Answer play_arrow
Diazepam (Valium) suppress which part of out body-
A)
Reticular activating system of brain
done
clear
B)
T-killer cell
done
clear
C)
B-cell
done
clear
D)
Pain receptor
done
clear
View Answer play_arrow
Which of following anticancer drug used in AIDS-
A)
Cyclosporin
done
clear
B)
Vincristin
done
clear
C)
Zidovudine
done
clear
D)
Alprazolam
done
clear
View Answer play_arrow
Match the following
Column-I Column-II [A] \[\phi \,\,\times \,\,174\] (i) 48502 bp [B] Lambda phage (ii) 5386 Nucleotides [C] E.coli (iii) \[6.6\times {{10}^{9}}\,bp\] [D] Human somatic cell (iv) \[4.6\times {{10}^{6}}\,bp\]
A)
A-(i) B-(ii) C-(iv) D-(iii)
done
clear
B)
A-(ii) B-(i) C-(iv) D-(iii)
done
clear
C)
A-(i) B-(ii) C-(iii) D-(iv)
done
clear
D)
A-(iv) B-(iii) C-(ii) D-(i)
done
clear
View Answer play_arrow
Mimosaceae can be distinguished from Caesalpiniaceae on the basis of
A)
Number of anthers
done
clear
B)
Adhesion of anthers
done
clear
C)
Characters of carpels
done
clear
D)
Characters of calyx
done
clear
View Answer play_arrow
Due to the cross between \[TTRr\,\,\times \,\,ttrr\] the resultant progenies showed how many percent plants tall, red flowered-
A)
\[50%\]
done
clear
B)
\[75%\]
done
clear
C)
\[25%\]
done
clear
D)
\[100%\]
done
clear
View Answer play_arrow
Mangolian idiots are due to trisomy in 21st chromosome is called-
A)
Down's syndrome
done
clear
B)
Turner's syndrome
done
clear
C)
Kleinfelters syndrome
done
clear
D)
Triplex syndrome
done
clear
View Answer play_arrow
Which of the following statements is correct?
A)
Animals can show mitotic divisions in both haploid
done
clear
B)
After S phase the number of chromosomes becomes double i.e. 2n to 4n
done
clear
C)
During the \[{{G}_{2}}\] phase, proteins are synthesized in preparation for mitosis while cell growth continues.
done
clear
D)
S or synthesis phase marks the period during which RNA synthesis takes place.
done
clear
View Answer play_arrow
Genetics modified crops (GMC) are usefull in agriculture because-
A)
They are more tolerant to abiotic stresses
done
clear
B)
They increase reliance on chemical pesticide
done
clear
C)
They have reduced nutrilitional value
done
clear
D)
All the above
done
clear
View Answer play_arrow
In RNA interference (RNA)-
A)
The silencing of a specific mRNA due to a complementary dsRNA molecule binds to and prevent translation of the mRNA
done
clear
B)
The silencing of a specific mRNA due to ds DNA
done
clear
C)
The silencing of a specific mRNA due to tRNA
done
clear
D)
All the above.
done
clear
View Answer play_arrow
In which stage of cell cycle, DNA replication occurs-
A)
\[{{G}_{1}}\,\,-\,\,phase\]
done
clear
B)
S - phase
done
clear
C)
\[{{G}_{2}}\,\,-\,\,phase\]
done
clear
D)
M - phase
done
clear
View Answer play_arrow
Which photosynthetic pigment converts nascent oxygen to molecular oxygen:
A)
Chlorophyll-a
done
clear
B)
Carotenoids
done
clear
C)
Phycobilins
done
clear
D)
Chlorophyll-b
done
clear
View Answer play_arrow
What is true for plasmid?
A)
Plasmids are widely used in gene transfer
done
clear
B)
These are found in virus
done
clear
C)
Plasmid contain gene for vital activities
done
clear
D)
These are main part of chromosome
done
clear
View Answer play_arrow
Blood carries the \[C{{O}_{2}}\] in 3 forms. The correct percentages of \[C{{O}_{2}}\] in these forms are-
A)
As carbinohaemoglobin in RBC As bicarbonates Dissolved form in plasma 20-25% 70% 7%
done
clear
B)
As carbinohaemoglobin in RBC As bicarbonates Dissolved form in plasma 70% 20-25% 7%
done
clear
C)
As carbinohaemoglobin in RBC As bicarbonates Dissolved form in plasma 20-25% 7% 70%
done
clear
D)
As carbinohaemoglobin in RBC As bicarbonates Dissolved form in plasma 7% 20-25% 70%
done
clear
View Answer play_arrow
Identify correct statement-
(i) Chest expands because diaphragm expands (ii) Chest expands because air enters into lungs (iii) The affinity ofcarbondioxide for haemoglobin is higher than the affinity of oxygen for haemoglobin (iv) Carbaminohaemoglobin is a more stable compound than carboxyhaemoglobin
A)
(i) & (iv)
done
clear
B)
(iii) only
done
clear
C)
(ii) & (iii)
done
clear
D)
None
done
clear
View Answer play_arrow
The third stage of parturition is called "after- birth". In this stage
A)
excessive bleeding occurs
done
clear
B)
fetus is born and cervix and vagina contraction to normal condition happens
done
clear
C)
fetus is born and contraction of uterine wall prevents excessive bleeding
done
clear
D)
placenta is expelled out
done
clear
View Answer play_arrow
50 primary spermatocytes give rise to-
A)
200 spermatocytes
done
clear
B)
50 spermatocytes
done
clear
C)
200 spermatozoa
done
clear
D)
50 spermatozoa.
done
clear
View Answer play_arrow
Read the following statements regarding the various techniques used in cancer detection
(i) Cancer detection is based on biospy and histophathological studies of the tissue, and blood and bone marrow tests for increased cell counts in case of leukaemia (ii) In biopsy, a piece of the suspected tissue cut into thin sections is stained and examined under microscope by a pathologist (iii) Techniques like radiography (use of X-rays), CT (computed tomography) and MRI (magnetic resonance imaging) are very useful to detect cancers of the internal organs. (iv) Computed tomography uses strong magnetic fields and non-ionising radiations to detect physiological changes in living tissues. (v) MRI uses X-rays and ionising radiation to generate a 3-D image of the internal structure of an object. Which of the above statements are incorrect?
A)
(i) and (iii)
done
clear
B)
(ii) and (iv)
done
clear
C)
(iii) and (iv)
done
clear
D)
(iv) and (v)
done
clear
View Answer play_arrow
In foetus, ductus arteriosus connects
A)
aorta and pulmonary artery
done
clear
B)
aorta and pulmonary vein
done
clear
C)
hepatic and pancreatic duct
done
clear
D)
pulmonary artery and vein
done
clear
View Answer play_arrow
One of the following is not a component of kidney stones-
A)
calcium oxalate
done
clear
B)
calcium phosphate
done
clear
C)
cholesterol
done
clear
D)
none of above
done
clear
View Answer play_arrow
Which of the following statement is false?
A)
ADH helps in water elimination making urine hypotonic
done
clear
B)
Protein free fluid is filtered from blood plasma into the Bowman?s capsule
done
clear
C)
Glucose is actively reabsorbed in the proximal convoluted tubule
done
clear
D)
Henle?s loop plays an important role in concentrating the urine
done
clear
View Answer play_arrow
Identify the correct arrangement of periods of Palaeozoic era in ascending order in geological time scale
A)
Cambrian \[\to \] Devonian \[\to \] Ordovician \[\to \] Silurian \[\to \] Carboniferous \[\to \] Permian
done
clear
B)
Cambrian \[\to \] Ordovician \[\to \] Silurian \[\to \] Devonian \[\to \] Carboniferous \[\to \]Permian
done
clear
C)
Cambrian \[\to \] Ordovician \[\to \] Devonian \[\to \] Silurian \[\to \] Carboniferous \[\to \] Permian
done
clear
D)
Silurian \[\to \] Devonian \[\to \] Cambrian \[\to \] Ordovician \[\to \] Permian \[\to \] Carboniferous
done
clear
View Answer play_arrow
In the Oparin theory, the most important/ essential element was-
A)
Proteins
done
clear
B)
Oxygen
done
clear
C)
Amino acids
done
clear
D)
Hydrogen, ammonia and methane
done
clear
View Answer play_arrow
Consider the statements given below regarding contraception and answer as directed thereafter.
[a] Medical Termination of Pregnancy (MTP) during first trimester is generally safe [b] Generally chances of conception are nil until mother breast-feeds the infant upto two years [c] Intrauterine devices like copper-T are effective contraceptives [d] Contraceptive pills may be taken upto one week after coitus to prevent concention Which two of the above statements are correct?
A)
b, c
done
clear
B)
c, d
done
clear
C)
a, c
done
clear
D)
a, b
done
clear
View Answer play_arrow
Plants bend toward the light because-
A)
They need light for photosynthesis
done
clear
B)
They need light for respiration
done
clear
C)
Light attracts them
done
clear
D)
Cells on the shaded side elongate more
done
clear
View Answer play_arrow
 The reaction must be-
The reaction must be- 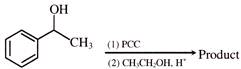
 is:
is: 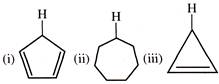
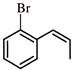
 is an example of:
is an example of: 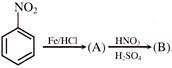 The major product is :
The major product is : 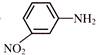
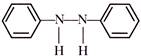
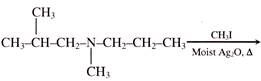 Product mixture \[\xrightarrow{{{O}_{3}}/Zn-{{H}_{2}}O}\] Final product mixture The final product mixture contains:
Product mixture \[\xrightarrow{{{O}_{3}}/Zn-{{H}_{2}}O}\] Final product mixture The final product mixture contains: 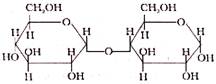
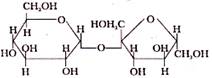
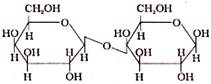
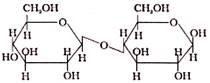
 Onion, Lemon, wheat, cabbage Primrose, Sunflower, Gulmohar, China Rose, Tulip
Onion, Lemon, wheat, cabbage Primrose, Sunflower, Gulmohar, China Rose, Tulip