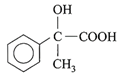
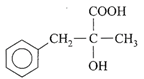
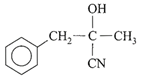
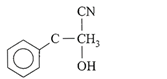
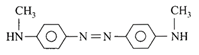The dimensional formula for Young's modulus is
A)
\[M{{L}^{-1}}{{T}^{-2}}\]
done
clear
B)
\[{{M}^{0}}L{{T}^{-2}}\]
done
clear
C)
\[ML{{T}^{-2}}\]
done
clear
D)
\[M{{L}^{2}}{{T}^{-2}}\]
done
clear
View Answer play_arrow
A man drops a ball downside from the roof of a tower of height 400 meters. At the same time another ball is thrown upside with a velocity 50 meter/sec from the surface of the tower, then they will meet at which height from the surface of the tower
A)
100 meters
done
clear
B)
320 meters
done
clear
C)
80 meters
done
clear
D)
240 meters
done
clear
View Answer play_arrow
A car moves from X to Y with a uniform speed \[{{v}_{u}}\]and returns to Y with a uniform speed \[{{v}_{d}}.\]The average speed for this round trip is:
A)
\[\frac{2{{v}_{d}}{{v}_{u}}}{{{v}_{d}}+{{v}_{u}}}\]
done
clear
B)
\[\sqrt{{{v}_{u}}{{v}_{d}}}\]
done
clear
C)
\[\frac{{{v}_{d}}{{v}_{u}}}{{{v}_{d}}+{{v}_{u}}}\]
done
clear
D)
\[\frac{{{v}_{u}}+{{v}_{d}}}{2}\]
done
clear
View Answer play_arrow
A particle of unit mass undergoes one-dimensional motion such that its velocity varies according to \[\upsilon (x)=\beta {{x}^{-2n}}\] where\[\beta \]and\[n\]are constants and x is the position of the particle. The acceleration of the particle as a function of \[x\]is given by
A)
\[-2n{{\beta }^{2}}{{x}^{-2n-1}}\]
done
clear
B)
\[-2n{{\beta }^{2}}{{x}^{-4n-1}}\]
done
clear
C)
\[-2n{{\beta }^{2}}{{x}^{-2n+1}}\]
done
clear
D)
\[-2n{{\beta }^{2}}{{e}^{-4n+1}}\]
done
clear
View Answer play_arrow
The vector sum of two forces is perpendicular to their vector differences. In that case, the force
A)
cannot be predicted
done
clear
B)
are equal to the each other
done
clear
C)
are equal to each other in magnitude
done
clear
D)
are not equal to each other in magnitude
done
clear
View Answer play_arrow
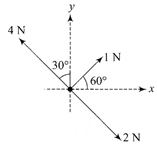
Three forces acting on a body are shown in the figure. To have the resultant force only along the \[y-\]direction, the magnitude of the minimum additional force needed is:
A)
\[0.5\,N\]
done
clear
B)
\[1.5\,N\]
done
clear
C)
\[\frac{\sqrt{3}}{4}N\]
done
clear
D)
\[\sqrt{3}\,N\]
done
clear
View Answer play_arrow
The position vector of a particle \[\vec{R}\]as a function of time is given by: \[\vec{R}=4\sin (2\pi t)\hat{i}+4\cos (2\pi t)\hat{j}\] Where R is in meters, \[t\]is in seconds and \[\hat{i}\]and \[\hat{j}\]denote unit vectors along \[x-\]and y- directions, respectively. Which one of the following statements is wrong for the motion of particle?
A)
Path of the particle is a circle of radius 4 meter
done
clear
B)
Acceleration vectors is along \[-\vec{R}\]
done
clear
C)
Magnitude of acceleration vector is \[\frac{{{v}^{2}}}{R}\] where v is the velocity of particle.
done
clear
D)
Magnitude of the velocity of particle is 8 meter/second
done
clear
View Answer play_arrow
A lift of mass 1000 kg is moving with an acceleration of\[1\text{ }m/{{s}^{2}}\]in upward direction. Tension developed in the string, which is connected to the lift, is
A)
9,800 N
done
clear
B)
10,000 N
done
clear
C)
10,800 N
done
clear
D)
11, 000 N
done
clear
View Answer play_arrow
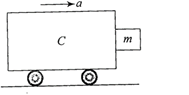
A block of mass m is in contact with the cart C as shown in the figure.
The coefficient of static friction between the block and the cart is p.. The acceleration a of the cart that will prevent the block from falling satisfies
A)
\[a>\frac{mg}{\mu }\]
done
clear
B)
\[a>\frac{g}{\mu m}\]
done
clear
C)
\[a\ge \frac{g}{\mu }\]
done
clear
D)
\[a<\frac{g}{\mu }\]
done
clear
View Answer play_arrow
A system consists of three masses \[{{m}_{1}},{{m}_{2}}\]and \[{{m}_{3}}\] connected by a string passing over a pulley P. The mass w; hangs freely and\[{{m}_{2}}\]and \[{{m}_{3}}\]are on a rough horizontal table (the coefficient of friction\[=\mu \]) The pulley is frictionless and of negligible mass. The downward acceleration of mass m, is
(Assume\[{{m}_{1}}={{m}_{2}}={{m}_{3}}=m\])
A)
\[\frac{g(1-g\mu )}{9}\]
done
clear
B)
\[\frac{2g\mu }{3}\]
done
clear
C)
\[\frac{g(1-2\mu )}{3}\]
done
clear
D)
\[\frac{g(1-2\mu )}{2}\]
done
clear
View Answer play_arrow
The mass of two substances are 4 gm and 9 gm respectively. If their kinetic energies are the same, then the ratio of their momenta will be
A)
4:9
done
clear
B)
9:4
done
clear
C)
3:2
done
clear
D)
2:3
done
clear
View Answer play_arrow
A vertical spring with force constant k is fixed on a table. A ball of mass m at a height h above the free upper end of the spring falls vertically on the spring, so that the spring is compressed by a distance d. The net work done in the process is
A)
\[mg(h+d)+\frac{1}{2}k{{d}^{2}}\]
done
clear
B)
\[mg(h+d)-\frac{1}{2}k{{d}^{2}}\]
done
clear
C)
\[mg(h-d)-\frac{1}{2}k{{d}^{2}}\]
done
clear
D)
\[mg(h-d)+\frac{1}{2}k{{d}^{2}}\]
done
clear
View Answer play_arrow
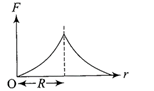
Force F on a particle moving in a straight line varies with distance d as shown in the figure. The work done on the particle during its displacement of 12 m is
A)
21 J
done
clear
B)
26 J
done
clear
C)
13 J
done
clear
D)
18 J
done
clear
View Answer play_arrow
The heart of a man pumps 5 litres of blood through the arteries per minute at a pressure of 150 mm of mercury. If the density of mercury be \[13.6\times {{10}^{3}}kg/{{m}^{3}}\text{ }and\text{ }g=10m/{{s}^{2}}\]then the power of heart in watt is:
A)
1.50
done
clear
B)
1.70
done
clear
C)
2.35
done
clear
D)
3.0
done
clear
View Answer play_arrow
A composite disc is to be made using equal masses of aluminium and iron so that it has as high a moment of inertia as possible. This is possible when
A)
the surfaces of the discs are made of iron with aluminium inside
done
clear
B)
the whole of aluminium is kept in the core and the iron at the outer rim of the disc
done
clear
C)
the whole of the iron is kept in the core and the aluminium at the outer rim of the disc
done
clear
D)
the whole disc is made with thin alternate sheets of iron and aluminium
done
clear
View Answer play_arrow
A particle of mass m moves in the AY plane with a velocity v along the straight line AB. If the angular momentum of the particle with respect to origin 0 is \[{{L}_{A}}\]when it is at A and \[{{L}_{B}}\]when it is at B, then
A)
\[{{L}_{A}}>{{L}_{B}}\]
done
clear
B)
\[{{L}_{A}}={{L}_{B}}\]
done
clear
C)
the relationship between\[{{L}_{A}}\]and\[{{L}_{B}}\]depends upon the slope of the line AB
done
clear
D)
\[{{L}_{A}}<{{L}_{B}}\]
done
clear
View Answer play_arrow
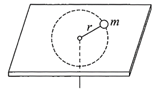
A small mass attached to a string rotates on frictionless table top as shown. If the tension of the string is increased by pulling the string causing the radius of the circular motion to decrease by a factor of 2, the kinetic energy of the mass will be
A)
remain constant
done
clear
B)
increase by a factor of 2
done
clear
C)
increase by a factor of 4
done
clear
D)
decrease by a factor of 2
done
clear
View Answer play_arrow
A rod PQ of mass M and length L is hinged at end P. The rod is kept horizontal by a massless string tied to point Q as shown in the figure. When string is cut, the initial angular acceleration of the rod is
A)
\[\frac{3g}{2L}\]
done
clear
B)
\[\frac{g}{L}\]
done
clear
C)
\[\frac{2g}{L}\]
done
clear
D)
\[\frac{2g}{3L}\]
done
clear
View Answer play_arrow
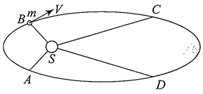
The figure shows elliptical orbit of a planet m about the sun S. The shaded area SCD is twice the shaded area SAB. If \[{{t}_{1}}\]is the time for the planet to move from C to D and\[{{t}_{2}}\] is the time to move from A to B, then
A)
\[{{t}_{1}}>{{t}_{2}}\]
done
clear
B)
\[{{t}_{1}}=4{{t}_{2}}\]
done
clear
C)
\[{{t}_{1}}=2{{t}_{2}}\]
done
clear
D)
\[{{t}_{1}}={{t}_{2}}\]
done
clear
View Answer play_arrow
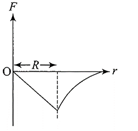
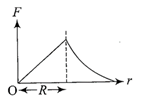
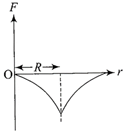
Dependence of intensity of gravitational field (E) of earth with distance (r) from centre of earth is correctly represented by
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
A capillary tube of radius R is immersed in water and water rises in it to a height H. Mass of water in the capillary tube is M. If the radius of the tube is doubled, mass of water that will rise in the capillary tube will now be
A)
M
done
clear
B)
2M
done
clear
C)
M/2
done
clear
D)
4M
done
clear
View Answer play_arrow
Two identical metal balls at temperature \[\text{200 }\!\!{}^\circ\!\!\text{ C}\] and \[\text{400 }\!\!{}^\circ\!\!\text{ C}\] kept in air at\[\text{27 }\!\!{}^\circ\!\!\text{ C}\]. The ratio of net heat loss by these bodies is
A)
1/4
done
clear
B)
1/2
done
clear
C)
1/16
done
clear
D)
\[\frac{{{473}^{4}}-{{300}^{4}}}{{{673}^{4}}-{{300}^{4}}}\]
done
clear
View Answer play_arrow
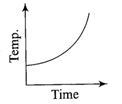
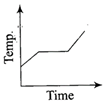
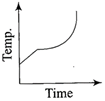
Liquid oxygen at 50 K is heated to 300 K at constant pressure of 1 atm. The rate of heating is constant. Which one of the following graphs represents the variation of temperature with time?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
An ideal gas heat engine operates in a Carnot cycle between \[\text{27 }\!\!{}^\circ\!\!\text{ C}\] and\[\text{127 }\!\!{}^\circ\!\!\text{ C}\]. It absorbs 6 kcal at the higher temperature. The amount of heat (in kcal) converted into work is equal to
A)
3.5
done
clear
B)
1.6
done
clear
C)
1.2
done
clear
D)
4.8
done
clear
View Answer play_arrow
At \[\text{10 }\!\!{}^\circ\!\!\text{ C}\] the value of the density of a fixed mass of an ideal gas divided by its pressure is\[x.\]At \[\text{110 }\!\!{}^\circ\!\!\text{ C}\] this ratio is
A)
\[x\]
done
clear
B)
\[\frac{383}{283}x\]
done
clear
C)
\[\frac{10}{110}x\]
done
clear
D)
\[\frac{283}{383}x\]
done
clear
View Answer play_arrow
An ideal gas goes from state A to state B via three different processes as indicated in the p -V diagram If \[{{Q}_{1}},{{Q}_{2}},{{Q}_{3}}\] indicate the heat absorbed by the gas along the three processes and \[\Delta {{U}_{1}},\]
\[\Delta {{U}_{2}},\Delta {{U}_{3}}\]indicate the change in internal energy along the three processes respectively, then
A)
\[{{Q}_{1}}>{{Q}_{2}}>{{Q}_{3}}\]and\[\Delta {{U}_{1}}=\Delta {{U}_{2}}=\Delta {{U}_{3}}\]
done
clear
B)
\[{{Q}_{3}}>{{Q}_{2}}>{{Q}_{1}}\]and\[\Delta {{U}_{1}}=\Delta {{U}_{2}}=\Delta {{U}_{3}}\]
done
clear
C)
\[{{Q}_{1}}={{Q}_{2}}={{Q}_{3}}\]and\[\Delta {{U}_{1}}>\Delta {{U}_{2}}>\Delta {{U}_{3}}\]
done
clear
D)
\[{{Q}_{3}}={{Q}_{2}}={{Q}_{1}}\]and\[\Delta {{U}_{1}}>\Delta {{U}_{2}}>\Delta {{U}_{3}}\]
done
clear
View Answer play_arrow
Figure below shows two paths that may be taken by a gas to go from a state A to a state C. In process AB, 400 J of heat is added to the system and in process BC, 100 J of heat is added to the system. The heat absorbed by the system in the process AC will be
A)
380 J
done
clear
B)
500 J
done
clear
C)
460 J
done
clear
D)
300 J
done
clear
View Answer play_arrow
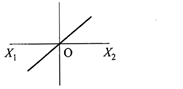
A particle of mass m oscillates with simple harmonic motion between points\[{{x}_{1}}\]and\[{{x}_{2}},\]the equilibrium position being O. Its potential energy is plotted. It will be as given below in the graph
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
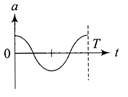
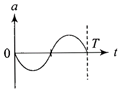
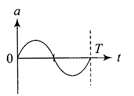
The oscillation of a body on a smooth horizontal surface is represented by the equation, \[X=A\cos (\omega t)\] where X = displacement at time t \[\omega =\]frequence of oscillation Which one of the following graphs shows correctly the variation a with f?
Note:- Here a = acceleration at time t T= time period
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
A source and an observer move away from each other with a velocity of 10 m/s with respect to ground. If the observer finds the frequency of sound coming from the source as 1950 Hz, then actual frequency of the source is (velocity of sound in air = 340 m/s)
A)
1950 Hz
done
clear
B)
2068 Hz
done
clear
C)
2132 Hz
done
clear
D)
2486 Hz
done
clear
View Answer play_arrow
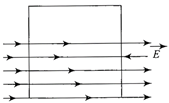
A square surface of side L m is in the plane of the paper. A uniform electric field \[\vec{E}(V/m),\]also in the plane of the paper, is limited only to the lower half of the square surface (see figure). The electric flux in SI units associated with the surface is:
A)
\[E{{L}^{2}}/(2{{\varepsilon }_{0}})\]
done
clear
B)
\[E{{L}^{2}}/2\]
done
clear
C)
zero
done
clear
D)
\[E{{L}^{2}}\]
done
clear
View Answer play_arrow
If identical charges (-q) are placed at each corner of a cube of side b, then electric potential energy of charge (+q) which is placed at centre of the cube will be
A)
\[\frac{8\sqrt{2}{{q}^{2}}}{4\pi {{\varepsilon }_{0}}b}\]
done
clear
B)
\[\frac{-8\sqrt{2}{{q}^{2}}}{\pi {{\varepsilon }_{0}}b}\]
done
clear
C)
\[\frac{-4\sqrt{2}{{q}^{2}}}{\pi {{\varepsilon }_{0}}b}\]
done
clear
D)
\[\frac{-4{{q}^{2}}}{\sqrt{3}\pi {{\varepsilon }_{0}}b}\]
done
clear
View Answer play_arrow
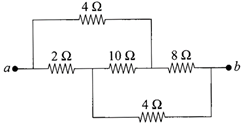
Find the equivalent resistance between the points a and b
A)
\[2\,\Omega \]
done
clear
B)
\[4\,\Omega \]
done
clear
C)
\[8\,\Omega \]
done
clear
D)
\[16\,\Omega \]
done
clear
View Answer play_arrow
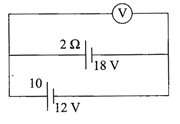
Two batteries, one of emf 18V and internal resistance \[2\,\Omega \] and the other of emf 12 V and
internal resistance \[1\,\Omega ,\] are connected as shown. The voltmeter V will record a reading of
A)
15 V
done
clear
B)
30 V
done
clear
C)
14 V
done
clear
D)
18 V
done
clear
View Answer play_arrow
An electron enters a magnetic field whose direction is perpendicular to the velocity of the electron. Then
A)
The speed of the electron will increase
done
clear
B)
The speed of the electron will decrease
done
clear
C)
The speed of the electron will remain the same
done
clear
D)
The velocity of the electron will remain the same
done
clear
View Answer play_arrow
A diamagnetic material in a magnetic field moves
A)
From weaker to the stronger parts of the field
done
clear
B)
Perpendicular to the field
done
clear
C)
From stronger to the weaker parts of the field
done
clear
D)
In none of the above directions
done
clear
View Answer play_arrow
In a coil of resistance\[1\,\Omega ,\]the induced current developed by changing magnetic flux through it, is shown in figure as a function of time. The magnitude of change in flux through the coil in weber is
A)
8
done
clear
B)
2
done
clear
C)
6
done
clear
D)
4
done
clear
View Answer play_arrow
In a circuit, L, C and R are connected in series with an alternating voltage source of frequency \[f.\] The current leads the voltage by \[\text{45 }\!\!{}^\circ\!\!\text{ }\]. The value of C is
A)
\[\frac{1}{2\pi f(2\pi fL+R)}\]
done
clear
B)
\[\frac{1}{\pi f(2\pi fL+R)}\]
done
clear
C)
\[\frac{1}{2f(2\pi fL-R)}\]
done
clear
D)
\[\frac{1}{\pi f(2\pi fL-R)}\]
done
clear
View Answer play_arrow
The electric and magnetic field of an electro- magnetic wave is
A)
in phase and parallel to each other
done
clear
B)
in opposite phase and perpendicular to each other
done
clear
C)
in opposite phase and parallel to each other
done
clear
D)
in phase and perpendicular to each other
done
clear
View Answer play_arrow
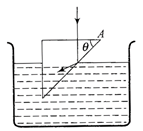
The refractive indices of the material of the prism and liquid are 1.56 and 1.32 respectively. What will be the value of \[\theta \] for the following refraction?
A)
\[\sin \theta \ge \frac{13}{11}\]
done
clear
B)
\[\sin \theta \ge \frac{11}{13}\]
done
clear
C)
\[\sin \theta \ge \frac{\sqrt{3}}{2}\]
done
clear
D)
\[\sin \theta \ge \frac{1}{\sqrt{2}}\]
done
clear
View Answer play_arrow
A parallel beam of fast moving electrons is incident normally on a narrow slit. A fluorescent screen is placed at a large distance from the slit. If the speed of the electrons is increased, which of the following statements is correct?
A)
Diffraction pattern is not observed on the screen in the case of electrons
done
clear
B)
The angular width of the central maximum of the diffraction pattern will increase
done
clear
C)
The angular width of the central maximum will decrease
done
clear
D)
The angular width of the central maximum will be unaffected
done
clear
View Answer play_arrow
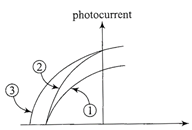
The figure shows a plot of photo current versus anode potential for a photosensitive surface for three different radiations. Which one of the following is a correct statement?
Retarding potential Anode potential
A)
Curves a and b represent incident radiations of different frequencies and different intensities
done
clear
B)
Curves a and b represent incident radiations of the same frequency but of different intensities
done
clear
C)
Curves b and c represent incident radiations of different frequencies and different intensities
done
clear
D)
Curves b and c represent incident radiations of the same frequency having the same intensity
done
clear
View Answer play_arrow
Electron in hydrogen atom first jumps from third excited state to second excited state and then from second excited state to first excited state. The ratio of wavelengths\[{{\lambda }_{1}}:{{\lambda }_{2}}\]emited in two cases is
A)
7/5
done
clear
B)
27/20
done
clear
C)
27/5
done
clear
D)
20/7
done
clear
View Answer play_arrow
During mean life of a radioactive element, the fraction that disintegrates is
A)
e
done
clear
B)
\[\frac{1}{e}\]
done
clear
C)
\[\frac{e-1}{e}\]
done
clear
D)
\[\frac{e}{e-1}\]
done
clear
View Answer play_arrow
For the given circuit of p-n junction diode, which of the following statement is correct:
A)
In forward biasing the voltage across R is V
done
clear
B)
In forward biasing the voltage across R is 2V
done
clear
C)
In reverse biasing the voltage across R is V
done
clear
D)
In reverse biasing the voltage across R is 2V
done
clear
View Answer play_arrow
The maximum number of molecules is present in:
A)
15 L of \[{{H}_{2}}\]gas at STP
done
clear
B)
5 L of \[{{N}_{2}}\]gas at STP
done
clear
C)
0.5 g of \[{{H}_{2}}\]gas
done
clear
D)
10 g of \[{{O}_{2}}\]gas
done
clear
View Answer play_arrow
If uncertainty in position and momentum are equal, then uncertainty in velocity is
A)
\[\sqrt{\frac{h}{2\pi }}\]
done
clear
B)
\[\frac{l}{m}\sqrt{\frac{h}{\pi }}\]
done
clear
C)
\[\sqrt{\frac{h}{\pi }}\]
done
clear
D)
\[\frac{1}{2m}\sqrt{\frac{h}{\pi }}\]
done
clear
View Answer play_arrow
Among the following series of transition metals ions, the one where all metal ions have \[3{{d}^{2}}\] electronic configuration is:
A)
\[T{{i}^{3+}},{{V}^{2+}},C{{r}^{3+}},M{{n}^{4+}}\]
done
clear
B)
\[T{{i}^{+}},{{V}^{4+}},C{{r}^{6+}},M{{n}^{7a+}}\]
done
clear
C)
\[T{{i}^{4+}},{{V}^{3+}},C{{r}^{2+}},M{{n}^{3+}}\]
done
clear
D)
\[T{{i}^{2+}},{{V}^{3+}},C{{r}^{4+}},M{{n}^{5+}}\]
done
clear
View Answer play_arrow
Which of the following two are isostructural?
A)
\[Xe{{F}_{2}},IF_{2}^{-}\]
done
clear
B)
\[N{{H}_{3,}}B{{F}_{3}}\]
done
clear
C)
\[CO_{3}^{2-},SO_{3}^{2-}\]
done
clear
D)
\[PC{{l}_{5}},IC{{l}_{5}}\]
done
clear
View Answer play_arrow
Vander Waals' real gas acts as an ideal gas at which conditions?
A)
High temperature, low pressure
done
clear
B)
Low temperature, high pressure
done
clear
C)
High temperature, high pressure
done
clear
D)
Low temperature, low pressure
done
clear
View Answer play_arrow
Change in enthalpy for reaction \[2{{H}_{2}}{{O}_{2}}(l)\xrightarrow{{}}2{{H}_{2}}O(l)+{{O}_{2}}(g)\] if heat of formation of \[{{H}_{2}}{{O}_{2}}(l)\]and \[{{H}_{2}}O(l)\] are -188 and -286 kJ/mol respectively is
A)
-196 kJ/mol
done
clear
B)
+196 kJ/mol
done
clear
C)
+948 kJ/mol
done
clear
D)
-948 kJ/mol
done
clear
View Answer play_arrow
For the reaction, \[C{{H}_{4}}(g)+2{{O}_{2}}(g)\rightleftharpoons C{{O}_{2}}(g)+2{{H}_{2}}O(l),\] \[{{\Delta }_{r}}H=-170.8\,kJ\,mo{{l}^{-1}}\] Which of the following statements is not true?
A)
Addition of\[C{{H}_{4}}\left( g \right)\]or\[{{O}_{2}}(g)\]at equilibrium will cause a shift to the right
done
clear
B)
The reaction is exothermic
done
clear
C)
At equilibrium, the concentrations of\[C{{O}_{2}}(g)\]and \[{{H}_{2}}O(l)\]are not equal
done
clear
D)
The equilibrium constant for the reaction is given by: \[{{K}_{p}}=\frac{[C{{O}_{2}}]}{[C{{H}_{4}}][{{O}_{2}}]}\]
done
clear
View Answer play_arrow
Ionisation constant of \[C{{H}_{3}}COOH\]is \[1.7\times {{10}^{-5}}\]and concentration of \[{{H}^{+}}\]ions is \[3.4\times {{10}^{-4}}.\] Then, find out initial concentration of \[C{{H}_{3}}COOH\]molecules.
A)
\[3.4\times {{10}^{-4}}\]
done
clear
B)
\[3.4\times {{10}^{-1}}\]
done
clear
C)
\[6.8\times {{10}^{-4}}\]
done
clear
D)
\[6.8\times {{10}^{-3}}\]
done
clear
View Answer play_arrow
The oxidation states of sulphur in the anions \[SO_{3}^{2-},{{S}_{2}}O_{4}^{2-}\] and \[{{S}_{2}}O_{6}^{2-}\]follow the order
A)
\[{{S}_{2}}O_{4}^{2-}<SO_{3}^{2-}<{{S}_{2}}O_{6}^{2-}\]
done
clear
B)
\[SO_{3}^{2-}<{{S}_{2}}O_{4}^{2-}<{{S}_{2}}O_{6}^{2-}\]
done
clear
C)
\[{{S}_{2}}O_{4}^{2-}<{{S}_{2}}O_{6}^{2-}<SO_{3}^{2-}\]
done
clear
D)
\[{{S}_{2}}O_{6}^{2-}<{{S}_{2}}O_{4}^{2-}<SO_{3}^{2-}\]
done
clear
View Answer play_arrow
The dielectric constant of \[{{H}_{2}}O\]is 80. The electrostatic force of attraction between \[N{{a}^{+}}\] and Cl will be
A)
reduced to 1/80 in water than in air
done
clear
B)
reduced to 1/40 in water than in air
done
clear
C)
will be increased to 80 in water than in air
done
clear
D)
will remain unchanged
done
clear
View Answer play_arrow
Which one of the alkali metals forms only the normal oxide, \[{{M}_{2}}O,\]on heating in air?
A)
Li
done
clear
B)
Na
done
clear
C)
Rb
done
clear
D)
K
done
clear
View Answer play_arrow
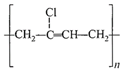
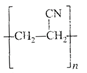
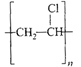
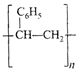
The straight chain polymer is formed by
A)
hydrolysis of\[{{(C{{H}_{3}})}_{2}}SiC{{l}_{2}}\]followed by condensation
done
clear
B)
hydrolysis of\[{{(C{{H}_{3}})}_{3}}SiCl\]followed by condensation
done
clear
C)
hydrolysis of \[C{{H}_{3}}SiC{{l}_{3}}\]followed by condensation polymerisation
done
clear
D)
hydrolysis of \[{{(C{{H}_{3}})}_{4}}\] Si by addition polymerization
done
clear
View Answer play_arrow
Lassaigne's test for the detection of nitrogen will fail in case of:
A)
\[N{{H}_{2}}CON{{H}_{2}}\]
done
clear
B)
\[{{H}_{2}}NCONHN{{H}_{2}}.HCl\]
done
clear
C)
\[{{H}_{2}}N.N{{H}_{2}}.2HCl\]
done
clear
D)
\[{{C}_{6}}{{H}_{5}}NHN{{H}_{2}}.2HCl\]
done
clear
View Answer play_arrow
Which one of the following is the most reactive towards electrophilic attack?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
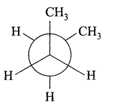
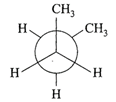
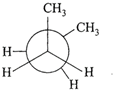
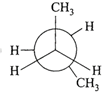
In the following the most stable conformation of \[n-\]butane is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The compound \[C{{H}_{3}}-\overset{C{{H}_{3}}}{\mathop{\overset{|}{\mathop{C}}\,}}\,=CH-C{{H}_{3}}\] on reaction with \[NaI{{O}_{4}}\]in the presence of \[KMn{{O}_{4}}\]gives
A)
\[C{{H}_{3}}CHO+C{{O}_{2}}\]
done
clear
B)
\[C{{H}_{3}}COC{{H}_{3}}\]
done
clear
C)
\[C{{H}_{3}}COC{{H}_{3}}+C{{H}_{3}}COOH\]
done
clear
D)
\[C{{H}_{3}}COC{{H}_{3}}+C{{H}_{3}}CHO\]
done
clear
View Answer play_arrow
Given
The enthalpy of the hydrogenation of these compounds will be in the order as:
A)
\[III > II > I\]
done
clear
B)
\[II>III>I\]
done
clear
C)
\[II>I>III\]
done
clear
D)
\[I>II>III\]
done
clear
View Answer play_arrow
\[{{\,}_{92}}{{U}^{238}}\]nucleus absorbs a neutron and disintegrates into \[{{\,}_{54}}{{X}^{139}},{{\,}_{38}}S{{r}^{94}}\]and X. So what will be the product X?
A)
3-neutrons
done
clear
B)
2-neutrons
done
clear
C)
or-particle
done
clear
D)
\[\beta -\]particle
done
clear
View Answer play_arrow
CsBr crystallises in a body centred cubic lattice. The nit cell length is 436.6 pm. Given that the atomic mass of Cs =133 and that of Br = 80 amu and Avogadro number being \[6.02\times {{10}^{23}}\,mo{{l}^{1}},\] the density of CsBr is
A)
\[8.25\text{ }g/c{{m}^{3}}\]
done
clear
B)
\[4.25\text{ }g/c{{m}^{3}}\]
done
clear
C)
\[~42.5\text{ }g/c{{m}^{3}}\]
done
clear
D)
\[0.425\text{ }g/c{{m}^{3}}\]
done
clear
View Answer play_arrow
A 0.5 molal aqueous solution of a weak acid (HX) is 20 per cent ionized. The lowering in freezing point of this solution is (\[{{K}_{f}}=1.86\,K/m\]for water)
A)
1.12 K
done
clear
B)
-0.56
done
clear
C)
0.56 K
done
clear
D)
-1.12K
done
clear
View Answer play_arrow
An electric current is passed through silver nitrate solution using silver electrodes. 10.79 g of silver was found to be deposited on the cathode if the same amount of electricity is passed through copper sulphate solution using copper electrodes, the weight of copper deposited on the cathode is
A)
6.4 g
done
clear
B)
2.3 g
done
clear
C)
12.8g
done
clear
D)
3.2g
done
clear
View Answer play_arrow
The electrode potentials for
\[C{{u}^{2+}}(aq)+{{e}^{-}}\xrightarrow{{}}C{{u}^{+}}(aq)\] and \[C{{u}^{+}}(aq)+{{e}^{-}}\xrightarrow{{}}Cu(s)\]
are +0.15 V and + 0.50 V respectively. The value of \[E_{C{{u}^{2+}}/Cu}^{o}\]will be
A)
0.150V
done
clear
B)
0.325V
done
clear
C)
0.500V
done
clear
D)
0.650V
done
clear
View Answer play_arrow
Aqueous solution of which of the following compounds is the best conductor of electric current?
A)
Ammonia, \[N{{H}_{3}}\]
done
clear
B)
Fuctose,\[{{C}_{6}}{{H}_{12}}{{O}_{6}}\]
done
clear
C)
Acetic acid,\[{{C}_{2}}{{H}_{4}}{{O}_{2}}\]
done
clear
D)
Hydrochloric acid, HCl
done
clear
View Answer play_arrow
For the reaction; \[2{{N}_{2}}{{O}_{5}}\to 4N{{O}_{2}}+{{O}_{2}},\]rate and rate constant are \[1.02\times {{10}^{-4}}M{{\sec }^{-1}}\]and \[3.4\times {{10}^{-5}}{{\sec }^{-1}}M\]respectively, then concentration of \[{{N}_{2}}{{O}_{5}},\]at that time will be
A)
1.732 M
done
clear
B)
3 M
done
clear
C)
\[1.02\times {{10}^{-4}}\,M\]
done
clear
D)
\[3.5\times {{10}^{5}}M\]
done
clear
View Answer play_arrow
The equality relationship between \[\frac{d[N{{H}_{3}}]}{dt}\]and \[-\frac{d[{{H}_{2}}]}{dt}\]is
A)
\[-\frac{d[N{{H}_{3}}]}{dt}=-\frac{3}{2}\left[ \frac{d[{{H}_{2}}]}{dt} \right]\]
done
clear
B)
\[\frac{d\left[ N{{H}_{3}} \right]}{dt}=-\frac{d\left[ {{H}_{2}} \right]}{dt}\]
done
clear
C)
\[\frac{d\left[ N{{H}_{3}} \right]}{dt}=-\frac{1}{3}\frac{d\left[ {{H}_{2}} \right]}{dt}\]
done
clear
D)
\[+\frac{d\left[ N{{H}_{3}} \right]}{dt}=-\frac{2}{3}\frac{d\left[ {{H}_{2}} \right]}{dt}\]
done
clear
View Answer play_arrow
For the reaction, \[{{N}_{2}}+3{{H}_{2}}\to 2N{{H}_{3}},\]if \[\frac{d\left[ N{{H}_{3}} \right]}{dt}\] \[=2\times {{10}^{-4}}mol\,{{L}^{-1}}{{s}^{-1}},\]the value of \[\frac{-d[{{H}_{2}}]}{dt}\] would be:
A)
\[1\times {{10}^{-4}}mol\,{{L}^{-1}}{{s}^{-1}}\]
done
clear
B)
\[3\times {{10}^{-4}}mol\,{{L}^{-1}}{{s}^{-1}}\]
done
clear
C)
\[4\times {{10}^{-4}}mol\,{{L}^{-1}}{{s}^{-1}}\]
done
clear
D)
\[6\times {{10}^{-4}}mol\,{{L}^{-1}}{{s}^{-1}}\]
done
clear
View Answer play_arrow
Activation energy \[({{E}_{a}})\] and rate constants (\[{{k}_{1}}\] and\[{{k}_{2}}\]) of a chemical reaction at two different temperatures (\[{{T}_{1}}\] and\[{{T}_{2}}\]) are related by
A)
In\[\frac{{{k}_{2}}}{{{k}_{1}}}=-\frac{{{E}_{a}}}{R}\left( \frac{1}{{{T}_{1}}}-\frac{1}{{{T}_{2}}} \right)\]
done
clear
B)
In\[\frac{{{k}_{2}}}{{{k}_{1}}}=-\frac{{{E}_{a}}}{R}\left( \frac{1}{{{T}_{2}}}-\frac{1}{{{T}_{1}}} \right)\]
done
clear
C)
In\[\frac{{{k}_{2}}}{{{k}_{1}}}=-\frac{{{E}_{a}}}{R}\left( \frac{1}{{{T}_{2}}}+\frac{1}{{{T}_{1}}} \right)\]
done
clear
D)
In \[\frac{{{k}_{2}}}{{{k}_{1}}}=\frac{{{E}_{a}}}{R}\left( \frac{1}{{{T}_{1}}}-\frac{1}{{{T}_{2}}} \right)\]
done
clear
View Answer play_arrow
In Freundlich adsorption isotherm, the value of \[1/n\] is
A)
1 in case of physical adsorption
done
clear
B)
1 in case of chemisorption
done
clear
C)
between 0 and 1 in all cases
done
clear
D)
between 2 and 4 in all cases
done
clear
View Answer play_arrow
Sulphide ores of metals are usually concentrated by froth floatation process. Which one of the following sulphide ores offers an exception and is concentrated by leaching?
A)
Argentite
done
clear
B)
Copper pyrite
done
clear
C)
Sphalerite
done
clear
D)
Galena
done
clear
View Answer play_arrow
Each of the following is true for white and red phosphorus except that they
A)
are both soluble in \[C{{S}_{2}}\]
done
clear
B)
can be oxidised by heating in air
done
clear
C)
consists of same kind of atoms
done
clear
D)
can be converted into one another
done
clear
View Answer play_arrow
Bleaching action of \[S{{O}_{2}}\]is due to
A)
Reduction
done
clear
B)
Oxidation
done
clear
C)
Hydrolysis
done
clear
D)
Its acidic nature
done
clear
View Answer play_arrow
Which of the statements given below is incorrect?
A)
ONF is isoelectronic with \[{{O}_{2}}{{N}^{-}}\]
done
clear
B)
\[O{{F}_{2}}\]is an oxide of fluorine
done
clear
C)
\[C{{l}_{2}}{{O}_{7}}\]is an. anhydride of perchloric acid
done
clear
D)
\[{{O}_{3}}\] molecule is bent
done
clear
View Answer play_arrow
\[B{{r}^{-}}\]is converted into \[B{{r}_{2}}\]by using
A)
\[C{{l}_{2}}\]
done
clear
B)
Conc. HCl
done
clear
C)
HBr
done
clear
D)
\[{{H}_{2}}S\]
done
clear
View Answer play_arrow
Acid strength of oxy acids of chlorine follows the order:
A)
\[HClO<HCl{{O}_{2}}<HCl{{O}_{3}}<HCl{{O}_{4}}\]
done
clear
B)
\[HCl{{O}_{4}}<HCl{{O}_{3}}<HCl{{O}_{2}}<HClO\]
done
clear
C)
\[HCl{{O}_{4}}<HCl{{O}_{4}}<HClO<HCl{{O}_{2}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
Among the fluorides below, the one which does not' exist is
A)
\[Xe{{F}_{4}}\]
done
clear
B)
\[~He{{F}_{4}}\]
done
clear
C)
\[~S{{F}_{4}}\]
done
clear
D)
\[~C{{F}_{4}}\]
done
clear
View Answer play_arrow
The temperature of blast furnace to produce iron from its ore \[F{{e}_{2}}{{O}_{3}}\]varies from \[\text{500 }\!\!{}^\circ\!\!\text{ C}\]at the top of the furnace to about \[\text{1900 }\!\!{}^\circ\!\!\text{ C}\]at the bottom of the furnace. The reaction between the ore \[F{{e}_{2}}{{O}_{3}}\]and CO at the lowest temperature\[(-500{{\,}^{o}}C)\]is
A)
\[3F{{e}_{2}}{{O}_{3}}+CO\to 2F{{e}_{3}}{{O}_{4}}+C{{O}_{2}}\]
done
clear
B)
\[F{{e}_{2}}{{O}_{3}}+CO\to 2FeO+C{{O}_{2}}\]
done
clear
C)
\[F{{e}_{2}}{{O}_{3}}+3CO\to 2Fe+3C{{O}_{2}}\]
done
clear
D)
\[F{{e}_{2}}{{O}_{3}}+CO\to 2Fe+C{{O}_{2}}+\frac{1}{2}{{O}_{2}}\]
done
clear
View Answer play_arrow
The oxidation number of Cr in \[[Cr{{(N{{H}_{3}})}_{6}}]C{{l}_{3}}\]is
A)
8
done
clear
B)
6
done
clear
C)
4
done
clear
D)
3
done
clear
View Answer play_arrow
Phenol reacts with \[CHC{{l}_{3}}\]and NaOH (at 340 K) to give
A)
o-chlorophenol
done
clear
B)
salicylaldehyde
done
clear
C)
benzaldehyde
done
clear
D)
chlorobenzene
done
clear
View Answer play_arrow
Which of the following reagents convert the propeneto 1-propanol
A)
\[{{H}_{2}}O,{{H}_{2}}S{{O}_{4}}\]
done
clear
B)
Aqueous KOH
done
clear
C)
\[MgS{{O}_{4}},NaB{{H}_{4}}/{{H}_{2}}O\]
done
clear
D)
\[{{B}_{2}}{{H}_{6}},{{H}_{2}}{{O}_{2}},O{{H}^{-}}\]
done
clear
View Answer play_arrow
In a set reactions acid yielded a product D \[C{{H}_{3}}COOH\xrightarrow{SOC{{l}_{2}}}A\] \[\xrightarrow[Anhr.\,AlC{{l}_{3}}]{Benzene}B\xrightarrow{HCN}C\xrightarrow{HOH}D.\]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
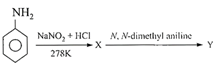
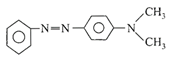
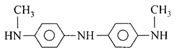
Aniline in a set of the following reactions yielded a coloured compound Y:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The pair in which both species have iron is
A)
nitrogenase, cytochromes
done
clear
B)
carboxypeptidase, haemoglobin
done
clear
C)
haemocyanin, nitrogenase
done
clear
D)
haemoglobin, cytochromes
done
clear
View Answer play_arrow
Which of the following represents neoprene polymer?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Glycerol is not used in
A)
explosive
done
clear
B)
cosmetics
done
clear
C)
soaps
done
clear
D)
matches
done
clear
View Answer play_arrow
0.45 g of an acid (mol wt. = 90) required 20 ml of 0.5 N KOH for complete neutralization. Basicity of acid is
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
Angiosperms have dominated the land flora primarily because of their
A)
Property of producing large number of seeds
done
clear
B)
Nature of self-pollination
done
clear
C)
Domestication by man
done
clear
D)
Power of adaptability in diverse habitat
done
clear
View Answer play_arrow
Biological organization starts with:
A)
Atomic level
done
clear
B)
Submicroscopic molecular level
done
clear
C)
Cellular level
done
clear
D)
Organismic level
done
clear
View Answer play_arrow
The living organisms can be unexceptionally distinguished from the non-living things on the basis of their ability for:
A)
Growth and movement
done
clear
B)
Responsiveness to touch
done
clear
C)
Interaction with the environment and progressive evolution
done
clear
D)
Reproduction
done
clear
View Answer play_arrow
Which one of the following animals is correctly matched with its particular named taxonomic category?
A)
Housefly - Musca, an order
done
clear
B)
Tiger - tigris, the species
done
clear
C)
Cuttlefish - Mollusca, a class
done
clear
D)
Humans - Primata, the family
done
clear
View Answer play_arrow
Plant decomposers are:
A)
Monera and fungi
done
clear
B)
Fungi and plants
done
clear
C)
Protista and Animalia
done
clear
D)
Anibalia and Mogna
done
clear
View Answer play_arrow
The chief advantage of encystment to an amoeba is:
A)
The ability to survive during adverse physical conditions
done
clear
B)
The ability to live for some time without ingesting food
done
clear
C)
Protection from parasites and predators
done
clear
D)
The chance to get rid of accumulated waste products
done
clear
View Answer play_arrow
Which antibiotic inhibits interaction between tRNA and mRNA during bacterial protein synthesis?
A)
Erythromycin
done
clear
B)
Neomycin
done
clear
C)
Streptomycin
done
clear
D)
Tetracycline
done
clear
View Answer play_arrow
Ringworm in humans is caused by:
A)
Bacteria
done
clear
B)
Fungi
done
clear
C)
Nematodes
done
clear
D)
Viruses
done
clear
View Answer play_arrow
Which one of the following matches is correct?
A)
Agaricus Parasitic Fungus Basidiomy-cetes
done
clear
B)
Phytophthora Aseptate mycelium Basidiomy-cetes
done
clear
C)
Alternaria Sexual reproduction absent Deuteromy-cetes
done
clear
D)
Mucor Reproduction by Conjugation Ascomyce-tes
done
clear
View Answer play_arrow
Which of the following plants produces seeds but not flowers?
A)
Maize
done
clear
B)
Mint
done
clear
C)
Peepal
done
clear
D)
Pinus
done
clear
View Answer play_arrow
Which one of the following is heterosporous?
A)
Adiantum
done
clear
B)
Equisetum
done
clear
C)
Dryopteris
done
clear
D)
Salvinia
done
clear
View Answer play_arrow
Read the following five statements (A-E) and answer as asked next to them.
A. In Equisetum the female gametophyte is retained on the parent sporophyte B. In Ginkgo male gametophyte is not independent C. The sporophyte in Riccia is more developed than that in Polytrichum D. Sexual reproduction in Volvox is isogamous E. The spores of slime molds lack cell walls
How many of the above statements are correct?
A)
Three
done
clear
B)
Four
done
clear
C)
One
done
clear
D)
Two
done
clear
View Answer play_arrow
In which of the following haemocyanin pigment is found:
A)
Annelida
done
clear
B)
Echinodermata
done
clear
C)
Insecta
done
clear
D)
Lower chordate
done
clear
View Answer play_arrow
Which of the following pairs are correctly matched?
Animals Morphological features A. Crocodile 4-chambered heart B. Sea Urchin Parapodia C. Obelia Metagenesis D. Lemur Thecodont
A)
Only A and B
done
clear
B)
A, C and D
done
clear
C)
B, C and D
done
clear
D)
Only A and D
done
clear
View Answer play_arrow
One example of animals having a single opening to the outside that serves both as mouth and as anus is:
A)
Octopus
done
clear
B)
Asterias
done
clear
C)
Ascidia
done
clear
D)
Fasciola
done
clear
View Answer play_arrow
In which one of the following the genus name, its two characters and its class/phylum are correctly matched?
A)
Genus name Two characters Class/Phylum Awelia (a) Cnidoblasts Coelenterata (b) Organ level of organization
done
clear
B)
Genus name Two characters Class/Phylum Ascaris (a) Body segmented Annelida (b) Males and females distinct
done
clear
C)
Genus name Two characters Class/Phylum Salamandra (a) A tympanum represents ear Amphibia (b) Fertilization is external
done
clear
D)
Genus name Two characters Class/Phylum Pteropus (a) Skin possesses hair Mammalia (b) Oviparous
done
clear
View Answer play_arrow
Which of the following represents the correct combination without any exception?
A)
Characteristics Class Body covered with feathers; skin moist and glandular; fore-limbs form wings; lungs with air sacs Aves
done
clear
B)
Characteristics Class Mammary gland; hair on body; pinnae; two pairs of limbs Mammalia
done
clear
C)
Characteristics Class Mouth ventral; gills without operculum; skin with placoid scales; persistent notochord Chondrichthyes
done
clear
D)
Characteristics Class Sucking and circular mouth; jaws absent, integument without scales; paired appendages Cyclostomata
done
clear
View Answer play_arrow
Roots of which plant contains a red pigment which have affinity for oxygen:
A)
Carrot
done
clear
B)
Soyabean
done
clear
C)
Mustard
done
clear
D)
Radish
done
clear
View Answer play_arrow
Ovary is half-inferior in the flowers of
A)
Guava
done
clear
B)
Plum
done
clear
C)
Brinjal
done
clear
D)
Cucumber
done
clear
View Answer play_arrow
Placentation in tomato and lemon is
A)
Marginal
done
clear
B)
Axile
done
clear
C)
Parietal
done
clear
D)
Free central
done
clear
View Answer play_arrow
Flowers are unisexual in:
A)
Onion
done
clear
B)
Pea
done
clear
C)
Cucumber
done
clear
D)
China rose
done
clear
View Answer play_arrow
Four radial VB are found in:
A)
Dicot root
done
clear
B)
Monocot root
done
clear
C)
Dicot stem
done
clear
D)
Monocot stem
done
clear
View Answer play_arrow
Vascular tissues in flowering plants develop from:
A)
periblem
done
clear
B)
dermatogens
done
clear
C)
phellogen
done
clear
D)
plerome
done
clear
View Answer play_arrow
The chief water conducting elements of xylem in gymnosperms are:
A)
Vessels
done
clear
B)
Fibres
done
clear
C)
Transfusion tissue
done
clear
D)
Tracheids
done
clear
View Answer play_arrow
Interfascicular cambium develops from the cells of:
A)
Medullary rays
done
clear
B)
Xylem parenchyma
done
clear
C)
Endodermis
done
clear
D)
Pericycle
done
clear
View Answer play_arrow
A column of water within xylem vessels of tall trees does not break under its weight because of:
A)
Positive root pressure
done
clear
B)
Dissolved sugars in water
done
clear
C)
Tensile strength of water
done
clear
D)
Lignification of xylem vessels
done
clear
View Answer play_arrow
ATP ase enzyme needed for muscle contraction is located in:
A)
Troponin
done
clear
B)
Myosin
done
clear
C)
Actin
done
clear
D)
Actinin
done
clear
View Answer play_arrow
The guts of cow and buffalo possess:
A)
Cyanobacteria
done
clear
B)
Fucus spp.
done
clear
C)
Chlorella spp.
done
clear
D)
Methanogens
done
clear
View Answer play_arrow
Which of the following occurs more than one and less than five in a chromosome?
A)
Chromatid
done
clear
B)
Chromomere
done
clear
C)
Centromere
done
clear
D)
Telomere
done
clear
View Answer play_arrow
Keeping in view the "fluid mosaic model" for the structure of cell membrane, which one of the following statements is correct with respect to the movement of lipids and proteins from one lipid monolayer to the other (described as flip- flop movement)?
A)
While proteins can flip-flop, lipids cannot
done
clear
B)
neither lipids nor proteins can flip-flop
done
clear
C)
Both lipids and proteins can flip-flop
done
clear
D)
While lipids can rarely flip-flop proteins cannot
done
clear
View Answer play_arrow
Peptide synthesis inside a cell takes place in:
A)
Ribosomes
done
clear
B)
Chloroplast
done
clear
C)
Mitochondria
done
clear
D)
Chromoplast
done
clear
View Answer play_arrow
Match the following and select the correct answer:
a. Centriole (i) Infoldings in mitochondria b. Chlorophyll (ii) Thylakoids c. Cristae (iii) Nucleic acids d. Ribozymes (iv) Basal body cilia or flagella
A)
A B C D (iv) (ii) (i) (iii)
done
clear
B)
A B C D (i) (ii) (iv) (iii)
done
clear
C)
A B C D (i) (iii) (ii) (iv)
done
clear
D)
A B C D (iv) (iii) (i) (ii)
done
clear
View Answer play_arrow
Balbiani rings are sites of:
A)
RNA and protein synthesis
done
clear
B)
Lipid synthesis
done
clear
C)
Nucleotide synthesis
done
clear
D)
Polysaccharide synthesis
done
clear
View Answer play_arrow
Collagen is:
A)
Fibrous protein
done
clear
B)
Globular protein
done
clear
C)
Lipid
done
clear
D)
Carbohydrate
done
clear
View Answer play_arrow
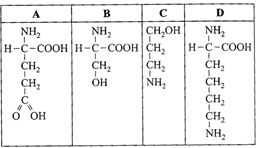
Which one out of A?D given below correctly represents the structural formula of the basic amino acid?
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
View Answer play_arrow
Which one of the following precedes reformation of the nuclear envelope during M phase of the cell cycle:
A)
Transcription from chromosomes and reassembly of the nuclear lamina
done
clear
B)
Formation of the contractile ring and formation of the phragmoplast
done
clear
C)
Formation of the contractile ring and transcription from chromosomes
done
clear
D)
Decondensation from chromosomes and reassembly of the nuclear lamina
done
clear
View Answer play_arrow
Guard cells help in:
A)
Fighting against infection
done
clear
B)
Protection against grazing
done
clear
C)
Transpiration
done
clear
D)
Guttation
done
clear
View Answer play_arrow
Enzyme first used for nitrogen fixation:
A)
Nitrogenase
done
clear
B)
Nitroreductase
done
clear
C)
Transferase
done
clear
D)
Transaminase
done
clear
View Answer play_arrow
Best defined function of manganese in green plants is:
A)
Nitrogen fixation
done
clear
B)
Water absorption
done
clear
C)
Photolysis of water
done
clear
D)
Calvin cycle
done
clear
View Answer play_arrow
Roots play insignificant role in the absorption of water in:
A)
Wheat
done
clear
B)
Sunflower
done
clear
C)
Pistia
done
clear
D)
Pea
done
clear
View Answer play_arrow
In photosynthesis energy from light reaction to dark reaction is transferred in the form of:
A)
ADP
done
clear
B)
ATP
done
clear
C)
RuDP
done
clear
D)
Chlorophyll
done
clear
View Answer play_arrow
Photoperiodism was first characterised in:
A)
Tobacco
done
clear
B)
Potato
done
clear
C)
Tomato
done
clear
D)
Cotton
done
clear
View Answer play_arrow
In photosynthesis, the light-independent reactions take place at:
A)
Stromal matrix
done
clear
B)
Thylakoid lumen
done
clear
C)
Photosystem I
done
clear
D)
Photosystem II
done
clear
View Answer play_arrow
Chemiosmotic theory of ATP synthesis in the chloroplasts and mitochondria is based on:
A)
Membrane potential
done
clear
B)
Accumulation of K ions
done
clear
C)
Proton gradient
done
clear
D)
Accumulation of Na ions
done
clear
View Answer play_arrow
In which one of the following processes \[C{{O}_{2}}\]is not released?
A)
Aerobic respiration in plants
done
clear
B)
Aerobic respiration in animals
done
clear
C)
Alcoholic fermentation
done
clear
D)
Lactate fermentation
done
clear
View Answer play_arrow
Which breaks dormancy of potato tuber?
A)
Gibberellin
done
clear
B)
IAA
done
clear
C)
ABA
done
clear
D)
Zeatin
done
clear
View Answer play_arrow
Typical growth curve in plants is:
A)
Parabolic
done
clear
B)
Sigmoid
done
clear
C)
Linear
done
clear
D)
Stair-steps shaped
done
clear
View Answer play_arrow
During prolonged fasting, in what sequence are the following organic compounds used up by the body:
A)
First carbohydrates, next fats and lastly proteins
done
clear
B)
First fats, next carbohydrates and lastly proteins
done
clear
C)
First carbohydrates, next proteins and lastly lipids
done
clear
D)
First proteins, next lipids and lastly carbohydrates
done
clear
View Answer play_arrow
Carrier ions like \[N{{a}^{+}}\]facilitate the absorption of substances like
A)
amino acids and glucose
done
clear
B)
glucose and fatty acids
done
clear
C)
fatty acids and glycerol
done
clear
D)
fructose and some amino acids
done
clear
View Answer play_arrow
Fructose is absorbed into the blood through mucosa cells of intestine by the process called
A)
active transport
done
clear
B)
facilitated transport
done
clear
C)
simple diffusion
done
clear
D)
co-transport mechanism
done
clear
View Answer play_arrow
Which one of the following mammalian cells is not capable of metabolizing glucose to carbon dioxide aerobically?
A)
Red blood cells
done
clear
B)
White blood cells
done
clear
C)
Unstriated muscle cells
done
clear
D)
Liver cells
done
clear
View Answer play_arrow
Examination of blood of a person suspected of having anemia shows large, immature, nucleated erythrocytes without haemoglobin, supple- menting his diet with which of the following is likely to alleviate his symptoms?
A)
Thiamine
done
clear
B)
Folic acid and cobalamin
done
clear
C)
Riboflavin
done
clear
D)
Iron compounds
done
clear
View Answer play_arrow
Figure shows schematic plant of blood circulation in humans with labels A to D. Identify the label and give its function/s.
A)
A-Pulmonary vein-takes impure blood from body parts,\[P{{O}_{2}}=60\,mm\,Hg\]
done
clear
B)
B-Pulmonary artery - takes blood from heart to lungs,\[P{{O}_{2}}=90\,mm\,Hg\]
done
clear
C)
C-Vena Cava - takes blood from body parts to right auricle,\[PC{{O}_{2}}=45\,mm\,Hg\]
done
clear
D)
D-Dorsal aorta - takes blood from heart to body parts, \[P{{O}_{2}}=95\,mm\,Hg\]
done
clear
View Answer play_arrow
Doctors use stethoscope to hear the sound; produced during each cardiac cycle. The second sound is heard when:
A)
AV node receives signal from SA node
done
clear
B)
AV valves open up
done
clear
C)
Ventricular walls vibrate due to gushing of blood from atria
done
clear
D)
Semilunar valves close down after the blood flows into vessels from ventricles
done
clear
View Answer play_arrow
When a fresh water protozoan possessing a contractile vacuole is placed in a glass containing marine water, the vacuole will:
A)
Disappear
done
clear
B)
Increase in size
done
clear
C)
Decrease in size
done
clear
D)
Increase in number
done
clear
View Answer play_arrow
Which one of the following correctly explains the function of a specific part of a human nephron?
A)
Afferent arteriole: carries the blood away from the glomerulus towards renal vein
done
clear
B)
Podocytes: Create minute spaces (slit pores) for the filtration of blood into the Bowman's capsule
done
clear
C)
Henle's loop: most reabsorption of the major substances from the glomerular filtrate
done
clear
D)
Distal convoluted tubule: re-absorption of\[{{K}^{+}}\] ions into the surrounding blood capillaries
done
clear
View Answer play_arrow
Removal of proximal convoluted tubule from the nephron will result in:
A)
No urine formation
done
clear
B)
More diluted urine
done
clear
C)
More concentrated urine
done
clear
D)
No change in quality and quantity of urine
done
clear
View Answer play_arrow
What used to be described as Nissi granules in a nerve cell are now identified as?
A)
Cell metabolites
done
clear
B)
Fat granules
done
clear
C)
Ribosomes
done
clear
D)
Mitochondria
done
clear
View Answer play_arrow
A diagram showing axon terminal and synapse is given. Identify correctly at least two of A-D.
A)
A-Receptor; C-Synaptic vesicles
done
clear
B)
B-Synaptic connection;\[D-{{K}^{+}}\]
done
clear
C)
A-Neurotransmitter; B-Synaptic cleft
done
clear
D)
C-Neurotransmitter; \[D-C{{a}^{++}}\]
done
clear
View Answer play_arrow
In mammalian eye, the 'fovea' is the center of the visual field, where:
A)
more rods than cones are found
done
clear
B)
high density of cones occur, but has no rods
done
clear
C)
the optic nerve leaves the eye
done
clear
D)
only rods are present
done
clear
View Answer play_arrow
When both the ovaries of rat are removed, then which hormone is decreased in blood?
A)
Oxytocin
done
clear
B)
Prolactin
done
clear
C)
Estrogen
done
clear
D)
Gonadotrophic releasing factor
done
clear
View Answer play_arrow
Which one of the following pair's correctly matches a hormone with a disease resulting from its deficiency:
A)
Insulin - Diabetes insipidus
done
clear
B)
Thyroxine - Tetany
done
clear
C)
Parathyroid hormone - Diabetes mellitus
done
clear
D)
Luteinizing hormone - Failure of ovulation
done
clear
View Answer play_arrow
Low \[C{{a}^{++}}\] in the body fluid may be the cause of:
A)
Tetany
done
clear
B)
Anaemia
done
clear
C)
Angina pectoris
done
clear
D)
Gout
done
clear
View Answer play_arrow
Fight-or-flight reaction causes activation of:
A)
the parathyroid glands, leading to increased metabolic rate.
done
clear
B)
the kidney, leading to suppression of renin angiotensin-aldosterone pathway.
done
clear
C)
the adrenal medulla, leading to increased secretion of epinephrine and norepinephrine
done
clear
D)
the pancreas leading to a reduction in the blood sugar levels.
done
clear
View Answer play_arrow
In angiosperms pollen tube liberate their male gametes into the:
A)
Central cell
done
clear
B)
Antipodal cells
done
clear
C)
Egg cell
done
clear
D)
Synergids
done
clear
View Answer play_arrow
The fruit is chambered, developed from inferior ovary and has seeds with succulent testa in:
A)
guava
done
clear
B)
cucumber
done
clear
C)
pomegranate
done
clear
D)
orange
done
clear
View Answer play_arrow
Transfer of pollen grains from the anther to the stigma of another flower of the sameplantis called:
A)
Xenogamy
done
clear
B)
Geitonogamy
done
clear
C)
Karyogamy
done
clear
D)
Autogamy
done
clear
View Answer play_arrow
Even in the absence of pollinating agents seed seting is assured in:
A)
Salvia
done
clear
B)
Fig
done
clear
C)
Commellina
done
clear
D)
Zostera
done
clear
View Answer play_arrow
Which of the following are the important flora rewards to the animal pollinators?
A)
Protein pellicle and stigmatic exudates
done
clear
B)
Colour and large size of flower
done
clear
C)
Nectar and pollen grains
done
clear
D)
Floral fragrance and calcium crystals
done
clear
View Answer play_arrow
Which one of the following fruits is parthenocarpic?
A)
Banana
done
clear
B)
Brinjal
done
clear
C)
Apple
done
clear
D)
Jackfruit
done
clear
View Answer play_arrow
Which one of the following statements is incorrect about menstruation?
A)
At menopause in the female, there is especially abrupt increase in gonadotropic hormones
done
clear
B)
The beginning of the cycle of menstruation is called menarche
done
clear
C)
During normal menstruation about 40 ml blood is lost
done
clear
D)
The menstrual fluid can easily clot
done
clear
View Answer play_arrow
The second maturation division of the mam- malian ovum occurs:
A)
Shortly after ovulation before the ovum makes entry into the fallopian tube
done
clear
B)
Until after the ovum has been penetrated by a sperm
done
clear
C)
Until the nucleus of the sperm has fused with that of the ovum
done
clear
D)
in the Graafian follicle following the first maturation division
done
clear
View Answer play_arrow
Vasa efferentia are the ductless leading from:
A)
Testicular lobules to rete testis
done
clear
B)
Rete testis to vas deferens
done
clear
C)
Vas deferens to epididymis
done
clear
D)
Epididymis to urethra
done
clear
View Answer play_arrow
Signals for parturition originate from:
A)
Placenta only
done
clear
B)
Fully developed foetus only
done
clear
C)
Both placenta and fully developed foetus
done
clear
D)
Oxytocin released from maternal pituitary
done
clear
View Answer play_arrow
Ectopic pregnancies are referred to as:
A)
Pregnancies terminated due to hormonal imbalance
done
clear
B)
Pregnancies with genetic abnormality.
done
clear
C)
Implantation of embryo at site other than uterus.
done
clear
D)
Implantation of defective embryo in the uterus
done
clear
View Answer play_arrow
A childless couple can be assisted to have a child through a technique called GIFT. The full form of this technique is:
A)
germ cell internal fallopian transfer
done
clear
B)
gamete inseminated fallopian transfer
done
clear
C)
gamete intra fallopian transfer
done
clear
D)
gamete internal fertilization and transfer
done
clear
View Answer play_arrow
When dominant and recessive alleles express itself together it is called:
A)
Codominance
done
clear
B)
Dominance
done
clear
C)
Amphidominance
done
clear
D)
Pseudodominance
done
clear
View Answer play_arrow
Genes for cytoplasmic male sterility in plants are generally located in:
A)
Chloroplast genome
done
clear
B)
Mitochondrial genome
done
clear
C)
Nuclear genome
done
clear
D)
Cytosol
done
clear
View Answer play_arrow
Haemophilia is more commonly seen in human males than in human females because:
A)
This disease is due to a Y-linked recessive mutation
done
clear
B)
This disease is due to an X-linked recessive mutation
done
clear
C)
This disease is due to an X-linked dominant mutation
done
clear
D)
A greater proportion of girls die in infancy
done
clear
View Answer play_arrow
Both sickle cell anemia and Huntington's chorea are:
A)
Bacteria-related diseases
done
clear
B)
Congenital disorders
done
clear
C)
Pollutant-induced disorders
done
clear
D)
Virus-related diseases
done
clear
View Answer play_arrow
A common test to find the genotype of a hybrid
A)
Crossing of one \[{{F}_{1}}\]progeny with male parent
done
clear
B)
Crossing of one\[{{F}_{2}}\]progeny with male parent
done
clear
C)
Crossing of one \[{{F}_{2}}\]progeny with female parent
done
clear
D)
Studying the sexual behaviour of \[{{F}_{1}}\]progenies
done
clear
View Answer play_arrow
Which one of the following cannot be explained on the basis of Mendel's Law of Dominance?
A)
The discrete unit controlling a particular character is called a factor
done
clear
B)
Out of one pair of factors one is dominant, and the other recessive
done
clear
C)
Alleles do not show any blending and both the characters recover as such in \[{{F}_{2}}\]generation
done
clear
D)
Factors occur in pairs
done
clear
View Answer play_arrow
Which of the following statements is not true of two genes that show 50% recombination frequency?
A)
The genes may be on different chromo- somes
done
clear
B)
The genes are tightly linked
done
clear
C)
The genes show independent assortment
done
clear
D)
If the genes are present on the same chromosome, they undergo more than one crossovers in every meiosis
done
clear
View Answer play_arrow
A colour blind man marries a woman with normal sight who has no history of colourblindness in her family. What is the probability of their grandson being colour blind?
A)
0.25
done
clear
B)
0.5
done
clear
C)
1
done
clear
D)
Nil
done
clear
View Answer play_arrow
In a DNA percentage of thymine is 20% then what is the percentage of guanine:
A)
20%
done
clear
B)
40%
done
clear
C)
30%
done
clear
D)
60%
done
clear
View Answer play_arrow
What would happen if in a gene encoding a polypeptide of 50 amino acids, 25th codon (UAU) is mutated to UAA:
A)
A polypeptide of 25 amino acids will be formed
done
clear
B)
Two polypeptides of 24 and 25 amino acids will be formed
done
clear
C)
A polypeptide of 49 amino acids will be formed
done
clear
D)
A polypeptide of 25 amino acids will be formed
done
clear
View Answer play_arrow
Crossing over that results in genetic recombination in higher organisms occurs between:
A)
Non-sister chromatids of a bivalent
done
clear
B)
Two daughter nuclei
done
clear
C)
Two different bivalents
done
clear
D)
Sister chromatids of a bivalent
done
clear
View Answer play_arrow
During transcription holoenzyme RNA polymerase binds to a DNA sequence and the DNA assumes a saddle like structure at the point. What is the sequence called
A)
CAAT box
done
clear
B)
GGTT box
done
clear
C)
AAAT box
done
clear
D)
TATA box
done
clear
View Answer play_arrow
Haploids are more suitable for mutation studies than the diploids. This is because:
A)
haploids are more abundant in nature than diploids
done
clear
B)
all mutations, whether dominant or reces-sive,are expressed in haploids
done
clear
C)
haploids are reproductively more stable than diploids
done
clear
D)
mutagens penetrate in haploids more effectively than in diploids
done
clear
View Answer play_arrow
Which one of the following does not follow the central dogma of molecular biology?
A)
Pea
done
clear
B)
Mucor
done
clear
C)
Chlamydomonas
done
clear
D)
HIV
done
clear
View Answer play_arrow
 The coefficient of static friction between the block and the cart is p.. The acceleration a of the cart that will prevent the block from falling satisfies
The coefficient of static friction between the block and the cart is p.. The acceleration a of the cart that will prevent the block from falling satisfies 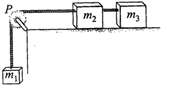 (Assume\[{{m}_{1}}={{m}_{2}}={{m}_{3}}=m\])
(Assume\[{{m}_{1}}={{m}_{2}}={{m}_{3}}=m\]) 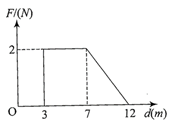


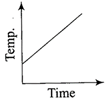
 \[\Delta {{U}_{2}},\Delta {{U}_{3}}\]indicate the change in internal energy along the three processes respectively, then
\[\Delta {{U}_{2}},\Delta {{U}_{3}}\]indicate the change in internal energy along the three processes respectively, then 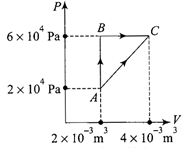
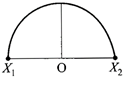
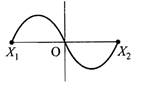


 internal resistance \[1\,\Omega ,\] are connected as shown. The voltmeter V will record a reading of
internal resistance \[1\,\Omega ,\] are connected as shown. The voltmeter V will record a reading of 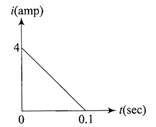
 Retarding potential Anode potential
Retarding potential Anode potential 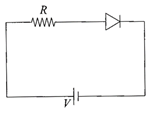
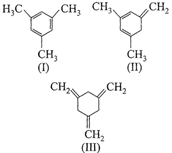 The enthalpy of the hydrogenation of these compounds will be in the order as:
The enthalpy of the hydrogenation of these compounds will be in the order as: