Two pendulum begin to swing simultaneously. The first pendulum makes 9 full oscillations while the other makes 7 in the same time. The ratio of length of the two pendulum is:
A)
\[\frac{9}{7}\]
done
clear
B)
\[\frac{7}{9}\]
done
clear
C)
\[\frac{49}{81}\]
done
clear
D)
\[\frac{81}{49}\]
done
clear
View Answer play_arrow
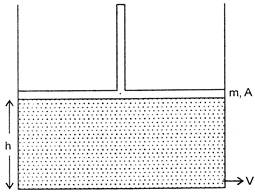
A cylindrical vessel contains a liquid of density P up to a height. The liquid is closed by a piston of mass m and area of cross-section A. There is a small hole at the bottom of the vessel. The speed V with which the liquid comes out of the hole is (neglect presence of atmosphere):
A)
\[\sqrt{2gh}\]
done
clear
B)
\[\sqrt{2\left( gh+\frac{mg}{P\,A} \right)}\]
done
clear
C)
\[\sqrt{2\left( gh+\frac{mg}{A} \right)}\]
done
clear
D)
\[\sqrt{\left( 2gh+\frac{mg}{A} \right)}\]
done
clear
View Answer play_arrow
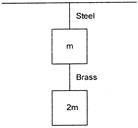
If the ratio of lengths, radii and Young?s modulus of steel and brass wires in the figure are a, b and c respectively, then the corresponding ratio of increase in their length would be:
A)
\[\frac{2{{a}^{2}}c}{b}\]
done
clear
B)
\[\frac{39}{2{{b}^{2}}c}\]
done
clear
C)
\[\frac{2ac}{{{b}^{2}}}\]
done
clear
D)
\[\frac{3c}{2a{{b}^{2}}}\]
done
clear
View Answer play_arrow
The escape velocity on the surface of the earth is\[11.2\,km/s\]. what would be the escape velocity on the surface of another planet of the same mass but \[{\scriptstyle{}^{1}/{}_{4}}\] times the radius of the earth
A)
\[44.8km/s\]
done
clear
B)
\[22.4km/s\]
done
clear
C)
\[5.6km/s\]
done
clear
D)
\[11.2km/s\]
done
clear
View Answer play_arrow
A projectile of mass m is thrown with velocity \[\upsilon \]making an angle of \[{{30}^{o}}\] with vertically. Neglecting air resistance the magnitude of change in momentum between the starting point and at the maximum height is
A)
\[\frac{m\upsilon }{2}\]
done
clear
B)
\[\frac{\sqrt{3}m\upsilon }{2}\]
done
clear
C)
\[m\upsilon \]
done
clear
D)
\[\frac{\sqrt{7}m\upsilon }{2}\]
done
clear
View Answer play_arrow
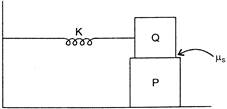
A block P of mass m is placed on a horizontal frictionless plane. A second block of same mass m is placed on it and is connected to a spring of spring constant K, the two blocks are pulled by distance A. Block Q oscillates without slipping. What is the maximum value of frictional force between the two blocks?
A)
\[\frac{KA}{2}\]
done
clear
B)
KA
done
clear
C)
\[{{H}_{5}}mg\]
done
clear
D)
zero
done
clear
View Answer play_arrow
A bullet makes n turns inside the barrel of length I of a rifle and emerges from it with a speed V. Assuming that the bullet moves inside the barrel with a uniform acceleration, and uniform angular acceleration, the angular velocity of the bullet about its longitudinal exis as it emerges from the barrel is
A)
\[\frac{V}{l}\]
done
clear
B)
\[\frac{2\pi nV}{l}\]
done
clear
C)
\[\frac{\pi n}{2V}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
A body when projected vertically up, covers a total distance D during its time of flight. If there were no gravity, the distance covered by it during the same time is equal to
A)
\[0\]
done
clear
B)
D
done
clear
C)
2D
done
clear
D)
4D
done
clear
View Answer play_arrow
A wire is of mass \[(0.3\pm 0.003)g.\]. The radius is \[(0.5\pm 0.005)\,mm\] and length is \[(6.0\pm 0.06)\,cm,\]then % error in density is:
A)
3
done
clear
B)
4
done
clear
C)
6
done
clear
D)
\[-2\]
done
clear
View Answer play_arrow
If we change unit of a physical quantity, then:
A)
Its dimensions change
done
clear
B)
Its dimensions remain same
done
clear
C)
It may change or may not change
done
clear
D)
Its magnitude changes
done
clear
View Answer play_arrow
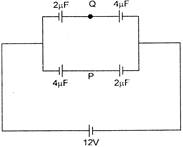
Find VPQ in the figure:
A)
12V
done
clear
B)
8V
done
clear
C)
4V
done
clear
D)
zero
done
clear
View Answer play_arrow
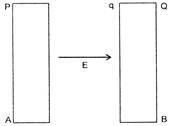
Two conducting plates P and Q with large surface area A are placed as shown in figure. A charge q is given to plate P. The electric field between the plates at any point is:
A)
\[\frac{q}{3A{{\varepsilon }_{0}}}\]
done
clear
B)
\[\frac{q}{2A{{\varepsilon }_{0}}}\]
done
clear
C)
\[\frac{q}{A{{\varepsilon }_{0}}}\]
done
clear
D)
\[\frac{2q}{A{{\varepsilon }_{0}}}\]
done
clear
View Answer play_arrow
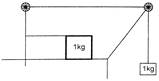
A block of mass 1kg is placed on a rough horizontal surface connected by a light string passing over two smooth pulleys as shown. Another block of 1kg is connected to the other end of the string. The acceleration of the system is: (Coefficient of friction \[\mu =0.2\])
A)
\[0.8g\text{ }m/{{s}^{2}}\]
done
clear
B)
\[0.4g\text{ }m/{{s}^{2}}\]
done
clear
C)
\[0.5g\text{ }m/{{s}^{2}}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
Three particles each of mass m are placed at the corners of an equilateral triangle of side d. The potential energy of the system is
A)
\[\frac{3G{{m}^{2}}}{d}\]
done
clear
B)
\[\frac{G{{m}^{2}}}{d}\]
done
clear
C)
\[\frac{-3G{{m}^{2}}}{d}\]
done
clear
D)
none of these
done
clear
View Answer play_arrow
A simple pendulum with a solid metal bob has a period T. The metal bob is now immersed in a liquid of density one-tenth that of the bob. The liquid is non viscous. Now the period of the same pendulum with its bob remaining all the time in the liquid will be:
A)
\[T\]
done
clear
B)
\[\left( \frac{9}{10} \right)T\]
done
clear
C)
\[\sqrt{\frac{10}{9}}T\]
done
clear
D)
\[\sqrt{\frac{9}{10}}T\]
done
clear
View Answer play_arrow
The angle which a vector \[i-j+\sqrt{2}K\] makes with y-axis is:
A)
\[{{60}^{o}}\]
done
clear
B)
\[{{120}^{o}}\]
done
clear
C)
\[{{150}^{o}}\]
done
clear
D)
\[{{\tan }^{-1}}\left( -\frac{1}{2} \right)\]
done
clear
View Answer play_arrow
We have ajar A filled with gas characterised by parameters P,V and T another jar B filled with gas with parameters \[2P,\frac{V}{4}\] and 2T, where the symbols have their usual meanings. The ratio of the number of molecules of jar A to those of jar B is:
A)
\[1:1\]
done
clear
B)
\[1:2\]
done
clear
C)
\[2:1\]
done
clear
D)
\[4:1\]
done
clear
View Answer play_arrow
If the temperature of the sun is increased from T to 2T and its radius from R to 2R, then the ratio of the radiant energy received on earth to what it was previously will be?
A)
\[4\]
done
clear
B)
\[16\]
done
clear
C)
\[32\]
done
clear
D)
\[64\]
done
clear
View Answer play_arrow
When two tuning forks A and B sounded together produce 4 beats per second. After felling of A and waxing of B, the number of beats remain unaltered. If initial frequency of A is 250 Hz, then the initial frequency of B is:
A)
\[246\text{ }Hz\]
done
clear
B)
\[250\text{ }Hz\]
done
clear
C)
\[254\text{ }Hz\]
done
clear
D)
\[242\text{ }Hz\]
done
clear
View Answer play_arrow
Two waves having the intensities in the rations of \[9:1\] produce interference. The ratio of maximum to minimum intensities is equal to:
A)
\[10:8\]
done
clear
B)
\[9:1\]
done
clear
C)
\[4:1\]
done
clear
D)
\[2:1\]
done
clear
View Answer play_arrow
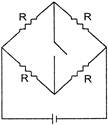
A triangular loop of side / carries a current I. It is placed in a magnetic field B, such that the plane of the loop is in the direction of B. The torque on the loop is:
A)
zero
done
clear
B)
\[IB{{l}^{2}}\]
done
clear
C)
\[\sqrt{\frac{3}{2}}IB{{l}^{2}}\]
done
clear
D)
\[\sqrt{\frac{3}{4}}IB{{l}^{2}}\]
done
clear
View Answer play_arrow
A proton and a-particle enter a uniform magnetic field with the same velocity. The period of rotation of the a-particle will be:
A)
four times that of the proton
done
clear
B)
two times that of the proton
done
clear
C)
three times that of the proton
done
clear
D)
same as that of the proton
done
clear
View Answer play_arrow
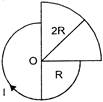
The magnetic field strength at O, due to current I in the figure below, is:
A)
\[\frac{7{{\mu }_{0}}I}{16\,R}\]
done
clear
B)
\[\frac{15{{\mu }_{0}}I}{16\,R}\]
done
clear
C)
\[\frac{11{{\mu }_{0}}I}{32\,R}\]
done
clear
D)
\[\frac{13{{\mu }_{0}}I}{32\,R}\]
done
clear
View Answer play_arrow
the meter bridge circuit shown in figure is balanced when jockey J divides wire AB in two parts AJ and BJ in the ratio of \[1:2\]. The unknown resistance Q has value:
A)
\[1\Omega \]
done
clear
B)
\[3\Omega \]
done
clear
C)
\[4\Omega \]
done
clear
D)
\[7\Omega \]
done
clear
View Answer play_arrow
A cell of emf \[\varepsilon \] is connected across a resistance R. The potential difference between the terminals of the cell is found to be \[\upsilon \]. The internal resistance of the cell must be:
A)
\[\frac{2(E-V)V}{R}\]
done
clear
B)
\[\frac{2(E-V)R}{E}\]
done
clear
C)
\[\frac{(E-V)R}{V}\]
done
clear
D)
\[(E-V)R\]
done
clear
View Answer play_arrow
In the balanced wheatstone bridge circuit as shown in the figure, when the key is pressed, what will be the change in the reading of the galvanometer?
A)
No change
done
clear
B)
Increased
done
clear
C)
Decreased
done
clear
D)
Zero
done
clear
View Answer play_arrow
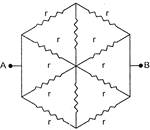
The resistance of haxagon circuit between A and B represented in figure is:
A)
r
done
clear
B)
\[0.5\,r\]
done
clear
C)
\[2\,r\]
done
clear
D)
\[3\,r\]
done
clear
View Answer play_arrow
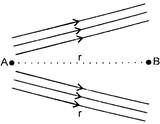
Figure shows the electric lines of force emerging from a charged body. If the electric field at A and B arc \[{{E}_{A}}\] and \[{{E}_{B}}\] respectively and if the displacement between A and B is r, then:
A)
\[{{E}_{A}}>{{E}_{B}}\]
done
clear
B)
\[{{E}_{A}}<{{E}_{B}}\]
done
clear
C)
\[{{E}_{A}}=\frac{{{E}_{B}}}{r}\]
done
clear
D)
\[{{E}_{A}}=\frac{{{E}_{B}}}{{{r}^{2}}}\]
done
clear
View Answer play_arrow
A charge \[+q\] is brought near an isolated metal cube having no charge initially:
A)
Cube becomes positively charged.
done
clear
B)
Cube becomes negatively charge.
done
clear
C)
External surface becomes negatively charged and interior becomes positively charged.
done
clear
D)
Interior remains charge free and the surface gets non-uniform charge distribution.
done
clear
View Answer play_arrow
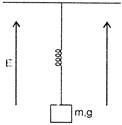
A block of mass 771 having charge q, strings by a spring of spring constant K in a verticle electrostatic field E. The string extension in equiliberium will be:
A)
\[\frac{mg}{K}\]
done
clear
B)
\[\frac{qE}{K}\]
done
clear
C)
\[\frac{mg+qE}{K}\]
done
clear
D)
\[\frac{mg-qE}{K}\]
done
clear
View Answer play_arrow
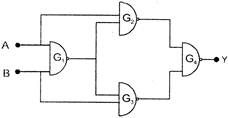
. Select the output Y of the combination of gates shown below for inputs \[A=1,\] \[B=0,\]\[A=1,\]\[B=1\]and \[A=0\],\[B=0\] respectively
A)
\[(001)\]
done
clear
B)
\[(101)\]
done
clear
C)
\[(111)\]
done
clear
D)
\[(100)\]
done
clear
View Answer play_arrow
In an electromagnetic wave, the electric and magnetising field are \[100\,V{{m}^{-1}}\] and \[0.265\,A{{m}^{-1}}.\]. The maximum rate of energy flow is:
A)
\[26.5w/{{m}^{2}}\]
done
clear
B)
\[36.5w/{{m}^{2}}\]
done
clear
C)
\[46.7\,w/{{m}^{2}}\]
done
clear
D)
\[765\,w/{{m}^{2}}\]
done
clear
View Answer play_arrow
In a common base transistor \[{{I}_{c}}={{I}_{a}}{{I}_{b}},\] load and plate resistance are \[4K\Omega \] and \[1K\Omega \]. The voltage gain of amplifier will be:
A)
\[80\]
done
clear
B)
\[4.2\]
done
clear
C)
\[3.8\]
done
clear
D)
\[76\]
done
clear
View Answer play_arrow
A freshly prepared radiactive source half-life 2h emits radiation of intensity which 64 times the permissible safe level. The minimum time after which it would be possible to work safely with this source is:
A)
\[6h\]
done
clear
B)
\[12h\]
done
clear
C)
\[42h\]
done
clear
D)
\[128h\]
done
clear
View Answer play_arrow
If the binding energy per nucleon in \[_{3}^{7}Li\] and \[2_{2}^{4}He\] nuclei are \[5.60\text{ }MeV\] and \[7.06\text{ }MeV\]respectively, then in the reaction \[_{1}^{1}H+_{3}^{7}Li\to 2_{2}^{4}He,\]energy of proton must be:
A)
\[39.2\text{ }MeV\]
done
clear
B)
\[28.24\text{ }MeV\]
done
clear
C)
\[17.28\text{ }MeV\]
done
clear
D)
\[1.46\text{ }MeV\]
done
clear
View Answer play_arrow
The ratio of de-Broglie wavelength of molecules of Hydrogen and helium which are at temperature \[{{27}^{o}}C\] and \[{{127}^{o}}C\] respectively will be
A)
\[\sqrt{\frac{4}{3}}\]
done
clear
B)
\[\sqrt{\frac{8}{3}}\]
done
clear
C)
\[\sqrt{\frac{3}{8}}\]
done
clear
D)
\[\sqrt{\frac{2}{4}}\]
done
clear
View Answer play_arrow
The electromagnetic waves that has highest wavelength is:
A)
X-rays
done
clear
B)
Ultraviolet rays
done
clear
C)
Infrared rays
done
clear
D)
Microwaves
done
clear
View Answer play_arrow
A p-type semiconductor has acceptor level \[57\text{ }MeV\] above the valence band. The maximum wavelength of light required to create a hole is:
A)
\[57\,A\]
done
clear
B)
\[57\times {{10}^{-3}}\overset{o}{\mathop{A}}\,\]
done
clear
C)
\[217100\overset{o}{\mathop{A}}\,\]
done
clear
D)
\[11.61\times {{10}^{-33}}\overset{o}{\mathop{A}}\,\]
done
clear
View Answer play_arrow
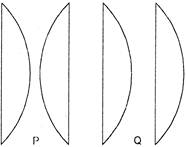
A liquid of refractive index \[1.33\] is placed between two identical plano-convex lenses, with reference index\[1.50\]. Two possible arrangement, P and Q are shown. The system is:
A)
Divergent in P, convergent in Q
done
clear
B)
Convergent in P, divergent in Q
done
clear
C)
Convergent in both
done
clear
D)
Divergent in both
done
clear
View Answer play_arrow
A concave lens of focal length f produces an image equal to \[\frac{1}{n}\]of size of object, the distance of the image, from the lens is:
A)
\[(n+1)f\]
done
clear
B)
\[(n-1)f\]
done
clear
C)
\[\left( \frac{n+1}{n} \right)f\]
done
clear
D)
\[\left( \frac{n-1}{n} \right)f\]
done
clear
View Answer play_arrow
A thin sheet of glass \[(\mu =1.5)\] of thickness 6 microns introduced in the path of one of interfering beamsd of a double slit experiment shifts the central frings to a position previously occupied by fifth bright fringe. Then, the wavelength of the light used is:
A)
\[6000\,\overset{o}{\mathop{A}}\,\]
done
clear
B)
\[3000\,\overset{o}{\mathop{A}}\,\]
done
clear
C)
\[45000\,\overset{o}{\mathop{A}}\,\]
done
clear
D)
\[7500\,\overset{o}{\mathop{A}}\,\]
done
clear
View Answer play_arrow
The principle of optical fibre is:
A)
Diffraction
done
clear
B)
Polarisation
done
clear
C)
Interference
done
clear
D)
Total internal reflection
done
clear
View Answer play_arrow
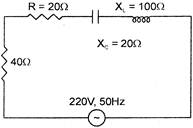
The power factor of the circuit shown in the figure is:
A)
\[0.4\]
done
clear
B)
\[0.2\]
done
clear
C)
\[0.8\]
done
clear
D)
\[0.6\]
done
clear
View Answer play_arrow
A current of i ampere flows along an infinitely long straight thin walled tube, then the magnetic induction at any point inside the tube at a distance r from the centre is:
A)
Infinite
done
clear
B)
zero
done
clear
C)
\[\frac{{{\mu }_{0}}}{4\pi }.\,\frac{2i}{r}\]
done
clear
D)
\[\frac{2i}{r}\].0
done
clear
View Answer play_arrow
The two coils of self- inductance 4H and 16H are wound on the same iron core. The coefficient of mutual inductance for them will be:
A)
\[8\,H\]
done
clear
B)
\[10\,H\]
done
clear
C)
\[20\,H\]
done
clear
D)
\[64\,H\]
done
clear
View Answer play_arrow
\[{{(N{{H}_{4}})}_{2}}C{{r}_{2}}{{O}_{7}}\] on heating gives a gas which is also given by
A)
Heating \[N{{H}_{4}}N{{O}_{2}}\]
done
clear
B)
Heating \[N{{H}_{4}}N{{O}_{3}}\]
done
clear
C)
\[M{{g}_{3}}{{N}_{2}}+{{H}_{2}}O\]
done
clear
D)
\[Na\] (comp.) \[+{{H}_{2}}{{O}_{2}}\]
done
clear
View Answer play_arrow
The pair of compounds having metals in their highest oxidation state is
A)
\[Mn{{O}_{2}},\,FeC{{l}_{3}}\]
done
clear
B)
\[{{[Mn{{O}_{4}}]}^{-}},\,Cr{{O}_{2}}C{{l}_{2}}\]
done
clear
C)
\[{{[Fe{{(CN)}_{6}}]}^{3-}},[Co{{(CN)}_{3}}]\]
done
clear
D)
\[{{[NiC{{l}_{4}}]}^{2-}},{{[CoC{{l}_{4}}]}^{-}}\]
done
clear
View Answer play_arrow
. The compound having tetrahedral geometry is
A)
\[{{[Ni{{(CN)}_{4}}]}^{2-}}\]
done
clear
B)
\[{{[Pd{{(CN)}_{4}}]}^{2-}}\]
done
clear
C)
\[{{[PdC{{l}_{4}}]}^{2-}}\]
done
clear
D)
\[{{[NiC{{l}_{4}}]}^{2-}}\]
done
clear
View Answer play_arrow
A sodium salt of an unknown anion when treated with \[MgC{{l}_{2}}\] gives white precipitate only on boiling, The anion is
A)
\[SO_{4}^{2-}\]
done
clear
B)
\[HCO_{3}^{-}\]
done
clear
C)
\[CO_{3}^{2-}\]
done
clear
D)
\[NO_{3}^{-}\]
done
clear
View Answer play_arrow
Which hydrogen like species will have the same radius as that of Bohr orbit of hydrogen atom?
A)
\[n=2,\,L{{i}^{2+}}\]
done
clear
B)
\[n=2,\,B{{e}^{3+}}\]
done
clear
C)
\[n=2,\,H{{e}^{+}}\]
done
clear
D)
\[n=3,\,L{{i}^{2+}}\]
done
clear
View Answer play_arrow
How many chiral compound are possible on mono chlorination of 2-methyl butane?
A)
2
done
clear
B)
8
done
clear
C)
4
done
clear
D)
8
done
clear
View Answer play_arrow
Which of the following statement is not true?
A)
The pressure of a gas is due to collision of the gas molecules with the walls of the container.
done
clear
B)
The molecular velocity of any gas is proportional to the square root of the absolute temperature.
done
clear
C)
The rate of diffusion of a gas is directly proportional to the density of the gas at constant pressure.
done
clear
D)
Kinetic energy of an ideal gas is directly proportional to the absolute temperature
done
clear
View Answer play_arrow
Positive deviation from ideal behaviour takes place because of
A)
molecular interaction between atoms and \[PV/nRT>1\]
done
clear
B)
molecular interaction between atoms and \[PV/nRT<1\]
done
clear
C)
finite size of the atoms and \[PV/nRT>1\]
done
clear
D)
finite size of the atoms and \[PV/nRT<1\]
done
clear
View Answer play_arrow
The presence of air bubble in blood stream is dangerous to life because
A)
Air combines with blood forming a complex
done
clear
B)
the flow of blood increases manifold due to concentration of surface active substances at the blood-air interface.
done
clear
C)
the flow of blood is obstructed due to the concentration of surface active substances at the blood-air interface.
done
clear
D)
none of these.
done
clear
View Answer play_arrow
In Kjeldahl's method of estimation of nitrogen copper salt acts as
A)
oxidising agent
done
clear
B)
reducing agent
done
clear
C)
catalytic agent
done
clear
D)
hydrolysis agent
done
clear
View Answer play_arrow
IUPAC name of the following \[C{{H}_{3}}-\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{N}}\,}}\,-\underset{{{C}_{2}}{{H}_{5}}}{\mathop{\underset{|}{\mathop{\overset{C{{H}_{3}}}{\mathop{\overset{|}{\mathop{C}}\,}}\,}}\,}}\,-C{{H}_{2}}-C{{H}_{3}}\]
A)
3-dimethylamino-3-methylpentane
done
clear
B)
3-(N, N-trimethyD-3-aminopentane
done
clear
C)
3-(N, N-trimethyD-3-pentanamine
done
clear
D)
3-(N, N-dimethyl) amino-3-methyl pentane
done
clear
View Answer play_arrow
Which among the following statements is correct with respect to the optical isomers?
A)
Enantiomers are non-super imposable mirror images
done
clear
B)
Diastereomers are superimposable mirror images
done
clear
C)
Eanatiomers are superimposable mirror images
done
clear
D)
Meso forms have no plane of symmetry
done
clear
View Answer play_arrow
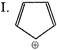
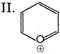
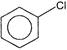
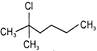
Which of the following shows \[{{S}_{N}}1\]reaction most readily?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Chloroform reacts with cone. \[HN{{O}_{3}}\] to give
A)
\[CC{{l}_{3}}N{{O}_{2}}\]
done
clear
B)
\[C{{H}_{3}}N{{O}_{2}}\]
done
clear
C)
\[C{{H}_{3}}CN\]
done
clear
D)
\[C{{H}_{2}}C{{H}_{2}}N{{O}_{2}}\]
done
clear
View Answer play_arrow
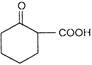
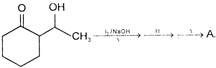
Compound A is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
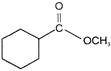
A is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Consider the following reactions
I. \[C{{H}_{3}}NC\xrightarrow{{{H}_{3}}{{O}^{+}}}\] II. \[CHC{{l}_{3}}\xrightarrow{NaOH/{{H}_{3}}{{O}^{+}}}\] III. \[CC{{l}_{4}}\xrightarrow{{{H}_{3}}{{O}^{+}}}\]
\[HCOOH\] is obtained in
A)
I, II, III
done
clear
B)
II, III
done
clear
C)
I, II
done
clear
D)
I, III
done
clear
View Answer play_arrow
Which of the following are aromatic?
A)
I, II, III
done
clear
B)
I, II
done
clear
C)
II, III
done
clear
D)
II
done
clear
View Answer play_arrow
Which of the following compound can lose a proton from methyl group most readily?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
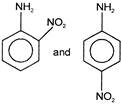
Aniline reacts with cone. \[HN{{O}_{3}}\] to given
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
A certain metal is used to prepare an antacid, this metal accidently catches fire which cannot be put out by using \[C{{O}_{2}}\] based extinguishers. The metal is
A)
\[Ca\]
done
clear
B)
\[Na\]
done
clear
C)
\[Mg\]
done
clear
D)
\[Al\]
done
clear
View Answer play_arrow
The high reactivity and high volatility of white phosphorus is because
A)
It contains tetrahedrally arranged \[{{P}_{4}}\] units
done
clear
B)
The bond angle is \[{{60}^{o}}\]
done
clear
C)
The vander waal?s forces of attraction are weak
done
clear
D)
Steric strain is large
done
clear
View Answer play_arrow
Which of the following has \[P\pi -d\pi \] bonding?
A)
\[NO_{3}^{-}\]
done
clear
B)
\[SO_{3}^{2-}\]
done
clear
C)
\[BO_{3}^{3-}\]
done
clear
D)
\[CO_{3}^{2-}\]
done
clear
View Answer play_arrow
A yellow metallic powder is burnt in a stream of fluorine to obtain a colourless gas X which is thermally stable and chemically inert. Its molecule has octahedral geometry. Another colourless gas Y with same constituent atoms as that of X is obtained when sulphur dichloride is heated with sodium fluoride. Its molecule has trigonal bipyramidal geometry. Gases X and Y are respectively.
A)
\[S{{F}_{4}}\] and \[{{S}_{2}}{{F}_{2}}\]
done
clear
B)
\[S{{F}_{4}}\] and \[S{{F}_{6}}\]
done
clear
C)
\[S{{F}_{6}}\] and \[S{{F}_{4}}\]
done
clear
D)
\[NaF\] and \[NaCl\]
done
clear
View Answer play_arrow
Which of the following is an explosive compound?
A)
\[XeO{{F}_{4}}\]
done
clear
B)
\[XeO{{F}_{2}}\]
done
clear
C)
\[Xe{{F}_{2}}\]
done
clear
D)
\[Xe{{O}_{3}}\]
done
clear
View Answer play_arrow
Which one of the following reactions represent 'developing? in photography?
A)
\[AgN{{O}_{3}}+NaBr\xrightarrow{{}}AgBr+NaN{{O}_{3}}\]
done
clear
B)
\[AgBr+2N{{a}_{2}}{{S}_{2}}{{O}_{3}}\xrightarrow{{}}N{{a}_{3}}\left[ Ag\,{{\left( {{S}_{2}}{{O}_{3}} \right)}_{2}} \right]+NaBr\]
done
clear
C)
\[AgBr+hv\xrightarrow{{}}AgBr*\]
done
clear
D)
\[{{C}_{6}}{{H}_{4}}{{(OH)}_{2}}+2AgBr*\xrightarrow{{}}{{C}_{6}}{{H}_{4}}{{O}_{2}}+2HBr+2Ag\]
done
clear
View Answer play_arrow
Which method of purification is represented by the following equations? \[Ti+2{{I}_{2}}\xrightarrow{773K}Ti{{I}_{4}}\xrightarrow{1675\,K}Ti+2{{I}_{2}}\]
A)
Cupellation
done
clear
B)
Polling
done
clear
C)
Van arkel
done
clear
D)
Zone refining
done
clear
View Answer play_arrow
A certain radioactive substance has half life period of 10 days. How long will it take for its activity to reduce to \[\frac{1}{8}\] of its original value?
A)
40 days
done
clear
B)
20 days
done
clear
C)
1.25 days
done
clear
D)
30 days
done
clear
View Answer play_arrow
Among cellulose, polyvinyl chloride, nylon and natural rubber, the polymer in which the inter molecular force of attraction is weakest in
A)
nylon
done
clear
B)
polyvinyl chloride
done
clear
C)
cellulose
done
clear
D)
natural rubber
done
clear
View Answer play_arrow
Detergents are known to pollute rivers and water ways. However, detergents can be made biodegradable and pollution free by taking
A)
Cyclic hydrocarbon chain
done
clear
B)
Shorter hydrocarbon chain
done
clear
C)
Unbranched hydrocarbon chain
done
clear
D)
hydrocarbon with more branching
done
clear
View Answer play_arrow
Which of the following statement is not true?
A)
Tetrazine is harmful edible colour
done
clear
B)
Alitame is an artificial sweetncr
done
clear
C)
BUT is an antioxidant
done
clear
D)
Sodium alkyi sulphate is cationic detergent
done
clear
View Answer play_arrow
Nitrobenzene on reduction with zinc and \[N{{H}_{4}}Cl\] gives
A)
azobenzene
done
clear
B)
aniline
done
clear
C)
hydrazobenzene
done
clear
D)
N-phenyl hydroxylamine
done
clear
View Answer play_arrow
The molecule which does not exhibit dipole moment is
A)
\[CHC{{l}_{3}}\]
done
clear
B)
\[CC{{l}_{4}}\]
done
clear
C)
\[{{H}_{2}}O\]
done
clear
D)
\[N{{H}_{3}}\]
done
clear
View Answer play_arrow
Which of the following oxides is amphoteric in character?
A)
\[MgO\]
done
clear
B)
\[C{{O}_{2}}\]
done
clear
C)
\[Si{{O}_{2}}\]
done
clear
D)
\[Sn{{O}_{2}}\]
done
clear
View Answer play_arrow
For \[{{N}_{2(g)}}+3{{H}_{2(g)}}2N{{H}_{3}}(g),\,\Delta H\] is equal to
A)
\[\Delta E+2RT\]
done
clear
B)
\[\Delta E-2RT\]
done
clear
C)
\[\Delta E+RT\]
done
clear
D)
\[\Delta E-RT\]
done
clear
View Answer play_arrow
Which expression is wrong for first order reaction?
A)
\[k=\frac{2.303}{t}\log \,\left( \frac{{{A}_{0}}}{{{A}_{t}}} \right)\]
done
clear
B)
\[k=\frac{t}{2.303}\log \,\left( \frac{{{A}_{0}}}{{{A}_{t}}} \right)\]
done
clear
C)
\[-k=\frac{t}{2.303}\log \,\left( \frac{{{A}_{t}}}{{{A}_{0}}} \right)\]
done
clear
D)
Rate \[=k[A]\]
done
clear
View Answer play_arrow
The rate of reaction for the reaction \[2A+B\xrightarrow{{}}C\] is found to be, rate \[=K[A]\,[B]\].The correct statement in relation to this reaction is that the
A)
unit of K must be \[{{S}^{-1}}\]
done
clear
B)
\[{{t}_{1/2}}\propto \frac{1}{a}\]
done
clear
C)
rate of formation of C is twice the rate of disappearance of A.
done
clear
D)
None of the above
done
clear
View Answer play_arrow
If solubility of calcium hydroxide is \[\sqrt{3}\] then its solubility product will be
A)
27
done
clear
B)
3
done
clear
C)
9
done
clear
D)
\[12\sqrt{3}\]
done
clear
View Answer play_arrow
Which one has the highest osmotic pressure?
A)
\[\frac{M}{10}HCl\]
done
clear
B)
\[\frac{M}{10}\]urea
done
clear
C)
\[\frac{M}{10}BaC{{l}_{2}}\]
done
clear
D)
\[\frac{M}{10}\] glucose
done
clear
View Answer play_arrow
Which one of the following statement is correct?
A)
Normality does not change with temperature.
done
clear
B)
Molarity does not change with temperature.
done
clear
C)
Molality does not change with temperature
done
clear
D)
None of these
done
clear
View Answer play_arrow
What is the standard free energy change at \[{{25}^{o}}C\] for the reaction
\[C{{d}_{(s)}}+P{{b}^{2+}}_{(aq)}\xrightarrow{{}}C{{d}^{2+}}_{(aq)}+P{{b}_{(s)}}\] Given \[E_{{\scriptstyle{}^{C{{d}^{2+}}}/{}_{Cd}}}^{o}=-0.403\,V\] \[E_{{\scriptstyle{}^{P{{b}^{2+}}}/{}_{Pb}}}^{o}=-0.125\,V\]
A)
\[-5.346\text{ }kJ\]
done
clear
B)
\[53.46\text{ }kJ\]
done
clear
C)
\[-10.69\text{ }kJ\]
done
clear
D)
\[-106.92\text{ }kJ\]
done
clear
View Answer play_arrow
The mass of carbon anode consumed (giving only carbon dioxide) in the production of 270 kg of aluminium metal from bauxite by the Hall process is (Atomic mass of \[Al=27\])
A)
\[180\,kg\]
done
clear
B)
\[270\text{ }kg\]
done
clear
C)
\[540\,kg\]
done
clear
D)
\[90\,kg\]
done
clear
View Answer play_arrow
Which one of the following will have the highest coagulating power for a ferric hydroxide solution?
A)
\[NaCl\]
done
clear
B)
\[BaC{{l}_{2}}\]
done
clear
C)
\[{{K}_{2}}Cr{{O}_{4}}\]
done
clear
D)
\[{{K}_{3}}[Fe\,{{(CN)}_{6}}]\]
done
clear
View Answer play_arrow
Intermediate formed by heating microcosmic salt and which forms coloured bead with coloured cation is
A)
\[N{{H}_{3}}\]
done
clear
B)
\[BaC{{l}_{2}}\]
done
clear
C)
\[{{K}_{2}}Cr{{O}_{4}}\]
done
clear
D)
lift
done
clear
View Answer play_arrow
Excess of \[KI\] reacts with \[CuS{{O}_{4}}\] solution and then \[N{{a}_{2}}{{S}_{2}}{{O}_{3}}\] solution is added to it. Which of the statements is incorrect for this reaction?
A)
\[C{{u}_{2}}{{I}_{2}}\] is formed
done
clear
B)
\[N{{a}_{2}}S{{O}_{3}}\] is oxidized
done
clear
C)
\[Cu{{I}_{2}}\] is formed
done
clear
D)
Evolved \[{{I}_{2}}\] is reduced
done
clear
View Answer play_arrow
Halophytes are able to live under high salt conditions due to all, except
A)
absence of sap vacuole
done
clear
B)
occurrence of mucilage cover on cell wall
done
clear
C)
high internal salt content
done
clear
D)
absence of branched chain lipids in cell membrane
done
clear
View Answer play_arrow
Choose incorrect option with respect to lichens
A)
Soredia arc most efficient spores to reproduce asexually
done
clear
B)
Isidia are primarily meant to increase surface area
done
clear
C)
All lichens are capable of \[{{N}_{2}}\] fixation
done
clear
D)
They do not grow in polluted areas
done
clear
View Answer play_arrow
Select the correct match
A)
Product Member Class Agar Gelidium Phaeophyceae
done
clear
B)
Product Member Class Carrageen Chondrus Rhodophyceae
done
clear
C)
Product Member Class Algin Chlorella Chorophyceae
done
clear
D)
Product Member Class Agar Sargassum Rhodophyceae
done
clear
View Answer play_arrow
Match the following
Column - I
Column - II
A. Pamela
1. Selaginella
B. Organ suigcneris
2. Equisetum
C. Sphenopsida
3. Psilotum
D. Psilopsida
4. Dryopteris
A)
A-4 B-1 C-2 D-3
done
clear
B)
A-1 B-4 C-3 D-2
done
clear
C)
A-3 B-2 C-1 D-4
done
clear
D)
A-2 B-1 C-4 D-3
done
clear
View Answer play_arrow
Flattened or cylindrical green stems of unlimited growth which have taken over the function of photosynthesis and help the plants to grow in dry habitats are
A)
Cladodes
done
clear
B)
Phylloclades
done
clear
C)
Phyllodes
done
clear
D)
Cladophylls
done
clear
View Answer play_arrow
Choose incorrect with respect to true nuts
A)
Litchi
done
clear
B)
Cashew nut
done
clear
C)
Water Chestnut
done
clear
D)
Coconut
done
clear
View Answer play_arrow
Plasma membrane is asymmetric on account of all, except
A)
Amount and types of extrinsic proteins are difference on two sides of membrane
done
clear
B)
Human RBC membrane has 52% proteins and 40% lipids
done
clear
C)
Lipids are different on two sides of membrane
done
clear
D)
Glycocalyx present only on outer side of Membrane
done
clear
View Answer play_arrow
Multienzyme complex called recombinase appear over the bivalents of facilitate exchange of chromatid segments in
A)
Diplotene
done
clear
B)
Pachytene
done
clear
C)
Zygotene
done
clear
D)
Leptotene
done
clear
View Answer play_arrow
When a plant cell is placed in hypertonic solution its cell membrane shrinks away from the cell wall and the cell becomes plasmolysed. Which is not true for this phenomenon?
A)
Hypertonic solution causes exomosis
done
clear
B)
Water moves out first from the vacuole
done
clear
C)
External solution occupies the space between cell wall and shrunken protoplast
done
clear
D)
DPD of cell becomes negative
done
clear
View Answer play_arrow
Toxicity of \[Mn\] in plant tissue
A)
inhibits absorption of \[Mg\] only
done
clear
B)
competes with \[Fe\] for binding with enzyme
done
clear
C)
inhibits absorption of \[Fe\] and \[Mg\]
done
clear
D)
inhibits translocation of \[Ca\] and \[Fe\] in shoot apices
done
clear
View Answer play_arrow
Mobile electron carriers in ETS of chloroplast are
A)
PQ, PC
done
clear
B)
UQ and Cyt. C
done
clear
C)
\[Cyt\text{ }{{b}_{6}}-f,\text{ }PC\]
done
clear
D)
Fd, PQ and \[Cyt.{{a}_{3}}\]
done
clear
View Answer play_arrow
Select correct statement with respect to fermentation
A)
less than 7% energy of glucose is released
done
clear
B)
\[C{{O}_{2}}\] is released essentially
done
clear
C)
can produce 8 ATP per mole of glucose
done
clear
D)
metabolic water is a by-product like aerobic respiration
done
clear
View Answer play_arrow
Plant harmone which helps to prevent fruit and leaf drop at early stages but promotes the abscission of older mature leaves and fruits is
A)
ABA
done
clear
B)
Auxins
done
clear
C)
GA,
done
clear
D)
Cytokinin
done
clear
View Answer play_arrow
Interruption of dark period by a flash of light prevent flowering in
A)
Cucumber
done
clear
B)
Spinach
done
clear
C)
Cocklebur
done
clear
D)
Larkspur
done
clear
View Answer play_arrow
Pollen tube content is discharged in
A)
degenerated synergid
done
clear
B)
egg cell
done
clear
C)
persistent synergid
done
clear
D)
central cell
done
clear
View Answer play_arrow
Endosperm in angiosperms are triploid but it is diploid in
A)
Maize
done
clear
B)
Pussiflora
done
clear
C)
Oenothera
done
clear
D)
Mango
done
clear
View Answer play_arrow
Proportion of offsprings with genotype AabbCc is ___ if parents are Aabbcc and aaBbCc.
A)
4/64
done
clear
B)
8/64
done
clear
C)
16/64
done
clear
D)
2/64
done
clear
View Answer play_arrow
Which of these is sex-limited trait in human beings?
A)
Hypertrichosis
done
clear
B)
Antlers in mole deer
done
clear
C)
Pattern baldness
done
clear
D)
Haemophilia
done
clear
View Answer play_arrow
Enzyme involved in removal ofthymidine dimer, filling of gaps and shows \[5'\to 3'\] exonuclease activity during DNA replication in E.coli is
A)
DNA P I
done
clear
B)
DNA P II
done
clear
C)
Komberg enzyme
done
clear
D)
More than one option is correct
done
clear
View Answer play_arrow
Ambiguous codon in genetic code is
A)
GUG
done
clear
B)
AUG
done
clear
C)
AGG
done
clear
D)
UAA
done
clear
View Answer play_arrow
Variety of wheat improved in India through mutation breeding is
A)
HUW - 468
done
clear
B)
Kalyan Sona
done
clear
C)
NP - 836
done
clear
D)
PV -18
done
clear
View Answer play_arrow
Excessive growth of Klamath weed was cheved in USA by introduction of
A)
Chrysolina beetle
done
clear
B)
Cochineal insect
done
clear
C)
Lady bird beetles
done
clear
D)
Praying mantis
done
clear
View Answer play_arrow
Predators are important in an ecosyster because they
A)
control the population of prey
done
clear
B)
are prudent by behaviour
done
clear
C)
are involved in transfer of energy from one to other trophic level
done
clear
D)
more than one option is correct
done
clear
View Answer play_arrow
Pyramid of number in a tree ecosystem terminating in parasitic food chain is
A)
Inverted
done
clear
B)
Upright
done
clear
C)
Spindle shaped
done
clear
D)
Triangular
done
clear
View Answer play_arrow
Illegal introduction of____ for aquaculture purposes is posing a serious threat to indigenous catfish in our country
A)
Clarias gariepinus
done
clear
B)
Nile Perch
done
clear
C)
Water hyacinth
done
clear
D)
Congress grass
done
clear
View Answer play_arrow
Photochomical smog
A)
occurs at low temperature
done
clear
B)
consists mainly ozone, PAN and \[N{{O}_{2}}\]
done
clear
C)
makes the environment reducing
done
clear
D)
mainly contributed by \[{{H}_{2}}S\] and \[S{{O}_{2}}\]
done
clear
View Answer play_arrow
Viroids known to cause disease in plants only
A)
arc larger than virus
done
clear
B)
can replicate in cell free medium
done
clear
C)
have no protein coat
done
clear
D)
have double stranded RNA as genetic material
done
clear
View Answer play_arrow
Asexual spores are generally absent in
A)
Algal fungi
done
clear
B)
Club fungi
done
clear
C)
Sac fungi
done
clear
D)
Conjugation fungi
done
clear
View Answer play_arrow
Select correct with respect to Euglenoids
A. Fresh water habitats
B. Produce auxospores
C. lias pigment Astaxanthin
D. Indicate sewage pollution
E. Have ridge or frustule
F. Chl. a, b and carotenoids are photosynthetic pigment
A)
B, C, D are correct
done
clear
B)
C, D, E are correct
done
clear
C)
A, D, E are correct
done
clear
D)
A, C, F are correct
done
clear
View Answer play_arrow
With respect to five kingdom classification select the option which correctly fills up the blanks in the given table
Character Cell wall Body organs
Monera (A) Cellular
Protista Present Cellular
Fungi Present (C)
Plantae (B) Tissue/Organ
A)
A - Cellulosic, B - Cellulose + Pectin, C - Loose tissue
done
clear
B)
A. - Peptidoglycan, B - Non cellulosic, C - Tissue organ
done
clear
C)
A - Polysaccharide, B Cellulose, C Loose tissue
done
clear
D)
A - Sugar + Amino acid, B - Present in some, C- Multicellular
done
clear
View Answer play_arrow
Select the correct statement with respect to Rhodophyccae.
A)
Internal fertilization and embryo formation are present
done
clear
B)
Non sulphated phycocolloids are present in cell Wall
done
clear
C)
No flagellated stage throughout the life cycle
done
clear
D)
Do not reproduce through asexual spore
done
clear
View Answer play_arrow
The position of youngest secondary phloem is
A)
Just outside the pith
done
clear
B)
Just inside the vascular cambium
done
clear
C)
Just outside the vascular cambium
done
clear
D)
Just inside the pericyde
done
clear
View Answer play_arrow
An elaborate mechanism of spore dispersal is found in
A)
Anthoceros
done
clear
B)
Riccia
done
clear
C)
Anthocerotoc
done
clear
D)
Funaria
done
clear
View Answer play_arrow
Which of these are features of solan aceae family are correct?
A. Racemose type of inflorescence B. Epipetalous stamen C. Zygomorphic flowers D. Marginal placentation E. Many ovules and swollen placenta F. Berry or capsule fruit
A)
E, F, B
done
clear
B)
A, B, C
done
clear
C)
D, E, F
done
clear
D)
B, F, E
done
clear
View Answer play_arrow
Distribution of living pteridophytes is restricted to narrow geographical regions because
A)
they have vascular tissue in gametophytes
done
clear
B)
they have diplontic life cycle
done
clear
C)
gametophyte is non vascular
done
clear
D)
gametophyte is not free living
done
clear
View Answer play_arrow
Gene therapy replaces faculty gene by normal gene, one of the following disease is treated by gene therapy
A)
AIDS
done
clear
B)
Leukemia
done
clear
C)
SCID
done
clear
D)
Haemophilia
done
clear
View Answer play_arrow
Most commonly used vector for cloning genes in plants
A)
pUC19
done
clear
B)
pBR322
done
clear
C)
Retro virus
done
clear
D)
Ti plasmid
done
clear
View Answer play_arrow
Select the answer with incorrect matching of disease, causative agent and investigation
A)
Tuberculosis Bacterial Mantouxtest
done
clear
B)
Syphilis test Bacterial Wasserman
done
clear
C)
Typhoid Bacterial Widaltest
done
clear
D)
Diphtheria Bacterial Dick test
done
clear
View Answer play_arrow
Which of following technique is not used to detect cancer of internal organs?
A)
Radiography
done
clear
B)
Computed tomography
done
clear
C)
Electroencephalography
done
clear
D)
Magnetic resonance imaging
done
clear
View Answer play_arrow
Which of the following are involved in the erythropoiesis in an adult body?
[A] Bone marrow [B] Kidneys [C] Diet [D] Liver [E] Spleen
A)
A & C only
done
clear
B)
A, B & C
done
clear
C)
A, B, D, E
done
clear
D)
D & E only
done
clear
View Answer play_arrow
Select the answer with correct matching
A)
IgA Dimer - in all our body secretions
done
clear
B)
IgD Dimer - contains 'J' chain
done
clear
C)
IgM Pentamer - First to be secreted during secondary response
done
clear
D)
IgG Monomer - Active immunity to developing foetus
done
clear
View Answer play_arrow
Natural silk fibre is chemically a protein, it's a product of
A)
Salivary gland of the larva
done
clear
B)
Salivary gland of the adult male
done
clear
C)
Salivary gland of the adult female
done
clear
D)
Cuticle of the adult
done
clear
View Answer play_arrow
Peculiar birds from Galapagos islands that were named Darwin finches, are excellent example of
A)
Seasonal migration
done
clear
B)
Adaptive radiation
done
clear
C)
Brood parasitism
done
clear
D)
Connecting links
done
clear
View Answer play_arrow
Biogeographically earth been divided into six realms. All are separated by oceans, except
A)
Oriental and Australin
done
clear
B)
Palacarctic and Oriental
done
clear
C)
Nearctic and Palaearctic
done
clear
D)
Neotropical and ethopian
done
clear
View Answer play_arrow
The fall in number of sperms per ml of semen leads to infertility. What are the other reasons that may cause infertility, except?
A)
Cryptorchidism
done
clear
B)
Gonadotropin deficiency
done
clear
C)
Infection like mumps after puberty
done
clear
D)
Inguinal hernia
done
clear
View Answer play_arrow
Select the incorrect combination, with respect to phyla of animal kingdom
A)
Phylum-Cnidana, Organisation-Tissue Symmetry-Radial Coclom- Absent
done
clear
B)
Phylum-Porifora, Organisation-Cellular Symmetry-Generally radial Coclom-Absent
done
clear
C)
Phylum-Platyhelminthes, Organisation-Organ Symmetry-Bilateral Coclom-Absent
done
clear
D)
Phylum-Ascahelminthes, Organisation-Organ system Symmetry-Bilateral Coclom-Pseudo coelomate
done
clear
View Answer play_arrow
Select incorrect match
A)
Conus arteriosus - Dorsal side of frog's heart
done
clear
B)
Integumentary nephridia - Segment 3rd to last
done
clear
C)
In female cockroach - 7th sternum is boat shaped
done
clear
D)
Blood glands in earth worm - 4th, 5th and 6th segments
done
clear
View Answer play_arrow
Select the correct set
A)
Mesodermal - Microglia cells of C.N.S.
done
clear
B)
Ligaments and tendons - Irregular dense tissue
done
clear
C)
Uninucleated with central nucleus - Striated muscles
done
clear
D)
Pharynx, Vagina - Stratified keratini sed squamous epithelium
done
clear
View Answer play_arrow
Select correct with respect to lymph
A)
It never clots
done
clear
B)
No RBC so it is colourless
done
clear
C)
It has more protein than blood
done
clear
D)
Among various types of leucocytes, the majority are -- basophils.
done
clear
View Answer play_arrow
Choose the incorrect one, with respect to comparison between Red and White muscles
A)
Feature White Muscle Red Muscle Diametre Smaller Bigger
done
clear
B)
Feature White Muscle Red Muscle Mitochondria Less in Number More in number
done
clear
C)
Feature White Muscle Red Muscle Blood capillaries Less More
done
clear
D)
Feature White Muscle Red Muscle Sarcoplasmic reticulam Less More
done
clear
View Answer play_arrow
Choose incorrect match
A)
Castle intrinsic factor - Oxyntic cell
done
clear
B)
\[HCl\]- Oxyntic cell
done
clear
C)
Helicobacterium - Peptic ulcer
done
clear
D)
Protein deficiency - Only marasmus
done
clear
View Answer play_arrow
A viscous, enzyme free, alkaline and watery fluid is secreted by \[(\underline{X})\] under the stimulus of \[(\underline{Y})\] and \[(\underline{X}),\] \[(\underline{Y})\] select \[(\underline{X})\], \[(\underline{Y})\] and \[(\underline{Z})\]. respectively.
A)
Crypts oflieberkuhn, Vagus, CCK
done
clear
B)
Pancreas, Adrenaline, Secretin
done
clear
C)
Burners gland, Vagus, Gastrointestinal hormone
done
clear
D)
Gastric glands, Histamine, Serotonin
done
clear
View Answer play_arrow
Select correct match
A)
Mountain sickness - \[{{O}_{2}}\] deficiency at high altitude - Fatigue, Tachychardia and Tachypnoea
done
clear
B)
Tidal volume - 500 ml - value is more in male than in female.
done
clear
C)
Surfactant - Phospholipid - Deficiency cause emphysema.
done
clear
D)
Smoking - Bronchiogenic carcinoma - most common cancer in male in India.
done
clear
View Answer play_arrow
Select the correct term for following disorders of circulatory system
A. When the heart muscle is suddenly damaged by inadequate blood supply B. When heart is not pumping blood effectively enough to meet the need of body C. When the heart stops beating D. When cardiac impulse is not passed to the ventricles properly due to defect of conducting system of heart
A)
Heart block, Heart failure, Cardiac arrest, Heart attack
done
clear
B)
Heart attack, Heart failure, Cardiac arrest, Heart block
done
clear
C)
Heart attack. Heart block, Heart failure, Cardiac arrest
done
clear
D)
Heart attack. Heart failure, Heart block, Cardiac arrest
done
clear
View Answer play_arrow
Select incorrect for gene therapy
A)
Permanent treatment of SCID is enzyme replacement therapy
done
clear
B)
Lymphocytes from patients blood are cultured
done
clear
C)
A functional ADA cDNA is introduced into the cultured lymphocytes
done
clear
D)
Lymphocytes then introduced into body of patients
done
clear
View Answer play_arrow
Select age related disorder which is characterized by reduced bone mass and increased chance of fractures
A)
Osteoporosis
done
clear
B)
Tetany
done
clear
C)
Muscular dystrophy
done
clear
D)
My asthenia gravis
done
clear
View Answer play_arrow
The digestive enzyme responsible for digestion of disaccharide only include
A)
Erepsin, Steapsin, Entcrokinase
done
clear
B)
Sucrase, Maltase, Lactase
done
clear
C)
Pepsin, Trypsin, Amylospsin
done
clear
D)
Amylopsin, Ptyalin, Sucrase
done
clear
View Answer play_arrow
A phenomenon when two species of different ancestry come close and resemble each other as a result of adaptation, it is termed as
A)
Mega evolution
done
clear
B)
Adaptive evolution
done
clear
C)
Divergent evolution
done
clear
D)
Convergent evolution
done
clear
View Answer play_arrow
Polypeptide chain absent in genetically engineered insulin is/are
A)
C - Polypeptide
done
clear
B)
B - Polypeptide
done
clear
C)
A - Polypeptide
done
clear
D)
A and B Polypeptide
done
clear
View Answer play_arrow
Blood vessels near a wound dilate and become more permeable in response to which substance released from damaged cells
A)
Pyrogens
done
clear
B)
Antibodies
done
clear
C)
Histamines
done
clear
D)
Antihistamines
done
clear
View Answer play_arrow
Human and Chimpanzee exhibits close resembiance common origin of these two is best shown by
A)
Opposable pollex
done
clear
B)
Cranial capacity
done
clear
C)
Stereoscopic vision
done
clear
D)
Banding pattern in chromosome 3 and 6
done
clear
View Answer play_arrow
Which of the following is correct?
A)
P - wave represents of depolarization of atria
done
clear
B)
QRS complex represents depolarisation of ventricle
done
clear
C)
The end of T-wave marks the end of systole
done
clear
D)
All of these
done
clear
View Answer play_arrow
\[\alpha -1\] Antitrypsin enzyme produced by genetic engineering as a cure for disease like
A)
Emphysema
done
clear
B)
Haemophilia
done
clear
C)
Leukemia
done
clear
D)
Asbestosis
done
clear
View Answer play_arrow
Select the answer with correct matching
A)
Round worm - Male longer than female - Pseudocoeclomate
done
clear
B)
Tapeworm - Eucoelomate - Segmentation
done
clear
C)
L - donovani - Sand fly - Sleepsing - Sickness
done
clear
D)
P - falciparum - Sporozoan - Relapse of malarial fever absent;
done
clear
View Answer play_arrow
Outer membrane, Inner membrane matrix and Elementary particles are part of
A)
Plastid
done
clear
B)
Mitoefaojadria
done
clear
C)
Ribosome
done
clear
D)
GERL
done
clear
View Answer play_arrow
Metapha.se of a mitotically dividing cell is characterized by
A)
Duplication of DNA and centrosome
done
clear
B)
Splitting of centromere
done
clear
C)
Equatorial movement of chromosome
done
clear
D)
Presence of ER, golgi complex, nuclear membrane
done
clear
View Answer play_arrow
Essential element that activates enzyme catal are
A)
\[Mg\]
done
clear
B)
N
done
clear
C)
\[Fe\]
done
clear
D)
K
done
clear
View Answer play_arrow
Emerson's photosynthetic enhancement effect proves
A)
Occurrence of two inter-connected photo-system
done
clear
B)
Photosynthesis as eight quanta process
done
clear
C)
Occurrence of single photo-system in light phase
done
clear
D)
None of these
done
clear
View Answer play_arrow
How many molecules of ATP are formed through ETS only when two molecules of 2 KGA undergo complete oxidation
A)
17
done
clear
B)
8
done
clear
C)
11
done
clear
D)
16
done
clear
View Answer play_arrow
A plant hormone that is used to increase length of stalk in grape
A)
Ethylene
done
clear
B)
Gibberellins
done
clear
C)
Auxins
done
clear
D)
ABA
done
clear
View Answer play_arrow
Anther wall layers that performs the function of protection and help in dehiscence of anther to release the pollen are
A)
Tapetum and middle layers
done
clear
B)
Epidermis, endothecium and middle layers
done
clear
C)
Only endothecium
done
clear
D)
Epidermis and endothecium
done
clear
View Answer play_arrow
Sex determination in grasshopper is
A)
ZZ - ZO type
done
clear
B)
XX - XY type
done
clear
C)
XX - XO type
done
clear
D)
ZW - ZZ type
done
clear
View Answer play_arrow
Find the correct match
A)
Chilly - Pusa komal
done
clear
B)
Flat bean - Pusa sawani
done
clear
C)
Mange - Pusa swarnim
done
clear
D)
Rape seed - Pusa Gaurav
done
clear
View Answer play_arrow
Total number of hot spots present in India is
A)
5
done
clear
B)
2
done
clear
C)
1
done
clear
D)
3
done
clear
View Answer play_arrow
Total landscape or geographical diversity is called
A)
P diversity
done
clear
B)
a diversity
done
clear
C)
y diversity
done
clear
D)
All are correct
done
clear
View Answer play_arrow
Select correct option with respect to \[{{N}_{2}}\] base
A)
A - Guanine, B - Thymine
done
clear
B)
A - Adenine, B - Uracil
done
clear
C)
A - Adenine, B - Thymine
done
clear
D)
A - Guanine, B - Uridine
done
clear
View Answer play_arrow
Which of these is incorrectly matched?
A)
Creative phosphate - Energy stored in muscles
done
clear
B)
Myosin - Contractile protein
done
clear
C)
Tropomyosin - Regulatory protein
done
clear
D)
Troponine - Ca-binding protein in smooth muscles
done
clear
View Answer play_arrow
Select incorrect match
A)
Hinge joint - Atlanta axial joint
done
clear
B)
Strongest cartilage - Fibrous cartilage
done
clear
C)
Median valleolus - Tibia
done
clear
D)
Pivot joint - Superior radio ulnar joint
done
clear
View Answer play_arrow
In the presence of ADH the greatest fraction of glomerular filterate is absorbed in the
A)
Collecting duct
done
clear
B)
D.C.T.
done
clear
C)
P.C.T.
done
clear
D)
Loop of Henie
done
clear
View Answer play_arrow
Male secondary sexual characters start developing with the onset of puberty, which of the following is not a male secondary sexual characteristics
A)
An enlarged penis
done
clear
B)
Increased fat in buttocks
done
clear
C)
Beard
done
clear
D)
Deep voice
done
clear
View Answer play_arrow
Which of the following hormone is synthesized in brain, gut as well as in pancreas?
A)
Somatostatin
done
clear
B)
Insulin
done
clear
C)
Dopamine
done
clear
D)
Vasopressin
done
clear
View Answer play_arrow
Which of the following group will show divergent evolution?
A)
Horn of cattle and tail of horse
done
clear
B)
Forelimb of whale, tiger, bat, human
done
clear
C)
Tentacles of hydra and arms of starfish
done
clear
D)
Wings of birds and butterfly
done
clear
View Answer play_arrow
Bulk of released from body tissue into the blood present as
A)
70% carbamino haemoglobin
done
clear
B)
Bicarbonates in blood plasma and RBC
done
clear
C)
Free \[C{{O}_{2}}\] in blood plasma
done
clear
D)
Carbamino haemoglobin in RBC
done
clear
View Answer play_arrow
What will be the \[P{{O}_{2}}\] and \[PC{{O}_{2}}\] in the atmospheric air compared to those in the alveolar air
A)
Both higher
done
clear
B)
Both lesser
done
clear
C)
\[P{{O}_{2}}\] higher, \[PC{{O}_{2}}\] lesser
done
clear
D)
\[P{{O}_{2}}\] lesser, \[PC{{O}_{2}}\] higher
done
clear
View Answer play_arrow
Select incorrect match
A)
6800 ml - Total lung capacity
done
clear
B)
2500 ml - Functional residual capacity
done
clear
C)
3500 ml -Inspiratory capacity
done
clear
D)
4500 ml - Vital capacity
done
clear
View Answer play_arrow
Select option not associated with epithelium is
A)
compactly packed cells with little I space
done
clear
B)
helps in protection, secretion, absorption, respiration etc.
done
clear
C)
form linings of external and internal surface
done
clear
D)
Highly vascularized
done
clear
View Answer play_arrow
Deficiency of Vit. \[{{B}_{7}}\] caused
A)
Pellagra
done
clear
B)
Anaemia
done
clear
C)
Neuritis
done
clear
D)
Dry skin
done
clear
View Answer play_arrow
Beriberi is due to deficiency of
A)
Iron
done
clear
B)
Vit. \[{{B}_{7}}\]
done
clear
C)
\[{{B}_{12}}\]
done
clear
D)
Vit. C
done
clear
View Answer play_arrow
ANF causes
A)
Vasodilation and increased B.P.
done
clear
B)
Vaso constriction
done
clear
C)
Vaso dilation and decreased B.P.
done
clear
D)
Reduced B.P. only
done
clear
View Answer play_arrow
China rose belongs to family
A)
Solanaceale
done
clear
B)
Malvaceae
done
clear
C)
Rubiaceael
done
clear
D)
Crucifereae
done
clear
View Answer play_arrow

 A is
A is