-
question_answer1)
Amides may be converted into amines by reaction named after [CPMT 1974; MP PET 1992; CBSE PMT 1999]
A)
Perkin done
clear
B)
Claisen done
clear
C)
Hoffmann done
clear
D)
Kolbe done
clear
View Solution play_arrow
-
question_answer2)
Reaction \[C{{H}_{3}}CON{{H}_{2}}\xrightarrow{NaOBr}\] gives [CPMT 1983, 93, 97]
A)
\[C{{H}_{3}}Br\] done
clear
B)
\[C{{H}_{4}}\] done
clear
C)
\[C{{H}_{3}}COBr\] done
clear
D)
\[C{{H}_{3}}N{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer3)
Acetamide is treated separately with the following reagents. Which would give methyl amine [IIT 1983; CPMT 1988, 94; MP PET 1993; MP PMT 1996; AIIMS 1998]
A)
\[PC{{l}_{5}}\] done
clear
B)
\[NaOH+B{{r}_{2}}\] done
clear
C)
Sodalime done
clear
D)
Hot conc. \[{{H}_{2}}S{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer4)
The amine formed from an amide by means of bromine and alkali has
A)
Same number of C atoms as that of amide done
clear
B)
One less C atom than that of amide done
clear
C)
One more C atom than that of amide done
clear
D)
Two more C atoms than that of amide done
clear
View Solution play_arrow
-
question_answer5)
\[C{{H}_{3}}CN\xrightarrow{Na+{{C}_{2}}{{H}_{5}}OH}X\] The compound X is [MP PMT 1983; BHU 1984]
A)
\[C{{H}_{3}}CON{{H}_{2}}\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}N{{H}_{2}}\] done
clear
C)
\[{{C}_{2}}{{H}_{6}}\] done
clear
D)
\[C{{H}_{3}}NHC{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer6)
Ethylamine can be prepared by the action of bromine and caustic potash on [CPMT 1994]
A)
Acetamide done
clear
B)
Propionamide done
clear
C)
Formamide done
clear
D)
Methyl cyanide done
clear
View Solution play_arrow
-
question_answer7)
Ethylamine can be obtained by the [CPMT 1985]
A)
Action of \[N{{H}_{3}}\] on ethyl iodide done
clear
B)
Action of \[N{{H}_{3}}\] on ethyl alcohol done
clear
C)
Both A and B done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer8)
Aniline is usually purified by [CPMT 1983, 93; JIPMER 1997]
A)
Steam distillation done
clear
B)
Simple distillation done
clear
C)
Vacuum distillation done
clear
D)
Extraction with a solvent done
clear
View Solution play_arrow
-
question_answer9)
Reduction of nitroalkanes yields
A)
Acid done
clear
B)
Alcohol done
clear
C)
Amine done
clear
D)
Diazo compounds done
clear
View Solution play_arrow
-
question_answer10)
Acetamide changes into methylamine by
A)
Hofmann bromamide reaction done
clear
B)
Hofmann reaction done
clear
C)
Friedel-Craft's reaction done
clear
D)
Hinsberg reaction done
clear
View Solution play_arrow
-
question_answer11)
When methyl iodide is heated with ammonia, the product obtained is
A)
Methylamine done
clear
B)
Dimethylamine done
clear
C)
Trimethylamine done
clear
D)
A mixture of the above three amines done
clear
View Solution play_arrow
-
question_answer12)
Acetanilide can be prepared from aniline and which of the following
A)
Ethanol done
clear
B)
Acetaldehyde done
clear
C)
Acetone done
clear
D)
Acetic anhydride done
clear
View Solution play_arrow
-
question_answer13)
Reduction of nitroalkanes in neutral medium (e.g. Zn / \[N{{H}_{4}}Cl\]) forms mainly
A)
\[R-N{{H}_{2}}\] done
clear
B)
\[R-NHOH\] done
clear
C)
\[R-N=N-Cl\] done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer14)
Nitrosobenzene can be prepared by oxidizing aniline from
A)
\[{{H}_{2}}S{{O}_{4}}\] done
clear
B)
\[{{H}_{2}}S{{O}_{5}}\] done
clear
C)
\[{{H}_{2}}S{{O}_{3}}\] done
clear
D)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] done
clear
View Solution play_arrow
-
question_answer15)
The Hinsberg's method is used for
A)
Preparation of primary amines done
clear
B)
Preparation of secondary amines done
clear
C)
Preparation of tertiary amines done
clear
D)
Separation of amine mixtures done
clear
View Solution play_arrow
-
question_answer16)
Which one of the following compound gives a secondary amine on reduction
A)
Nitromethane done
clear
B)
Nitrobenzene done
clear
C)
Methyl isocyanide done
clear
D)
Methyl cyanide done
clear
View Solution play_arrow
-
question_answer17)
Chloropicrin is manufactured by the reaction between \[C{{l}_{2}},\,\,NaOH\] and
A)
Nitromethane done
clear
B)
Nitroethane done
clear
C)
Nitrophenol done
clear
D)
Nitrostyrene done
clear
View Solution play_arrow
-
question_answer18)
In the reaction \[R-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-OH\xleftarrow{{{H}_{3}}{{O}^{+}}}X\xrightarrow{[H]}RC{{H}_{2}}N{{H}_{2}};\] 'X' is [MP PMT 1990]
A)
Isonitrile done
clear
B)
Nitrile done
clear
C)
Nitrite done
clear
D)
Oxime done
clear
View Solution play_arrow
-
question_answer19)
When ethanol is mixed with ammonia and passed over alumina the compound formed is [CBSE PMT 1990]
A)
\[{{C}_{2}}{{H}_{5}}N{{H}_{2}}\] done
clear
B)
\[{{C}_{2}}{{H}_{4}}\] done
clear
C)
\[{{C}_{2}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}\] done
clear
D)
\[C{{H}_{3}}OC{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer20)
Which of the following reactions does not yield an amine [CPMT 1989, 93]
A)
\[RX+N{{H}_{3}}\xrightarrow{{}}\] done
clear
B)
\[RCH=NOH+[H]\underset{{{C}_{2}}{{H}_{5}}OH}{\mathop{\xrightarrow{Na}}}\,\] done
clear
C)
\[RCN+{{H}_{2}}O\xrightarrow{{{H}^{+}}}\] done
clear
D)
\[RCON{{H}_{2}}+4H\xrightarrow{LiAl{{H}_{4}}}\] done
clear
View Solution play_arrow
-
question_answer21)
Identify 'B' in the reaction Acetamide \[\underset{\Delta }{\mathop{\xrightarrow{{{P}_{2}}{{O}_{5}}}}}\,A\xrightarrow{4H}B\] [MP PET 1995]
A)
\[C{{H}_{3}}N{{H}_{2}}\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}N{{H}_{2}}\] done
clear
C)
\[C{{H}_{3}}CN\] done
clear
D)
\[C{{H}_{3}}COON{{H}_{4}}\] done
clear
View Solution play_arrow
-
question_answer22)
Which of the following gives primary amine on reduction [MP PMT 1995]
A)
\[C{{H}_{3}}-C{{H}_{2}}-\overset{O}{\mathop{\overset{||}{\mathop{N}}\,}}\,\to O\] done
clear
B)
\[C{{H}_{3}}-C{{H}_{2}}-O-N=O\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}N{{O}_{3}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer23)
Which of the following is converted into an alcohol on treatment with \[HN{{O}_{2}}\] [MP PET 1996; MP PMT 1999]
A)
Methyl amine done
clear
B)
Aniline done
clear
C)
Dimethyl amine done
clear
D)
Triethyl amine done
clear
View Solution play_arrow
-
question_answer24)
Which of the following gives RNC, when reacted with \[CHC{{l}_{3}}\] and KOH [MP PET 1996]
A)
\[RN{{H}_{2}}\] done
clear
B)
\[{{R}_{2}}NH\] done
clear
C)
\[{{R}_{3}}N\] done
clear
D)
\[{{R}_{4}}{{N}^{+}}C{{l}^{-}}\] done
clear
View Solution play_arrow
-
question_answer25)
When aniline reacts with \[NaN{{O}_{2}}\] and dil. HCl at \[{{0}^{o}}-{{5}^{o}}C,\] the product formed is [MP PMT 1996; AIIMS 1996]
A)
Nitroaniline done
clear
B)
Benzene diazonium chloride done
clear
C)
Benzene done
clear
D)
Trinitroaniline done
clear
View Solution play_arrow
-
question_answer26)
Starting from propanoic acid, the following reactions were carried out \[\text{Propanoic acid }\xrightarrow{SOC{{l}_{2}}}X\xrightarrow{N{{H}_{3}}}Y\xrightarrow{B{{r}_{2}}+KOH}Z\] What is the compound Z
A)
\[C{{H}_{3}}-C{{H}_{2}}-Br\] done
clear
B)
\[C{{H}_{3}}-C{{H}_{2}}-N{{H}_{2}}\] done
clear
C)
D)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-N{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer27)
In the reaction \[C{{H}_{3}}COOH\xrightarrow{PC{{l}_{5}}}(A)\xrightarrow{N{{H}_{3}}}(B)\xrightarrow{NaBrO}(C).\] the final product C is
A)
Ammonium acetate done
clear
B)
Acetamide done
clear
C)
Amino methane done
clear
D)
Ethanal done
clear
View Solution play_arrow
-
question_answer28)
In the following reaction, X is \[X\xrightarrow{\text{Bromination}}Y\xrightarrow{NaN{{O}_{2}}+HCl}Z\underset{{{C}_{2}}{{H}_{5}}OH}{\mathop{\xrightarrow{\text{Boiling}}}}\,\text{Tribromobenzene}\] [CPMT 1999]
A)
Benzoic acid done
clear
B)
Salicylic acid done
clear
C)
Phenol done
clear
D)
Aniline done
clear
View Solution play_arrow
-
question_answer29)
Which of the following reactions will not give primary amine [CPMT 1999]
A)
\[C{{H}_{3}}CON{{H}_{2}}\xrightarrow{KOH.B{{r}_{2}}}\] done
clear
B)
\[C{{H}_{3}}CN\xrightarrow{LiAl{{H}_{4}}}\] done
clear
C)
\[C{{H}_{3}}NC\xrightarrow{LiAl{{H}_{4}}}\] done
clear
D)
\[C{{H}_{3}}CON{{H}_{2}}\xrightarrow{LiAl{{H}_{4}}}\] done
clear
View Solution play_arrow
-
question_answer30)
Carbylamine reaction is given by [BHU 1996; EAMCET 1990]
A)
\[{{1}^{o}}\] amine done
clear
B)
\[{{3}^{o}}\] amine done
clear
C)
\[{{2}^{o}}\] amine done
clear
D)
Quarternary salts done
clear
View Solution play_arrow
-
question_answer31)
The reaction \[{{C}_{6}}{{H}_{5}}N{{H}_{2}}+CHC{{l}_{3}}+3KOH\to {{C}_{6}}{{H}_{5}}NC+3KCl+2{{H}_{2}}O\] is known as [BHU 1996]
A)
Carbylamine reaction done
clear
B)
Reimer-Tiemann reaction done
clear
C)
Kolbe reaction done
clear
D)
Hofmann's degradation done
clear
View Solution play_arrow
-
question_answer32)
\[C{{H}_{3}}CON{{H}_{2}}\xrightarrow{Na+ROH}Z+{{H}_{2}}O.\] What is Z ? [CPMT 1996]
A)
\[C{{H}_{3}}C{{H}_{2}}N{{H}_{2}}\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}NC\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{3}}\] done
clear
D)
\[N{{H}_{2}}CON{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer33)
Which of the following reacts with chloroform and a base to form phenyl isocyanide [AFMC 1997]
A)
Aniline done
clear
B)
Phenol done
clear
C)
Benzene done
clear
D)
Nitrobenzene done
clear
View Solution play_arrow
-
question_answer34)
Aromatic primary amine when treated with cold \[HN{{O}_{2}}\] gives [Pb. CET 2002; DCE 1999]
A)
Benzyl alcohol done
clear
B)
Nitro benzene done
clear
C)
Benzene done
clear
D)
Diazonium salt done
clear
View Solution play_arrow
-
question_answer35)
Which of the following compound is the strongest base [BHU 1999]
A)
Ammonia done
clear
B)
Aniline done
clear
C)
Methylamine done
clear
D)
N-methyl aniline done
clear
View Solution play_arrow
-
question_answer36)
Nitrobenzene combines with hydrogen in the presence of platinum to produce [BHU 1999]
A)
Toluene done
clear
B)
Benzene done
clear
C)
Aniline done
clear
D)
Azobenzene done
clear
View Solution play_arrow
-
question_answer37)
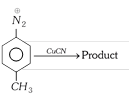
The product is [RPET 2000]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer38)
Ethyl amine on heating with \[C{{S}_{2}}\] in presence of \[HgC{{l}_{2}}\] forms [MP PET 2000]
A)
\[{{C}_{2}}{{H}_{5}}NCS\] done
clear
B)
\[{{({{C}_{2}}{{H}_{5}})}_{2}}S\] done
clear
C)
\[{{({{C}_{2}}{{H}_{5}})}_{2}}CS\] done
clear
D)
\[{{C}_{2}}{{H}_{5}}{{(CS)}_{2}}\] done
clear
View Solution play_arrow
-
question_answer39)
Which of the following reacts with \[NaN{{O}_{2}}+HCl\] to give phenol [MP PMT 2000]
A)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}NHC{{H}_{3}}\] done
clear
B)
\[{{(C{{H}_{3}})}_{2}}NH\] done
clear
C)
\[C{{H}_{3}}N{{H}_{2}}\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer40)
Which of the following reactions give \[RCON{{H}_{2}}\] [Roorkee 2000]
A)
\[R-C\equiv N+{{H}_{2}}O\xrightarrow{HCl}\] done
clear
B)
\[RCOON{{H}_{4}}\xrightarrow{\,\,\,\,\,\text{heat}\,\,\,\,\,\,\,\,\,\,\,\,}\] done
clear
C)
\[R-COCl+N{{H}_{3}}\xrightarrow{\,\,\,\,\,\,\,\,\,\,\,}\] done
clear
D)
\[{{(RCO)}_{2}}O+N{{H}_{3}}\xrightarrow{\,\,\,\,\,\,\,\,\,\,\,\,}\] done
clear
View Solution play_arrow
-
question_answer41)
When chlorobenzene is treated with \[N{{H}_{3}}\] in presence of \[C{{u}_{2}}O\] in xylene at 570 K. The product obtained is [Pb. PMT 2000]
A)
Benzylamine done
clear
B)
Diazonium salt done
clear
C)
Schiff?s base done
clear
D)
Aniline done
clear
View Solution play_arrow
-
question_answer42)
Nitrobenzene can be prepared from benzene by using a mixture of conc.\[HN{{O}_{3}}\] and conc.\[{{H}_{2}}S{{O}_{4}}\]. In the nitrating mixture, \[HN{{O}_{3}}\] acts as a [BHU 2001]
A)
Base done
clear
B)
Acid done
clear
C)
Catalyst done
clear
D)
Reducing agent done
clear
View Solution play_arrow
-
question_answer43)
The rate determining step for the preparation of nitrobenzene from benzene is [AIIMS 2001]
A)
Removal of \[\overset{+\,\,\,\,\,\,}{\mathop{N{{O}_{2}}}}\,\] done
clear
B)
Removal of \[\overset{+\,\,\,\,\,\,}{\mathop{N{{O}_{3}}}}\,\] done
clear
C)
Formation of \[\overset{+\,\,\,\,\,\,}{\mathop{N{{O}_{2}}}}\,\] done
clear
D)
Formation of \[\overset{+\,\,\,\,\,\,}{\mathop{N{{O}_{3}}}}\,\] done
clear
View Solution play_arrow
-
question_answer44)
In this reaction \[{{C}_{6}}{{H}_{5}}N{{H}_{2}}+HCl+NaN{{O}_{2}}\to X\]. Product X is [RPMT 2002; AFMC 2002]
A)
Aniline hydrochloride done
clear
B)
Nitro aniline done
clear
C)
Benzenediazonium chloride done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer45)
The diazonium salts are the reaction products in presence of excess of mineral acid with nitrous acid and [MP PET 2002]
A)
Primary aliphatic amine done
clear
B)
Secondary aromatic amine done
clear
C)
Primary aromatic amine done
clear
D)
Tertiary aliphatic amine done
clear
View Solution play_arrow
-
question_answer46)
In acid medium nitrobenzene is reduced to aniline as shown in the reaction \[{{C}_{6}}{{H}_{5}}-N{{O}_{2}}+6[H]\to {{C}_{6}}{{H}_{5}}-N{{H}_{2}}+2{{H}_{2}}O\]The reducing agent used in this reaction is ??. [Orissa JEE 2002]
A)
\[LiAl{{H}_{4}}\] done
clear
B)
\[Sn/HCl\] done
clear
C)
Na/alcohol done
clear
D)
\[{{H}_{2}}/Ni\] done
clear
View Solution play_arrow
-
question_answer47)
When aniline is treated with sodium nitrite and hydrochloric acid at 0oC, it gives [Orissa JEE 2003]
A)
Phenol and \[{{N}_{2}}\] done
clear
B)
Diazonium salt done
clear
C)
Hydrazo compound done
clear
D)
No reaction takes place done
clear
View Solution play_arrow
-
question_answer48)
\[C{{H}_{3}}N{{O}_{2}}\xrightarrow{Sn+HCl}C{{H}_{3}}X,\] the ?X? contain [CPMT 2003]
A)
\[-N{{H}_{2}}\] done
clear
B)
\[-\,COOH\] done
clear
C)
\[-\,CHO\] done
clear
D)
\[{{(C{{H}_{3}}CO)}_{2}}O\] done
clear
View Solution play_arrow
-
question_answer49)
In the series of reaction \[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\underset{0-5{}^\circ C}{\mathop{\xrightarrow{NaN{{O}_{2}}/HCl}}}\,X\underset{C{{H}_{2}}O}{\mathop{\xrightarrow{HN{{O}_{2}}}}}\,Y+{{N}_{2}}+HCl\] X and Y are respectively [EAMCET 2003]
A)
\[{{C}_{6}}{{H}_{5}}-N=N-{{C}_{6}}{{H}_{5}}\,,\,{{C}_{6}}{{H}_{5}}N_{2}^{\oplus }C{{l}^{\Theta }}\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}N_{2}^{\oplus }C{{l}^{\Theta }},\,{{C}_{6}}{{H}_{5}}-N=N-{{C}_{6}}{{H}_{5}}\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}N_{2}^{\oplus }C{{l}^{\Theta }},\,{{C}_{6}}{{H}_{5}}N{{O}_{2}}\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}N{{O}_{2}},\,{{C}_{6}}{{H}_{6}}\] done
clear
View Solution play_arrow
-
question_answer50)
Aromatic nitriles (ArCN) are not prepared by reaction [AIIMS 2004]
A)
ArX + KCN done
clear
B)
\[ArN_{2}^{+}+CuCN\] done
clear
C)
\[ArCON{{H}_{2}}+{{P}_{2}}{{O}_{5}}\] done
clear
D)
\[ArCON{{H}_{2}}+SOC{{l}_{ 2}}\] done
clear
View Solution play_arrow
-
question_answer51)
An organic amino compound reacts with aqueous nitrous acid at low temperature to produce an oily nitroso amine. The compound is : [DCE 2003]
A)
\[C{{H}_{3}}N{{H}_{2}}\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}N{{H}_{2}}\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}NH.C{{H}_{2}}C{{H}_{3}}\] done
clear
D)
\[{{(C{{H}_{3}}C{{H}_{2}})}_{3}}\] done
clear
View Solution play_arrow
-
question_answer52)
Azo-dyes are prepared from : [BHU 2004; Pb. CET 2001]
A)
Aniline done
clear
B)
Salicylic acid done
clear
C)
Benzaldehyde done
clear
D)
Chlorobenzene done
clear
View Solution play_arrow
-
question_answer53)
Gabriel's phthalimide synthesis is used for the preparation of [CPMT 1982; DPMT 1983]
A)
Primary aromatic amine done
clear
B)
Secondary amine done
clear
C)
Primary aliphatic amine done
clear
D)
Tertiary amine done
clear
View Solution play_arrow
-
question_answer54)
For the preparation of p-nitroiodobenzene from p-nitroaniline, the best method is [Orissa JEE 2005]
A)
\[NaN{{O}_{2}}/HCl\] followed by \[KI\]. done
clear
B)
\[NaN{{O}_{2}}/HCl\] followed by \[CuCN\] done
clear
C)
\[LiAl{{H}_{4}}\] followed by \[{{I}_{2}}\] done
clear
D)
\[NaB{{H}_{4}}\] followed by \[{{I}_{2}}\] done
clear
View Solution play_arrow
-
question_answer55)
\[KCN\] reacts readily to give a cyanide with [J & K 2005]
A)
Ethyl alcohol done
clear
B)
Ethyl bromide done
clear
C)
Bromobenzene done
clear
D)
Chlorobenzene done
clear
View Solution play_arrow
 The product is [RPET 2000]
The product is [RPET 2000]  done
clear
done
clear
 done
clear
done
clear
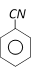 done
clear
done
clear
