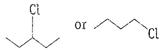 Or
Write the structures and names of the compounds formed
when compound A with molecular formula
Or
Write the structures and names of the compounds formed
when compound A with molecular formula 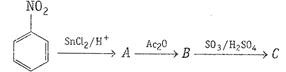
|
|
Time (min) |
| 0.400 0.289 0.209 0.151 0.109 | 0.00 20.00 40.00 60.00 80.00 |
You need to login to perform this action.
You will be redirected in
3 sec
