-
question_answer1)
Molar conductance of a 1.5 M solution of an electrolyte is found to be \[138.9\text{ }S\text{ }c{{m}^{2}}\]. The specific conductance of this solution is
A)
\[0.208\text{ S}\,\text{c}{{\text{m}}^{-1}}\] done
clear
B)
\[0.102\text{ S}\,\text{c}{{\text{m}}^{-1}}\] done
clear
C)
\[0.320\text{ S}\,\text{c}{{\text{m}}^{-1}}\] done
clear
D)
\[0.152\text{ S}\,\text{c}{{\text{m}}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer2)
| Given the following half reactions: |
| \[{{A}^{2+}}+{{e}^{-}}\to {{A}^{+}},{{E}^{o}}=+1.70V\] |
| \[{{B}^{2+}}+2{{e}^{-}}\to {{B}^{+}},{{E}^{o}}=+0.60V\] |
| \[{{C}^{3+}}+{{e}^{-}}\to {{C}^{2+}},{{E}^{o}}=+0.90V\] |
| The only reaction that could be used in titration would be |
A)
\[{{A}^{+}}\] with \[{{B}^{4+}}\] done
clear
B)
\[{{A}^{+}}\] with \[{{C}^{3+}}\] done
clear
C)
\[{{A}^{2+}}\] with \[{{C}^{2+}}\] done
clear
D)
\[{{C}^{2+}}\] with \[{{B}^{4+}}\] done
clear
View Solution play_arrow
-
question_answer3)
| Emf of the cell |
| \[Ni|N{{i}^{2+}}(0.1\,M)||A{{u}^{3+}}\,(1.0\,M)|\,Au\] will be |
| \[(E_{Ni/N{{i}^{2+}}}^{{}^\circ }=0.25,\,E_{Au/A{{u}^{3+}}}^{{}^\circ }=1.5\,V)\] |
A)
1.75 V done
clear
B)
\[+\]1.7795 V done
clear
C)
\[+\]0.7795 V done
clear
D)
\[-\]1.7795 V done
clear
View Solution play_arrow
-
question_answer4)
\[Zn|Z{{n}^{+2}}({{C}_{1}})||Z{{n}^{+2}}({{C}_{2}})|Zn\] For this cell \[\Delta G\] is negative if
A)
\[{{C}_{1}}={{C}_{2}}\] done
clear
B)
\[{{C}_{1}}>{{C}_{2}}\] done
clear
C)
\[{{C}_{2}}>{{C}_{1}}\] done
clear
D)
Both [a] and [c] are correct done
clear
View Solution play_arrow
-
question_answer5)
Equivalent conductivity of \[BaC{{l}_{2}},{{H}_{2}}S{{O}_{4}}\] and HCl are \[{{y}_{1}},{{y}_{2}}\] and \[{{y}_{3}}S\,c{{m}^{-1}}e{{q}^{-1}}\] at infinite dilution. If conductivity of saturated \[BaS{{O}_{4}}\] solution is \[y\,S\,c{{m}^{-1}},\] then find \[{{K}_{sp}}\] of \[BaS{{O}_{4}}\].
A)
\[\frac{{{y}^{2}}}{{{({{y}_{1}}+{{y}_{2}}-{{y}_{3}})}^{2}}}\] done
clear
B)
\[\frac{2.5{{y}^{-2}}}{{{({{y}_{1}}+{{y}_{2}}-{{y}_{3}})}^{2}}}\] done
clear
C)
\[\frac{500}{{{y}_{1}}+{{y}_{2}}-{{y}_{3}}}\] done
clear
D)
\[\frac{2.5\times {{10}^{5}}{{y}^{2}}}{{{({{y}_{1}}+{{y}_{2}}-{{y}_{3}})}^{2}}}\] done
clear
View Solution play_arrow
-
question_answer6)
A current of 2.0 A when passed for 5 hours through a molten metal salt deposits 22.2 g of metal of atomic weight 177. The oxidation state of the metal in the metal salt is
A)
+ 1 done
clear
B)
+ 2 done
clear
C)
+ 3 done
clear
D)
+ 4 done
clear
View Solution play_arrow
-
question_answer7)
A current of 0.250 A is passed through 400 mL of a 2, 0 M solution of NaCl for 35 minutes. What will be the pH of the solution after the current is turned off?
A)
12.98 done
clear
B)
12.13 done
clear
C)
10.48 done
clear
D)
9.24 done
clear
View Solution play_arrow
-
question_answer8)
Acidulated water is electrolysed by 1 A current for 16 min and 5s using inert electrodes, The volume of gases liberated at STP will be
A)
22.4 mL done
clear
B)
112 mL done
clear
C)
168 mL done
clear
D)
336 mL done
clear
View Solution play_arrow
-
question_answer9)
Equivalent conductance of saturated \[BaS{{O}_{4}}\] is \[400\,{{\Omega }^{-1}}\,c{{m}^{2}}eq{{u}^{-1}}\] and specific conductance is\[8\times {{10}^{-5}}\,{{\Omega }^{-1}}\,c{{m}^{-1}}\]. Hence, \[{{K}_{sp}}\] of \[BaS{{O}_{4}}\] is
A)
\[4\times {{10}^{-8}}{{M}^{2}}\] done
clear
B)
\[1\times {{10}^{-8}}{{M}^{2}}\] done
clear
C)
\[2\times {{10}^{-4}}{{M}^{2}}\] done
clear
D)
\[1\times {{10}^{-4}}{{M}^{2}}\] done
clear
View Solution play_arrow
-
question_answer10)
The reduction potential of hydrogen half-cell will be negative if
A)
\[p({{H}_{2}})=2\,atm\,and\,[{{H}^{+}}]=2.0M\] done
clear
B)
\[p({{H}_{2}})=1\,atm\,and\,[{{H}^{+}}]=2.0M\] done
clear
C)
\[p({{H}_{2}})=1\,atm\,and\,[{{H}^{+}}]=1.0M\] done
clear
D)
\[p({{H}_{2}})=2\,atm\,and\,[{{H}^{+}}]=1.0M\] done
clear
View Solution play_arrow
-
question_answer11)
| EMF of which of the following cells at 298 K is highest? |
| Given, \[E_{\left( M{{g}^{2+}}/mg \right)}^{0}=-2.37V;\] |
| \[E_{\left( C{{u}^{2}}/Cu \right)}^{0}=+2.34V;\] |
| \[E_{\left( F{{e}^{2+}}/Fe \right)}^{0}=-0.44V;\] |
| \[E_{\left( S{{n}^{2+}}/Sn \right)}^{0}=-0.14V;\] |
| \[E_{\left( \frac{1}{2}B{{r}_{2}}/B{{r}^{-}} \right)}^{0}=+1.08V;\] |
A)
\[Mg(s)|M{{g}^{2+}}(0.001M)|\]\[|C{{u}^{2+}}(0.0001M)|Cu(s)\] done
clear
B)
\[Fe(s)|F{{e}^{2+}}(0.001M)|\]\[|{{H}^{+}}(1M)|{{H}_{2}}(g)(1bar)|Pt(s)\] done
clear
C)
\[Sn(s)S{{n}^{2+}}(0.050M)|\]\[|{{H}^{+}}(0.020M)|{{H}_{2}}(g)(1bar)|Pt(s)\] done
clear
D)
\[Pt(s)|B{{r}_{2}}(l)|B{{r}^{+}}(0.010M)|\]\[|{{H}^{+}}(0.030M)|{{H}_{2}}(g)(1bar)|Pt(s)\] done
clear
View Solution play_arrow
-
question_answer12)
On electrolyzing the molten chloride of metal \[725\text{ }ml\] of \[C{{l}_{2}}\] liberated at \[25{}^\circ C\] and 740 mm Hg pressure at anode for every one gm of metal deposite at cathode. The formula of metal chloride is (GAM of metal \[=52.01\])
A)
\[MCI\] done
clear
B)
\[MC{{I}_{2}}\] done
clear
C)
\[MC{{I}_{3}}\] done
clear
D)
\[MC{{I}_{4}}\] done
clear
View Solution play_arrow
-
question_answer13)
The standard electrode potential for the following reaction is + 1.33 V. What is the potential at pH = 2? \[C{{r}_{2}}O_{7}^{2-}(aq1M)+14{{H}^{\oplus }}(aq)+6{{e}^{-}}\]\[\xrightarrow{\,}\,2C{{r}^{3+}}(aq1M)+7{{H}_{2}}O(l)\]
A)
+1.82 V done
clear
B)
+1.99 V done
clear
C)
+1.608 V done
clear
D)
+1.054 V done
clear
View Solution play_arrow
-
question_answer14)
Molar conductance's of \[BaC{{l}_{2}},{{H}_{2}}S{{O}_{4}}\] and HCl at infinite dilutions are \[{{x}_{1}},{{x}_{2}}\] and \[{{x}_{3}}\] respectively. Equivalent conductance of \[BaS{{O}_{4}}\] at infinite dilution will be:
A)
\[({{x}_{1}}+{{x}_{2}}-{{x}_{3}})/2\] done
clear
B)
\[{{x}_{1}}+{{x}_{2}}-2{{x}_{3}}\] done
clear
C)
\[({{x}_{1}}-{{x}_{2}}-{{x}_{3}})/2\] done
clear
D)
\[({{x}_{1}}+{{x}_{2}}-2{{x}_{3}})/2\] done
clear
View Solution play_arrow
-
question_answer15)
Galvanic cell involves
A)
conversion of thermal energy (Heating) into electrical energy done
clear
B)
conversion of electrical energy into chemical energy done
clear
C)
conversion of chemical energy into thermal energy done
clear
D)
conversion of chemical energy into electrical energy done
clear
View Solution play_arrow
-
question_answer16)
Which of the following is correct statement-
A)
Conductivity & molar conductivity of solution increase on dilution done
clear
B)
Voltage of button cell remain unchanged throughout its life time done
clear
C)
On electrolysis of aqueous \[CuS{{O}_{4}}\] using platinum electrode its concentration \[[C{{u}^{2+}}]\] remain constant done
clear
D)
On electrolysis of aqueous \[NaCl,\] solution becomes acidic done
clear
View Solution play_arrow
-
question_answer17)
The resistance of 0.1N acetic acid when measured in a cell with cell constant \[1.5\,c{{m}^{-1}}\] is \[5.250\,\Omega \]. The value of \[{{\lambda }_{eq}}\] of \[0.1\,N\,AcOH\] is
A)
60 done
clear
B)
90 done
clear
C)
18.4 done
clear
D)
0.023 done
clear
View Solution play_arrow
-
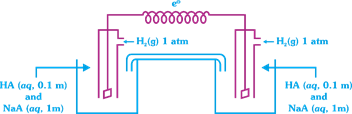
question_answer18)
A cell diagram shown below contains one litre of Buffer solution \[HA(p{{K}_{a}}=8)\] and NaA in both compartment. What is the cell emf?
A)
0.81 V done
clear
B)
0.071 V done
clear
C)
0.0591 V done
clear
D)
1.182 V done
clear
View Solution play_arrow
-
question_answer19)
Given the following molar conductivities at \[{{250}^{o}}C;HCl,425\,{{\Omega }^{-1}}c{{m}^{2}}mo{{l}^{-1}};\]\[NaCl\,\,125{{\Omega }^{-1}}\]\[c{{m}^{2}}mo{{l}^{-1}}\]NaC (sodium crotonate), \[82\,{{\Omega }^{-1}}c{{m}^{2}}mo{{l}^{-1}};\] what is the ionisation constant of crotonic acid? If the conductivity of a 0.001 M crotonic acid solution is \[3.82\times {{10}^{-4}}{{\Omega }^{-1}}c{{m}^{-1}}\]?
A)
\[1.5\times {{10}^{-4}}\] done
clear
B)
\[1.11\times {{10}^{-5}}\] done
clear
C)
\[2.11\times {{10}^{-5}}\] done
clear
D)
\[1.8\times {{10}^{-5}}\] done
clear
View Solution play_arrow
-
question_answer20)
| Given |
| (i) \[Mn{{O}_{4}}^{-}+8{{H}^{+}}+5{{e}^{-}}\xrightarrow{{}}\]\[M{{n}^{2+}}+4{{H}_{2}}\,O\] |
| (ii) \[Mn{{O}_{2}}+4{{H}^{+}}+2{{e}^{-}}\xrightarrow{{}}\]\[M{{n}^{2+}}+2{{H}_{2}}\,O{{E}^{0}}={{x}_{2}}V\] |
| Find \[{{E}^{0}}\] for the following reaction |
| \[Mn{{O}_{4}}^{-}+4{{H}^{+}}+3{{e}^{-}}\xrightarrow{{}}Mn{{O}_{2}}+2{{H}_{2}}\,O\] |
A)
\[{{x}_{2}}-{{x}_{1}}\] done
clear
B)
\[{{x}_{1}}-{{x}_{2}}\] done
clear
C)
\[\frac{5{{x}_{1}}-2{{x}_{2}}}{3}\] done
clear
D)
\[\frac{2{{x}_{1}}-5{{x}_{2}}}{3}\] done
clear
View Solution play_arrow
-
question_answer21)
3F of electricity passes through three electrolytic cells connected in series containing \[A{{g}^{+}},C{{a}^{2+}}\] and \[A{{l}^{3+}}\] ions, respectively. The molar ratio in which the three metal ions are liberated at the electrodes is
A)
1 : 2 : 3 done
clear
B)
2 : 3 : 1 done
clear
C)
6 : 3 : 2 done
clear
D)
3 : 4 : 2 done
clear
View Solution play_arrow
-
question_answer22)
Match List I with List II and select the correct answer using the code given below the lists:
| List I | List II |
| \[Zn|Z{{n}^{2+}}(0.01M)|\,\]\[|Z{{n}^{2+}}(0.1M)|Zn\] | (p) primary cell |
| 1 Coulomb | (q) Secondary cell |
| Dry Cell | (r) \[6.24\times {{10}^{18}}\]electrons |
| Lead storage cell | (s) Concentration cell |
A)
A-s, B-r, C-p, D-q done
clear
B)
A-p, B-q, C-r, D-s done
clear
C)
A-q, B-r, C-s, D-p done
clear
D)
A-s, B-p, C-q, D-r done
clear
View Solution play_arrow
-
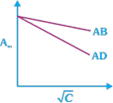
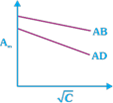
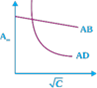
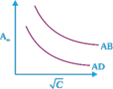
question_answer23)
Which of the following graphs are correct for strong electrolyte AD and AB.
A)
B)
C)
D)
View Solution play_arrow
-
question_answer24)
Salts of A (atomic weight: 7), B (atomic weight: 27), and C (atomic weight: 48) were electrolyzed under identical condition using the same quantity of electricity. It was found that when 2.1 got A was deposited, the weights of B and C deposited were 2.7 and 7.2 g, respectively. The valencies of A, B and C, respectively are:
A)
3, 1 and 2 done
clear
B)
1, 3 and 2 done
clear
C)
3, 1 and 3 done
clear
D)
2, 3 and 2 done
clear
View Solution play_arrow
-
question_answer25)
| The temperature coefficient of a cell whose operation is based on the reaction |
| \[Pb(s)+HgC{{l}_{2}}(aq)\xrightarrow{{}}PbC{{l}_{2}}(aq)+Hg(l)\] |
| \[{{\left( \frac{dE}{dT} \right)}_{p}}=1.5\times {{10}^{-4}}V/K\,at\,298K\] |
| The change in entropy (in J/K mol) during the operation is: |
A)
8627 done
clear
B)
57.9 done
clear
C)
28.95 done
clear
D)
14.475 done
clear
View Solution play_arrow
-
question_answer26)
| The electrolysis of acetate solution produces ethane according to reaction: |
| \[2C{{H}_{3}}CO{{O}^{-}}\xrightarrow{{}}{{C}_{2}}{{H}_{6}}(g)+2C{{O}_{2}}(g)+2{{e}^{-}}\] |
| The current efficiency of the process is 80%. What volume of gases would be produced at \[\text{27}{}^\circ \text{C}\] and 740 torr, if the current of 0.5 A is passed through the solution for 96.45 min? |
A)
6.0 L done
clear
B)
0.60 L done
clear
C)
1.365 L done
clear
D)
0.91 L done
clear
View Solution play_arrow
-
question_answer27)
A hydrogen electrode placed in a buffer solution of \[C{{H}_{3}}COONa\] and acetic acid in the ratio x : y and y : x has electrode potential value \[{{E}_{1}}\] volts and \[{{E}_{2}}\] volts, respectively at \[\text{25}{}^\circ \text{C}\]. The \[p{{K}_{a}}\] value of acetic acid is (\[{{E}_{1}}\] and \[{{E}_{2}}\] are oxidation potential):
A)
\[\frac{{{E}_{1}}+{{E}_{2}}}{0.118}\] done
clear
B)
\[\frac{{{E}_{2}}-{{E}_{1}}}{0.118}\] done
clear
C)
\[-\frac{{{E}_{1}}+{{E}_{2}}}{0.118}\] done
clear
D)
\[\frac{{{E}_{1}}-{{E}_{2}}}{0.118}\] done
clear
View Solution play_arrow
-
question_answer28)
Resistance of a conductivity cell filled with a solution of an electrolyte of concentration 0.1 M is 100 ohm. The conductivity of this solution is 1.29 S/m. Resistance of the same cell filled with 0.02 M of the same solution if the electrolyte is 520 ohm. The molar conductivity of 0.02 M solution of electrolyte would be:
A)
\[124\times {{10}^{-4}}S-{{m}^{2}}/mol\] done
clear
B)
\[1240\times {{10}^{-4}}S-{{m}^{2}}/mol\] done
clear
C)
\[1.24\times {{10}^{-4}}S-{{m}^{2}}/mol\] done
clear
D)
\[12.4\times {{10}^{-4}}S-{{m}^{2}}/mol\] done
clear
View Solution play_arrow
-
question_answer29)
The standard electrode potential for the following reaction is +1.33V. What is the potential at \[pH=2.0\]? \[C{{r}_{2}}{{O}_{7}}^{2-}(aq.1M)+14\,{{H}^{+}}(aq)+6{{e}^{-}}\xrightarrow{{}}\] \[2C{{r}^{3+}}(aq.\,1\,M)+7{{H}_{2}}O(\ell )\]
A)
+1.820 V done
clear
B)
+1.990 V done
clear
C)
+1.608 V done
clear
D)
+1.0542 V done
clear
View Solution play_arrow
-
question_answer30)
| For the following cell reaction |
| \[Pb(s)+H{{g}_{2}}S{{O}_{4}}(s)PbS{{O}_{4}}(s)+2Hg(l)\] \[E_{cell}^{o}=0.92V\] |
| \[{{K}_{sp}}(PbS{{O}_{4}})=2\times {{10}^{-8}},\]\[{{K}_{sp}}(H{{g}_{2}}S{{O}_{4}})=1\times {{10}^{-6}}\] |
| Hence, \[{{E}_{cell}}\] is |
A)
0.92 V done
clear
B)
0.89 V done
clear
C)
1.04 V done
clear
D)
0.95 V done
clear
View Solution play_arrow
-
question_answer31)
Zn-Hg is prepared by electrolysis of aqueous \[ZnC{{l}_{2}}\] using Hg cathode (9g). How much current is to be passed through \[ZnC{{l}_{2}}\] solution for 1000 s to prepare a Zn-Hg with 25% Zn by weight (Zn = 65.4):
A)
5.6 A done
clear
B)
7.2 A done
clear
C)
8.85 A done
clear
D)
11.2 A done
clear
View Solution play_arrow
-
question_answer32)
For the cell \[Pt|{{H}_{2}}(g)|\] solution \[X||KCI\] (saturated) \[|H{{g}_{2}}C{{l}_{2}}|Hg|\,Pt\] the observed EMF at \[25{}^\circ C\] was \[600\text{ }mV.\] When solution X was replaced by a standard phosphate buffer with \[pH=7.00,\] the EMF was \[777\text{ }mV\]. Find the pH of solution X.
A)
3 done
clear
B)
4 done
clear
C)
5 done
clear
D)
6 done
clear
View Solution play_arrow
-
question_answer33)
The same quantity of electricity was passed through two electrolytic cells containing a salt of metal X and zinc sulphate solution respectively using platinum electrode. 0.468 g of metal X and 1.532 g of Zn were deposited. The equivalent mass of X is (At. wt. of Zn is 65.4 u)
A)
9.98 g/equiv done
clear
B)
3.2 g/equiv done
clear
C)
5.9 g/equiv done
clear
D)
0.99 g/equiv done
clear
View Solution play_arrow
-
question_answer34)
The standard EMF of a cell having one electron change is found to be 0.591 V at \[\text{25}{}^\circ \text{C}\]. The equilibrium constant of the reaction is:
A)
\[1.0\times {{10}^{30}}\] done
clear
B)
\[1.0\times {{10}^{5}}\] done
clear
C)
\[1.0\times {{10}^{10}}\] done
clear
D)
\[1.0\times {{10}^{1}}\] done
clear
View Solution play_arrow
-
question_answer35)
| Consider following sets: |
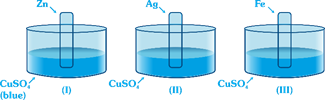 |
| Blue colour solution changes to colourless (or fades) in |
A)
I, II, III done
clear
B)
I, II done
clear
C)
II, III done
clear
D)
I, III done
clear
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear

