-
How will you determine the standard electrode potential of the system Mg2+/Mg?
View Answer play_arrow
-
Can you store copper sulphate solution in a zinc pot?
View Answer play_arrow
-
On the basis of standard electrode potentials suggest three substances that can oxidize ferrous ions under suitable conditions.
View Answer play_arrow
-
Calculate the potential of hydrogen electrode contact with a solution whose pH is 10.
View Answer play_arrow
-
Calculate
the emf of the cell in which t-following reaction takes place
Ni(s)
+ 2Ag+ (0.002M)

Ni2+
(0.160 M) + 2Ag(s)
Given that E°cell = 1.05 V.
-
The
cell in which the following reaction occurs :
2Fe3+
(aq) + 2I? (aq)

2Fe2+
(aq) + I2(s)
has
E0cell = 0.236 V at 298 K. Calculate the standard Gibb's energy and
the equilibrium constant of the cell reaction.
-
Why does the conductivity of a solution decrease with dilution?
View Answer play_arrow
-
Suggest a way to determine the
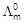
value of
water.
-
The molar conductivity of 0.025 mol L?1
methanoic acid is 46.1 S cm2 mol?1. Calculate its degree
of dissociation and dissociation constant.
Given
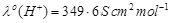
and
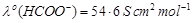
.
-
If a current of 0.5 ampere flows through a metallic wire for 2 hours, then how many electrons flow through the wire?
View Answer play_arrow
-
Suggest a list of metals that are extracted electrolytically.
View Answer play_arrow
-
Consider the reaction :
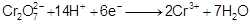
What is the quantity of electricity in coulombs needed to
reduce 1 mol of Cr2
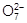
?
-
Write the chemistry of recharging the lead storage battery, highlighting all the materials that are involved during recharging.
View Answer play_arrow
-
Suggest two materials other than hydrogen that can be used as fuels in fuel cells.
View Answer play_arrow
-
Explain how rusting of iron is envisaged as setting up of an electrochemical cell.
View Answer play_arrow
-
Arrange the following metals in the order in which they displace each other from the solution of their salts. Al, Cu, Fe, Mg and Zn.
View Answer play_arrow
-
Given the standard electrode potential.
K+ /K = – 2.93V, Ag+/Ag = 0.80 V.
Hg2+/Hg = 0.79 V
Mg2+/Mg = – 2.37 V, Cr3+/Cr = – 0.74 V
Arrange these metals in their increasing order of reducing power.
View Answer play_arrow
-
Depict the galvanic cell in which the reaction
Zn (s) + 2Ag+ (aq)

Zn2+
(aq) + 2Ag(s) takes place. Further tell
(i) Which of the electrode is
negatively charged.
(ii) The carriers of the current
in the cell
(iii) Individual reaction at each electrode.
-
Calculate the standard cell potentials of galvanic
cell in which the following reactions take place :
(i) 2Cr (s) + 3 Cd2+
(aq)
® 2 Cr3+ (aq) + 3 Cd
E0Cr3+ /Cr
= ? 0.74 V and E0Cd 2+/Cd = ? 0.40V
(ii) Fe2+ (aq) + Ag+
(aq)

Fe3+
(aq) + Ag (s)
E0Ag+ /Ag =
0.80 V and E0Fe 3+ /Fe = 0.77V
Calculate the
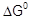
and
equilibrium constant of the reactions.
-
Write
the Nernst equation and rind e.m.f. of the following cells at 298 K.
Mg(s)/Mg2+
(0.001 M) || Cu2+s(0.0001 M) | Cu(s)
E0Mg2+/Mg
= ? 2.37 V, E0Cu2+ /Cu = 0.34 V
(ii) Fe (s)/Fe2+
(0.001 M) || H+ (1M)/H2 (g) (1 bar) | Pt (s)
E0Fe+2/Fe
= 0.44 V
(iii) Sn (s)/Sn2+
(0.050 M) || H+ (0.020 M) | H2 (g) (1 bar) I Pt (s)

(iv) Pt (s)/Br2
(1)/Br?(0.010 M) || H+ (0.030 M) |H2 (g) (1
bar) I Pt (s)

-
In the button cells widely used in watches and
other devices the following reaction takes place:
Zn(s) + Ag2O(s) + H2O(l)

Zn2+(aq)
+ 2Ag(s) + 2OH? (aq).
Determine
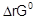
and E°
for the reaction. Given E0Ag+/Ag = 0.344V and E0Zn2+/Zn
= ?0.76V
-
Define conductivity and molar conductivity for the solution of an electrolyte. Discuss their variation with concentration.
View Answer play_arrow
-
The conductivity of 0.20 M solution of KCI at 298 K is 0.0248 S cm–1. Calculate its molar conductivity.
View Answer play_arrow
-
The resistance of a conductivity cell containing 0.001 M KCI
solution at 298 K is 1500
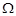
. What is
the cell constant if conductivity of 0.001 M KCl solution at 298 K is 0.146 x
10?3 S cm?1?
-
The
conductivity of sodium chloride at 298 K has been determined at different concentration
and the results are given below?
|
Concentration/M
|
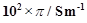
|
|
0.001
|
1.237
|
|
0.010
|
11.85
|
|
0.020
|
23.15
|
|
0.050
|
55.53
|
|
0.100
|
106.74
|
Calculate
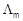
for all
concentrations and draw a plot between
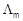
and c1/2.
Find the value of
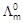
-
Conductivity of 0.00241 M acetic
acid is 7.896 × 10?5 S cm?1. Calculate its molar
conductivity and if
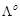
for
acetic acid is 390.5S cm2 mol?1, what is its dissociation
constant?
-
How much charge is required for the following
reduction of
(i) 1 mol of Al3+ to
Al.
(ii) 1 mol of Cu2+ to
Cu.
(iii) 1 mol of
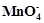
to Mn2+
-
How much electricity in terms of Faraday is required to produce
(i) 20.0 g of Cu from molten CaCl2
(ii) 40.0 g of Al from molten Al2O3.
View Answer play_arrow
-
How much electricity is required in coulomb for the oxidation of
(i) 1 mol of H2O to O2?
(ii) 1 tool FeO to Fe2O3?
View Answer play_arrow
-
A solution of Ni(NO3)2 is electrolysed between platinum electrodes using a current of 5 amperes for 20 minutes. What mass of Ni is deposited at the cathode?
View Answer play_arrow
-
Three electrolytic cells A, B and
C containing solution of ZnSO4, AgNO3 and CuSO4,
respectively are connected in series. A steady current of 1.5 amperes was
passed through them until 1.45 g of silver deposited at the cathode of cell it.
How long did the current flow? What mass of copper and of zinc were deposited?
(Atomic mass : Zn = 65.4u, Ag = 108u, Cu = 63.54).
View Answer play_arrow
-
Using the standard electrode potentials given below
predict if the reaction between the following is feasible.
(i) Fe3+ (aq) and I?
(aq)
(ii) Ag+ (aq) and
Cu(s)
(iii) Fe3+ (aq) and Br?
(aq)
(iv) Ag (s) and Fe3+
(aq)
(v) Br2 (aq) and Fe2+
(aq)
Given E01/2 I2/I?
= 0.54V, E0Cu2+/Cu = 0.34 V
E01/2 Br2 /
Br? = 1.09 V, E0Ag+ Ag = 0.80 V and
E0Fe3+ /Fe2+ = 0.77 V
View Answer play_arrow
-
Predict the products of electrolysis in each of the
following :
(i) An aqueous solution of AgNO3
with silver electrodes.
(ii) An aqueous solution of AgNO3
with platinum electrodes.
(iii) A dilute solution of H2SO4
with platinum electrodes.
(iv) An aqueous solution of CuCl2
with platinum electrodes
(Given E0Ag+ /Ag = + 0.80 V,
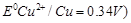
-
question_answer34)
Which cell will measure standard electrode
potential of copper electrode?
(a) (b)
(b) 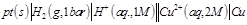 (c)
(c) 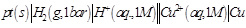 (d)
(d) 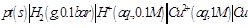
View Answer play_arrow
-
question_answer35)
Electrode potential for Mg electrode varies
according to the equation
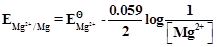 .The graph
.The graph 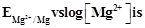
View Answer play_arrow
-
question_answer36)
Which of the following statement is correct?
(a) 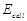 and
and
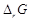 of cell reaction both are
extensive properties
(b)
of cell reaction both are
extensive properties
(b) 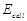 and
and
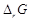 of cell reaction both are
intensive properties
(c)
of cell reaction both are
intensive properties
(c) 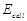 is
an intensive property while
is
an intensive property while 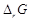 of
cell reaction is an extensive property
(d)
of
cell reaction is an extensive property
(d) 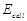 is
an extensive property while
is
an extensive property while 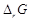 of
cell reaction is an intensive property
of
cell reaction is an intensive property
View Answer play_arrow
-
question_answer37)
The difference between the electrode potentials
of two electrodes when no current is drawn through the cell is called.........
(a) cell potential (b)
cell emf
(c) potential
difference (d) cell voltage
View Answer play_arrow
-
question_answer38)
Which of the following statement is not correct
about an inert electrode in a cell?
(a) It does not participate in the cell
reaction
(b) It provides surface either
for oxidation or for reduction reaction
(c) It provides surface for
conduction of electrons
(d) It provides surface for
redox reaction
View Answer play_arrow
-
question_answer39)
An electrochemical cell can behave like an
electrolytic cell when.........
(a) 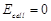 (b)
(b)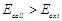 (C)
(C) 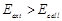 (d)
(d)
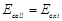
View Answer play_arrow
-
question_answer40)
Which of the statements about solutions of
electrolytes is not correct?
(a) Conductivity of solution depends
upon size of ions
(b) Conductivity depends upon
viscosity of solution
(c) Conductivity does not depend
upon solvation of ions present in solution
(d) Conductivity of solution
increases with temperature
View Answer play_arrow
-
question_answer41)
Using the data given below find out the
strongest reducing agent.
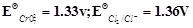
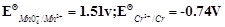 (a)
(a) 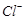 (b)
Cr
(c)
(b)
Cr
(c) 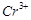 (d)
(d)
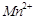
View Answer play_arrow
-
question_answer42)
Use the data given in Q. 8 and find out which of
the following is the strongest oxidising agent?
(a) 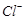 (d)
(d)
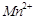 (c)
(c) 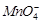 (d)
(d)
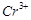
View Answer play_arrow
-
question_answer43)
Using the data given in Q.8 find out in which
option the order of reducing power is correct,
(a) 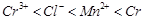 (b)
(b) 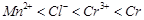 (c)
(c) 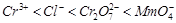 (d)
(d) 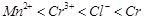
View Answer play_arrow
-
question_answer44)
Use the data given in Q. 8 and find out the most
stable ion in its reduced form.
(a) CI- (b)
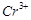 (c) Cr (d) Mn2+
(c) Cr (d) Mn2+
View Answer play_arrow
-
question_answer45)
Use the data of Q. 8 and find out the most
stable oxidised species...
(a) Cr3+ (b)
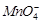 (c)
(c) 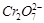 (d)
Mn2+
(d)
Mn2+
View Answer play_arrow
-
question_answer46)
The quantity of charge required to obtain one
mole of aluminium from AL203 is........ .
(a)1F (b)6F
(03F (d) 2 F
View Answer play_arrow
-
question_answer47)
The cell constant of a conductivity cell..........
(a) changes with change of electrolyte
(b) changes with change of
concentration of electrolyte
(c) changes with temperature of
electrolyte
(d) remains constant for a cell
View Answer play_arrow
-
question_answer48)
While charging the lead storage battery..........
(a) PbSO4 anode is reduced to
Pb
(b) PbSO4 cathode is
reduced to Pb
(c) PbSO4 cathode is
oxidised to Pb
(d) PbSO4 anode is
oxidised to PbO2
View Answer play_arrow
-
question_answer49)
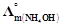 is
equal to ......... .
(a)
is
equal to ......... .
(a) 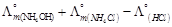 (b)
(b) 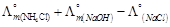 (c)
(c) 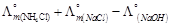 (d)
(d) 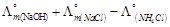
View Answer play_arrow
-
question_answer50)
In the electrolysis of
aqueous sodium chloride solution which of the half-cell reaction will occur at
anode?
(a) 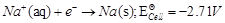 (b)
(b) 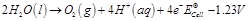 (c)
(c) 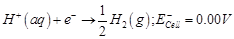 (d)
(d) 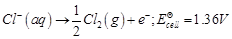
View Answer play_arrow
-
question_answer51)
The positive value of the standard electrode
potential of Cu2+/Cu indicates that...........
(a) this redox couple is a stronger
reducing agent than the H+ /H2 couple
(b) this redox couple is a
stronger oxidising agent than H+ /H2
(c) Cu can displace H2
from acid
(d) Cu cannot displace H2
from acid
View Answer play_arrow
-
question_answer52)
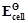 for
some half-cell reactions are given below. On the basis of these mark the
correct answer.
(a)
for
some half-cell reactions are given below. On the basis of these mark the
correct answer.
(a) 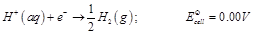 (b)
(b) 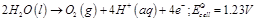 (c)
(c) 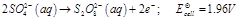 (a) In dilute sulphuric acid
solution, hydrogen will be reduced at cathode
(b) In concentrated sulphuric
acid solution, water will be oxidised at anode
(c) In dilute sulphuric acid
solution, water will be oxidised at anode
(d) In dilute sulphuric acid
solution,
(a) In dilute sulphuric acid
solution, hydrogen will be reduced at cathode
(b) In concentrated sulphuric
acid solution, water will be oxidised at anode
(c) In dilute sulphuric acid
solution, water will be oxidised at anode
(d) In dilute sulphuric acid
solution, 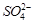 ion will be oxidised to
tetrathionate ion at anode
ion will be oxidised to
tetrathionate ion at anode
View Answer play_arrow
-
question_answer53)
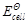 = 1.1
V or Daniel cell. Which of the following expressions are correct description of
state of equilibrium in this cell?
(a)
= 1.1
V or Daniel cell. Which of the following expressions are correct description of
state of equilibrium in this cell?
(a) 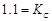 (b)
(b) 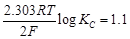 (c)
(c) 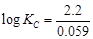 (d)
(d) 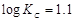
View Answer play_arrow
-
question_answer54)
Conductivity of an electrolytic solution depends on
(a) nature of electrolyte
(b) concentration of electrolyte
(c) power of AC source
(d) distance between the
electrodes
View Answer play_arrow
-
question_answer55)
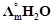 is
equal to.......
(a)
is
equal to.......
(a) 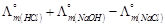 (b)
(b) 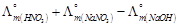 (c)
(c) 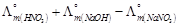 (d)
(d) 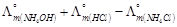
View Answer play_arrow
-
question_answer56)
What will happen during the electrolysis of
aqueous solution ofCuS04 by using platinum electrodes?
(a) Copper will deposit at cathode
(b) Copper will deposit at anode
(c) Oxygen will be released at
anode
(d) Copper will dissolve at
anode
View Answer play_arrow
-
question_answer57)
What will happen during the electrolysis of
aqueous solution ofCuS04 in the presence of Cu electrodes?
(a) Copper will deposit at cathode
(b) Copper will dissolve at
anode
(c) Oxygen will be released at
anode
(d) Copper will deposit at anode
View Answer play_arrow
-
question_answer58)
Conductivity k , is equal to.........
(a) 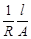 (b)
(b)
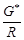 (c)
(c) 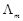 (d)
(d)
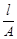
View Answer play_arrow
-
question_answer59)
Molar conductivity of ionic solution depends on..........
(a) temperature
(b) distance between electrodes
(c) concentration of
electrolytes in solution
(d) surface area of electrodes
View Answer play_arrow
-
question_answer60)
For the given cell, 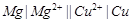 (a) Mg is cathode
(b) Cu is cathode
(c) The cell reaction is
(a) Mg is cathode
(b) Cu is cathode
(c) The cell reaction is
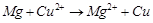 (d) Cu is the oxidising agent
(d) Cu is the oxidising agent
View Answer play_arrow
-
question_answer61)
Can absolute electrode potential of an electrode
be measured?
View Answer play_arrow
-
question_answer62)
Can 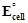 or
or
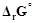 for cell reaction ever
be equal to zero?
for cell reaction ever
be equal to zero?
View Answer play_arrow
-
question_answer63)
Under what condition is 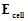 = 0 or
= 0 or 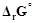 = 0?
= 0?
View Answer play_arrow
-
question_answer64)
What does the negative sign in the expression 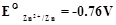 mean?
mean?
View Answer play_arrow
-
question_answer65)
Aqueous copper sulphate solution and aqueous
silver nitrate solution are electrolysed by 1 ampere current for 10 minutes in
separate electrolytic cells. Will the mass of copper and silver deposited on
the cathode be same or different? Explain your answer.
View Answer play_arrow
-
question_answer66)
Depict the galvanic cell in which the cell
reaction is
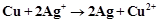
View Answer play_arrow
-
question_answer67)
Value of standard electrode potential for the
oxidation of Cl- ions is more positive than that of water, even then
in the electrolysis of aqueous sodium chloride, why is Cl- oxidised
at anode instead of water?
View Answer play_arrow
-
question_answer68)
What is electrode potential?
View Answer play_arrow
-
question_answer69)
Consider the following diagram in which an
electrochemical cell is coupled to an electrolytic cell. What will be
the polarity of electrodes 'A' and 'B' in the electrolytic cell?
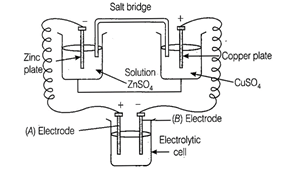
View Answer play_arrow
-
question_answer70)
Why is alternating current used for measuring
resistance of an electrolytic solution?
View Answer play_arrow
-
question_answer71)
A galvanic cell has electrical potential of
1.1V. If an opposing potential of 1.1V is applied to this cell, what will
happen to the cell reaction and current flowing through the cell?
View Answer play_arrow
-
question_answer72)
How will the pH of brine {aq NaCI solution) be
affected when it is electrolysed?
View Answer play_arrow
-
question_answer73)
Unlike dry cell, the mercury cell has a constant
cell potential throughout its useful life. Why?
View Answer play_arrow
-
question_answer74)
Solutions of two electrolytes A and B are
diluted. The 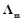 of 'B'
increases 1.5 times while that of A increases 25 times. Which of the two is a
strong electrolyte? Justify your answer.
of 'B'
increases 1.5 times while that of A increases 25 times. Which of the two is a
strong electrolyte? Justify your answer.
View Answer play_arrow
-
question_answer75)
When acidulated water (dil. 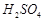 solution) is
electrolysed, with pH of the solution be affected? Justify your answer.
solution) is
electrolysed, with pH of the solution be affected? Justify your answer.
View Answer play_arrow
-
question_answer76)
In an aqueous solution how does specific
conductivity of electrolytes change with addition of water?
View Answer play_arrow
-
question_answer77)
Which reference electrode is used to measure the
electrode potential of other electrodes?
View Answer play_arrow
-
question_answer78)
Consider a cell given below
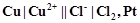 Write
the reactions that occur at anode and cathode.
Write
the reactions that occur at anode and cathode.
View Answer play_arrow
-
question_answer79)
Write the Nernst equation for the cell reaction
in the Daniel cell. How will the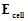 be
affected when concentration of
be
affected when concentration of 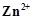 ions
is increased?
ions
is increased?
View Answer play_arrow
-
question_answer80)
What advantage do the fuel cells have over
primary and secondary batteries?
View Answer play_arrow
-
question_answer81)
Write the cell reaction of a lead storage
battery when it is discharged. How does the density of the electrolyte change
when the battery is discharged?
View Answer play_arrow
-
question_answer82)
Why on dilution the 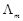 of
of 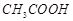 increases drastically,
while that of CH3COONa increases gradually?
increases drastically,
while that of CH3COONa increases gradually?
View Answer play_arrow
-
question_answer83)
Match the terms given in Column I with the units
given in Column II.
|
Column 1
|
Column 11
|
A. 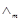
|
1. 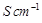
|
B. 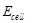
|
2. 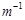
|
C. 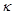
|
3. 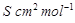
|
|
D. G*
|
4. V
|
View Answer play_arrow
-
question_answer84)
Match the terms given in Column I with the items
given in Column II.
|
|
Column 1
|
Column 11
|
|
A.
|
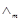
|
1. Intensive property
|
|
B.
|
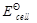
|
2. Depends on number of. ions/volume
|
|
C.
|
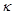
|
3. Extensive property
|
|
D.
|
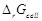
|
4. Increases with dilution
|
View Answer play_arrow
-
question_answer85)
Match the items of Column I and Column II.
|
|
Column 1
|
Column 11
|
|
A.
B.
C.
D.
|
Lead storage battery
Mercury cell
Fuel cell
Rusting
|
1. Maximum efficiency
2. Prevented by galvanisation
3. Gives steady potential
4. Pb is anode, PbO2 is cathode
|
View Answer play_arrow
-
question_answer86)
Match the items of Column I and Column II.
|
Column I
|
Column II
|
A. 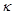
|
1. I ´
t
|
B. 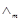
|
2. 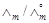
|
C. 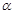
|
3. 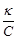
|
|
D. Q
|
4. 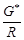
|
View Answer play_arrow
-
question_answer87)
Match the items of Column I and Column II.
|
Column 1
|
Column 11
|
|
A. Lechlanche cell
B. Ni-Cd cell
C. Fuel ceil
D. Mercury cell
|
1. Cell reaction 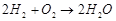 2. Does not involve any ion in solution and is
used in hearing aids.
3. Rechargeable
4. Reaction at anode,
2. Does not involve any ion in solution and is
used in hearing aids.
3. Rechargeable
4. Reaction at anode, 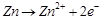 5 .Converts energy of combustion into '
electrical energy
5 .Converts energy of combustion into '
electrical energy
|
View Answer play_arrow
-
question_answer88)
Match the items of Column I and Column II on the
basis of data given below
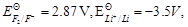
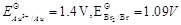
View Answer play_arrow
-
question_answer89)
Assertion (A) Cu is less reactive than
hydrogen.
Reason (R) 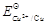 is negative.
is negative.
View Answer play_arrow
-
question_answer90)
Assertion (A) 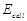 should
have a positive value for the cell to function.
Reason (R)
should
have a positive value for the cell to function.
Reason (R) 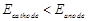
View Answer play_arrow
-
question_answer91)
Assertion (A) Conductivity of all
electrolytes decreases on dilution.
Reason (R) On dilution number of
ions per unit volume decreases.
View Answer play_arrow
-
question_answer92)
Assertion (A) 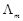 for
weak electrolytes shows a sharp increase when the electrolytic solution is
diluted.
Reason (R) For weak electrolytes
degree of dissociation increases with dilution of solution.
for
weak electrolytes shows a sharp increase when the electrolytic solution is
diluted.
Reason (R) For weak electrolytes
degree of dissociation increases with dilution of solution.
View Answer play_arrow
-
question_answer93)
Assertion (A) Mercury cell does not give
steady potential.
Reason (R) In the cell reaction,
ions are not involved in solution.
View Answer play_arrow
-
question_answer94)
Assertion (A) Electrolysis of NaCI solution
gives chlorine at anode instead of O2.
Reason (R) Formation of oxygen at
anode requires over voltage.
View Answer play_arrow
-
question_answer95)
Assertion (A) For measuring resistance of an
ionic solution an AC source is used.
Reason (R) Concentration of ionic
solution will change if DC source is used.
View Answer play_arrow
-
question_answer96)
Assertion (A) Current stops flowing when 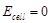 Reason (R) Equilibrium of the
cell reaction is attained.
Reason (R) Equilibrium of the
cell reaction is attained.
View Answer play_arrow
-
question_answer97)
Assertion (A) 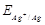 increase
with increase in concentration of
increase
with increase in concentration of 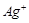 ions.
Reason(R)
ions.
Reason(R) 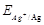 has a positive value.
has a positive value.
View Answer play_arrow
-
question_answer98)
Assertion (A) Copper sulphate can be stored
in zinc vessel.
Reason (R) Zinc is less reactive
than copper.
View Answer play_arrow
-
question_answer99)
Consider the figure and answer the following
questions.
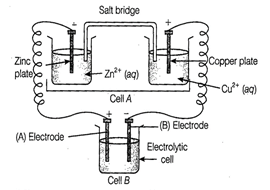 (i) Cell 'A' has
(i) Cell 'A' has 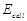 -2V and Cell 'B' has
-2V and Cell 'B' has 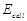 =1.1V which of the two
cell 'A' or 'B' will act as an electrolytic cell. Which electrode reactions
will occur in this cell?
(ii) If cell 'A' has
=1.1V which of the two
cell 'A' or 'B' will act as an electrolytic cell. Which electrode reactions
will occur in this cell?
(ii) If cell 'A' has 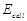 = 0.5V and cell 'B' has
= 0.5V and cell 'B' has 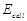 =1.1V then what WUL be the
reactions at anode and cathode?
=1.1V then what WUL be the
reactions at anode and cathode?
View Answer play_arrow
-
question_answer100)
Consider figure from the above question and
answer the questions (i) to (vi) given below.
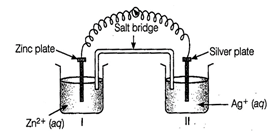 (i) Redraw the diagram to show
the direction of electron flow.
(ii) Is silver plate the anode
or cathode?
(iii) What will happen if salt
bridge is removed?
(iv) When will the cell stop
functioning?
(v) How will concentration of Zn2+
ions and Ag+ ions be affected when the cell functions?
(vi) How will the concentration
of Zn2+ ions and Ag+ ions be affected after the cell becomes
'dead'?
(i) Redraw the diagram to show
the direction of electron flow.
(ii) Is silver plate the anode
or cathode?
(iii) What will happen if salt
bridge is removed?
(iv) When will the cell stop
functioning?
(v) How will concentration of Zn2+
ions and Ag+ ions be affected when the cell functions?
(vi) How will the concentration
of Zn2+ ions and Ag+ ions be affected after the cell becomes
'dead'?
View Answer play_arrow
-
question_answer101)
What is the relationship between Gibbs free
energy of the cell reaction in a galvanic cell and the emf of the cell? When
will the maximum work be obtained from a galvanic cell?
View Answer play_arrow
![]() .The graph
.The graph ![]()

![]() is
equal to ......... .
(a)
is
equal to ......... .
(a) ![]() (b)
(b) ![]() (c)
(c) ![]() (d)
(d) ![]()
![]() is
equal to.......
(a)
is
equal to.......
(a) ![]() (b)
(b) ![]() (c)
(c) ![]() (d)
(d) ![]()
![]() (b)
(b)
![]() (c)
(c) ![]() (d)
(d)
![]()
![]() or
or
![]() for cell reaction ever
be equal to zero?
for cell reaction ever
be equal to zero?
![]() = 0 or
= 0 or ![]() = 0?
= 0?
![]() mean?
mean?
![]()
![]() Write
the reactions that occur at anode and cathode.
Write
the reactions that occur at anode and cathode.
![]()
![]()
![]() is negative.
is negative.
![]() should
have a positive value for the cell to function.
Reason (R)
should
have a positive value for the cell to function.
Reason (R) ![]()
