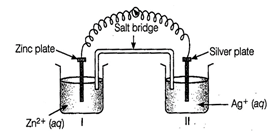
 (i) Redraw the diagram to show
the direction of electron flow.
(ii) Is silver plate the anode
or cathode?
(iii) What will happen if salt
bridge is removed?
(iv) When will the cell stop
functioning?
(v) How will concentration of Zn2+
ions and Ag+ ions be affected when the cell functions?
(vi) How will the concentration
of Zn2+ ions and Ag+ ions be affected after the cell becomes
'dead'?
(i) Redraw the diagram to show
the direction of electron flow.
(ii) Is silver plate the anode
or cathode?
(iii) What will happen if salt
bridge is removed?
(iv) When will the cell stop
functioning?
(v) How will concentration of Zn2+
ions and Ag+ ions be affected when the cell functions?
(vi) How will the concentration
of Zn2+ ions and Ag+ ions be affected after the cell becomes
'dead'?
Answer:
(i) Electrons move from Zn to Ag as
E° is more negative for Zn, so Zn undergoes oxidation and Ag+ undergoes
reduction.
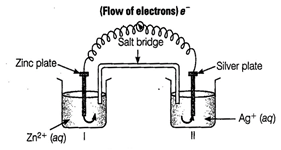 (ii) Ag is the cathode as it is
the site of reduction where Ag+ takes electrons from medium and
deposit at cathode.
(iii) Cell will stop
functioning because cell potential drops to zero. At E = 0 reaction reaches
equilibrium.
(iv) When
(ii) Ag is the cathode as it is
the site of reduction where Ag+ takes electrons from medium and
deposit at cathode.
(iii) Cell will stop
functioning because cell potential drops to zero. At E = 0 reaction reaches
equilibrium.
(iv) When ![]() because at this condition
reaction reaches to equilibrium.
(v) Concentration of Zn2+
ions will increase and concentration of Ag+ ions will decrease because
Zn is converted into Zn2+and Ag+ is converted into Ag.
(vi) When
because at this condition
reaction reaches to equilibrium.
(v) Concentration of Zn2+
ions will increase and concentration of Ag+ ions will decrease because
Zn is converted into Zn2+and Ag+ is converted into Ag.
(vi) When ![]() equilibrium is reached
and concentration of Zn2+ ions and Ag+ will not change.
equilibrium is reached
and concentration of Zn2+ ions and Ag+ will not change.
You need to login to perform this action.
You will be redirected in
3 sec
