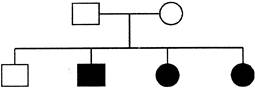
A geometry consists of a square and a disc as shown in figure. It is made up of an iron sheet of uniform density. The Y-coordinate of centre of mass of the geometry with respect to point o is:
A)
\[\frac{4a+3\pi a}{4+\pi }\]
done
clear
B)
\[\frac{4a+3\pi a}{4+3\pi }\]
done
clear
C)
\[(4+3\pi )a\]
done
clear
D)
\[3a\]
done
clear
View Answer play_arrow
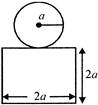
A block Q of mass m is placed on a horizontal frictionless surface AB and a body P of mass m is released on its frictionless slope. As P slides by 9 length L on this slope of inclination \[\theta \], the block Q would slide by distance
A)
\[\frac{m}{M}L\,\cos \theta \]
done
clear
B)
\[\frac{m}{M+m}L\]
done
clear
C)
\[\frac{M+m}{mL\,\cos \theta }\]
done
clear
D)
\[\frac{mL\,\cos \theta }{m+M}\]
done
clear
View Answer play_arrow
Maximum acceleration of an object in simple harmonic motion is \[24m{{s}^{-2}}\] and maximum velocity is \[16m{{s}^{-1}}\]. The amplitude of object is:
A)
\[\frac{32}{3}m\]
done
clear
B)
\[\frac{3}{32}m\]
done
clear
C)
\[\frac{2}{3}m\]
done
clear
D)
\[\frac{3}{2}m\]
done
clear
View Answer play_arrow
An open vessel containing water is given a constant acceleration a in the horizontal direction. Then, the free surface of water gets sloped with the horizontal at an angle \[\theta \] given by:
A)
\[\theta ={{\tan }^{-1}}\left( \frac{a}{g} \right)\]
done
clear
B)
\[\theta ={{\tan }^{-1}}\left( \frac{g}{a} \right)\]
done
clear
C)
\[\theta ={{\sin }^{-1}}\left( \frac{a}{g} \right)\]
done
clear
D)
\[\theta ={{\cos }^{-1}}\left( \frac{g}{a} \right)\]
done
clear
View Answer play_arrow
Work done in splitting a drop of 1mm radius into 64 droplets is (surface tension of water is\[72\times {{10}^{-3}}J/{{m}^{2}}\])
A)
\[20\times {{10}^{-6}}J\]
done
clear
B)
\[2.7\times {{10}^{-6}}J\]
done
clear
C)
\[4\times {{10}^{-6}}J\]
done
clear
D)
\[5.4\times {{10}^{-6}}J\]
done
clear
View Answer play_arrow
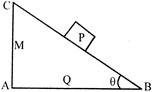
A planet is moving in an elliptical path around the sun as shown in figure. Speed of planed in positions P and Q are \[{{V}_{1}}\] and \[{{V}_{2}}\]respectively with \[{{S}_{P}}={{r}_{1}}\]and \[{{S}_{Q}}={{r}_{2}},\] then \[{{V}_{1}}/{{V}_{2}}\]is equal to:
A)
\[\frac{{{r}_{1}}}{{{r}_{2}}}\]
done
clear
B)
\[\frac{{{r}_{2}}}{{{r}_{1}}}\]
done
clear
C)
constant
done
clear
D)
\[{{\left( \frac{{{r}_{1}}}{{{r}_{2}}} \right)}^{2}}\]
done
clear
View Answer play_arrow
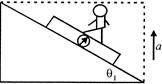
A man of mass \[m=60kg\] is standing on weighing machine fixed on a triangular wedge of angle \[\theta ={{60}^{o}}\] with horizontal as shown in the figure. The wedge is moving up with an upward acceleration\[a=2m{{s}^{-2}}\]. The weight registered by machine is:
A)
600N
done
clear
B)
1440N
done
clear
C)
360N
done
clear
D)
240N
done
clear
View Answer play_arrow
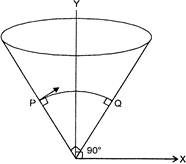
A particle is projected from point P with velocity \[5\sqrt{2}m{{s}^{-1}},\] perpendicular to the surface of a hollow right angle cone whose axis is verticle. It colides at Q normally. The time of the flight of the particle is:
A)
1S
done
clear
B)
\[\sqrt{2}S\]
done
clear
C)
\[2\sqrt{2}S\]
done
clear
D)
2S
done
clear
View Answer play_arrow
The pitch of a screw gauge is 1mm and there are 100 divisions on its circular scale. When nothing is put in between its jaws, the zero of the circular scale lies 4 divisions below the reference line when a steel wire is placed between the jaws, two main scale divisioins are clearly visible and 67 divisions on the circular scale are observed. The diameter of the wire is:
A)
\[2.71\,mm\]
done
clear
B)
\[2.67\,mm\]
done
clear
C)
\[2.63\,mm\]
done
clear
D)
\[2.65\,mm\]
done
clear
View Answer play_arrow
The dimesions of \[\frac{1}{\sqrt{{{\mu }_{0}}{{\varepsilon }_{0}}}}\] are:
A)
\[[{{I}^{o}}{{M}^{o}}L{{T}^{-1}}]\]
done
clear
B)
\[[{{I}^{2}}{{M}^{1}}{{L}^{-3}}T]\]
done
clear
C)
\[[{{I}^{-2}}{{T}^{-2}}LM]\]
done
clear
D)
\[2[{{I}^{o}}{{M}^{o}}{{L}^{-1}}T]\]
done
clear
View Answer play_arrow
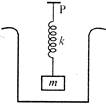
A cube of mass m and density D is suspended from the point P, by a spring of stiffness K. The system is kept inside a beaker filled with a liquid of density d. The elongation in the spring (assuming \[D>d\]) is:
A)
\[\frac{mg}{K}\left( 1-\frac{d}{D} \right)\]
done
clear
B)
\[\frac{mg}{K}\left( 1-\frac{D}{d} \right)\]
done
clear
C)
\[\frac{mg}{K}\left( 1-\frac{d}{D} \right)\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
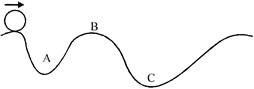
A body moves along an uneven surface with constant speed at all points. The normal reaction exert by the road on the body is:
A)
Maximum at A
done
clear
B)
Minimum at A
done
clear
C)
Maximum at C
done
clear
D)
the same at A,B and C
done
clear
View Answer play_arrow
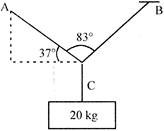
A block of mass 20 kg is balanced by three strings A, B and C as shows in figure. Ratio of tensions in strings A and B \[({{T}_{A}}/{{T}_{B}})\]: is
A)
\[\frac{5}{8}\]
done
clear
B)
\[\frac{5\sqrt{3}}{8}\]
done
clear
C)
\[\frac{5}{6}\]
done
clear
D)
\[\frac{8}{5}\]
done
clear
View Answer play_arrow
If the velocity of a particle is \[(10+2{{t}^{2}})m{{s}^{-1}},\] then the average acceleration of the particle between 2s and 5s is:
A)
\[2m{{s}^{-2}}\]
done
clear
B)
\[4m{{s}^{-2}}\]
done
clear
C)
\[12m{{s}^{-2}}\]
done
clear
D)
\[14m{{s}^{-1}}\]
done
clear
View Answer play_arrow
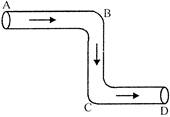
The tube shown in the figure is of uniform cross-section liquid flows through it at a constant speed in the direction shown by the arrows. The liquid exerts:
A)
a net force to the right on the tube
done
clear
B)
a net force to the left on the tube
done
clear
C)
a clockwise torque on the tube
done
clear
D)
an anti-clockwise torque on the tube
done
clear
View Answer play_arrow
Vectors \[A=2i-j+3k\] and \[B=Pi-3k+j\] are perpendicular for ?P? equal to:
A)
zero
done
clear
B)
\[-2\]
done
clear
C)
\[-3\]
done
clear
D)
5
done
clear
View Answer play_arrow
A body cools from \[{{60}^{o}}C\] to \[{{50}^{o}}C\] in 10 min. If the room temperature is \[{{25}^{o}}C\]and assuming Newton's law of cooling to hold good, the temperature of the body at the end of the next 10 min will be:
A)
\[{{38.5}^{o}}C\]
done
clear
B)
\[{{40}^{o}}C\]
done
clear
C)
\[{{42.85}^{o}}C\]
done
clear
D)
\[{{45}^{o}}C\]
done
clear
View Answer play_arrow
P-V diagram of a diatomic gas is a straight line passing through origin. The molar heat capacity of the gas in the process will be:
A)
\[4R\]
done
clear
B)
\[2.5R\]
done
clear
C)
\[3R\]
done
clear
D)
\[4R/3\]
done
clear
View Answer play_arrow
For the stationary wave \[Y=4\,\sin \,\left( \frac{\pi x}{15} \right)\cos \,(96\pi t),\] (X and Y are in cm and t in secondary the distance between a node and the next anti-node is:
A)
\[7.5\,cm\]
done
clear
B)
\[15\,cm\]
done
clear
C)
\[22.5\,cm\]
done
clear
D)
\[30\,cm\]
done
clear
View Answer play_arrow
A train is moving with a constant speed along a circular track. The engine of the train emits a sound of frequency F. The frequency heard by the guard at rear end of the train:
A)
is less than f
done
clear
B)
is greater than f
done
clear
C)
is equal to f
done
clear
D)
may be greater than, less than or equal to f depending on the factors like speed of train, length of train and radius of circular track:
done
clear
View Answer play_arrow
A straight wire of mass 200g and length \[1.5m\] caries a current of 2A. It is suspended in mid-air by a uniform horizontal magnetic field B. The magnitude of B (in tesla) is\[(g=9.8\,m{{s}^{-2}})\] :
A)
2
done
clear
B)
\[1.5\]
done
clear
C)
\[0.55\]
done
clear
D)
\[0.66\]
done
clear
View Answer play_arrow
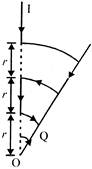
Figure shows a conductor carrying a current I. The magnetic field intensity at the point 0 (common centre of all the three arcs) is (\[\theta \] is radian):
A)
\[\frac{5{{\mu }_{0}}I\theta }{24\,\pi r}\]
done
clear
B)
\[\frac{{{\mu }_{0}}I\theta }{24\,\pi r}\]
done
clear
C)
\[\frac{11{{\mu }_{0}}I\theta }{24\,\pi r}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
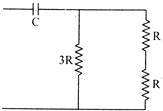
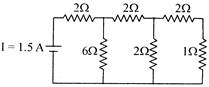
The time constant of an R-C circuit shown in the figure is:
A)
\[3\text{ }RC\]
done
clear
B)
\[2/3\text{ }RC\]
done
clear
C)
\[6RC/S\]
done
clear
D)
\[2RC\]
done
clear
View Answer play_arrow
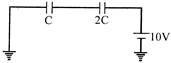
The emf of the battery shown in the figure is:
A)
\[6V\]
done
clear
B)
\[12V\]
done
clear
C)
\[18V\]
done
clear
D)
\[8V\]
done
clear
View Answer play_arrow
The potentiometer wire AB is 600cm long. At what distance from A should the jockey J touch the wire to get zero deflection in the galvanometer?
A)
\[320\,cm\]
done
clear
B)
\[120\,cm\]
done
clear
C)
\[20\,cm\]
done
clear
D)
\[450\,cm\]
done
clear
View Answer play_arrow
In the circuit shown in the figure, \[C=6\mu F\]. The charge stored in the capacitor of capacity C is:
A)
zero
done
clear
B)
\[90\mu C\]
done
clear
C)
\[40\mu C\]
done
clear
D)
\[60\mu C\]
done
clear
View Answer play_arrow
A body has a charge of one coulomb. The number of excess (or lesser) electrons on it from its normal state will be:
A)
\[\infty \]
done
clear
B)
\[1.6\times {{10}^{-19}}\]
done
clear
C)
\[1.6\times {{10}^{19}}\]
done
clear
D)
\[6.25\times {{10}^{18}}\]
done
clear
View Answer play_arrow
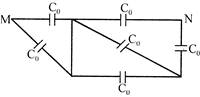
The equivalent capacitance between points M and N is:
A)
\[\frac{10}{11}{{C}_{0}}\]
done
clear
B)
\[2{{C}_{0}}\]
done
clear
C)
\[{{C}_{0}}\]
done
clear
D)
None of above
done
clear
View Answer play_arrow
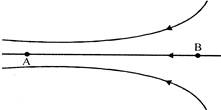
Figure shows some of the electric field lines corresponding to an electric field. The figure suggests that (\[\varepsilon \] = electric field, V = potential)
A)
\[{{V}_{A}}={{V}_{B}}\]
done
clear
B)
\[{{\varepsilon }_{A}}={{\varepsilon }_{B}}\]
done
clear
C)
\[{{V}_{A}}>{{V}_{B}}\]
done
clear
D)
\[{{V}_{A}}<{{V}_{B}}\]
done
clear
View Answer play_arrow
Three points charges \[q-2q\] and \[-2q\] are placed at the verticles of an equilateral traingle of side a. The work done by some external forces to slowly increase their separation to 2a will be:
A)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}.\frac{2{{q}^{2}}}{a}\]
done
clear
B)
\[\frac{{{q}^{2}}}{4\pi {{\varepsilon }_{0}}a}\]
done
clear
C)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}.\frac{3{{q}^{2}}}{3R}\]
done
clear
D)
zero
done
clear
View Answer play_arrow
Kirchhoff?s second law is based on the law of conversation of:
A)
momentum
done
clear
B)
charge
done
clear
C)
energy
done
clear
D)
sum of mass and energy
done
clear
View Answer play_arrow
As per Bohr model, the minimum energy (in eV) required to remove an electron from the ground state of doubly ionized \[Li\,(Z=3)\] is:
A)
\[1.51\]
done
clear
B)
\[13.6\]
done
clear
C)
\[40.8\]
done
clear
D)
\[122.4\]
done
clear
View Answer play_arrow
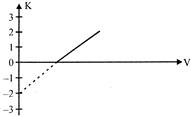
Figure represents the graph of kinetic energy (K) of photo electrons (in eV) and frequency (V) for Q metal used as cathode in photoelectric experiment. The work function of metal is:
A)
\[1eV\]
done
clear
B)
\[1.5eV\]
done
clear
C)
\[2eV\]
done
clear
D)
\[3eV\]
done
clear
View Answer play_arrow
The ratio ionization energy of Bohr's hydrogen atom and Bohr's hydrogen like lithium atom is:
A)
\[1:1\]
done
clear
B)
\[1:3\]
done
clear
C)
\[1:9\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
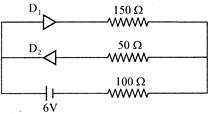
The circuit shown in the figure contains two diodes each with a forward resistance of \[50\Omega \]. and with infinite backward resistance. If the battery voltage is 6V, the current through the \[100\Omega \] resistance (in ampere) is:
A)
zero
done
clear
B)
\[0.02A\]
done
clear
C)
\[0.03A\]
done
clear
D)
\[0.036A\]
done
clear
View Answer play_arrow
Which of the following process represents a Y - decay?
A)
\[^{A}{{X}_{Z}}+\gamma {{\to }^{A}}{{X}_{Z-1}}+a+b\]
done
clear
B)
\[^{A}{{X}_{Z}}{{+}^{1}}{{n}_{0}}{{\to }^{-3}}{{X}_{Z-2}}+c\]
done
clear
C)
\[^{A}{{X}_{Z}}{{\to }^{A}}{{X}_{Z}}+\gamma \]
done
clear
D)
\[^{A}{{X}_{Z}}{{+}_{-1}}{{e}^{0}}{{\to }^{A}}{{X}_{Z-1}}+g\]
done
clear
View Answer play_arrow
A half life of \[^{215}At\] is \[100\mu s\]is. The time taken for the radioactive of a sample of \[^{215}At\] to decay to 1/16 of its initial value is:
A)
\[400\,s\]
done
clear
B)
\[6.3\,s\]
done
clear
C)
\[40\,s\]
done
clear
D)
\[300\,s\]
done
clear
View Answer play_arrow
For a medical the refractive indices for red. violet and yellow colour light are respectively \[1.52,1.64\]and\[1.60\]. The dispersive power of the material is:
A)
2
done
clear
B)
\[0.45\]
done
clear
C)
\[0.2\]
done
clear
D)
\[0.045\]
done
clear
View Answer play_arrow
Two polaroids are kept crossed to each other. Now, one of them is rotated through an angle of \[{{45}^{o}}\]. The percentage of unpolarized incident, now transmitted through the system is:
A)
15%
done
clear
B)
25%
done
clear
C)
50%
done
clear
D)
60%
done
clear
View Answer play_arrow
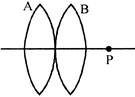
Two convex lenses placed in contact from the image of a distance of object at P. If the lens B is moved to the right, the image will:
A)
move to the left
done
clear
B)
move to the right
done
clear
C)
remain at P
done
clear
D)
move either to the left or right, depending upon focal lengths of the lenses.
done
clear
View Answer play_arrow
An alienating voltage given by \[V=300\sqrt{2}\sin \] (volts in connected across a \[1\mu f\] capacitor through an AC ammeter. The reading of the ammeter will be:
A)
\[10\,MA\]
done
clear
B)
\[40\,MA\]
done
clear
C)
\[100\,MA\]
done
clear
D)
\[15\,MA\]
done
clear
View Answer play_arrow
The emf is induced in a \[1\text{ }mH\] inductor in which the current changes from 5A to 3A in \[{{10}^{-3}}s\] is:
A)
\[2\times {{10}^{-6}}V\]
done
clear
B)
\[8\times {{10}^{-6}}V\]
done
clear
C)
\[2\,V\]
done
clear
D)
\[8\,V\]
done
clear
View Answer play_arrow
A \[50\text{ }mH\] coil carries a current of 2A, the energy stored in it (in joule) s
A)
\[0.05\]
done
clear
B)
\[0.1\]
done
clear
C)
\[0.5\]
done
clear
D)
1
done
clear
View Answer play_arrow
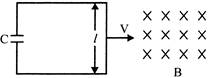
A capacitance, C is connected to a conducing rod of length Z moving with a velocity v in a transverse magnetic field B then the charge developed in the capacitor is:
A)
zero
done
clear
B)
\[BlvC\]
done
clear
C)
\[\frac{BlvC}{2}\]
done
clear
D)
\[\frac{BlvC}{3}\]
done
clear
View Answer play_arrow
Moment of inertia of ring about any of the diameters is \[{{I}_{0}}\]. The moment of inertia of the ring about any tangent perpendicular to the plane is:
A)
\[{{I}_{0}}\]
done
clear
B)
\[2{{I}_{0}}\]
done
clear
C)
\[3{{I}_{0}}\]
done
clear
D)
\[4{{I}_{0}}\]
done
clear
View Answer play_arrow
If the absolute temperature of a gas is doubled and the pressure is reduced to one half, the volume of the gas will:
A)
remain unchanged
done
clear
B)
be doubled
done
clear
C)
increase four times
done
clear
D)
be halved
done
clear
View Answer play_arrow
The order of acidic strength of boron trihalides is
A)
\[B{{F}_{3}}<BC{{l}_{3}}<B{{B}_{3}}<B{{I}_{3}}\]
done
clear
B)
\[B{{I}_{3}}<BB{{r}_{3}}<BC{{l}_{3}}<B{{F}_{3}}\]
done
clear
C)
\[B{{F}_{3}}<B{{I}_{3}}<BC{{l}_{3}}<BB{{r}_{3}}\]
done
clear
D)
\[BC{{l}_{3}}<BB{{r}_{3}}<B{{I}_{3}}<B{{F}_{3}}\]
done
clear
View Answer play_arrow
The carbide which reacts with water to form ethyne is:
A)
\[SiC\]
done
clear
B)
\[A{{l}_{4}}{{C}_{3}}\]
done
clear
C)
\[B{{e}_{2}}C\]
done
clear
D)
\[Ca{{C}_{2}}\]
done
clear
View Answer play_arrow
Density of a crystal remains unchanged as a result of:
A)
ionic defect
done
clear
B)
schottky defect
done
clear
C)
frenkel defect
done
clear
D)
crystal defect
done
clear
View Answer play_arrow
The dissociation constant of acetic acid \[{{K}_{a}}\] is \[1.74\times {{10}^{-5}}\]at 29 K. The pH of a solution of \[0.1\text{ }M\]acetic acid is:
A)
\[2.88\]
done
clear
B)
\[3.6\]
done
clear
C)
\[4.0\]
done
clear
D)
\[1.0\]
done
clear
View Answer play_arrow
By adding \[20\text{ }ml\] of \[0.1\text{ }NHCl\]to \[20\text{ }ml\] of \[0.001\text{ }N\text{ }NaOH\] solution. The pH of the resulting solution would be
A)
1
done
clear
B)
5
done
clear
C)
\[1.3\]
done
clear
D)
9
done
clear
View Answer play_arrow
Which one of the following pairs of elements is called 'chemical twins' because of their very similar chemical properties?
A)
\[Mn\] and W
done
clear
B)
\[Mo\] and \[Tc\]
done
clear
C)
\[Fe\] and \[Co\]
done
clear
D)
\[Hf\] and \[Zr\]
done
clear
View Answer play_arrow
Barbituric acid is used as:
A)
an antipyretic
done
clear
B)
an antiseptic
done
clear
C)
a tranquilizer
done
clear
D)
an analgesic
done
clear
View Answer play_arrow
Invar an alloy of \[Fe\] and \[Ni\] is used in watches and meter scale. Its characteristic property is:
A)
small coefficient of expansion
done
clear
B)
resistance of corrosion
done
clear
C)
magnetic nature
done
clear
D)
hardness and elasticity
done
clear
View Answer play_arrow
The acid which contains the aldehyde group is
A)
acetic acid
done
clear
B)
formic acid
done
clear
C)
ben zoic acid
done
clear
D)
picric acid
done
clear
View Answer play_arrow
Pyrolusite is an ore of
A)
magnesium
done
clear
B)
manganese
done
clear
C)
zinc
done
clear
D)
iron
done
clear
View Answer play_arrow
Acrolein is formed from glycerol by heating with to
A)
acidic \[KMn{{O}_{4}}\]
done
clear
B)
\[B{{r}_{2}}\] water
done
clear
C)
\[KHS{{O}_{4}}\]
done
clear
D)
\[HN{{O}_{3}}\]
done
clear
View Answer play_arrow
An aqueous solution of \[6.3g\]of oxalic acid dihydrate is made up to 250 ml. The volume of \[0.1\text{ }N\text{ }NaOH\] required to completely neutralise \[10\text{ }ml\] of this solution is
A)
\[40\text{ }ml\]
done
clear
B)
\[20\text{ }ml\]
done
clear
C)
\[10\text{ }ml\]
done
clear
D)
\[\text{4 }ml\]
done
clear
View Answer play_arrow
How many grams of \[KCl\]should be added to \[1.0kg\] of water to lower its freezing point to\[-{{8.0}^{o}}C\]? (\[{{K}_{f}}\] for water \[={{1.86}^{o}}C\,kg\,mo{{l}^{-1}}\] )
A)
\[16.02\text{ }g\]
done
clear
B)
\[1.602\text{ }g\]
done
clear
C)
\[0.1602\,g\]
done
clear
D)
\[160.2\text{ }g\]
done
clear
View Answer play_arrow
The green house effect is caused by
A)
\[NO\]
done
clear
B)
\[N{{O}_{2}}\]
done
clear
C)
\[CO\]
done
clear
D)
\[C{{O}_{2}}\]
done
clear
View Answer play_arrow
A piece of wood from an archaeological source shows a \[^{14}C\] activity which is 60% of the acitivity found in a fresh wood today. Calculate the age of the archaeological sample. Half-life period of C-14 is 5770 year
A)
3215 year
done
clear
B)
6216 year
done
clear
C)
4258 year
done
clear
D)
2129 year
done
clear
View Answer play_arrow
The bond order in the species \[{{O}_{2}},O_{2}^{+}\] and \[O_{2}^{-}\]follows the order
A)
\[{{O}_{2}}>O_{2}^{+}>O_{2}^{-}\]
done
clear
B)
\[O_{2}^{+}>{{O}_{2}}>O_{2}^{-}\]
done
clear
C)
\[O_{2}^{-}>{{O}_{2}}>O_{2}^{+}\]
done
clear
D)
\[O_{2}^{+}>O_{2}^{-}>{{O}_{2}}\]
done
clear
View Answer play_arrow
Which of the following is a buffer solution?
A)
\[KOH\] and \[KCl\]
done
clear
B)
\[{{K}_{2}}S{{O}_{4}}\] and \[{{H}_{2}}S{{O}_{4}}\]
done
clear
C)
\[NaOH\] and \[KOH\]
done
clear
D)
\[C{{H}_{3}}COOH\] and \[C{{H}_{3}}COONa\]
done
clear
View Answer play_arrow
Which of the following is true concerning first order reaction?
A)
The rate falls to zero after two half-life
done
clear
B)
The rate is independent of initial concentration
done
clear
C)
The rate is proportional to temperature
done
clear
D)
The half-life is independent of the initial concentration.
done
clear
View Answer play_arrow
When a mixture of ammonium chloride and sodium nitrite is heated, the gas evolved is:
A)
nitrogen
done
clear
B)
oxygen
done
clear
C)
\[N{{H}_{3}}\]
done
clear
D)
nitrous oxide
done
clear
View Answer play_arrow
\[C{{H}_{3}}C{{H}_{2}}I\xrightarrow{KCN}A\xrightarrow[Partial]{{{H}^{2}},{{H}_{2}}O}B\xrightarrow{B{{r}_{2}}/KOH}C\]The product C is
A)
\[{{C}_{2}}{{H}_{5}}N{{H}_{2}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{5}}CON{{H}_{2}}\]
done
clear
C)
\[{{C}_{8}}{{H}_{7}}N{{H}_{2}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{5}}COOH\]
done
clear
View Answer play_arrow
Which of the following is not correct?
A)
Enzymes are proteins produced by living organisms
done
clear
B)
They catalyse specific reactions
done
clear
C)
Temperature affects enzyme activity
done
clear
D)
pH of the solution does not affect enzyme activity
done
clear
View Answer play_arrow
The EMF of a cell is realed to the equilibrium constant of the cell reaction as
A)
In \[{{K}_{C}}=\frac{nFE_{cell}^{''}}{RT}\]
done
clear
B)
\[{{K}_{C}}=\frac{nFE_{cell}^{o}}{RT}\]
done
clear
C)
\[E_{cell}^{o}\frac{-RT}{nF}\] In \[{{K}_{C}}\]
done
clear
D)
\[{{K}_{C}}=\frac{RT}{nF}\] In \[E_{cell}^{o}\]
done
clear
View Answer play_arrow
If the standard electrode potenital of \[C{{u}^{2+}}/Cu\]electrode is \[0.34\text{ }V,\] what is the electrode potential of \[0.01\text{ }M\] concentration of \[C{{u}^{2+}}\]? (T= 298 K)
A)
\[0.399\,V\]
done
clear
B)
\[0.281\,V\]
done
clear
C)
\[0.222\,V\]
done
clear
D)
\[0.176\,V\]
done
clear
View Answer play_arrow
Standard entropy of \[{{X}_{2}},{{Y}_{2}}\] and \[X{{Y}_{3}}\] are 60, 40 and \[50\,J{{K}^{-1}}mo{{l}^{-1}}\] respectively. For the reaction, \[\frac{1}{2}{{X}_{2}}+\frac{3}{2}{{Y}_{2}}\xrightarrow{{}}X{{Y}_{3}},\] \[\Delta H=-30\,kJ\]to be at equilibrium, the temperature will be
A)
\[1250\text{ }K\]
done
clear
B)
\[500\,K\]
done
clear
C)
\[750\text{ }K\]
done
clear
D)
\[1000\text{ }K\]
done
clear
View Answer play_arrow
In which of the following octahedral complexes of \[Co\] (at no. 27) will the magnitude of Ay be the highest?
A)
\[{{[Co\,{{(CN)}_{6}}]}^{3-}}\]
done
clear
B)
\[{{[Co\,{{({{C}_{2}}{{O}_{4}})}_{3}}]}^{3-}}\]
done
clear
C)
\[{{[Co\,{{({{H}_{2}}O)}_{6}}]}^{3+}}\]
done
clear
D)
\[{{[Co\,{{(N{{H}_{3}})}_{6}}]}^{3+}}\]
done
clear
View Answer play_arrow
The organic chloro compound which shows complete stereo chemical inversion during a \[{{S}_{N}}^{2}\]reaction is
A)
\[{{({{C}_{2}}{{H}_{5}})}_{2}}CHCl\]
done
clear
B)
\[{{(C{{H}_{3}})}_{3}}CCl\]
done
clear
C)
\[{{(C{{H}_{3}})}_{2}}CHCl\]
done
clear
D)
\[C{{H}_{3}}Cl\]
done
clear
View Answer play_arrow
Which one of the following constitutes a group of the isoelectronic species?
A)
\[C_{2}^{2-},O_{2}^{-},CO,NO\]
done
clear
B)
\[N{{O}^{-}},C_{2}^{2-},C{{N}^{-}},{{N}_{2}}\]
done
clear
C)
\[C{{N}^{-}},{{N}_{2}},O_{2}^{2},CO_{2}^{2-}\]
done
clear
D)
\[{{N}_{2}},O_{2}^{-},N{{O}^{+}},CO\]
done
clear
View Answer play_arrow
Amount of oxalic acid present in a solution can be determined by its titration with \[KMn{{O}_{4}}\] solution in the presence of\[{{H}_{2}}S{{O}_{4}}\]. The titration gives unsatisfactory result when carried out in the presence of \[HCl\] because \[HCl\]
A)
gets oxidised by oxalic acid to chlorine
done
clear
B)
frunishes \[{{H}^{+}}\] ions in addition to those from oxalic acid
done
clear
C)
reduces permanganate to \[M{{n}^{2+}}\]
done
clear
D)
Oxidises oxalic acid to carbon dioxide and water
done
clear
View Answer play_arrow
Among the following substituted silanes the one which will give rise to cross linked silicone polymer on hydrolysis is
A)
\[RuSi\]
done
clear
B)
\[{{R}_{3}}SiCl\]
done
clear
C)
\[{{R}_{2}}SiC{{l}_{2}}\]
done
clear
D)
\[RSiC{{l}_{3}}\]
done
clear
View Answer play_arrow
Which of the following pair of molecules will have permanent dipole moment for both members?
A)
\[N{{O}_{2}}\] and \[C{{O}_{2}}\]
done
clear
B)
\[N{{O}_{2}}\] and \[{{O}_{3}}\]
done
clear
C)
\[Si{{F}_{4}}\] and \[C{{O}_{2}}\]
done
clear
D)
\[Si{{F}_{4}}\] and \[N{{O}_{2}}\]
done
clear
View Answer play_arrow
One gram sample of \[N{{H}_{4}}N{{O}_{3}}\] is decomposed in a bomb calorimeter, the temperature of the calorimeter increases by\[6.12\text{ }K\]. The heat capacity of the system is\[1.23\text{ }kJ/g/deg\]. What is the molar heat of decomposition for \[N{{H}_{4}}N{{O}_{3}}\]?
A)
\[-7.53\text{ }kJ/mol\]
done
clear
B)
\[-398.1\text{ }kJ/mol\]
done
clear
C)
\[-16.1\text{ }kJ/mol\]
done
clear
D)
\[-\text{ }602\text{ }kJ/mol\]
done
clear
View Answer play_arrow
With increase in temperature, ionic product of water
A)
decreases
done
clear
B)
increases
done
clear
C)
may increase or decrease
done
clear
D)
remains same
done
clear
View Answer play_arrow
When hydrogen peroxide is treated with cold acidified \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] solution containing ether, a blue colour is obtained. This is due to
A)
Chromium sulphate
done
clear
B)
Potassium chromate
done
clear
C)
Perchromic acid
done
clear
D)
Chromium trioxide
done
clear
View Answer play_arrow
Which of the following statements is correct.
A)
Elemental sodium can be prepared and isolated by electrolysing an aqueous solution of \[NaCl\].
done
clear
B)
Elemental Na is a strong oxidising agent.
done
clear
C)
Elemental Na is insoluble in \[N{{H}_{3}}\]
done
clear
D)
Elemental Na is easily oxidised.
done
clear
View Answer play_arrow
Pick out the incorrect statement regarding ozone.
A)
The central oxygen in \[{{O}_{3}}\] is \[S{{P}^{2}}\]- hybridized.
done
clear
B)
It causes tailing of mercury.
done
clear
C)
The two \[O-O\] bonds in \[{{O}_{3}}\] are equal due to resonance.
done
clear
D)
With \[Ba{{O}_{2}}\] it reacts to produce \[{{H}_{2}}{{O}_{2}}\].
done
clear
View Answer play_arrow
Water transported through lead pipes becomes poisonous due to the formation of
A)
\[Pb{{(OH)}_{2}}\]
done
clear
B)
\[P{{b}_{3}}{{O}_{4}}\]
done
clear
C)
\[Pb{{O}_{2}}\]
done
clear
D)
\[PbO\]
done
clear
View Answer play_arrow
Which of the following species does not exist?
A)
\[{{[GeC{{l}_{6}}]}^{2-}}\]
done
clear
B)
\[{{[\text{SnC}{{\text{l}}_{\text{6}}}]}^{2-}}\]
done
clear
C)
\[{{[\text{SiC}{{\text{l}}_{\text{6}}}]}^{2-}}\]
done
clear
D)
\[{{[CC{{l}_{6}}]}^{2-}}\]
done
clear
View Answer play_arrow
The substance used as a smoke screen in warfare is
A)
\[SiC{{l}_{4}}\]
done
clear
B)
\[AlC{{l}_{3}}\]
done
clear
C)
\[P{{H}_{3}}\]
done
clear
D)
Acetylene
done
clear
View Answer play_arrow
The stability of the hydrides follows the order
A)
\[N{{H}_{3}}>P{{H}_{3}}>As{{H}_{3}}>Sb{{H}_{3}}\]
done
clear
B)
\[N{{H}_{3}}<P{{H}_{3}}<As{{H}_{3}}<Sb{{H}_{3}}\]
done
clear
C)
\[P{{H}_{3}}<N{{H}_{3}}<As{{H}_{3}}<Sb{{H}_{3}}\]
done
clear
D)
\[As{{H}_{3}}>N{{H}_{3}}>P{{H}_{3}}>Sb{{H}_{3}}\]
done
clear
View Answer play_arrow
Both methane and ehtane can be prepared in one step from
A)
\[C{{H}_{3}}C{{H}_{2}}I\]
done
clear
B)
\[C{{H}_{3}}Br\]
done
clear
C)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
D)
\[C{{H}_{3}}OH\]
done
clear
View Answer play_arrow
Aniline reacts with phosgene and \[KOH\] to form
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Soft drinks and baby feeding bottles are generally made up of
A)
Polyester
done
clear
B)
Polyamide
done
clear
C)
Polystyrene
done
clear
D)
Polyurethane
done
clear
View Answer play_arrow
reagent x is
A)
air
done
clear
B)
\[KMn{{O}_{4}}/{{H}_{2}}S{{O}_{4}}\]
done
clear
C)
\[{{K}_{2}}{{S}_{2}}{{O}_{8}}\]
done
clear
D)
\[{{K}_{2}}S{{O}_{5}}\]
done
clear
View Answer play_arrow
The enzymes which are used to convert starch into ethyl alcohol are
A)
maltase, diastase
done
clear
B)
diastase, maltase, zymase
done
clear
C)
invertase, zymase
done
clear
D)
invertase, diastase, maltase
done
clear
View Answer play_arrow
Select incorrect with respect to property of cardiac muscles
A)
Exhibits Tetanus
done
clear
B)
Exhibits rhymicity
done
clear
C)
Immune to fatigue
done
clear
D)
It is involuntary and striated
done
clear
View Answer play_arrow
Find the incorrect match with respect to epithelium and function
A)
Cuboidal epithelium - duct of gland and tubular part of Nephron
done
clear
B)
Squamous epithelium - wall of blood vessel and alveoli of lungs
done
clear
C)
Columnar epithelium - esophagus and pharynx
done
clear
D)
Cilliated epithelium - Bronchioles and fallopian tubes
done
clear
View Answer play_arrow
Setal in earthworm
A)
are maximum in clitelum
done
clear
B)
are female genitals
done
clear
C)
each segment or section has muscles and bristles called setae
done
clear
D)
None of the above
done
clear
View Answer play_arrow
14th segment in Earthworm is
A)
Male genital pore
done
clear
B)
Female genital pore
done
clear
C)
Testis in earth worm
done
clear
D)
Spermathecal apertures
done
clear
View Answer play_arrow
Select correct with respect to Arthropoda
A)
Schiocoelom present, circulation is open type
done
clear
B)
Enterocoelom present, segmentation present
done
clear
C)
Close type circulation, Schizocoelumpresent
done
clear
D)
Schizocoelom present, segmentation absent
done
clear
View Answer play_arrow
A. Collagen is the most abundant protein in animal world. B. Rubisco is the most abundant protein in whole biosphere.
A)
Both A and B are correct
done
clear
B)
Both A and B are incorrect
done
clear
C)
A is correct, B is incorrect
done
clear
D)
A is incorrect, B is correct
done
clear
View Answer play_arrow
Polysaccharides are the components of cell wall in plants and
A)
Insects
done
clear
B)
Fungi
done
clear
C)
Monera
done
clear
D)
More than one is correct
done
clear
View Answer play_arrow
Human beings have 2 sets of teeth during their life, this type of definition is called
A)
Diphyodont
done
clear
B)
Thecodont
done
clear
C)
Premolar
done
clear
D)
Acrodont
done
clear
View Answer play_arrow
A. Jejunum opens into large intestine. B. Pyrolic portion opens into the first part of small intestine.
A)
Both A and B are correct
done
clear
B)
Only A is incorrect
done
clear
C)
Both are incorrect
done
clear
D)
Only B is incorrect
done
clear
View Answer play_arrow
Volume of air inspired or expired during a normal respiration is
A)
Tidal volume
done
clear
B)
Total lung capacity
done
clear
C)
Expiratory reserve volume
done
clear
D)
Inspiratory reserve volume
done
clear
View Answer play_arrow
Every 100 ml of oxygenated blood can deliver __of \[{{O}_{2}}\] to tissues in normal condition
A)
\[10\text{ }ml\]
done
clear
B)
\[15\text{ }ml\]
done
clear
C)
\[\text{20 }ml\]
done
clear
D)
\[\text{30 }ml\]
done
clear
View Answer play_arrow
All the following statements regarding WBC are correct except
A)
Phagocytic - Monocyte, Neutrophil
done
clear
B)
\[6000\text{ -}\,\,8000/m{{m}^{3}}\] - Normal concentration
done
clear
C)
Granulocytes - Basophil, eosinophil, neutrophil
done
clear
D)
Eosinophil secrete histamine serotonin and heparin
done
clear
View Answer play_arrow
Select incorrect statement with respect to wheat and pea plants
A)
Belonging to different order of same division
done
clear
B)
Represent same division
done
clear
C)
Arc included in same order
done
clear
D)
Belong to different classes
done
clear
View Answer play_arrow
Funaria sporophyte not has
A)
Rhizoid
done
clear
B)
Leaves
done
clear
C)
Axis
done
clear
D)
All of these
done
clear
View Answer play_arrow
Select odd one with respect to Archaebacteria
A)
Thermoacidophiles are facultative anaerobe
done
clear
B)
Halococcus, cell membrane has ATP are activity
done
clear
C)
Methanogens are aerobic chemoautotrophs.
done
clear
D)
Cell wall helps them to withstand in extreme environmental conditions
done
clear
View Answer play_arrow
Select true statement
A)
Mitotic division are found in Bacteria
done
clear
B)
True sexual reproduction is found in bacteria
done
clear
C)
Bacteria reproduce mainly by fission
done
clear
D)
Budding is most common method of reproduction in bacteria
done
clear
View Answer play_arrow
Select incorrect with respect to sexual reproduction in bryophytes
A)
Archegonium is female sex organ
done
clear
B)
Sperms are twisted and comma-shaped with two flagella
done
clear
C)
Sex organs are multicellular and non-jacketed
done
clear
D)
Male sex organ is Antheridium
done
clear
View Answer play_arrow
For the given pedigree chart select correct statement
A)
Male parent is homozygous autosomal dominant
done
clear
B)
Trait under study is colour blindness
done
clear
C)
Both parents have heterozygous condition
done
clear
D)
Female parent is reussive
done
clear
View Answer play_arrow
A plant in which petiole is modified into flat green and photosynthetic structure
A)
Acacia
done
clear
B)
Casuarina
done
clear
C)
Opuntia
done
clear
D)
Hydrilla
done
clear
View Answer play_arrow
Select correct with respect to anatomy of monocot stem
A)
No endodermis
done
clear
B)
One or few layered parenchymatous pericycle
done
clear
C)
Hypodermis is collenchymatous
done
clear
D)
Vascular bundles are conjoint and open
done
clear
View Answer play_arrow
Mitochondria are the sites of
A)
Aerobic respiration
done
clear
B)
Glycolysis
done
clear
C)
E.TS. in E. coli
done
clear
D)
More than one are correct
done
clear
View Answer play_arrow
The endomembrane system does not includes
A)
ER
done
clear
B)
Golgibody
done
clear
C)
uacuole
done
clear
D)
Glyoxysome
done
clear
View Answer play_arrow
Middle layer is mainly made of__,_
A)
Calcium sulphate
done
clear
B)
Calcium pectate
done
clear
C)
Magnesium phosphate
done
clear
D)
Sulphur
done
clear
View Answer play_arrow
Plant part used for tissue culture is sterilized by
A)
Heat
done
clear
B)
Methiolate
done
clear
C)
UV-rays
done
clear
D)
X-Rays
done
clear
View Answer play_arrow
Dissolution of synaptonemal complex is the beginning
A)
Zygatene
done
clear
B)
Diakinesis
done
clear
C)
Diplotene
done
clear
D)
Pachytene
done
clear
View Answer play_arrow
Select the correct match with respect to mineral and its deficiency symptom
A)
Magnesium - Brown heart of turnip
done
clear
B)
Calcium - Degeneration of meristem
done
clear
C)
Potassium - Loss of apical dominance
done
clear
D)
Nitrogen - Chlorosis first in young leaf
done
clear
View Answer play_arrow
Select correct sequence of \[{{C}_{3}}\] cycle
A)
Regeneration, Carboxylation, Reduction
done
clear
B)
Carboxylation, Regeneration, Reduction
done
clear
C)
Reduction, Regeneration, Carboxylation
done
clear
D)
Carboxylation, Reduction, Regeneration
done
clear
View Answer play_arrow
4C compound is converted into 3C compound in (with respect to C4 plants)
A)
Chloroplast
done
clear
B)
Mesophyll cytoplasm
done
clear
C)
Bundle sheath cells
done
clear
D)
Mesophyll chloroplast
done
clear
View Answer play_arrow
Which hormone promote rapid internode or petiole elongation in deep water rice plants
A)
ABA
done
clear
B)
Auxin
done
clear
C)
Ethylene
done
clear
D)
Kinetin
done
clear
View Answer play_arrow
Select incorrect match
A)
Allium Bulb
done
clear
B)
Agave - Bulbil
done
clear
C)
Eichornia - Runner
done
clear
D)
Corm - Crocus
done
clear
View Answer play_arrow
Select incorrect match
A)
Molecular diagnosis - Genetherapy
done
clear
B)
Vaccine safety - Transgenic mice
done
clear
C)
Emphysema treatment - \[{{\alpha }_{1}}\] antitrysin
done
clear
D)
p carotene rich transgenic rice - Golden rice
done
clear
View Answer play_arrow
Select odd one with respect to honeybee
A)
Polymorphic, Social, Useful
done
clear
B)
A natural product of honey bee is honey
done
clear
C)
Metathoracic leg has pollen basket
done
clear
D)
Bee hive has only one queen
done
clear
View Answer play_arrow
Key concept of Darwinian theory of evolution is
A)
Use and disuse of organs
done
clear
B)
Natural selection
done
clear
C)
Branching descent
done
clear
D)
Both [b] and [c]
done
clear
View Answer play_arrow
Wolf and Tasmanian welfare example of
A)
Adaptive radiation
done
clear
B)
Convergent evolution
done
clear
C)
Genetic drift
done
clear
D)
Hardy Weinberg principles
done
clear
View Answer play_arrow
Darwin Finches are example of
A)
Transfection
done
clear
B)
Divergent evolution
done
clear
C)
Adaptive radiation
done
clear
D)
Convergent evolution
done
clear
View Answer play_arrow
The first clinical gene therapy was applied for which of following disease
A)
SCID
done
clear
B)
AIDs
done
clear
C)
Multiple sclerosis
done
clear
D)
Haemophilia
done
clear
View Answer play_arrow
A method of introduction of r-DNA in animal cell is to the mix it up with charged substance like calcium phosphate and spread it over the host cell is called
A)
Macroprojection
done
clear
B)
Transfection
done
clear
C)
Electroporation
done
clear
D)
Transduction
done
clear
View Answer play_arrow
As ELISA is for AIDs, then Wassermann test is for
A)
Treponema
done
clear
B)
Syphilis
done
clear
C)
VDRL
done
clear
D)
Typhoid
done
clear
View Answer play_arrow
VDRL is investigation for
A)
Pneumonia
done
clear
B)
Typhoid
done
clear
C)
Syphilis
done
clear
D)
HIV
done
clear
View Answer play_arrow
During embryonic development of foetus, genital' organs are well developed at the
A)
End of 10 weeks
done
clear
B)
End of 12 weeks
done
clear
C)
End of 2nd month
done
clear
D)
Starting of 1st month
done
clear
View Answer play_arrow
First sign of growing factor is
A)
Listening of heart sound
done
clear
B)
Movement in womb
done
clear
C)
Bipedal sense
done
clear
D)
Crying
done
clear
View Answer play_arrow
First menstruation is
A)
Spermiation
done
clear
B)
Menarche
done
clear
C)
Spermiogenesis
done
clear
D)
Oogenesis
done
clear
View Answer play_arrow
Transformation of spermatids into sperm is
A)
Spermicide
done
clear
B)
Spermiogenesis
done
clear
C)
Ovulation
done
clear
D)
Gametogenesis
done
clear
View Answer play_arrow
Function of Bulb urethral gland is
A)
Testosterone secretion
done
clear
B)
Lubricant secretion
done
clear
C)
Releasing of sperm
done
clear
D)
Egg releasing
done
clear
View Answer play_arrow
Cortisol
A)
stimulate erythropoisis
done
clear
B)
stimulate secretion of \[HCl\] and pepsinogen
done
clear
C)
Suppreses the immune response
done
clear
D)
more than one are correct
done
clear
View Answer play_arrow
Gastrin
A)
Secrete fructose
done
clear
B)
Secrets testosterone
done
clear
C)
Stimulate secretion of pepsinogen
done
clear
D)
Stimulate thy mosin
done
clear
View Answer play_arrow
Differentiation of T-Lymphocyte is due to
A)
Parathyroid
done
clear
B)
Thyrocalcitonin
done
clear
C)
Thymosin
done
clear
D)
Oxytocin
done
clear
View Answer play_arrow
Introduction of Nileperch in lake Victoria of East Africa resulted in
A)
Excessive growth of Cichlid fish
done
clear
B)
Elimination of many species of Cichlid fish
done
clear
C)
Removal of water areas
done
clear
D)
Increase in water hyacinth
done
clear
View Answer play_arrow
Most effective technique for separation of particulate matter from polluted air
A)
ESPs
done
clear
B)
Cyclone Collectors
done
clear
C)
Trajectory separators
done
clear
D)
Wet scrubbers
done
clear
View Answer play_arrow
Select correct option with respect to changes during ecological succession
A)
Decrease in stability
done
clear
B)
Biomass reduction
done
clear
C)
Increase in structural complexity
done
clear
D)
Decrease in species diversity
done
clear
View Answer play_arrow
Select the incorrect match
A)
Flora - Index of plant species of. a particular area
done
clear
B)
Botanical garden - Quick referral system
done
clear
C)
Museum - Dead specimens of plant and animals
done
clear
D)
Zoological park - condition similar to natural habitats
done
clear
View Answer play_arrow
That process which is not basic to classical taxonomy is
A)
Evolutionary relationship
done
clear
B)
Identification
done
clear
C)
Characterization
done
clear
D)
Classification
done
clear
View Answer play_arrow
The male and female gametophytes do not have an independent free living existence in
A)
Liverworts
done
clear
B)
Pteridophytes
done
clear
C)
Mosses
done
clear
D)
Gymnosperms
done
clear
View Answer play_arrow
Select the incorrect statement
A)
All seed bearing plants follow diplontic life cycle
done
clear
B)
Sporophytic generation is represented only by one called zygote in haplontic life cycle
done
clear
C)
A dominant, independent, photosynthetic phase is represented by a diploid gametophyte in bryophytes
done
clear
D)
Laminaria show hapio - diplontic life cycle
done
clear
View Answer play_arrow
Roots that help some plants growing in swampy areas to get oxygen for respiration are called
A)
Prop root
done
clear
B)
Pneumatophores
done
clear
C)
Stilt roots
done
clear
D)
Adventitious roots
done
clear
View Answer play_arrow
Worled phyllotaxy is seen in
A)
Alstonia
done
clear
B)
Colotropic
done
clear
C)
Sunflower
done
clear
D)
Mustard
done
clear
View Answer play_arrow
In epigynous flowers, the ovary is _ as in __
A)
Superior, Rose
done
clear
B)
Inferior, sunflower
done
clear
C)
Half inferior, cucumber
done
clear
D)
Half superior, Guava
done
clear
View Answer play_arrow
Ascospore is normally
A)
Haploid
done
clear
B)
Diploid
done
clear
C)
Triploid
done
clear
D)
Tetraploid
done
clear
View Answer play_arrow
Perianth is found in
A)
Petunia, Datura
done
clear
B)
Trifolium, Indigofera
done
clear
C)
Asparagus, Lily
done
clear
D)
Tobacco, Mustard
done
clear
View Answer play_arrow
Which of the following is a secondary lateral meristem?
A)
Interfascicular cambium
done
clear
B)
Intrafascicular cambium
done
clear
C)
Apical meristem
done
clear
D)
Intercalary meristem
done
clear
View Answer play_arrow
Select the incorrect statement
A)
Parenchyma forms major component within organs
done
clear
B)
Mechanical support to petiole of a leaf and herbaceous
done
clear
C)
Sclerenchyma are usually dead and have protoplast
done
clear
D)
Sclerenchyma can be in the form of fibres of sclereids
done
clear
View Answer play_arrow
Select the correct match
A)
Lentical - Produced by differentiation of phellem
done
clear
B)
Heart wood - Peripheral region of secondary xylem
done
clear
C)
Companion cells - Specialized parenchymatous cells
done
clear
D)
Trichomes - Always unbranched unicellular and stiff
done
clear
View Answer play_arrow
The major site for the synthesis of lipid in a eukaryotic cell is
A)
Golgi apparatus
done
clear
B)
Smooth endoplasmic reticulum
done
clear
C)
Microbodies
done
clear
D)
Ccntrosome
done
clear
View Answer play_arrow
Select the incorrect statement
A)
Cell membrane provides selective entry to molecules
done
clear
B)
RER is frequently observed in cells involved in protein synthesis and secretion
done
clear
C)
ER, Pcroxisomes, Golgi complex and lysosomes from the endomembrane system
done
clear
D)
Golgi apparatus performs the function of packaging of material
done
clear
View Answer play_arrow
Select the odd one out with respect to lysosomes
A)
Rich in all types of hydrolytic enzymes
done
clear
B)
Optimally active at acidic pH
done
clear
C)
Performs catalase activity
done
clear
D)
Formed by the process of packaging in golgi apparatus
done
clear
View Answer play_arrow
The basal body of cilia or flagella are structurally equivalent to
A)
Centriole
done
clear
B)
Kinetochore
done
clear
C)
Centromere
done
clear
D)
Muclear matrix
done
clear
View Answer play_arrow
Select the odd one out with respect to S-shape of cell cycle
A)
Amount of DNA per cell doubles
done
clear
B)
Centriole duplicates in cytoplasm of animal cells
done
clear
C)
Number of chromosomes remains same
done
clear
D)
Tubulin proteins are synthesized
done
clear
View Answer play_arrow
Which stage of meiosis prophase-I is characterized by the appearance of recombination nodules?
A)
Zygotene
done
clear
B)
Pachytene
done
clear
C)
Diplotene
done
clear
D)
Leptotene
done
clear
View Answer play_arrow
Select the correct statement
A)
Golgi, ER, nucleolus and nuclear envelope are partially visible at the end of mitotic prophase
done
clear
B)
Cells are metabolically inactive in quiescent stage
done
clear
C)
Growth of multicellular organisms is due to mitosis
done
clear
D)
Sister chromatids separate during anaphase-I of meiosis
done
clear
View Answer play_arrow
Match items in Column-I with Column-II
Column-I Column-II A. Aquaporins I. By vascular System B. Long distance transport II. Uphill transport C. Facilitated diffusion III. Passive movement D. Active transport IV. Water channels
A)
A-I B-IV C-II D-III
done
clear
B)
A-IV B-I C-III D-II
done
clear
C)
A-IV B-III C-II D-I
done
clear
D)
A-I B-II C-III D-IV
done
clear
View Answer play_arrow
Select the odd one with respect to solute potential
A)
For a solution at atmospheric pressure water potential is equal to solute potential
done
clear
B)
Solute potential is always negative
done
clear
C)
More the solute molecules, more negative is the solute potential
done
clear
D)
All solutions have a higher solute potential than pure water
done
clear
View Answer play_arrow
Select the incorrect match
A)
Root Pressure - Guttation
done
clear
B)
Imbibition - Osmotic phenomenon
done
clear
C)
Osmosis - Diffusion of water across SPM
done
clear
D)
Transpiration - Cohesion - Adhesion
done
clear
View Answer play_arrow
The essential mineral element in one that
A)
is indirectly involved in metabolism of plant
done
clear
B)
cannot be replaced by any other element in plants
done
clear
C)
is most abundant in the plant
done
clear
D)
can enter the plants through roots
done
clear
View Answer play_arrow
Which of the following is incorrect?
A)
Rhizobium and Frankia can be free living in soil
done
clear
B)
Nitrogen is the limiting nutrient for natural and agricultural ecosystem
done
clear
C)
Nitrogenase is highly sensitive to molecular oxygen, it requires aerobic conditions of function
done
clear
D)
Ammonia is the first stable product of nitrogen fixation
done
clear
View Answer play_arrow
Which of the following statement about hydroponics is incorrect?
A)
It is the technique of growing plants is a nutrient solution
done
clear
B)
By this method essential elements and their deficiency symptoms can be studied
done
clear
C)
The technique has been successfully employed for commercial production of tomato seedless cucumber and lettuce
done
clear
D)
The growth is optimum when nutrient solution is not aerated
done
clear
View Answer play_arrow
The regions of light wavelength at which there is maximum absorption by chlorophyll-a is
A)
Red and green light
done
clear
B)
Blue and Violet light
done
clear
C)
Blue and Red
done
clear
D)
Green and Orange
done
clear
View Answer play_arrow
Consider the following statements in the context of cyclic photophosphorylation
A. Only PS 1 functions B. NADPH + \[{{H}^{+}}\] is formed C. Occurs when light of wavelength beyond 680 nm is available for excitation D. Oxygen is evolved of these statement:
A)
B & D are correct
done
clear
B)
A & C are correct
done
clear
C)
All are correct
done
clear
D)
A, B & D are correct
done
clear
View Answer play_arrow
The ATP and \[NADPH+{{H}^{+}}\] requirement for reduction of one molecule of \[C{{o}_{2}}\] in \[{{C}_{3}}\] plants in the form of ratio (\[C{{o}_{2}}\] : ATP : \[NADP{{H}_{2}}\]) is
A)
\[1:3:2\]
done
clear
B)
\[1:5:2\]
done
clear
C)
\[1:2:1\]
done
clear
D)
\[1:6:1\]
done
clear
View Answer play_arrow
Plant showing hatch and slack path are related to all, except
A)
They have a special type of leaf anatomy
done
clear
B)
They tolerate higher temperatures
done
clear
C)
They have greater productivity of biomass
done
clear
D)
They have phosphoenol pyruvate as the first stable product
done
clear
View Answer play_arrow
Photorespiration is not related to
A)
No synthesis of sugar and ATP
done
clear
B)
It is due to binding of \[{{O}_{2}}\] to RUB is \[Co\]
done
clear
C)
Common occurrence in \[{{C}_{4}}\] plants
done
clear
D)
It is a wasteful process
done
clear
View Answer play_arrow
Which of the following is the major limiting factor for photosynthesis?
A)
Light
done
clear
B)
Carbon dioxide
done
clear
C)
Temperature
done
clear
D)
Water
done
clear
View Answer play_arrow
Select the odd one out with respect to glycolysis
A)
it is the only process of respiration in anaerobic organisms
done
clear
B)
no occurrence of decarboxylation reaction
done
clear
C)
occurs in cytoplasm of cell and is present in all organisms
done
clear
D)
PEP is the end product of glcolysis
done
clear
View Answer play_arrow
The end of aerobic respiration is
A)
\[C{{O}_{2}}\] + water + energy
done
clear
B)
\[C{{o}_{2}}+NAD{{H}_{2}}\] energy
done
clear
C)
3PGA + water + energy
done
clear
D)
Ethyl alcohol + energy
done
clear
View Answer play_arrow
In aerobic respiration, oxygen
A)
Drives the whole process by removing hydrogen from the system and termilly receiving \[{{e}^{-}}\]
done
clear
B)
Acts as the activator of \[FAD{{H}_{2}}\]
done
clear
C)
Acts as initial e' acceptor in ETS
done
clear
D)
Produces two molecules of ATP
done
clear
View Answer play_arrow
In tricarboxylic acid cycle, substrate level phosphorylation takes place during conversion of
A)
Malic acid into oxaloacetic acid
done
clear
B)
Succinyl CoA to succinic acid
done
clear
C)
Citric acid into \[\alpha \] - Ketoglutaric acid
done
clear
D)
OAA into citric acid
done
clear
View Answer play_arrow
The synthetic auxin which is widely used to kill dicotyledonous weeds is
A)
Indole - butyric acid
done
clear
B)
2,4-D
done
clear
C)
Naphthalene acetic acid
done
clear
D)
Indole acetic acid
done
clear
View Answer play_arrow
The plant growth regulator which causes fruit like apple to elongate and also improve its shape is
A)
Ethylcne
done
clear
B)
Abscisic Acid
done
clear
C)
Gibberellins
done
clear
D)
Cytokinin
done
clear
View Answer play_arrow
Select the odd one with respect to vernalisation
A)
it prevents precocious reproductive development
done
clear
B)
enables plants to have sufficient time to reach maturity
done
clear
C)
refers specially to the promotion of flowering by a period of low temperature
done
clear
D)
it is not required by in biennial plants
done
clear
View Answer play_arrow
The site of perception of light/dark in the plants are
A)
Shoot apices
done
clear
B)
Flowering apices
done
clear
C)
Leaves
done
clear
D)
Meristems
done
clear
View Answer play_arrow
Which plants growth substance helps seeds to withstand desiccation and other factors unfavourable for growth
A)
Auxin
done
clear
B)
Gibberellins
done
clear
C)
Abscissic Acid
done
clear
D)
Ethylene
done
clear
View Answer play_arrow

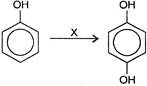 reagent x is
reagent x is