-
question_answer1)
The values of electronegativity of atoms A and B are 1.20 and 4.0 respectively. The percentage of ionic character of A ? B bond is [MP PET 2003]
A)
50 % done
clear
B)
43 % done
clear
C)
55.3 % done
clear
D)
72.24% done
clear
View Solution play_arrow
-
question_answer2)
\[O_{2}^{2-}\]is the symbol of ?.. ion [EAMCET 2003]
A)
Oxide done
clear
B)
Superoxide done
clear
C)
Peroxide done
clear
D)
Monoxide done
clear
View Solution play_arrow
-
question_answer3)
The number of electrons that are paired in oxygen molecule is [IIT 1995]
A)
7 done
clear
B)
8 done
clear
C)
14 done
clear
D)
16 done
clear
View Solution play_arrow
-
question_answer4)
When \[{{N}_{2}}\] goes to \[N_{2}^{+},\] the \[N-N\] bond distance ..... and when \[{{O}_{2}}\] goes to \[O_{2}^{+},\] the \[O-O\] bond distance ....... [IIT 1996]
A)
Decrease, increase done
clear
B)
Increase, decrease done
clear
C)
Increase, increase done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer5)
Which of the following contains a coordinate covalent bond [UPSEAT 2001]
A)
\[{{N}_{2}}H_{5}^{+}\] done
clear
B)
\[BaC{{l}_{2}}\] done
clear
C)
HCl done
clear
D)
\[{{H}_{2}}O\] done
clear
View Solution play_arrow
-
question_answer6)
Which combination is best explained by the co-ordinate covalent bond [JIPMER 2001; CBSE PMT 1990]
A)
\[{{H}^{+}}+{{H}_{2}}O\] done
clear
B)
Cl + Cl done
clear
C)
\[Mg+\frac{1}{2}{{O}_{2}}\] done
clear
D)
\[{{H}_{2}}+{{I}_{2}}\] done
clear
View Solution play_arrow
-
question_answer7)
Arrange the following compounds in order of increasing dipole moment. (I) Toluene (II) \[m-\]dichlorobenzene (III) \[o-\]dichlorobenzene (IV) \[p-\]dichlorobenzene [IIT 1996]
A)
\[I<IV<II<III\] done
clear
B)
\[IV<I<II<III\] done
clear
C)
\[IV<I<III<II\] done
clear
D)
\[IV<II<I<III\] done
clear
View Solution play_arrow
-
question_answer8)
The correct order of dipole moment is [Roorkee 1999]
A)
\[C{{H}_{4}}<N{{F}_{3}}<N{{H}_{3}}<{{H}_{2}}O\] done
clear
B)
\[N{{F}_{3}}<C{{H}_{4}}<N{{H}_{3}}<{{H}_{2}}O\] done
clear
C)
\[N{{H}_{3}}<N{{F}_{3}}<C{{H}_{4}}<{{H}_{2}}O\] done
clear
D)
\[{{H}_{2}}O<N{{H}_{3}}<N{{F}_{3}}<C{{H}_{4}}\] done
clear
View Solution play_arrow
-
question_answer9)
Which of the following has the highest dipole moment [AIIMS 2002]
A)
B)
\[\underset{\,C{{H}_{3}}}{\overset{H\,\,\,\,\,}{\mathop{\underset{|\,\,\,\,\,\,\,}{\overset{|\,\,\,\,\,\,\,}{\mathop{C\,\,\,\,}}}\,}}}\,=\,\,\ \underset{H\,\,\,\,\,}{\mathop{\overset{C{{H}_{3}}}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,\,\,\,\,\,}}\,}}\,\] done
clear
C)
\[\underset{C{{H}_{3}}\,H}{\overset{C{{H}_{3}}\,H}{\mathop{\underset{|\ \ \ \ \ \,\ \ |}{\overset{|\ \ \ \ \ \ \ \ |}{\mathop{C=C}}}\,}}}\,\] done
clear
D)
\[\underset{C{{H}_{3}}\,}{\overset{Cl\,\,\,\,\,}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,\,\,\,\,\,\,}}}\,=\ \ \underset{Cl\,\,\,}{\overset{\,\,C{{H}_{3}}}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,\,\,\,\,\,}}}\,\] done
clear
View Solution play_arrow
-
question_answer10)
Which of the following arrangement of molecules is correct on the basis of their dipole moments [AIIMS 2002]
A)
\[B{{F}_{3}}>N{{F}_{3}}>N{{H}_{3}}\] done
clear
B)
\[N{{F}_{3}}>B{{F}_{3}}>N{{H}_{3}}\] done
clear
C)
\[N{{H}_{3}}>B{{F}_{3}}>N{{F}_{3}}\] done
clear
D)
\[N{{H}_{3}}>N{{F}_{3}}>B{{F}_{3}}\] done
clear
View Solution play_arrow
-
question_answer11)
The type of hybrid orbitals used by the chlorine atom in \[ClO_{2}^{-}\] is [IIT 1992]
A)
\[s{{p}^{3}}\] done
clear
B)
\[s{{p}^{2}}\] done
clear
C)
\[sp\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer12)
Among the following species, identify the isostructural pairs, \[N{{F}_{3}},\,\,NO_{3}^{-},\,\,B{{F}_{3}},\,\,{{H}_{3}}{{O}^{+}},\,\,H{{N}_{3}}\] [IIT 1996]
A)
\[[N{{F}_{3}},\,\,NO_{3}^{-}]\] and \[[B{{F}_{3}},\,\,{{H}_{3}}{{O}^{+}}]\] done
clear
B)
\[[N{{F}_{3}},\,\,H{{N}_{3}}]\] and \[[NO_{3}^{-},\,\,B{{F}_{3}}]\] done
clear
C)
\[[N{{F}_{3}},\,\,{{H}_{3}}{{O}^{+}}]\] and \[[NO_{3}^{-},\,\,B{{F}_{3}}]\] done
clear
D)
\[[N{{F}_{3}},\,\,{{H}_{3}}{{O}^{+}}]\] and \[[H{{N}_{3}},\,\,B{{F}_{3}}]\] done
clear
View Solution play_arrow
-
question_answer13)
In the compound \[C{{H}_{2}}=CH-C{{H}_{2}}-C{{H}_{2}}-C\equiv CH,\] the \[{{C}_{2}}-{{C}_{3}}\] bond is of the type [IIT 1999]
A)
\[sp-s{{p}^{2}}\] done
clear
B)
\[s{{p}^{3}}-s{{p}^{3}}\] done
clear
C)
\[sp-s{{p}^{3}}\] done
clear
D)
\[s{{p}^{2}}-s{{p}^{3}}\] done
clear
View Solution play_arrow
-
question_answer14)
The correct order of increasing \[C-O\] bond length of \[CO,\,\,CO_{3}^{2-},\,\,C{{O}_{2}}\] is [IIT 1999]
A)
\[CO_{3}^{2-}<C{{O}_{2}}<CO\] done
clear
B)
\[C{{O}_{2}}<CO_{3}^{2-}<CO\] done
clear
C)
\[CO<CO_{3}^{2-}<C{{O}_{2}}\] done
clear
D)
\[CO<C{{O}_{2}}<CO_{3}^{2-}\] done
clear
View Solution play_arrow
-
question_answer15)
In the dichromate dianion [IIT 1999]
A)
\[4\,Cr-O\]bonds are equivalent done
clear
B)
\[6\,Cr-O\]bonds are equivalent done
clear
C)
All \[Cr-O\] bonds are equivalent done
clear
D)
All \[Cr-O\] bonds are non-equivalent done
clear
View Solution play_arrow
-
question_answer16)
Bond length of ethane (I), ethene (II), acetylene (III) and benzene (IV) follows the order [CPMT 1999]
A)
\[I>II>III>IV\] done
clear
B)
\[I>II>IV>III\] done
clear
C)
\[I>IV>II>III\] done
clear
D)
\[III>IV>II>I\] done
clear
View Solution play_arrow
-
question_answer17)
Hybridisation state of chlorine in \[Cl{{F}_{3}}\] is [RPET 1999]
A)
\[s{{p}^{3}}\] done
clear
B)
\[s{{p}^{3}}d\] done
clear
C)
\[s{{p}^{3}}{{d}^{2}}\] done
clear
D)
\[s{{p}^{3}}{{d}^{3}}\] done
clear
View Solution play_arrow
-
question_answer18)
Molecular shapes of \[S{{F}_{4}},\,C{{F}_{4}}\] and \[Xe{{F}_{4}}\]are [IIT Screening 2000]
A)
The same with 2, 0 and 1 lone pairs of electrons respectively done
clear
B)
The same, with 1, 1 and 1 lone pairs of electrons respectively done
clear
C)
Different, with 0, 1 and 2 lone pairs of electrons respectively done
clear
D)
Different, with 1, 0 and 2 lone pairs of electrons respectively done
clear
View Solution play_arrow
-
question_answer19)
Structure of \[IF_{4}^{+}\] and hybridization of iodine in this structure are [UPSEAT 2001]
A)
\[s{{p}^{3}}d\], Linear done
clear
B)
\[s{{p}^{3}}{{d}^{2}}\], T-shaped done
clear
C)
\[s{{p}^{3}}d\], Irregular tetrahedral done
clear
D)
\[s{{p}^{3}}{{d}^{2}}\], Octahedral done
clear
View Solution play_arrow
-
question_answer20)
In which of the following the central atom does not use sp3 hybrid orbitals in its bonding [UPSEAT 2001, 02]
A)
\[BeF_{3}^{-}\] done
clear
B)
\[OH_{3}^{+}\] done
clear
C)
\[NH_{2}^{-}\] done
clear
D)
\[N{{F}_{3}}\] done
clear
View Solution play_arrow
-
question_answer21)
The magnetic moment of \[{{K}_{3}}[Fe{{(CN)}_{6}}]\] is found to be 1.7 B.M. How many unpaired electron (s) is/are present per molecule [Orissa JEE 2003]
A)
1 done
clear
B)
2 done
clear
C)
3 done
clear
D)
4 done
clear
View Solution play_arrow
-
question_answer22)
\[{{N}_{2}}\] and \[{{O}_{2}}\] are converted into monocations \[N_{2}^{+}\] and \[O_{2}^{+}\] respectively. Which is wrong [CBSE PMT 1997]
A)
In \[{{N}_{2}},\] the \[N-N\]bond weakens done
clear
B)
In \[{{O}_{2}},\] the \[O-O\] bond order increases done
clear
C)
In \[{{O}_{2}},\] paramagnetism decreases done
clear
D)
\[N_{2}^{+}\] becomes diamagnetic done
clear
View Solution play_arrow
-
question_answer23)
The common features among the species \[C{{N}^{-}}\], CO and \[N{{O}^{+}}\] are [IIT Screening 2001]
A)
Bond order three and isoelectronic done
clear
B)
Bond order three and weak field ligands done
clear
C)
Bond order two and p-acceptors done
clear
D)
Isoelectronic and weak field ligands done
clear
View Solution play_arrow
-
question_answer24)
The number of \[SS\] bonds in sulphur trioxide trimer \[{{S}_{3}}O{}_{9}\] is [IIT Screening 2001]
A)
Three done
clear
B)
Two done
clear
C)
One done
clear
D)
Zero done
clear
View Solution play_arrow
-
question_answer25)
Strongest intermolecular hydrogen bond is present in the following molecules pairs [IIT 1981; DCE 2000]
A)
\[Si{{H}_{4}}\] and \[SiF\] done
clear
B)
\[C{{H}_{3}}-\overset{\overset{O}{\mathop{||}}\,}{\mathop{C}}\,-C{{H}_{3}}\] and \[CHC{{l}_{3}}\] done
clear
C)
\[H-\overset{\overset{O}{\mathop{||}}\,}{\mathop{C}}\,-OH\] and \[C{{H}_{3}}-\overset{\overset{O}{\mathop{||}}\,}{\mathop{C}}\,-OH\] done
clear
D)
\[{{H}_{2}}O\] and \[{{H}_{2}}{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer26)
A compound contains atoms \[X,\,\,Y,\,\,Z.\] The oxidation number of \[X\]is \[sp-s{{p}^{2}}\] is + 5 and \[Z\] is \[-\,2\]. Therefore, a possible formula of the compound is [CPMT 1988]
A)
\[XY{{Z}_{2}}\] done
clear
B)
\[{{X}_{2}}\,{{\left( Y{{Z}_{3}} \right)}_{2}}\] done
clear
C)
\[{{X}_{3}}\,{{\left( Y{{Z}_{4}} \right)}_{2}}\] done
clear
D)
\[{{X}_{3}}\,{{\left( {{Y}_{4}}Z \right)}_{2}}\] done
clear
View Solution play_arrow
-
question_answer27)
Bonds present in \[CuS{{O}_{4}}.5{{H}_{2}}O\]is [IIT 1983; DCE 2001]
A)
Electrovalent and covalent done
clear
B)
Electrovalent and coordinate done
clear
C)
Electrovalent, covalent and coordinate done
clear
D)
Covalent and coordinate done
clear
View Solution play_arrow
-
question_answer28)
The ionization of hydrogen atom would give rise to [UPSEAT 2001]
A)
Hybrid ion done
clear
B)
Hydronium ion done
clear
C)
Proton done
clear
D)
Hydroxyl ion done
clear
View Solution play_arrow
-
question_answer29)
Which can be described as a molecule with residual bonding capacity [JIPMER 2000]
A)
\[BeC{{l}_{2}}\] done
clear
B)
\[NaCl\] done
clear
C)
\[C{{H}_{4}}\] done
clear
D)
\[{{N}_{2}}\] done
clear
View Solution play_arrow
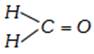 done
clear
done
clear
