-
question_answer1)
Which of the following halides is not correct according to the name and classification?
A)
\[C{{H}_{3}}C{{H}_{2}}C{{\left( C{{H}_{3}} \right)}_{2}}C{{H}_{2}}I\]-1-Iodo-2,2-dimethyl- butane, Primary haloalkane done
clear
B)
\[{{\left( C{{H}_{3}} \right)}_{2}}CHCH\left( Cl \right)C{{H}_{3}}\]-2-Chloro-3-methyl- butane, Secondary haloalkane done
clear
C)
\[C{{H}_{3}}C\left( Cl \right)\left( {{C}_{2}}{{H}_{5}} \right)C{{H}_{2}}C{{H}_{3}}\]- 2-Chloro-2- ethyl butane, Secondary haloalkane done
clear
D)
View Solution play_arrow
-
question_answer2)
Which of the following is halogen exchange reaction?
A)
\[RX+NaI\to RI+NaX\] done
clear
B)
C)
\[R-OH+HX\xrightarrow{ZnC{{l}_{2}}}R-X+{{H}_{2}}O\] done
clear
D)
View Solution play_arrow
-
question_answer3)
| Halogen acids react with alcohols to form alkyl halides. The reaction follows a nucleophilic substitution mechanism. What will be the major product of the following reaction? |
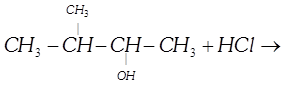 |
A)
B)
C)
D)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}Cl\] done
clear
View Solution play_arrow
-
question_answer4)
The negative part of the addendum (the molecule to be added) adds on to the carbon atom of the double bond containing the least number of hydrogen atoms. This rule is known as
A)
Saytzeff's rule done
clear
B)
Peroxide rule done
clear
C)
Markovnikov's rule done
clear
D)
van't Hoff rule done
clear
View Solution play_arrow
-
question_answer5)
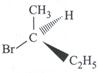
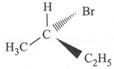
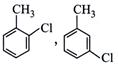
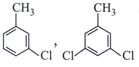
| Which of the following structures is enantiomeric with the molecule (P) given below : |
 |
A)
B)
C)
D)
View Solution play_arrow
-
question_answer6)
| The end product (Q) in the following sequence of reactions is |
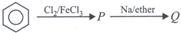 |
A)
B)
C)
D)
View Solution play_arrow
-
question_answer7)
| The reaction |
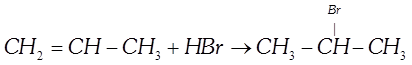 is an example of is an example of |
A)
nucleophilic addition done
clear
B)
free radical addition done
clear
C)
electrophilic addition done
clear
D)
electrophilic substitution. done
clear
View Solution play_arrow
-
question_answer8)
| Classify the following compounds as primary, secondary and tertiary halides. |
| (i) 1-Bromobut-2-ene |
| (ii) 4-Bromopent-2-ene |
| (iii) 2-Bromo-2-methylpropane |
A)
(i)-secondary, (ii)-tertiary, (iii)-primary done
clear
B)
(i)-secondary, (ii)-primary, (iii)-tertiary done
clear
C)
(i)-primary, (ii)-tertiary, (iii)-secondary done
clear
D)
(i)-primary, (ii)-secondary, (iii)-tertiary done
clear
View Solution play_arrow
-
question_answer9)
The order of reactivity of various alkyl halides towards nucleophilic substitution follows the order
A)
\[R-I>R-Br>R-Cl>R-F\] done
clear
B)
\[R-F>R-Cl>R-Br>R-I\] done
clear
C)
\[R-Cl>R-Br>R-I>R-F\] done
clear
D)
\[R-Br>R-I>R-Cl>R-F\] done
clear
View Solution play_arrow
-
question_answer10)
| Arrange the following compounds in the increasing order of their densities. |
(i) 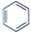 |
(ii) 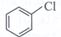 |
(iii) 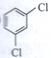 |
(iv) 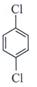 |
A)
i < ii < iii < iv done
clear
B)
i < iii < iv < ii done
clear
C)
iv < iii < ii < i done
clear
D)
ii < iv < iii < i done
clear
View Solution play_arrow
-
question_answer11)
Arrange the following alkyl halides in order of dehydrohalogenation; \[{{C}_{2}}{{H}_{5}}I,\,{{C}_{2}}{{H}_{5}}Cl,\,{{C}_{2}}{{H}_{5}}Br,\,{{C}_{2}}{{H}_{5}}F\]
A)
\[{{C}_{2}}{{H}_{5}}F>{{C}_{2}}{{H}_{5}}Cl>{{C}_{2}}{{H}_{5}}Br>{{C}_{2}}{{H}_{5}}I\] done
clear
B)
\[{{C}_{2}}{{H}_{5}}I>{{C}_{2}}{{H}_{5}}Br>{{C}_{2}}{{H}_{5}}Cl>{{C}_{2}}{{H}_{5}}F\] done
clear
C)
\[{{C}_{2}}{{H}_{5}}I>{{C}_{2}}{{H}_{5}}Cl>{{C}_{2}}{{H}_{5}}Br>{{C}_{2}}{{H}_{5}}F\] done
clear
D)
\[{{C}_{2}}{{H}_{5}}F>{{C}_{2}}{{H}_{5}}I>{{C}_{2}}{{H}_{5}}Br>{{C}_{2}}{{H}_{5}}Cl\] done
clear
View Solution play_arrow
-
question_answer12)
| Haloalkanes contain halogen atom(s) attached to the \[s{{p}^{3}}\] hybridised carbon atom of an alkyl group. Identify haloalkane from the following compounds. |
| (i) 2-Bromopentane |
| (ii) Vinyl chloride (chloroethene) |
| (iii) Chlorobenzene |
| (iv) Trichloromethane |
A)
Only (ii) done
clear
B)
(ii) and (iv) done
clear
C)
(i) and (iv) done
clear
D)
(i), (ii) and (iii) done
clear
View Solution play_arrow
-
question_answer13)
Which of the following haloalkanes react; with aqueous KOH most easily?
A)
1-Bromobutane done
clear
B)
2-Bromobutane done
clear
C)
2-Bromo-2-methylpropane done
clear
D)
2-Chlorobutane done
clear
View Solution play_arrow
-
question_answer14)
Which of the following is a primary halide
A)
iso-Propyl iodide done
clear
B)
sec-Butyl iodide done
clear
C)
ter-Butyl bromide done
clear
D)
neo-Hexyl chloride done
clear
View Solution play_arrow
-
question_answer15)
Which of the following is most reactive towards aqueous NaOH?
A)
\[{{C}_{6}}{{H}_{5}}Cl\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}C{{H}_{2}}Cl\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}Br\] done
clear
D)
\[Br{{C}_{6}}{{H}_{4}}Br\] done
clear
View Solution play_arrow
-
question_answer16)
Which of the following products does no
A)
B)
C)
D)
View Solution play_arrow
-
question_answer17)
| Arrange the following compounds in decreasing order of their boiling points. |
| (i) \[C{{H}_{3}}Br\] |
| (ii) \[C{{H}_{3}}C{{H}_{2}}Br\] |
| (iii) \[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Br\] |
| (iv) \[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}Br\] |
A)
(i) > (ii) > (iii) > (iv) done
clear
B)
(iv) > (iii) > (ii) > (i) done
clear
C)
(i) > (iii) > (ii) > (iv) done
clear
D)
(iii) > (iv) > (i) > (ii) done
clear
View Solution play_arrow
-
question_answer18)
| Arrange the following compounds in order of their reactivity towards \[{{S}_{N}}2\] reaction. |
| (i) \[C{{H}_{3}}{{\left( C{{H}_{2}} \right)}_{3}}C{{H}_{2}}Br\] |
| (ii) \[{{\left( C{{H}_{3}} \right)}_{2}}CHC{{H}_{2}}C{{H}_{2}}Br\] |
(iii) 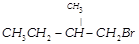 |
(iv) 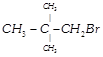 |
A)
(i) > (ii) > (iii) > (iv) done
clear
B)
(ii) > (iii) > (iv) > (i) done
clear
C)
(iii) > (i) > (ii) > (iv) done
clear
D)
(iv) > (ii) > (i) > (iii) done
clear
View Solution play_arrow
-
question_answer19)
| In the following pairs of halogen compounds, which compound undergoes faster \[{{S}_{N}}1\] reaction? |
(i) 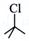 and and 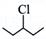 |
(ii) 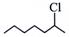 and and  |
A)
B)
C)
D)
View Solution play_arrow
-
question_answer20)
Among the choices of alkyl bromide, the least reactive bromide in \[{{S}_{N}}2\] reaction is
A)
1-bromopentane done
clear
B)
2-bromo-2-methylbutane done
clear
C)
1-bromo-3-methylbutane done
clear
D)
1-bromo-2-methylbutane. done
clear
View Solution play_arrow
-
question_answer21)
| Consider the following reaction : |
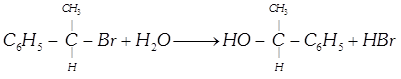 |
| The reaction proceeds with 98% racemisation. |
| The reaction may follow |
A)
\[{{S}_{N}}1\] mechanism done
clear
B)
\[{{S}_{N}}2\] mechanism done
clear
C)
E1 mechanism done
clear
D)
E2 mechanism. done
clear
View Solution play_arrow
-
question_answer22)
| Identify the products [A] and [B] in the reactions. |
| \[RX+AgCN\to (A)+AgX\]; |
| \[RX+KCN\to (B)+KX\] |
A)
\[(A)\to RCN,\,(B)\to RCN\] done
clear
B)
\[(A)\to RCN,\,(B)\to RNC\] done
clear
C)
\[(A)\to RNC,\,(B)\to RCN\] done
clear
D)
\[(A)\to RNC,\,(B)\to RNC\] done
clear
View Solution play_arrow
-
question_answer23)
Butane nitrile can be prepared by heating
A)
propyl alcohol with KCN done
clear
B)
butyl chloride with KCN done
clear
C)
butyl alcohol with KCN done
clear
D)
propyl chloride with KCN. done
clear
View Solution play_arrow
-
question_answer24)
Methyl bromide reacts with AgF to give methyl fluoride and silver bromide. This reaction is called
A)
Fittig reaction done
clear
B)
Swarts reaction done
clear
C)
Wurtz reaction done
clear
D)
Finkelstein reaction. done
clear
View Solution play_arrow
-
question_answer25)
The alkyl halide is converted into an alcohol by
A)
elimination done
clear
B)
dehydrohalogenation done
clear
C)
addition done
clear
D)
substitution. done
clear
View Solution play_arrow
-
question_answer26)
Which of the following compounds can yield only one monochlorinated product upon free radical chlorination?
A)
2, 2-Dimethylpropane done
clear
B)
2-Methylpropane done
clear
C)
2-Methylbutane done
clear
D)
n-Butane done
clear
View Solution play_arrow
-
question_answer27)
Which of the following statements is not correct about \[{{S}_{N}}2\] reactions of alkyl halides?
A)
Nucleophile attacks the carbon from the side opposite to where the leaving group is attached. done
clear
B)
The bond formation and bond breaking take place in one step. done
clear
C)
The rate of reaction depends upon the concentration of nucleophile. done
clear
D)
\[{{S}_{N}}2\] mechanism is predominant is tertiary alkyl halides. done
clear
View Solution play_arrow
-
question_answer28)
| In the reaction given below : |
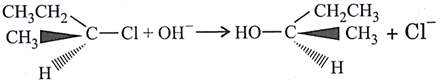 |
| Which of the following statements is correct? |
A)
The reaction proceeds via \[{{S}_{N}}2\] mechanism hence inversion of configuration takes place. done
clear
B)
The reaction proceeds via \[{{S}_{N}}1\] mechanism hence inversion of configuration takes place. done
clear
C)
The reaction proceeds via \[{{S}_{N}}2\] mechanism hence their is no change in the configuration. done
clear
D)
The reaction proceeds via \[{{S}_{N}}1\] mechanism hence there is no change in the configuration. done
clear
View Solution play_arrow
-
question_answer29)
2-Bromo-3, 3-dimethylbutane on reaction with aqueous KOH yields X as the major product. X is
A)
2, 3, 3-trimethylpropan-1-ol done
clear
B)
2, 2-dimethylbutan-3-ol done
clear
C)
2, 3-dimethylbutan-2-ol done
clear
D)
2, 2-dimethylpropan-2-ol. done
clear
View Solution play_arrow
-
question_answer30)
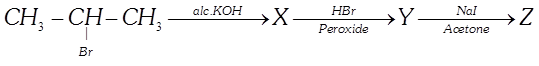 |
| In the given reaction what will be the final product? |
A)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}I\] done
clear
B)
\[C{{H}_{3}}CHIC{{H}_{2}}I\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}C{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}CH{{I}_{2}}\] done
clear
View Solution play_arrow
-
question_answer31)
A mixture of two haloalkanes was treated with sodium metal in ether solution. After the reaction, the product formed was 2-methylpropane. The two haloalkanes present in the mixture were
A)
2-chloropropane and chloromethane done
clear
B)
chloropropane and chloroethane done
clear
C)
2-chloropropane and chloroethane done
clear
D)
chloroethane and chloromethane. done
clear
View Solution play_arrow
-
question_answer32)
Tertiary alkyl halides are practically inert substitution by \[{{S}_{N}}2\] mechanism because
A)
the carbocation formed is unstable done
clear
B)
is steric hindrance done
clear
C)
is inductive effect done
clear
D)
the rate of reaction is faster in \[{{S}_{N}}2\] mechanism. done
clear
View Solution play_arrow
-
question_answer33)
An alkyl halide, RX reacts with KCN to give propane nitrile. RX is
A)
\[{{C}_{3}}{{H}_{7}}Br\] done
clear
B)
\[{{C}_{4}}{{H}_{9}}Br\] done
clear
C)
\[{{C}_{2}}{{H}_{5}}Br\] done
clear
D)
\[{{C}_{5}}{{H}_{11}}Br\] done
clear
View Solution play_arrow
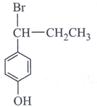
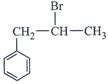
-
question_answer34)
Which of the following will give enantiomeric pair on reaction with water due to presence of asymmetric carbon atom?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer35)
Which of the following is the most reactive towards nucleophilic substitution reaction?
A)
\[ClC{{H}_{2}}-CH=C{{H}_{2}}\] done
clear
B)
\[C{{H}_{2}}=CH-Cl\] done
clear
C)
\[C{{H}_{3}}CH=CH-Cl\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}Cl\] done
clear
View Solution play_arrow
-
question_answer36)
\[{{S}_{N}}1\]reaction is fastest in
A)
\[C{{H}_{3}}C{{H}_{2}}Br\] done
clear
B)
C)
D)
View Solution play_arrow
-
question_answer37)
Ethyl alcohol is obtained when ethyl chloride is boiled with
A)
alcoholic KOH done
clear
B)
aqueous KOH done
clear
C)
water done
clear
D)
aqueous \[KMn{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer38)
The IUPAC name of tertiary butyl chloride is
A)
2-chloro-2-methypropane done
clear
B)
3-chlorobutane done
clear
C)
4-chlorobutane done
clear
D)
1, 2-chloro-3-methylpropane. done
clear
View Solution play_arrow
-
question_answer39)
Which of the following reactions does not take place?
A)
\[{{C}_{2}}{{H}_{5}}Br+KN{{O}_{2}}\to {{C}_{2}}{{H}_{5}}-O-N=O+KBr\] done
clear
B)
C)
\[{{C}_{2}}{{H}_{5}}Br+AgCN\to {{C}_{2}}{{H}_{5}}NC+AgBr\] done
clear
D)
\[{{C}_{2}}{{H}_{5}}Br+KCN\to {{C}_{2}}{{H}_{5}}NC+KBr\] done
clear
View Solution play_arrow
-
question_answer40)
A compound X with molecular formula \[{{C}_{7}}{{H}_{8}}\] is treated with \[C{{l}_{2}}\] in presence of \[FeC{{l}_{3}}\]. Which of the following compounds are formed during the reaction?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer41)
| Among the isomers of \[{{C}_{5}}{{H}_{11}}Cl\], the one which is chiral is |
(i) 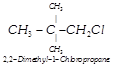 |
(ii) 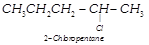 |
(iii) 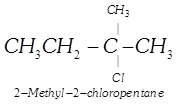 |
(iv) 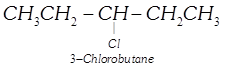 |
A)
(i) and (ii) done
clear
B)
(i), (ii) and (iii) done
clear
C)
(i) and (iii) done
clear
D)
only (ii) done
clear
View Solution play_arrow
-
question_answer42)
| Which of the following reactions will give the major and minor products? |
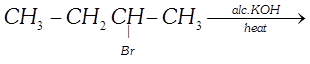 |
| \[C{{H}_{3}}-CH\underset{(A)}{\mathop{=}}\,CH-C{{H}_{3}}+C{{H}_{3}}-C{{H}_{2}}\underset{(B)}{\mathop{-}}\,CH=C{{H}_{2}}\] |
A)
[A] is major product and [B] is minor product. done
clear
B)
[A] is minor product and [B] is major product. done
clear
C)
Both [A] and [B] are major products. done
clear
D)
Only [B] is formed and [A] is not formed. done
clear
View Solution play_arrow
-
question_answer43)
Match the reactions given in column I with the type of reaction mentioned in column II and mark the appropriate choice.
| | Column I | | Column II |
| [A] | 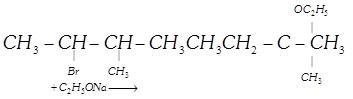 | (i) | \[\beta \]-elimination |
| [B] | \[C{{H}_{3}}C{{H}_{2}}Br\xrightarrow{AgOH}\]\[C{{H}_{3}}C{{H}_{2}}OH\] | (ii) | \[{{S}_{N}}1\]nucleophilic substitution |
| [C] | \[C{{H}_{3}}CH=C{{H}_{2}}+HBr\] \[\xrightarrow{Peroxide}\] \[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Br\] | (iii) | \[{{S}_{N}}2\]nucleophilic substitution |
| [D] | \[C{{H}_{3}}-C{{H}_{2}}Br+\] \[alc.KOH\to \] \[C{{H}_{2}}=C{{H}_{2}}\] | (iv) | Kharasch effect |
A)
[A] \[\to \] (iv), [B]\[\to \] (i), [C] \[\to \](ii), [D] \[\to \](iii) done
clear
B)
[A] \[\to \] (ii), [B] \[\to \] (iii), [C] \[\to \] (iv), [D] \[\to \] (i) done
clear
C)
[A] \[\to \] (i), [B] \[\to \] (ii), [C] \[\to \] (iv), [D] \[\to \] (iii) done
clear
D)
[A] \[\to \] (iii), [B] \[\to \] (i), [C] \[\to \] (ii), [D] \[\to \] (iv) done
clear
View Solution play_arrow
-
question_answer44)
| An alkyl halide with molecular formula \[{{C}_{6}}{{H}_{13}}Br\] on dehydrohalogenation gives two isomeric alkenes X and Y with molecular formula \[{{C}_{6}}{{H}_{12}}\]. On reductive ozonolysis X and Y gave four compounds \[C{{H}_{3}}COC{{H}_{3}}\], \[C{{H}_{3}}CHO\], \[C{{H}_{3}}C{{H}_{2}}CHO\] and \[{{\left( C{{H}_{3}} \right)}_{2}}CHCHO\]. The alkyl halide is |
A)
4-bromo-2-methylpentane done
clear
B)
3-bromo-2-methylpentane done
clear
C)
2-bromo-2, 3-dimethylbutane done
clear
D)
2, 2-dimethyl-1-bromobutane. done
clear
View Solution play_arrow
-
question_answer45)
Which of the following is an example of vic-dihalide?
A)
Dichloromethane done
clear
B)
1, 2-Dichloroethane done
clear
C)
Ethylidene chloride done
clear
D)
Allyl chloride done
clear
View Solution play_arrow
-
question_answer46)
In \[{{S}_{N}}2\] reactions the sequence of bond breaking and bond formation is as follows
A)
bond breaking is followed by formation done
clear
B)
bond formation is followed by breaking done
clear
C)
bond breaking and formation are simultaneously done
clear
D)
bond breaking and formation take place randomly. done
clear
View Solution play_arrow
-
question_answer47)
 |
| X in the reaction is |
A)
B)
C)
D)
View Solution play_arrow
-
question_answer48)
Which of the following alkyl halides undergoes hydrolysis with aqueous KOH at the fastest rate?
A)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Cl\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}Cl\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}Cl\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}CH(Br)C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer49)
A mixture of 1-chloropropane and 2-chloropropane when treated with alcoholic KOH gives
A)
prop-1-ene done
clear
B)
prop-2-ene done
clear
C)
a mixture of prop-1-ene and prop-2-ene done
clear
D)
propanol. done
clear
View Solution play_arrow
-
question_answer50)
Consider the following reaction and identify X and Y. \[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}I\xrightarrow{alc.\,KOH}X\xrightarrow{B{{r}_{2}}}Y\]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer51)
2-Chloro-2-methylpropane on reaction with alc. KOH gives X as the major product X is
A)
but-2-ene done
clear
B)
2-methylbut-1-ene done
clear
C)
2-methylbutane-1-ene done
clear
D)
2-methylbutan-2-ol. done
clear
View Solution play_arrow
-
question_answer52)
Grignard reagent, a very useful starting compound for a number of organic reactions can be prepared by
A)
reaction of alkyl halides with a solution of magnesium hydroxide done
clear
B)
reaction of alkyl halides with dry magnesium powder in presence of dry ether done
clear
C)
reaction of \[MgC{{l}_{2}}\] with ether and alcohol done
clear
D)
reaction of alkyl halide with magnesium in presence of alcohol. done
clear
View Solution play_arrow
-
question_answer53)
Which of the following reactions follows Martovnikov's rule?
A)
\[{{C}_{2}}{{H}_{4}}+HBr\] done
clear
B)
\[{{C}_{3}}{{H}_{6}}+C{{l}_{2}}\] done
clear
C)
\[{{C}_{3}}{{H}_{6}}+HBr\] done
clear
D)
\[{{C}_{3}}{{H}_{6}}+B{{r}_{2}}\] done
clear
View Solution play_arrow
-
question_answer54)
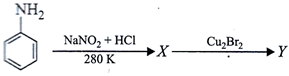 |
| X and Y in the reaction are |
A)
B)
C)
D)
View Solution play_arrow
-
question_answer55)
| Identify the products [A] and [B]. |
(i) 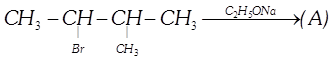 |
(ii) 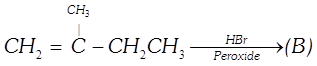 |
A)
B)
C)
D)
View Solution play_arrow
-
question_answer56)
Aryl halides are less reactive towards nucleophilic substitution reactions as compared to alkyl halides due to
A)
formation of a less stable carbonium ion in aryl halides done
clear
B)
resonance stabilisation in aryl halides done
clear
C)
presence of double bonds in alkyl halides done
clear
D)
inductive effect in aryl halides. done
clear
View Solution play_arrow
-
question_answer57)
Which of the following reactions is not correctly matched?
A)
\[2{{C}_{2}}{{H}_{5}}Br+2Na\xrightarrow{dry\,ether}{{C}_{4}}{{H}_{10}}+2NaBr\]: Wurtz reaction done
clear
B)
\[C{{H}_{3}}Br+AgF\to C{{H}_{3}}F+AgBr\]: Etards reaction done
clear
C)
\[{{C}_{6}}{{H}_{5}}Br+2Na+Br{{C}_{2}}{{H}_{5}}\xrightarrow{dry\,ether}{{C}_{6}}{{H}_{5}}{{C}_{2}}{{H}_{5}}+2NaBr\]: Wurtz-Fitting reaction done
clear
D)
\[2{{C}_{6}}{{H}_{5}}Br+2Na\xrightarrow{dry\,ether}{{C}_{6}}{{H}_{5}}-{{C}_{6}}{{H}_{5}}+2NaBr\]: Fitting reaction done
clear
View Solution play_arrow
-
question_answer58)
The main difference in C - X bond of a haloalkane and a haloarene is
A)
C - X bond in haloalkanes is shorter than haloarenes done
clear
B)
in haloalkanes the C attached to halogen in C - X bond is \[s{{p}^{3}}\] hybridised while in haloarenes it is \[s{{p}^{2}}\] hybridized done
clear
C)
C-X bond in haloarenes acquires a double bond character due to higher electronegativity of X than haloalkanes done
clear
D)
haloalkanes are less reactive than haloarenes due to difficulty in C-X cleavage in haloalkanes. done
clear
View Solution play_arrow
-
question_answer59)
Match the column I with column II and mark the appropriate choice.
| | Column - I | | Column - II |
| [a] | \[C{{H}_{3}}CHC{{l}_{2}}\] | (i) | Vinyl halide |
| [b] | \[C{{H}_{2}}ClC{{H}_{2}}Cl\] | (ii) | Alkylidene halide |
| [c] | \[CHCl=C{{H}_{2}}\] | (iii) | Alkylene dihalide |
| [d] | \[ClC{{H}_{2}}-CH=C{{H}_{2}}\] | (iv) | Allyl halide |
A)
[A] \[\to \] (i), [B] \[\to \] (ii), [C] \[\to \] (iv), [D] \[\to \] (iii) done
clear
B)
[A] \[\to \] (ii), [B] \[\to \] (iii), [C] \[\to \] (i), [D] \[\to \] (iv) done
clear
C)
[A] \[\to \] (iii), [B] \[\to \] (iv), [C] \[\to \] (ii), [D] \[\to \] (i) done
clear
D)
[A]\[\to \] (iv), [B] \[\to \] (i), [C] \[\to \] (iii), [D] \[\to \] (ii) done
clear
View Solution play_arrow
-
question_answer60)
Which is the correct increasing order of boiling points of the following compounds? 1-Iodobutane, 1-Bromobutane, 1-Chlorobutane, Butane
A)
Butane < 1-Chlorobutane < 1-Bromobutane < 1-Iodobutane done
clear
B)
1-Iodobutane < 1-Bromobutane < 1-Chlorobutane < Butane done
clear
C)
Butane < 1-Iodobutane < 1-Bromobutane < 1-Chlorobutane done
clear
D)
Butane < 1-Chlorobutane < 1-Iodobutane < 1-Bromobutane done
clear
View Solution play_arrow
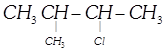 done
clear
done
clear
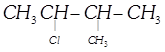 done
clear
done
clear
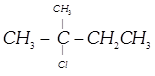 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
![]()
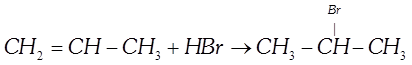 is an example of
is an example of 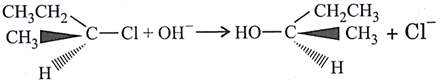
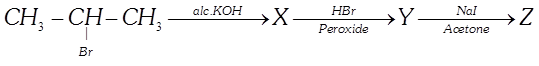
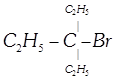 done
clear
done
clear
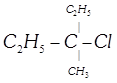 done
clear
done
clear
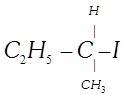 done
clear
done
clear
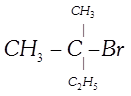 done
clear
done
clear
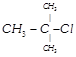 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear

 done
clear
done
clear
 done
clear
done
clear
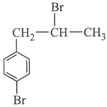 done
clear
done
clear
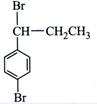 done
clear
done
clear
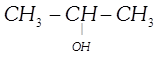
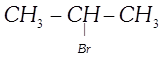

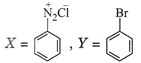 done
clear
done
clear
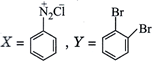 done
clear
done
clear
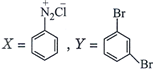 done
clear
done
clear
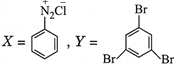 done
clear
done
clear
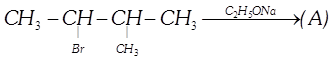
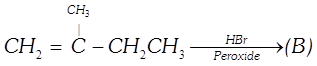
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
