A) C - X bond in haloalkanes is shorter than haloarenes
B) in haloalkanes the C attached to halogen in C - X bond is \[s{{p}^{3}}\] hybridised while in haloarenes it is \[s{{p}^{2}}\] hybridized
C) C-X bond in haloarenes acquires a double bond character due to higher electronegativity of X than haloalkanes
D) haloalkanes are less reactive than haloarenes due to difficulty in C-X cleavage in haloalkanes.
Correct Answer: B
Solution :
In haloarenes, carbon of C-X is \[s{{p}^{2}}\]hybridised while in haloalkanes it is \[s{{p}^{3}}\]hybridised.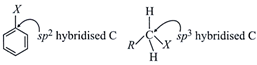
You need to login to perform this action.
You will be redirected in
3 sec
