When a metal surface is illuminated by light of wavelengths 400 nm and 250 nm, the maximum velocities of the photoelectrons ejected are \[\upsilon \] and \[2\upsilon \] respectively. The work function of the metal is (h = Planck's constant, c = velocity of light in air)
A)
\[2\,\,hc\times {{10}^{6}}J\]
done
clear
B)
\[1.5\,\,hc\times {{10}^{6}}J\]
done
clear
C)
\[hc\times {{10}^{6}}J\]
done
clear
D)
\[0.5\,\,hc\times {{10}^{6}}J\]
done
clear
View Answer play_arrow
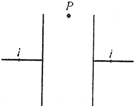
An electron (mass \[=9\times {{10}^{-31}}kg,\] charge\[=1.6\times {{10}^{-19}}C\]) moving with a velocity of \[{{10}^{6}}m/s\]enters a magnetic field. If it describes a circle of radius 0.1m, then strength of magnetic field must be
A)
\[4.5\times {{10}^{-5}}T\]
done
clear
B)
\[1.4\times {{10}^{-5}}T\]
done
clear
C)
\[5.5\times {{10}^{-5}}T\]
done
clear
D)
\[2.6\times {{10}^{-5}}T\]
done
clear
View Answer play_arrow
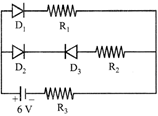
Figure shows a circuit in which three identical diodes are used. Each diode has forward resistance of \[20\,\,\Omega \] and infinite backward resistance. Resistors\[={{R}_{1}}={{R}_{2}}=50\Omega .\]Battery voltage is 6 V. The current through \[{{R}_{3}}\] is :
A)
50 mA
done
clear
B)
100 mA
done
clear
C)
60 mA
done
clear
D)
25 mA
done
clear
View Answer play_arrow
When an electron jumps from the fourth orbit to the second orbit, one gets the
A)
second line of Lyman series
done
clear
B)
second line of Paschen series
done
clear
C)
second line of Balmer series
done
clear
D)
first line of P fund series
done
clear
View Answer play_arrow
In a two dimensional motion, instantaneous speed \[{{v}_{0}}\] is a positive constant. Then which of the following is necessarily true?
A)
The average velocity is not zero at any time
done
clear
B)
Average acceleration must always vanish
done
clear
C)
Displacements in equal time intervals are equal
done
clear
D)
Equal plane lengths are traversed in equal Intervals
done
clear
View Answer play_arrow
A ball of mass 150 g, moving with an acceleration \[20\,m/{{s}^{2}},\]is hit by a force, which acts on it for 0.1 sec. The impulsive force is
A)
0.5 N
done
clear
B)
0.1 N
done
clear
C)
0.3 N
done
clear
D)
1.2 N
done
clear
View Answer play_arrow
A man is watching two trains, one leaving and the other coming with equal speed of 4 m/s. If they sound their whistles each of frequency 240 Hz, the number of beats heard by man (velocity of sound in air = 320 m/s) will be equal to
A)
12
done
clear
B)
0
done
clear
C)
3
done
clear
D)
6
done
clear
View Answer play_arrow
Two identical galvanometers are taken, one is to be converted into an ammeter and other into a milliammeter. Shunt of milliammeter compared to ammeter is
A)
less
done
clear
B)
more
done
clear
C)
zero
done
clear
D)
None of these
done
clear
View Answer play_arrow
In a compound microscope, the intermediate image is
A)
virtual, erect and magnified
done
clear
B)
real, erect and magnified
done
clear
C)
real, inverted and magnified
done
clear
D)
virtual, erect and reduced
done
clear
View Answer play_arrow
Let \[{{E}_{a}}\] be the electric field due to a dipole in its axial plane distant \[\ell \] and \[{{E}_{q}}\] be the field in the equatorial plane distant \[\ell ',\] then the relation between \[{{E}_{a}}\] and \[{{E}_{q}}\] will be
A)
\[{{E}_{a}}=4{{E}_{q}}\]
done
clear
B)
\[{{E}_{q}}=2{{E}_{a}}\]
done
clear
C)
\[{{E}_{a}}=2{{E}_{q}}\]
done
clear
D)
\[{{E}_{q}}=3{{E}_{a}}\]
done
clear
View Answer play_arrow
A combination of capacitors is set up as shown in the figure. The magnitude of the electric field, due to a point charge Q (having a charge equal to the sum of the charges on the 4 \[\mu \] F and 9 \[\mu \] F capacitors), at a point distance 30m from it, would equal:
A)
420 N/C
done
clear
B)
480 N/C
done
clear
C)
240 N/C
done
clear
D)
360 N/C
done
clear
View Answer play_arrow
The moment of inertia of a disc of mass m and radius r about an axis, which is tangential to the circumference of the disc and parallel to its diameter is
A)
\[\frac{3}{2}m\,{{r}^{2}}\]
done
clear
B)
\[\frac{2}{3}m\,{{r}^{2}}\]
done
clear
C)
\[\frac{5}{4}m\,{{r}^{2}}\]
done
clear
D)
\[\frac{4}{5}m\,{{r}^{2}}\]
done
clear
View Answer play_arrow
A sphere of radius R has uniform volume charge density. The electric potential at a point \[(r<R)\] is
A)
due to the charge inside a sphere of radius r only
done
clear
B)
due to the entire charge of the sphere
done
clear
C)
due to the charge in the spherical shell of inner and outer radii r and R, only
done
clear
D)
independent of r
done
clear
View Answer play_arrow
A convex lens of focal length 40 cm is held co- axially 12 cm above a mirror of focal length 18 cm. An object held x cm above the lens gives rise to an image coincident with it. Then x is equal to:
A)
12 cm
done
clear
B)
15 cm
done
clear
C)
18 cm
done
clear
D)
30 cm
done
clear
View Answer play_arrow
A body of mass 3 kg is under a constant force which causes a displacement s in meter in it, given by the relation \[s=\frac{1}{3}{{t}^{2}},\] where t is in second. Work done by the force in 2 second is
A)
\[\frac{3}{8}J\]
done
clear
B)
\[\frac{8}{3}J\]
done
clear
C)
\[\frac{19}{5}J\]
done
clear
D)
\[\frac{5}{19}J\]
done
clear
View Answer play_arrow
Under a constant torque the angular momentum of a body changes from A to 4A in 4 second. The torque on the body will be
A)
\[1\,A\]
done
clear
B)
\[\frac{1}{4}A\]
done
clear
C)
\[\frac{4}{3}A\]
done
clear
D)
\[\frac{3}{4}A\]
done
clear
View Answer play_arrow
An electric bulb marked 40 W and 200V, is used in a circuit of supply voltage 100V. Now its power is
A)
10 W
done
clear
B)
20 W
done
clear
C)
40 W
done
clear
D)
100 W
done
clear
View Answer play_arrow
The height at which the acceleration due to gravity becomes \[\frac{g}{9}\] (where g = the acceleration due to gravity on the surface of the earth) in terms of R, the radius of the earth, is
A)
\[\frac{R}{\sqrt{2}}\]
done
clear
B)
\[R/2\]
done
clear
C)
\[\sqrt{2}R\]
done
clear
D)
\[2\,R\]
done
clear
View Answer play_arrow
Two coherent monochromatic light beams of intensities I and 4 I are superimposed. The maximum and minimum possible intensities in the resulting beam are
A)
5 I and I
done
clear
B)
5 I and 3 I
done
clear
C)
9 I and I
done
clear
D)
9 I and 3 I
done
clear
View Answer play_arrow
When a wave travels in a medium the particles displacement is given by the equation y = 0.03 \[\sin \,\pi (2t-0.01x),\] where x and y are in seconds. The wavelength of the wave is
A)
200 m
done
clear
B)
100 m
done
clear
C)
20 m
done
clear
D)
10 m
done
clear
View Answer play_arrow
Figure shows a parallel plate capacitor and the current in the connecting wires that is discharging the capacitor.
A)
The displacement current is leftward.
done
clear
B)
The displacement current is rightward
done
clear
C)
The electric field \[\overrightarrow{E}\] is rightward
done
clear
D)
The magnetic field at point P is out the page.
done
clear
View Answer play_arrow
The specific heat capacity of a metal at low temperature (T) is given as\[{{C}_{p}}(kJ{{K}^{-1}}k{{g}^{-1}})=32{{\left( \frac{T}{400} \right)}^{3}}.\] A 100 g vessel of this metal is to be cooled from 20 K to 4 K by a special refrigerator operating at room temperature\[\left( 27{}^\circ C \right).\] The amount of work required to cool in vessel is
A)
equal to 0.002 kJ
done
clear
B)
greater than 0.148 kJ
done
clear
C)
between 0.148 KJ and 0.028 kJ
done
clear
D)
less than 0.028 KJ
done
clear
View Answer play_arrow
Solar radiation emitted by sun resembles that emitted by a black body at a temperature of 6000 K. Maximum intensity is emitted at a wavelength of about 4800 \[\overset{{}^\circ }{\mathop{A.}}\,\] If the sun were to cool down from 6000 K to 3000K, then the peak intensity would occur at a wavelength
A)
\[4800\overset{{}^\circ }{\mathop{A}}\,\]
done
clear
B)
\[9600\overset{{}^\circ }{\mathop{A}}\,\]
done
clear
C)
\[7200\overset{{}^\circ }{\mathop{A}}\,\]
done
clear
D)
\[6400\overset{{}^\circ }{\mathop{A}}\,\]
done
clear
View Answer play_arrow
Two particles are oscillating along two close parallel straight lines side by side, with the same frequency and amplitudes. They pass each other, moving in opposite directions when their displacement is half of the amplitude. The mean positions of the two particles lie on a straight line perpendicular to the paths of the two particles. The phase difference is
A)
\[0\]
done
clear
B)
\[2\pi /3\]
done
clear
C)
\[\pi \]
done
clear
D)
\[\pi /6\]
done
clear
View Answer play_arrow
A particle of mass 10 g is kept on the surface of a uniform sphere of mass 100 kg and radius 10 cm. Find the work to be done against the gravitational force between them to take the particle far away from the sphere (Take \[G=6.67\times {{10}^{-11}}N{{m}^{2}}/k{{g}^{2}}\])
A)
\[3.33\times {{10}^{-10}}J\]
done
clear
B)
\[13.34\times {{10}^{-10}}J\]
done
clear
C)
\[6.67\times {{10}^{-10}}J\]
done
clear
D)
\[6.67\times {{10}^{-9}}J\]
done
clear
View Answer play_arrow
What is the acceleration of a projectile at its highest point
A)
maximum
done
clear
B)
minimum
done
clear
C)
zero
done
clear
D)
g
done
clear
View Answer play_arrow
A particle is moving in a circle of radius r under the action of a force \[F=\alpha {{r}^{2}}\] which is directed towards centre of the circle. Total mechanical energy (kinetic energy + potential energy) of the particle is (take potential energy = 0 for r = 0):
A)
\[\frac{1}{2}\alpha {{r}^{3}}\]
done
clear
B)
\[\frac{5}{6}\alpha {{r}^{3}}\]
done
clear
C)
\[\frac{4}{3}\alpha {{r}^{3}}\]
done
clear
D)
\[\alpha {{r}^{3}}\]
done
clear
View Answer play_arrow
A block A of mass 7 kg is placed on a frictionless table. A thread tied to it passes over a frictionless pulley and carries a body B of mass 3 kg at the other end. The acceleration of the system is (given\[g=10\,\,m{{s}^{-2}}\])
A)
\[100\,m{{s}^{-2}}\]
done
clear
B)
\[3\,m{{s}^{-2}}\]
done
clear
C)
\[10\,m{{s}^{-2}}\]
done
clear
D)
\[30\,m{{s}^{-2}}\]
done
clear
View Answer play_arrow
The kinetic theory of gases
A)
explains the behaviour of an ideal gas
done
clear
B)
describes the motion of a single atom or molecule
done
clear
C)
relates the temperature of the gas with K.E. of atoms of the gas
done
clear
D)
all of the above
done
clear
View Answer play_arrow
A uniform magnetic field acts at right angles to the direction of motion of electron. As a result, the electron moves in a circular path of radius 2 cm. If the speed of electron is doubled, then the radius of the circular path will be
A)
2.0 on
done
clear
B)
0.5cm
done
clear
C)
4.0 cm
done
clear
D)
1.0cm
done
clear
View Answer play_arrow
A photoelectric cell is illuminated by a point source of light 1 m away. When the source is shifted to 2 m, then
A)
number of electrons emitted is a quarter of the initial number
done
clear
B)
each emitted electron carries one quarter of the initial energy
done
clear
C)
number of electrons emitted is half the initial number
done
clear
D)
each emitted electron carries half the initial energy
done
clear
View Answer play_arrow
An engine has an efficiency of 1/6. When the temperature of sink is reduced by \[62{}^\circ C,\] its efficiency is doubled. Temperature of the source is
A)
\[37{}^\circ C\]
done
clear
B)
\[62{}^\circ C\]
done
clear
C)
\[99{}^\circ C\]
done
clear
D)
\[124{}^\circ C\]
done
clear
View Answer play_arrow
Two point dipoles of dipole moment \[{{\overrightarrow{p}}_{1}}\] and \[{{\overrightarrow{p}}_{2}}\] are at a distances from each other and \[{{\overrightarrow{p}}_{1}}||{{\overrightarrow{p}}_{2}}.\] The force between the dipoles is:
A)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\frac{4{{p}_{1}}{{p}_{2}}}{{{x}^{4}}}\]
done
clear
B)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\frac{3{{p}_{1}}{{p}_{2}}}{{{x}^{3}}}\]
done
clear
C)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\frac{6{{p}_{1}}{{p}_{2}}}{{{x}^{4}}}\]
done
clear
D)
\[\frac{1}{4\pi {{\varepsilon }_{0}}}\frac{8{{p}_{1}}{{p}_{2}}}{{{x}^{4}}}\]
done
clear
View Answer play_arrow
The Bulk modulus for an incompressible liquid is
A)
zero
done
clear
B)
unity
done
clear
C)
infinity
done
clear
D)
between 0 and 1
done
clear
View Answer play_arrow
If unit of length and force are increased 4 times. The unit of energy
A)
is increased by 4 times
done
clear
B)
is increased by 16 times
done
clear
C)
is increased by 8 times
done
clear
D)
remains unchanged
done
clear
View Answer play_arrow
A particle starting from rest falls from a certain height. Assuming that the acceleration due to gravity remain the same throughout the motion, its displacements in three successive half second intervals are \[{{S}_{1}},\]\[{{S}_{2}}\] and \[{{S}_{3}}\] then
A)
\[{{S}_{1}}:{{S}_{2}}:{{S}_{3}}=1:5:9\]
done
clear
B)
\[{{S}_{1}}:{{S}_{2}}:{{S}_{3}}=1:3:5\]
done
clear
C)
\[{{S}_{1}}:{{S}_{2}}:{{S}_{3}}=9:2:3\]
done
clear
D)
\[{{S}_{1}}:{{S}_{2}}:{{S}_{3}}=1:1:1\]
done
clear
View Answer play_arrow
A mass of 2kg is whirled in a horizontal circle by means of a string at initial speed of 5 revolution per minute. Keeping the radius constant, the tension in the string is doubled. The new speed is nearly
A)
2.25 rpm
done
clear
B)
7 rpm
done
clear
C)
10 rpm
done
clear
D)
14 rpm
done
clear
View Answer play_arrow
A transformer is employed to
A)
convert A.C. into D.C.
done
clear
B)
convert D.C. into A.C.
done
clear
C)
obtain a suitable A.C. voltage
done
clear
D)
obtain a suitable D.C. voltage
done
clear
View Answer play_arrow
A car runs at a constant speed on a circular track of radius 100 m, taking 62.8 seconds in every circular loop. The average velocity and average speed for each circular loop respectively, is
A)
0, 10 m/s
done
clear
B)
10 m/s, 10 m/s
done
clear
C)
10 m/s, 0
done
clear
D)
0, 0
done
clear
View Answer play_arrow
In the circuit shown, the internal resistance of the cell is negligible. The steady state current in the \[2\Omega \] resistor is
A)
0.6 A
done
clear
B)
0.9 A
done
clear
C)
1.2 A
done
clear
D)
1.5 A
done
clear
View Answer play_arrow
A coin is placed on a horizontal platform which undergoes vertical simple harmonic motion of angular frequency\[\omega .\] The amplitude of oscillation is gradually increased. The coin will leave contact with the platform for the first time
A)
at the mean position of the platform
done
clear
B)
for an amplitude of \[\frac{g}{{{\omega }^{2}}}\]
done
clear
C)
for an amplitude of \[\frac{{{g}^{2}}}{{{\omega }^{2}}}\]
done
clear
D)
at the highest position of the platform
done
clear
View Answer play_arrow
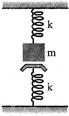
A mass m is suspended from a spring of force constant k and just touches another identical spring fixed to the floor as shown in the figure. The time period of small oscillations is
A)
\[2\pi \sqrt{\frac{m}{k}}\]
done
clear
B)
\[\pi \sqrt{\frac{m}{k}}+\pi \sqrt{\frac{m}{k/2}}\]
done
clear
C)
\[\pi \sqrt{\frac{m}{3k/2}}\]
done
clear
D)
\[\pi \sqrt{\frac{m}{k}}+\pi \sqrt{\frac{m}{2k}}\]
done
clear
View Answer play_arrow
Two wires of same material and length but cross- sections in the ratio 1: 2 are used to suspend the same loads. The extensions in them will be in the ratio of
A)
1 : 2
done
clear
B)
2 : 1
done
clear
C)
4 : 1
done
clear
D)
1 : 4
done
clear
View Answer play_arrow
The meniscus of a liquid contained in one of the limbs of a narrow U-tube is held in an electromagnet with the meniscus in line with the field. The liquid is seen to rise. This indicates that the liquid is
A)
ferromagnetic
done
clear
B)
paramagnetic
done
clear
C)
diamagnetic
done
clear
D)
non-magnetic
done
clear
View Answer play_arrow
The coefficient of self-inductance of a solenoid is 0.18 mH. If a core of soft iron of relative permeability 900 is inserted, then the coefficient of self-inductance will become nearly.
A)
5.4 mH
done
clear
B)
162 mH
done
clear
C)
0.006 mH
done
clear
D)
0.0002 mH
done
clear
View Answer play_arrow
The values of Planck's constant is \[6.63\times {{10}^{-34}}Js.\] The velocity of light is \[3.0\times {{10}^{8}}m\,{{s}^{-1}}.\] Which value is closest to the wavelength in nanometers of a quantum of light with frequency of \[8\times {{10}^{15}}{{s}^{-1}}?\]
A)
\[5\times {{10}^{-18}}\]
done
clear
B)
\[4\times {{10}^{1}}\]
done
clear
C)
\[3\times {{10}^{7}}\]
done
clear
D)
\[2\times {{10}^{-25}}\]
done
clear
View Answer play_arrow
Higher order (>3) reactions are rare due to:
A)
Shifting of equilibrium towards reactants due to elastic collisions.
done
clear
B)
Loss of active species on collision.
done
clear
C)
Low probability of simultaneous collision of all the reacting species.
done
clear
D)
Increase in entropy and activation energy as more molecules are involved.
done
clear
View Answer play_arrow
Which of the following order is wrong?
A)
\[N{{H}_{3}}<P{{H}_{3}}<As{{H}_{3}}\text{Acidic}\]
done
clear
B)
\[Li<Be<B<CI{{E}_{1}}\]
done
clear
C)
\[A{{l}_{2}}{{O}_{3}}<MgO<N{{a}_{2}}O<{{K}_{2}}O-\text{Basic}\]
done
clear
D)
\[L{{i}^{+}}<N{{a}^{+}}<{{K}^{+}}<C{{s}^{+}}-\text{Ionic}\,\,\text{radius}\]
done
clear
View Answer play_arrow
A compound of a metal ion \[{{M}^{x+}}(Z=24)\] has a spin only magnetic moment of \[\sqrt{15}\] Bohr Magnetons. The number of unpaired electrons in the compound are
A)
2
done
clear
B)
4
done
clear
C)
5
done
clear
D)
3
done
clear
View Answer play_arrow
In the following reaction sequence :
The compound I is:
A)
\[\underset{\underset{Cl}{\mathop{|}}\,}{\mathop{C{{H}_{2}}}}\,-\underset{\underset{Cl}{\mathop{|}}\,}{\mathop{CH}}\,-C{{H}_{3}}\]
done
clear
B)
\[\underset{\underset{Cl}{\mathop{|}}\,}{\mathop{C{{H}_{2}}}}\,-\underset{\underset{Cl}{\mathop{|}}\,}{\mathop{C{{H}_{2}}}}\,-C{{H}_{3}}\]
done
clear
C)
\[CH-\underset{\underset{Cl}{\mathop{|}}\,}{\mathop{CH}}\,-C{{H}_{2}}-C{{H}_{3}}\]
done
clear
D)
done
clear
View Answer play_arrow
In which of the following sets, all the given species are isostructural?
A)
\[C{{O}_{2}},\]\[N{{O}_{2}},\]\[Cl{{O}_{2}},\]\[Si{{O}_{2}}\]
done
clear
B)
\[PC{{l}_{3}},\]\[Al\,C{{l}_{3}},\]\[BC{{l}_{3}},\]\[SbC{{l}_{3}}\]
done
clear
C)
\[B{{F}_{3}},\]\[N{{F}_{3}},\]\[P{{F}_{3}},\]\[Al{{F}_{3}}\]
done
clear
D)
\[BF_{4}^{-},\]\[CC{{l}_{4}},\]\[NH_{4}^{+},\]\[PCl_{4}^{+}\]
done
clear
View Answer play_arrow
In the cyclic process shown in \[P-V\]diagram, the magnitude of the work done is :
A)
\[\pi {{\left( \frac{{{P}_{2}}-{{P}_{1}}}{2} \right)}^{2}}\]
done
clear
B)
\[\pi {{\left( \frac{{{V}_{2}}-{{V}_{1}}}{2} \right)}^{2}}\]
done
clear
C)
\[\frac{\pi }{4}({{P}_{2}}-{{P}_{1}})({{V}_{2}}-{{V}_{1}})\]
done
clear
D)
\[\pi ({{V}_{2}}-{{V}_{1}}^{2})\]
done
clear
View Answer play_arrow
On the basis of the information available from the reaction \[\frac{4}{3}Al+{{O}_{2}}\xrightarrow{{}}\frac{2}{3}A{{l}_{2}}{{O}_{3}},\]\[\Delta G=-827\,kJ\,mo{{l}^{-1}}\] of \[{{O}_{2}},\] the minimum e.m.f. required to carry out electrolysis of \[A{{l}_{2}}{{O}_{3}}\] is \[(F=96500\,C\,mo{{l}^{-1}})\]
A)
4.28 V
done
clear
B)
6.42 V
done
clear
C)
8.56 V
done
clear
D)
2.14 V
done
clear
View Answer play_arrow
Iron sulphide is heated in air to form A, an oxide of sulphur. A is dissolved in water to give an acid. The basicity of this acid is
A)
2
done
clear
B)
3
done
clear
C)
1
done
clear
D)
Zero
done
clear
View Answer play_arrow
Which of the following cannot be made by using Williamson's synthesis?
A)
Methoxybenzene
done
clear
B)
Benzyl-p-nitrophenyl ether
done
clear
C)
Methyl tertiary butyl ether
done
clear
D)
Di-tert-butyl ether
done
clear
View Answer play_arrow
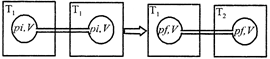
Two closed bulbs of equal volume (V) containing an ideal gas initially at pressure \[{{P}_{i}}\] and temperature \[{{T}_{1}}\] are connected through a narrow tube of negligible volume as shown in the figure below. The temperature of one of the bulbs is then raised to \[{{T}_{2}}.\] The final pressures \[{{P}_{f}}\] is :
A)
\[2{{p}_{i}}\left( \frac{{{T}_{2}}}{{{T}_{1}}+{{T}_{2}}} \right)\]
done
clear
B)
\[2{{p}_{i}}\left( \frac{{{T}_{1}}{{T}_{2}}}{{{T}_{1}}+{{T}_{2}}} \right)\]
done
clear
C)
\[{{p}_{i}}\left( \frac{{{T}_{1}}{{T}_{2}}}{{{T}_{1}}+{{T}_{2}}} \right)\]
done
clear
D)
\[2{{p}_{i}}\left( \frac{{{T}_{1}}}{{{T}_{1}}+{{T}_{2}}} \right)\]
done
clear
View Answer play_arrow
Which of the following is an expression of Raoults law if \[{{P}_{A}}\] is the partial pressure of the solvent in a solution. \[{{P}^{0}}_{A}\] is the partial pressure of pure solvent and if \[{{X}_{A}}\] and \[{{X}_{B}}\] are the mole fraction of the solute and the solvent respectively?
A)
\[{{P}_{A}}=P_{A}^{O}{{x}_{A}}\]
done
clear
B)
\[{{P}_{A}}=P_{A}^{O}{{x}_{A}}\left( \frac{1}{{{x}_{B}}} \right)\]
done
clear
C)
\[{{P}_{A}}=P_{A}^{O}{{x}_{B}}\]
done
clear
D)
\[P_{A}^{O}=P\left( \frac{{{x}_{A}}}{{{x}_{B}}} \right)\]
done
clear
View Answer play_arrow
Standard potentials \[\left( E{}^\circ \right)\] for some half-reactions are given below: (I) \[S{{n}^{4+}}+2e\xrightarrow{{}}S{{n}^{2+}};\]\[{{E}^{o}}=+0.15V\] (II) \[2H{{g}^{2+}}+2e\xrightarrow{{}}Hg_{2}^{2+};\]\[{{E}^{o}}=0.92V\] (III) \[Pb{{O}_{2}}+4{{H}^{+}}+2e\xrightarrow{{}}P{{b}^{2+}}+2{{H}_{2}}O;\]\[{{E}^{o}}=+1.45V\] based on the above, which one of the following statements is correct?
A)
\[S{{n}^{4+}}\] is a stronger oxidising agent than \[P{{b}^{4+}}\]
done
clear
B)
\[S{{n}^{2+}}\] is a stronger reducing agent than \[H{{g}_{2}}^{2+}\]
done
clear
C)
\[P{{b}^{2+}}\] is a stronger oxidisng agent than \[P{{b}^{4+}}\]
done
clear
D)
\[P{{b}^{2+}}\] is a stronger reducing agent than \[S{{n}^{2+}}\]
done
clear
View Answer play_arrow
Match the columns
Column-I Column-II [A] Compound formed when yellow \[CrO_{4}^{2-}\] acidified. (p) Acidified \[MnO_{4}^{-}\] [B] Reagent oxidises \[F{{e}^{2+}}\]to \[F{{e}^{3+}}.\] (q) \[C{{r}_{2}}O_{7}^{2-}\] [C] Compound produced when \[Mn{{O}_{2}}\] is fused with \[KN{{O}_{3}}.\] (r) \[{{K}_{2}}Mn{{O}_{4}}\] [D] Compound having dark purple crystals isostructural with \[KCl{{O}_{4}}.\] (s) \[KMn{{O}_{4}}\]
A)
A\[\to \](q), B\[\to \](p), C\[\to \](r), D\[\to \](s)
done
clear
B)
A\[\to \](p), B\[\to \](q), C\[\to \](r), D\[\to \](s)
done
clear
C)
A\[\to \](q), B\[\to \](r), C\[\to \](p), D\[\to \](s)
done
clear
D)
A\[\to \](q), B\[\to \](p), C\[\to \](s), D\[\to \](r)
done
clear
View Answer play_arrow
Phenol on reaction with \[B{{r}_{2}}\] in non-polar aprotic solvent furnishes
A)
2, 4, 6-Tribromophenol
done
clear
B)
p-Bromophenol
done
clear
C)
o-and p-Bromophenol
done
clear
D)
m-Bromophenol
done
clear
View Answer play_arrow
(1) \[{{N}_{2}}(g)+3{{H}_{2}}(g)2N{{H}_{3}}(g),\]\[{{K}_{1}}\] (2) \[{{N}_{2}}\left( g \right)+{{O}_{2}}\left( g \right)2NO\left( g \right),\]\[{{K}_{2}}\] (3) \[{{H}_{2}}(g)+\frac{1}{2}{{O}_{2}}(g){{H}_{2}}O(g),\]\[{{K}_{3}}\] The equation for the equilibrium constant of the reaction \[2N{{H}_{3}}(g)+\frac{5}{2}{{O}_{2}}(g)2NO(g)+3{{H}_{2}}O(g),\] \[({{K}_{4}})\] in terms of \[{{K}_{1}},\]\[{{K}_{2}}\] and \[{{K}_{3}}\] is:
A)
\[\frac{{{K}_{1}}.{{K}_{2}}}{{{K}_{3}}}\]
done
clear
B)
\[\frac{{{K}_{1}}.K_{3}^{2}}{{{K}_{2}}}\]
done
clear
C)
\[{{K}_{1}}{{K}_{2}}{{K}_{3}}\]
done
clear
D)
\[\frac{{{K}_{2}}.K_{3}^{3}}{{{K}_{1}}}\]
done
clear
View Answer play_arrow
A metallic crystal crystallizes into a lattice containing a sequence of layers AB AB AB......Any packing of spheres leaves out voids in the lattice. What percentage of volume of this lattice is empty space?
A)
74%
done
clear
B)
26%
done
clear
C)
50%
done
clear
D)
none of these.
done
clear
View Answer play_arrow
Which one of the following arrangements represents the correct order of solubilities of sparingly soluble salts \[H{{g}_{2}}C{{l}_{2}},C{{r}_{2}}{{(S{{O}_{4}})}_{3}},\]\[BaS{{O}_{4}}\]and \[CrC{{l}_{3}}\] respectively?
A)
\[BaS{{O}_{4}}>H{{g}_{2}}C{{l}_{2}}>C{{r}_{2}}{{(S{{O}_{4}})}_{3}}>CrC{{l}_{3}}\]
done
clear
B)
\[BaS{{O}_{4}}>H{{g}_{2}}C{{l}_{2}}>CrC{{l}_{3}}>C{{r}_{2}}{{(S{{O}_{4}})}_{3}}\]
done
clear
C)
\[BaS{{O}_{4}}>CrC{{l}_{3}}>H{{g}_{2}}C{{l}_{2}}>C{{r}_{2}}{{(S{{O}_{4}})}_{3}}\]
done
clear
D)
\[H{{g}_{2}}C{{l}_{2}}>BaS{{O}_{4}}>CrC{{l}_{3}}>C{{r}_{2}}{{(S{{O}_{4}})}_{3}}\]
done
clear
View Answer play_arrow
Which among the following factors is the most important in making fluorine the strongest oxidizing halogen?
A)
Hydration enthalpy
done
clear
B)
Ionization enthalpy
done
clear
C)
Electron affinity
done
clear
D)
Bond dissociation energy
done
clear
View Answer play_arrow
\[\alpha \]-D-Glucose and \[\beta \]-D-glucose differ from each other due to difference in one carbon with respect to its:
A)
Size of hemiacetal ring
done
clear
B)
Number of OH groups
done
clear
C)
Configuration
done
clear
D)
Conformation
done
clear
View Answer play_arrow
Identify the incorrect statement:
A)
The S-S-S bond angles in the \[{{S}_{8}}\] and \[{{S}_{6}}\] rings are the same.
done
clear
B)
Rhombic and monoclinic sulphur have \[{{S}_{8}}\] molecules.
done
clear
C)
\[{{S}_{2}}\] is paramagnetic like oxygen
done
clear
D)
\[{{S}_{8}}\] ring has a crown shape.
done
clear
View Answer play_arrow
In which of the following Tyndall effect is not observed?
A)
Suspensions
done
clear
B)
Emulsions
done
clear
C)
Sugar solution
done
clear
D)
Gold sol
done
clear
View Answer play_arrow
The most electropositive metals are isolated from their ores by:
A)
High temperature reduction with carbon.
done
clear
B)
Self reduction.
done
clear
C)
Thermal decomposition.
done
clear
D)
Electrolysis of fused ionic salts.
done
clear
View Answer play_arrow
The hypothetical complex chloro diaquatri- amminecobalt (III) chloride can be represented as
A)
\[[CoCl{{(N{{H}_{3}})}_{3}}{{({{H}_{2}}O)}_{2}}]C{{l}_{2}}\]
done
clear
B)
\[[Co{{\left( N{{H}_{3}} \right)}_{3}}\left( {{H}_{2}}O \right)C{{l}_{3}}]\]
done
clear
C)
\[[Co{{\left( N{{H}_{3}} \right)}_{3}}{{\left( {{H}_{2}}O \right)}_{2}}Cl]\]
done
clear
D)
\[[Co{{\left( N{{H}_{3}} \right)}_{3}}{{\left( {{H}_{2}}O \right)}_{3}}]C{{l}_{3}}\]
done
clear
View Answer play_arrow
Which of the following is not correctly matched?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Consider the following ionization enthalpies of two elements 'A' and 'B'.
Element Ionization Enthalpy (KJ/mol) 1st 2nd 3rd A 899 1757 14847 B 737 1450 7731
Which of the following statements is correct?
A)
Both ?A? and ?B? belong to group-1 where ?B? comes below ?A?.
done
clear
B)
Both ?A? and ?B? belong to group-1 where ?A? comes below ?B?.
done
clear
C)
Both ?A' and ?B? belong to group-2 where ?B? comes below ?A?.
done
clear
D)
Both ?A? and ?B? belong to group-2 where ?A? comes below ?B?.
done
clear
View Answer play_arrow
The pH of a solution is increased from 3 to 6; its \[{{H}^{+}}\]ion concentration will be
A)
reduced to half
done
clear
B)
doubled
done
clear
C)
reduced by 1000 times
done
clear
D)
increased by 1000 times
done
clear
View Answer play_arrow
The first ionization energies of alkaline earth metals are higher than those of the alkali metals. This is because
A)
There is an increase in the nuclear charge of the alkaline earth metals
done
clear
B)
There is a decrease in the nuclear charge of the alkaline earth metals
done
clear
C)
There is no change in the nuclear charge
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Identify the statement that is not correct as far as structure of diborane is concerned
A)
There are two bridging hydrogen atoms and four terminal hydrogen atoms in diborane
done
clear
B)
Each boron atom forms four bonds in diborane
done
clear
C)
The hydrogen atoms are not in the same plane in diborane
done
clear
D)
All, B - H bonds in diborane are similar
done
clear
View Answer play_arrow
The correct order of increasing acid strength of the compounds [A] \[C{{H}_{3}}C{{O}_{2}}H\] [B] \[MeOC{{H}_{2}}C{{O}_{2}}H\] [C] \[C{{F}_{3}}C{{O}_{2}}H\]
A)
\[D<A<B<C\]
done
clear
B)
\[A<D<B<C\]
done
clear
C)
\[B<D<A<C\]
done
clear
D)
\[D<A<C<B\]
done
clear
View Answer play_arrow
Hydrogen peroxide acts both as an oxidizing and as a reducing agent depending upon the nature of the reacting species. In which of the following cases \[{{H}_{2}}{{O}_{2}}\] acts as a reducing agent in acid medium?
A)
\[MnO_{4}^{-}\]
done
clear
B)
(b)\[C{{r}_{2}}O_{7}^{2-}\]
done
clear
C)
\[SO_{3}^{2-}\]
done
clear
D)
\[KI\]
done
clear
View Answer play_arrow
The enthalpy of formation for \[{{C}_{2}}{{H}_{4}}(g)C{{O}_{2}}(g)\] and \[{{H}_{2}}O(1)\] at \[25{}^\circ C\] and 1 atm pressure are 52, \[-394\] and \[-286\,kJ\,mo{{l}^{-1}}\] respectively The enthalpy of combustion of \[{{C}_{2}}{{H}_{4}}(g)\] will be
A)
\[+1412\,kJ\,mo{{l}^{-1}}\]
done
clear
B)
\[-1412\,kJ\,mo{{l}^{-1}}\]
done
clear
C)
\[141.2\,kJ\,mo{{l}^{-1}}\]
done
clear
D)
\[-141.2\,kJ\,mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
Consider the following standard electrode potentials (\[E{}^\circ \]in volts) in aqueous solution:
Element \[{{M}^{3+}}/M\] \[{{M}^{+}}/M\] Al \[-1.66\] \[+0.55\] TI \[+1.26\] \[-0.34\]
Based on these data, which of the following statements is correct?
A)
\[T{{l}^{+}}\] is more stable than \[A{{l}^{3+}}\]
done
clear
B)
\[A{{l}^{+}}\] is more stable than \[A{{l}^{3+}}\]
done
clear
C)
\[T{{l}^{+}}\] is more stable than \[A{{l}^{+}}\]
done
clear
D)
\[T{{l}^{3+}}\] is more stable than \[A{{l}^{3+}}\]
done
clear
View Answer play_arrow
Ozone is an important constituent of stratosphere because it:
A)
Destroys bacteria which are harmful to human life.
done
clear
B)
Prevents the formation of smog over large cities.
done
clear
C)
Removes poisonous gases of the atmosphere by reacting with them.
done
clear
D)
Absorbs ultraviolet radiation which is harmful to human life.
done
clear
View Answer play_arrow
Which of the following products is formed when benzaldehyde is treated with \[C{{H}_{3}}MgBr\] and the addition product so obtained is subjected to acid hydrolysis?
A)
A secondary alcohol
done
clear
B)
A primary alcohol
done
clear
C)
Phenol
done
clear
D)
tert-Butyl alcohol
done
clear
View Answer play_arrow
In which of the following pairs, there is greatest difference in the oxidation number of the underlined elements?
A)
\[\underline{N}{{O}_{2}}\] and \[{{\underline{N}}_{2}}{{O}_{4}}\]
done
clear
B)
\[{{\underline{P}}_{2}}{{O}_{5}}\] and \[{{\underline{P}}_{4}}{{O}_{10}}\]
done
clear
C)
\[{{\underline{N}}_{2}}O\] and \[\underline{N}O\]
done
clear
D)
\[\underline{S}{{O}_{2}}\] and \[\underline{S}{{O}_{3}}\]
done
clear
View Answer play_arrow
The boiling point of a solution of 0.11 g of a substance in 15 g of ether was found to be \[0.1{}^\circ C\] higher than that of pure ether. The molecular weight of the substance will be \[({{K}_{b}}=2.16\,K\,kg\,mo{{l}^{-1}})\]
A)
\[148\,g\,mo{{l}^{-1}}\]
done
clear
B)
\[158\,g\,mo{{l}^{-1}}\]
done
clear
C)
\[168\,g\,mo{{l}^{-1}}\]
done
clear
D)
\[178\,g\,mo{{l}^{-1}}\]
done
clear
View Answer play_arrow
Which one of the following is an example of homogeneous catalysis?
A)
Haber's process of synthesis of ammonia
done
clear
B)
Catalytic conversion of \[S{{O}_{2}}\]to \[S{{O}_{3}}\] in contact process
done
clear
C)
Catalytic hydrogenation of oils
done
clear
D)
Acid hydrolysis of methyl acetate
done
clear
View Answer play_arrow
The decomposition of a substance follows first order kinetics. Its concentration is reduced to 1/ 8th of its initial value in 24 minutes. The rate constant of the decomposition process is
A)
\[{1}/{24\,{{\min }^{-1}}}\;\]
done
clear
B)
\[\frac{0.692}{24}\,{{\min }^{-1}}\]
done
clear
C)
\[\frac{2.303}{24}\,\log \,\left( \frac{1}{8} \right)\,{{\min }^{-1}}\]
done
clear
D)
\[\frac{2.303}{24}\,\log \,\left( \frac{8}{1} \right)\,{{\min }^{-1}}\]
done
clear
View Answer play_arrow
The electronic configuration of elements A, B and C are \[[He]2{{s}^{1}},\] \[[Ne]3{{s}^{1}}\] and \[[Ar]4{{s}^{1}}\] respectively. Which one of the following order is correct for \[I{{E}_{1}}(in\,kJ\,mo{{l}^{-1}})\]of A, B and C?
A)
\[A>B>C\]
done
clear
B)
\[C>B>A\]
done
clear
C)
\[B>C>A\]
done
clear
D)
\[C>A>B\]
done
clear
View Answer play_arrow
An element (X) forms compounds of the formula \[XC{{l}_{3}},\] \[{{X}_{2}}{{O}_{5}}\] and \[C{{a}_{3}}{{X}_{2}}\] but does not form \[XC{{l}_{5}}.\] Which of the following is the element X?
A)
B
done
clear
B)
Al
done
clear
C)
N
done
clear
D)
P
done
clear
View Answer play_arrow
Which metal is present in brass, bronze and German silver?
A)
Zn
done
clear
B)
Mg
done
clear
C)
Cu
done
clear
D)
Al
done
clear
View Answer play_arrow
Both geometrical and optical isomerisms are shown by
A)
\[{{[Co{{(en)}_{2}}C{{l}_{2}}]}^{+}}\]
done
clear
B)
\[{{[Co{{(N{{H}_{3}})}_{5}}Cl]}^{2+}}\]
done
clear
C)
\[{{[Co{{(N{{H}_{3}})}_{4}}C{{l}_{2}}]}^{+}}\]
done
clear
D)
\[{{[Cr{{(ox)}_{3}}]}^{3-}}\]
done
clear
View Answer play_arrow
Which one of the following is most reactive towards nucleophilic substitution reaction?
A)
\[C{{H}_{2}}=CH-Cl\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}Cl\]
done
clear
C)
\[C{{H}_{3}}CH=CH-Cl\]
done
clear
D)
\[ClC{{H}_{2}}-CH=C{{H}_{2}}\]
done
clear
View Answer play_arrow
Which of the following is strongest Lewis base?
A)
\[C{{H}_{3}}^{-}\]
done
clear
B)
\[N{{H}_{2}}^{-}\]
done
clear
C)
\[O{{H}^{-}}\]
done
clear
D)
\[{{F}^{-}}\]
done
clear
View Answer play_arrow
The polynucleated condition appeared by free nuclear division without cytokinesis is
A)
Syncytium
done
clear
B)
Coenocytic
done
clear
C)
Found in amphibian oocyte
done
clear
D)
All of these
done
clear
View Answer play_arrow
What is the similarity between gymnosperms and angiosperms?
A)
Phloem of both have companian cells.
done
clear
B)
Endosperm is formed before fertilization in both.
done
clear
C)
Origin of ovule and seed is similar in both.
done
clear
D)
Both have leaves, stem and roots.
done
clear
View Answer play_arrow
Fibrous root system is better adapted than tap root system for
A)
transport of organic matter
done
clear
B)
absorption of water and minerals
done
clear
C)
storage of food
done
clear
D)
anchorage of plant to soil
done
clear
View Answer play_arrow
Which of the following element is necessary for translocation of sugars in plants?
A)
Boron
done
clear
B)
Molybdenum
done
clear
C)
Manganese
done
clear
D)
Iron
done
clear
View Answer play_arrow
In meiosis, actual haploidy in terms of DNA comes in
A)
Metaphase-I
done
clear
B)
Anaphase-II
done
clear
C)
Anaphase-I
done
clear
D)
Interkinesis
done
clear
View Answer play_arrow
Proton pumps in the plasma membranes of plant cells may
A)
generate a membrane potential that helps drive cations into the cell through their specific carriers.
done
clear
B)
be coupled to the movement of \[{{K}^{+}}\]into guard- cells.
done
clear
C)
drive the accumulation of sucrose in sieve- tube members.
done
clear
D)
be involved in all of the above
done
clear
View Answer play_arrow
The Z-scheme refers to
A)
the type of photosynthesis occurs in plants found in areas with minimal precipitation.
done
clear
B)
the pattern ofgrana within the chloroplasts of photosynthetic plants.
done
clear
C)
the carbon-fixation process which is also known as the Calvin cycle.
done
clear
D)
an energy diagram for the transfer of electrons in the light reactions of photosynthesis in plants.
done
clear
View Answer play_arrow
Which one of the following statement regarding auxin is/are correct?
A)
IAA and IBA are natural but NAA, 2, 4-D and 2, 4, 5-T are synthetic auxins.
done
clear
B)
IAA and NAA are natural but IBA, 2, 4, 5-T and 2, 4-D are synthetic auxin.
done
clear
C)
NAA and 2, 4, 5-T are natural but IAA, IBA and 2, 4-D are synthetic auxins.
done
clear
D)
IAA, NAA, IBA, 2, 4-D and 2, 4, 5-T are synthetic auxins.
done
clear
View Answer play_arrow
In angiospermic plant having chromosome number of 12 will have chromosome number in integuments and nucellus
A)
4
done
clear
B)
6
done
clear
C)
12
done
clear
D)
24
done
clear
View Answer play_arrow
When two gray-bodied fruit flies are mated, their offspring total 86 gray-bodied males, 81 yellow- bodied males, and 165 grey-bodied females. The allele for yellow body is
A)
sex-linked and dominant.
done
clear
B)
not sex-linked and dominant.
done
clear
C)
sex-linked and recessive.
done
clear
D)
not sex-linked and recessive.
done
clear
View Answer play_arrow
In negative operon
A)
co-repressor binds with represser
done
clear
B)
co-repressor does not bind with represser
done
clear
C)
co-repressor binds with inducer
done
clear
D)
cAMP have negative effect on lac operon
done
clear
View Answer play_arrow
Which of the following does not have stomata?
A)
Hydrophytes
done
clear
B)
Mesophytes
done
clear
C)
Xerophytes
done
clear
D)
Submerged hydrophytes
done
clear
View Answer play_arrow
Which of the following is pair of No-fertilizers?
A)
Azolla and BGA
done
clear
B)
Nostoc and legumes
done
clear
C)
Rhizobium and grasses
done
clear
D)
Salmonella and E. coli
done
clear
View Answer play_arrow
Transgenic plants are the ones:
A)
generated by introducing foreign DNA into a cell and regenerating a plant from that cell.
done
clear
B)
produced after protoplast fusion in artificial medium.
done
clear
C)
grown m artificial medium after hybridization in the field.
done
clear
D)
produced by a somatic embryo in artificial medium.
done
clear
View Answer play_arrow
Which one of the following is the matching pair of a sanctuary and its main protected wild animals:
A)
North Eastern Himalayan region \[-\] Sambar
done
clear
B)
Sunderbans \[-\] Rhino
done
clear
C)
Gir \[-\] Lion
done
clear
D)
Kaziranga \[-\] Musk deer
done
clear
View Answer play_arrow
The basis of karyotaxonomyis
A)
Number of nucleoli
done
clear
B)
Sedimentation rate ofribosomes
done
clear
C)
Chromosome banding
done
clear
D)
Chromosome number
done
clear
View Answer play_arrow
In Chlorophyceae, sexual reproduction occurs by
A)
isogamy and anisogamy
done
clear
B)
isogamy, anisogamy and oogamy
done
clear
C)
oogamy only
done
clear
D)
anisogamy and oogamy
done
clear
View Answer play_arrow
Velamen is found in
A)
roots of screwpine
done
clear
B)
aerial and terrestrial roots of orchids
done
clear
C)
leaves of Ficus elastic
done
clear
D)
only aerial roots of orchids
done
clear
View Answer play_arrow
Collenchyma differs from parenchyma in having
A)
living protoplasm
done
clear
B)
cellulose walls
done
clear
C)
vacuoles
done
clear
D)
pectin deposits at comers
done
clear
View Answer play_arrow
Which of the following amino acid has a hydroxyl group?
A)
Valine
done
clear
B)
Threonine
done
clear
C)
Leucine
done
clear
D)
Histidine
done
clear
View Answer play_arrow
Aquaporins are
A)
Cytoplasmic connections between cortex cells.
done
clear
B)
Openings in the lower epidermis of leaves through which water vapour escapes
done
clear
C)
Openings into root hairs through which water enters
done
clear
D)
Water specific channels in membranes that may regulate the rate of osmosis.
done
clear
View Answer play_arrow
Which one is a \[{{C}_{4}}\]-plant?
A)
Papaya
done
clear
B)
Pea
done
clear
C)
Potato
done
clear
D)
Maize/Corn
done
clear
View Answer play_arrow
Which of the following is an important intermediate found in all the types of respiration?
A)
Acetyl CoA
done
clear
B)
Pyruvic acid
done
clear
C)
Oxaloacetate
done
clear
D)
Tricarboxylic acid
done
clear
View Answer play_arrow
Tomatoes exhibiting delayed ripening were developed by a biotechnique that involved:
A)
DNA against enzymes
done
clear
B)
Antisense RNA
done
clear
C)
Mutation
done
clear
D)
None of the above
done
clear
View Answer play_arrow
In tertiary structure of DNA, what is a histone octamer?
A)
A complex consisting of eight positively charged histone proteins (two of each \[{{H}_{2}}A,\]\[{{H}_{2}}B,\]\[{{H}_{3}}\] and\[{{H}_{4}}\]) that aid in the packaging of DNA.
done
clear
B)
A complex consisting of eight negatively charged histone proteins (two of each \[{{H}_{2}}A,\] \[{{H}_{2}}B,\]\[{{H}_{3}}\] and\[{{H}_{4}}\]) that aid in the packaging of DNA.
done
clear
C)
A complex consisting of nine positively charged histone proteins (\[{{H}_{1}}\] and two of each \[{{H}_{2}}A,\]\[{{H}_{2}}B,\]\[{{H}_{3}}\]and\[{{H}_{4}}\]) that aid in the packaging DNA.
done
clear
D)
A complex consisting of nine negatively charged histone proteins (\[{{H}_{1}}\] and two of each \[{{H}_{2}}B,\]\[{{H}_{3}}\]and\[{{H}_{4}}\]) that aid in the packaging of DNA.
done
clear
View Answer play_arrow
Which one among the following chemicals is used for causing defoliation of forest trees?
A)
Phosphon-D
done
clear
B)
Malic hydrazide
done
clear
C)
2, 4-dichlorophenoxy acetic acid
done
clear
D)
Amo-1618
done
clear
View Answer play_arrow
If the forest cover is reduced to half, what is most likely to happen on a long term basis?
A)
Tribals living in these areas will starve to death
done
clear
B)
Cattle in these and adjoining areas will die due to lack of fodder
done
clear
C)
Large areas will become deserts
done
clear
D)
Crop breeding programmes will suffer due to a reduced availability of variety of germplasm
done
clear
View Answer play_arrow
Major aerosol pollutant in jet plane emission is
A)
Sulphur dioxide
done
clear
B)
Carbon monoxide
done
clear
C)
Methane
done
clear
D)
Fluorocarbon
done
clear
View Answer play_arrow
Which one is correct match
A. Soil I. Recharge B. Ground water II. Rotational grazing C. Forest management III. Reverine forest D. Grassland management IV. Taungya E. Wetland V. Erosion VI. Energy
A)
A\[\to \]V, B\[\to \]I, C\[\to \]IV, D\[\to \]II, E\[\to \]III
done
clear
B)
A\[\to \]I, B\[\to \]IV, C\[\to \]III, D\[\to \]II, E\[\to \]V
done
clear
C)
A\[\to \]V, B\[\to \]I, C\[\to \]IV, D\[\to \]III, E\[\to \]II
done
clear
D)
A\[\to \]V, B\[\to \]II, C\[\to \]III, D\[\to \]IV, E\[\to \]I
done
clear
View Answer play_arrow
The amalgamation for six-kingdom classification into domains given by Carl Woese was based upon
A)
Types of r-RNA
done
clear
B)
Sequence of ribosomal RNA genes
done
clear
C)
Sequence of genes on DNA
done
clear
D)
Cell wall composition
done
clear
View Answer play_arrow
Sexual reproduction in Spirogyra is an advanced feature because it shows
A)
physiologically differentiated sex organs
done
clear
B)
different sizes of motile sex organs
done
clear
C)
same size of motile sex organs
done
clear
D)
morphologically different sex organs
done
clear
View Answer play_arrow
Moniliform or beaded roots are storage root in
A)
Dahlia
done
clear
B)
Asparagus
done
clear
C)
Momordica
done
clear
D)
Portulaca
done
clear
View Answer play_arrow
Which of the following plant shows multiple epidermis?
A)
Croton
done
clear
B)
Allium
done
clear
C)
Nerium
done
clear
D)
Cucurbita
done
clear
View Answer play_arrow
Select the correct statement with respect to mitosis.
A)
Chromatids separate but remain in the centre of the cell in anaphase.
done
clear
B)
Chromatids start moving towards opposite poles in telophase.
done
clear
C)
Golgi complex and endoplasmic reticulum are still visible at the end of prophase.
done
clear
D)
Chromosomes move to the spindle equator and get align
done
clear
View Answer play_arrow
In an experiment, mature leaves on the plant were enclosed for a fixed amount of time in a transparent bag that had radioactive \[C{{O}_{2}}.\] In which part of the plant will maximum radioactivity be found after some time?
A)
Actively growing leaves.
done
clear
B)
Guard cells of all the leaves.
done
clear
C)
In mature leaves.
done
clear
D)
Senescing leaves and roots.
done
clear
View Answer play_arrow
In laboratory experiments, two species of the protist Paramecium were grown alone and in the presence of the other species. The following graphs show growth of species 1 (left) and species 2 (right), both alone and when in mixed culture.
Interpretation of these graphs shows that
A)
Competitive exclusion occurred in these experiments.
done
clear
B)
Both species are affected by interspecific competition but species 1 is affected less.
done
clear
C)
Both species are affected by interspecific competition but species 2 is affected less.
done
clear
D)
Both species are affected equally by interspecific competition.
done
clear
View Answer play_arrow
Which one of the following insecticides is more stable in the environment?
A)
DDT
done
clear
B)
Diazinon
done
clear
C)
Camphechlor
done
clear
D)
Malathion
done
clear
View Answer play_arrow
If by radiation all nitrogenase enzymes are inactivated, then there will be no
A)
fixation of nitrogen in legumes
done
clear
B)
fixation of atmospheric nitrogen
done
clear
C)
conversion from nitrate to nitrite in legumes
done
clear
D)
conversion from ammonium to nitrate in soil
done
clear
View Answer play_arrow
Which one of the following is an example of Ex- situ conservation?
A)
Wildlife sanctuary
done
clear
B)
Seed bank
done
clear
C)
Sacred groves
done
clear
D)
National park
done
clear
View Answer play_arrow
The class of enzymes that catalyze the removal of a group from a substrate without addition of water, leaving double bonds, is
A)
Transferases
done
clear
B)
Dehydrogenases
done
clear
C)
Hydrolases
done
clear
D)
Lyases
done
clear
View Answer play_arrow
The main organelle involved in modification and routing of newly synthesized proteins to their destinations is
A)
chloroplast
done
clear
B)
mitochondria
done
clear
C)
lysosome
done
clear
D)
endoplasmic reticulum
done
clear
View Answer play_arrow
Which of the following statements about nitrogen fixation is false?
A)
Nitrogenase is only catalytic under anaerobic conditions.
done
clear
B)
The energy for nitrogen fixation can be provided by either photosynthesis or respiration.
done
clear
C)
In nitrogen fixation, nitrogen is reduced by the addition of three successive pairs of hydrogen atoms.
done
clear
D)
Most nitrogen fixing microbes are aerobic.
done
clear
View Answer play_arrow
When a photosynthetic plant is transferred to an atmosphere of enriched \[{{O}_{2,}}\] its rate of
A)
photosynthesis would increase.
done
clear
B)
photosynthesis would decrease.
done
clear
C)
respiration would decrease.
done
clear
D)
osmosis would increase.
done
clear
View Answer play_arrow
The diagram below summarises the stages involved in respiratory metabolism of mammalian skeletal muscle.
Stage 4 in the diagram occurs in mammalian muscle cells under anaerobic conditions. Why is surge 4 necessary?
A)
to detoxfying, pyruvate
done
clear
B)
to form lactate which is immediately reconverted to glycogen.
done
clear
C)
to oxidise the reduced nicotinamide adenine di-nucleotide facilitating stage 2
done
clear
D)
to reduce the oxidised nicotinamide adenine dinucleotide facilitating stage 2
done
clear
View Answer play_arrow
Common enzyme in giycolysis and pentose phosphate pathways is
A)
hexokinase
done
clear
B)
aconitase
done
clear
C)
famarase
done
clear
D)
dehydrogenase
done
clear
View Answer play_arrow
Bacteria lack alternation of generation because there is
A)
neither syngamy nor reduction division.
done
clear
B)
distinct chromosomes are absent.
done
clear
C)
no conjugation.
done
clear
D)
no exchange of genetic material.
done
clear
View Answer play_arrow
Which one of the following categories of animals, is correctly described with no single exception in it?
A)
All sponges are marine and have collar cells.
done
clear
B)
All mammals are viviparous and possess diaphragm for breathing.
done
clear
C)
All bony fishes have four pairs of gills and an operculum on each side.
done
clear
D)
All reptiles possess scales, have a three chambered heart and are cold blooded (poikilothermal).
done
clear
View Answer play_arrow
Which word combination applies to stratified squamous epithelium?
A)
Mesoderm \[-\] calcification
done
clear
B)
Ecoderm \[-\] keratinization
done
clear
C)
Mesoderm \[-\] ossification
done
clear
D)
Endoderm \[-\] cornification
done
clear
View Answer play_arrow
Which of the following is a component of vitamin (thiamine, biotin), Acetyl CoA, cysteine, methionine and ferredoxin?
A)
Fe
done
clear
B)
S
done
clear
C)
Co
done
clear
D)
K
done
clear
View Answer play_arrow
About 70% of \[C{{O}_{2}}\] is transported as
A)
Carbonic acid
done
clear
B)
Carboxyhaemoglobin
done
clear
C)
Bicarbonates
done
clear
D)
Carbamino compounds
done
clear
View Answer play_arrow
In human heart, which of the following valve remains in contact of the oxygenated blood only?
A)
Tricuspid valve
done
clear
B)
Semilunar valve
done
clear
C)
Eustachian valve
done
clear
D)
Mitral valve
done
clear
View Answer play_arrow
The urine under normal conditions does not contain glucose because
A)
The normal blood sugar is fructose
done
clear
B)
Glucose of blood is not filtered in the glomerulus
done
clear
C)
Glucose in glomerular filtrate is reabsorbed in the uriniferous tubules
done
clear
D)
Glucose in glomerular filtrate is converted into glycogen.
done
clear
View Answer play_arrow
Which of the following sets of vertebrate hormones are all produced in the anterior pituitary gland?
A)
Somatostatin, vasopressin, insulin
done
clear
B)
Prolactin, growth hormone, enkephalins
done
clear
C)
Oxytocin, prolactin, adrenocorticotropin
done
clear
D)
Estrogen, progesterone, testosterone
done
clear
View Answer play_arrow
Which one of the following statements is incorrect about menstruation?
A)
During normal menstruation about 40 ml. blood is lost
done
clear
B)
The menstrual fluid can easily clot
done
clear
C)
At menopause in the female, there is especially abrupt increase in gonadotropic hormones
done
clear
D)
The beginning of the cycle of menstruation is called menarche
done
clear
View Answer play_arrow
Find the incorrect match
A)
Super bug \[-\]A.N. Chakroborty
done
clear
B)
Minisatellite \[-\] Alee Jeffrey
done
clear
C)
Genomics \[-\] Roderick
done
clear
D)
Dolly \[-\] Gurdon
done
clear
View Answer play_arrow
In which condition, the gene ratio remains constant for any species?
A)
Sexual selection
done
clear
B)
Random mating
done
clear
C)
Mutation
done
clear
D)
Gene flow
done
clear
View Answer play_arrow
Geometric representation of age structure is a characteristic of
A)
population
done
clear
B)
landscape
done
clear
C)
ecosystem
done
clear
D)
biotic community
done
clear
View Answer play_arrow
What is true for monoclonal antibodies?
A)
These antibodies obtained from one parent and for one antigen
done
clear
B)
These antibodies obtained from one parent and for two antigens
done
clear
C)
These antibodies obtained from one parent and for many antigens
done
clear
D)
These antibodies obtained from many parents and for many antigens
done
clear
View Answer play_arrow
Phosphorus is a sedimentary nutrient. This means it
A)
cycles very slowly.
done
clear
B)
never enters the atmosphere.
done
clear
C)
settles as sediment in the ocean.
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Genetic material of TMV is
A)
RNA
done
clear
B)
DNA
done
clear
C)
Both
done
clear
D)
Protein
done
clear
View Answer play_arrow
All but one of the following bones are found in the human appendicular skeleton. Select the exception.
A)
Finger bones
done
clear
B)
Upper arm bone
done
clear
C)
Jaw bone
done
clear
D)
Ankle bones
done
clear
View Answer play_arrow
In which of the following animals, respiration occurs without any respiratory organ?
A)
Frog
done
clear
B)
Fish
done
clear
C)
Cockroach
done
clear
D)
Earthworm
done
clear
View Answer play_arrow
Spending the dry hot period in an inactive state by an animal is
A)
camouflage
done
clear
B)
hibernation
done
clear
C)
aestivation
done
clear
D)
mullerian mimicry
done
clear
View Answer play_arrow
In animal cells, cytokinesis involves
A)
the separation of sister chromatids
done
clear
B)
the contraction of the contractile ring of microfilament
done
clear
C)
depolymerisation of kinetochore microtubules
done
clear
D)
a protein kinase that phosphorylates other enzymes
done
clear
View Answer play_arrow
Myocardial Infarction is caused by
A)
hardening of arteries
done
clear
B)
lumpy thickness develop in the inner walls of arteries
done
clear
C)
clot may occur in the lumen of a coronary artery
done
clear
D)
sudden interruption in blood flow towards a portion of heart
done
clear
View Answer play_arrow
Continued consumption of a diet rich in butter, red meat and eggs for a long period may lead to
A)
vitamin toxicity
done
clear
B)
kidney stones
done
clear
C)
hypercholesterolemia
done
clear
D)
urine laden with ketone bodies
done
clear
View Answer play_arrow
The scientific value of a "DNA chip" is that
A)
researchers can determine how an entire genome functions.
done
clear
B)
DNA can be monitored for the presence of particular mutations.
done
clear
C)
the activity of all the genes in an organism can be monitored at once.
done
clear
D)
All of the above
done
clear
View Answer play_arrow
In an egg, the type of cleavage is determined by
A)
shape and size of the sperm
done
clear
B)
size and location of the nucleus
done
clear
C)
amount and distribution of yolk
done
clear
D)
number of egg membranes
done
clear
View Answer play_arrow
If for some reason, the vasa efferentia in the human reproductive system gets blocked, the gametes will not be transported from
A)
testes to epididymis
done
clear
B)
epididymis to vas deferens
done
clear
C)
ovary to uterus
done
clear
D)
vagina to uterus
done
clear
View Answer play_arrow
Medical Termination of Pregnancy (MTP) is considered safe up to how many weeks of pregnancy?
A)
Eight weeks
done
clear
B)
Twelve weeks
done
clear
C)
Eighteen weeks
done
clear
D)
Six weeks
done
clear
View Answer play_arrow
During blood taping, agglutination indicates that the
A)
RBCs carry certain antigens
done
clear
B)
Plasma contains certain antigens
done
clear
C)
RBCs carry certain antibodies
done
clear
D)
Plasma contains certain antibodies
done
clear
View Answer play_arrow
Ultrasound of how much frequency is beamed into human body for sonography?
A)
15-30 MHz
done
clear
B)
1-15 MHz
done
clear
C)
45-70 MHz
done
clear
D)
30-45 MHz
done
clear
View Answer play_arrow
Cry I endotoxins obtained from Bacillus thuringiensis are effective against
A)
mosquitoes
done
clear
B)
flies
done
clear
C)
nematodes
done
clear
D)
boll worms
done
clear
View Answer play_arrow
Retention of larval characters even after sexual maturity is called
A)
Parthenogenesis
done
clear
B)
Ontogenesis
done
clear
C)
Paedogenesis
done
clear
D)
Neoteny
done
clear
View Answer play_arrow
Which of the following type of enzyme is not matched correctly with the molecule that it breaks down?
A)
Amylase-starch
done
clear
B)
Lipase-starch
done
clear
C)
Protease-proteins
done
clear
D)
Disaccharidase-sugars
done
clear
View Answer play_arrow
The alveoli contain specialized cells that secrete a phospholipoprotein complex known as pulmonary surfactant. The function of pulmonary surfactant is:
A)
to provide some rigidity to the alveoli to prevent lung collapse
done
clear
B)
to decrease the surface tension of the alveoli, making it easier to expand them
done
clear
C)
to facilitate the diffusion of \[{{O}_{2}}\] and \[C{{O}_{2}}\] by providing protein channels through which these gases flow
done
clear
D)
to provide a sticky surface upon which dust and microbes are trapped and disposed of
done
clear
View Answer play_arrow
The structure which prevents the entry of food into the windpipe is
A)
Gullet
done
clear
B)
Glottis
done
clear
C)
Tonsil
done
clear
D)
Epiglottis
done
clear
View Answer play_arrow
Layer of cells that secrete enamel of tooth is
A)
Osteoblast
done
clear
B)
Ameloblast
done
clear
C)
Odontoblast
done
clear
D)
Dentoblast
done
clear
View Answer play_arrow
Cow's milk is slightly yellowish in colour due to the presence of
A)
Carotene
done
clear
B)
Riboflavin
done
clear
C)
Xanthophyll
done
clear
D)
Xanthophyll and Carotene
done
clear
View Answer play_arrow
Blood plasma contains proteins but glomerular filtrate does not. Why does this difference in composition occur?
A)
Blood osmotic pressure is maintained by the presence of plasma proteins
done
clear
B)
Proteins are actively transported from the kidney tubule back into the blood capillaries
done
clear
C)
Proteins cannot pass through the membranes of the glomerular capillaries.
done
clear
D)
There is a high hydrostatic pressure in the blood within the glomerular capillaries.
done
clear
View Answer play_arrow
Which is not correct with respect to human kidney?
A)
The peripheral region is called cortex and central medulla
done
clear
B)
Malpighian capsules are present in the cortex region
done
clear
C)
Blood enters glomerulus through efferent arterioles
done
clear
D)
The concave part of kidney is called hilus
done
clear
View Answer play_arrow
Frequency of sound is discriminated by
A)
The intensity of movement of basilar fibres of cochlea
done
clear
B)
The type of fluid - perilymph or endolymph
done
clear
C)
The site at the cochlear coil
done
clear
D)
All of these
done
clear
View Answer play_arrow
Which one of the following is the correct difference between rod cells and cone cells of our retina?
A)
Distribution Rod Cells - More concentrated in centre of retina Cone Cells - Evently distributed all over retina
done
clear
B)
Visual acuity Rod Cells - High Cone Cells - Low
done
clear
C)
Visual pigment contained Rod Cells - Iodopsin Cone Cells - Rhodopsin
done
clear
D)
Over all function Rod Cells - Vision in poor light Cone Cells - Colour vision and detailed vision in bright light
done
clear
View Answer play_arrow
Which of the following glands grows to the maximum size at puberty and then diminishes gradually?
A)
Thymus
done
clear
B)
Pituitary
done
clear
C)
Thyroid
done
clear
D)
Adrenal
done
clear
View Answer play_arrow
Which of the following statements about human pregnancy and fetal development is falser
A)
The blastocyst is a stage in fetal development that appears early during the third trimester.
done
clear
B)
A vital connection between the fetal and maternal blood supplies occurs at the placenta.
done
clear
C)
Most of the growth in fetal size occurs during the second and third trimesters.
done
clear
D)
All major organ systems have formed by the end of the fetus's first three months of life.
done
clear
View Answer play_arrow
Saltatory conduction of nerve impulse takes place through:
A)
Myelinated fibre
done
clear
B)
Non-myelinated fibre
done
clear
C)
Gray fibres
done
clear
D)
None of these
done
clear
View Answer play_arrow
In which part of the brain corpora quadrigemina is located?
A)
Diencephalon
done
clear
B)
Mesencephalon
done
clear
C)
Prosencephalon
done
clear
D)
Rhombencephalon
done
clear
View Answer play_arrow
The joint between _____(i)_____ and ____(ii)____ forms ball and socket joint.
A)
(i) - Humerus, (ii) - Ulna
done
clear
B)
(i) - Humerus, (ii) - Scapula
done
clear
C)
(i) - Ulna, (ii) - Radius
done
clear
D)
(i) - Ulna, (ii) - Scapula
done
clear
View Answer play_arrow
Meningitis, a disease which is responsible for damage of the membrane of the brain, is caused by:
A)
Neisseria
done
clear
B)
Bordetella
done
clear
C)
Bacillus
done
clear
D)
Yersinia
done
clear
View Answer play_arrow
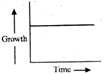
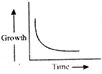
Which of the following shows the correct graph of arithmetic growth?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow


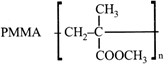
 The compound I is:
The compound I is: 

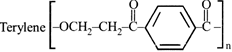
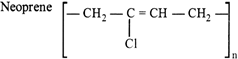
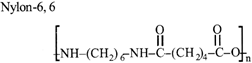
 Interpretation of these graphs shows that
Interpretation of these graphs shows that 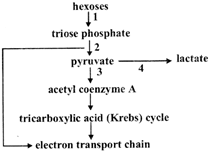 Stage 4 in the diagram occurs in mammalian muscle cells under anaerobic conditions. Why is surge 4 necessary?
Stage 4 in the diagram occurs in mammalian muscle cells under anaerobic conditions. Why is surge 4 necessary?