n identical light bulbs, each designed to draw P power from a certain voltage supply, are joined in series across that supply. The total power which they will draw is
A)
\[nP\]
done
clear
B)
\[P\]
done
clear
C)
\[\frac{P}{n}\]
done
clear
D)
\[\frac{P}{{{n}^{2}}}\]
done
clear
View Answer play_arrow
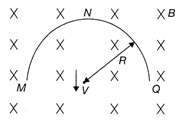
A thin semi-circular conducting ring of radius R is falling with its plane vertical in a horizontal magnetic induction\[\vec{B}.\]At the position MNQ, the speed of the ring is \[\upsilon .\]The potential difference developed across the ring is
A)
0
done
clear
B)
\[\frac{1}{2}B\upsilon \pi {{R}^{2}},\] and M is at a higher potential
done
clear
C)
\[\pi RB\upsilon ,\]and Q is at a higher potential
done
clear
D)
\[2RB\upsilon ,\] and Q is at a higher potential
done
clear
View Answer play_arrow
A particle with a specific charge s is fired with a speed\[\upsilon \]towards a wall at a distance d, perpendicular to the wall. What minimum magnetic field must exist in this region for the particle not to hit the wall?
A)
\[\frac{\upsilon }{sd}\]
done
clear
B)
\[\frac{2\upsilon }{sd}\]
done
clear
C)
\[\frac{\upsilon }{2sd}\]
done
clear
D)
\[\frac{\upsilon }{4sd}\]
done
clear
View Answer play_arrow
The activity of a sample of radioactive material is \[{{A}_{1}}\]at time \[{{t}_{1}}\]and \[{{A}_{2}}\]at time\[{{t}_{2}}({{t}_{2}}>{{t}_{1}}).\]Its mean life is T.
A)
\[{{A}_{1}}{{t}_{1}}={{A}_{2}}{{t}_{2}}\]
done
clear
B)
\[\frac{{{A}_{1}}-{{A}_{2}}}{{{t}_{2}}-{{t}_{1}}}=\text{constant}\]
done
clear
C)
\[{{A}_{2}}={{A}_{1}}{{e}^{\left( \frac{{{t}_{1}}-{{t}_{2}}}{T} \right)}}\]
done
clear
D)
\[{{A}_{2}}={{A}_{1}}{{e}^{\left( \frac{{{t}_{1}}}{T{{t}_{2}}} \right)}}\]
done
clear
View Answer play_arrow
Equations of two progressive waves at a certain point in a medium are given by \[{{y}_{1}}=a\sin (\omega t+{{\phi }_{1}})\]and\[{{y}_{2}}=a\sin (\omega t+{{\phi }_{2}}).\]If amplitude and time period of resultant wave formed by the superposition of these two waves is same as that of both the waves, then \[{{\phi }_{1}}-{{\phi }_{2}}\]is
A)
\[\frac{\pi }{3}\]
done
clear
B)
\[\frac{2\pi }{3}\]
done
clear
C)
\[\frac{\pi }{6}\]
done
clear
D)
\[\frac{\pi }{4}\]
done
clear
View Answer play_arrow
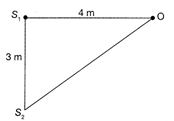
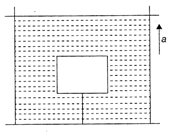
In the figure the intensity of waves arriving at D from two coherent sources\[{{s}_{1}}\]and \[{{s}_{2}}\] is \[{{I}_{0}}.\]The wavelength of the wave is \[\lambda =4\,m.\]Resultant intensity at D will be
A)
\[4{{I}_{0}}\]
done
clear
B)
\[{{I}_{0}}\]
done
clear
C)
\[2{{I}_{0}}\]
done
clear
D)
0
done
clear
View Answer play_arrow
Which of the following relation is correct.
A)
\[\sqrt{{{\varepsilon }_{0}}{{E}_{0}}}=\sqrt{{{\mu }_{0}}{{B}_{0}}}\]
done
clear
B)
\[\sqrt{{{\mu }_{0}}{{\varepsilon }_{0}}}{{E}_{0}}={{B}_{0}}\]
done
clear
C)
\[{{E}_{0}}=\sqrt{{{\mu }_{0}}{{\varepsilon }_{0}}{{B}_{0}}}\]
done
clear
D)
\[\sqrt{{{\mu }_{0}}{{E}_{0}}}=\sqrt{{{\varepsilon }_{0}}}{{B}_{0}}\]
done
clear
View Answer play_arrow
A piano convex lens fits exactly into a piano concave lens. Their plane surfaces are parallel to each other. If the lenses are made of different materials of refractive indices\[{{\mu }_{1}}\] and \[{{\mu }_{2}}\] and R is the radius of curvature of the curved surface of the lenses, then focal length of the combination is
A)
\[\frac{R}{2({{\mu }_{1}}+{{\mu }_{2}})}\]
done
clear
B)
\[\frac{R}{2({{\mu }_{1}}-{{\mu }_{2}})}\]
done
clear
C)
\[\frac{R}{({{\mu }_{1}}-{{\mu }_{2}})}\]
done
clear
D)
\[\frac{2R}{({{\mu }_{2}}-{{\mu }_{1}})}\]
done
clear
View Answer play_arrow
In Young's double slit experiment, the two equally bright slits are coherent, but of phase difference \[\pi /3.\]If maximum intensity on the screen is \[{{I}_{o}},\]the intensity at the point on the screen equidistant from the slits is
A)
\[{{I}_{o}}\]
done
clear
B)
\[\frac{{{I}_{o}}}{2}\]
done
clear
C)
\[\frac{{{I}_{o}}}{4}\]
done
clear
D)
\[\frac{3{{I}_{o}}}{4}\]
done
clear
View Answer play_arrow
Light described at a place by the equation \[E=100V\,{{m}^{-1}}[\sin (6\times {{10}^{15}}t)\]\[+\sin (8\times {{10}^{15}})]\]falls on a metal surface of work function 2 eV The maximum KE of the photoelectronsis: (Use\[h=6.6\times {{10}^{-34}}J-s\]
A)
2.25 eV
done
clear
B)
3.25 eV
done
clear
C)
4.24 eV
done
clear
D)
5.25 eV
done
clear
View Answer play_arrow
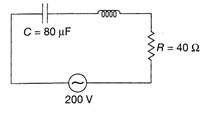
In the figure, a series LCR circuit is connected to a variable frequency 200 V source \[L=5\,H,C=80\,\mu F\]and \[R=40\,\Omega .\]Then the source frequency which drive the circuit at resonance is
A)
25 Hz
done
clear
B)
\[\frac{25}{\pi }HZ\]
done
clear
C)
\[50\,Hz\]
done
clear
D)
\[\frac{50}{\pi }\,Hz\]
done
clear
View Answer play_arrow
The binding energy of two nuclei\[{{P}^{n}}\] and \[{{Q}^{2n}}\]are \[x-\]joule and \[y-\]joule, respectively. Then the energy released in the reaction \[{{P}^{n}}+{{P}^{n}}={{Q}^{2n}}\]will be
A)
\[2x+y\]
done
clear
B)
\[y-2x\]
done
clear
C)
\[xy\]
done
clear
D)
\[x+y\]
done
clear
View Answer play_arrow
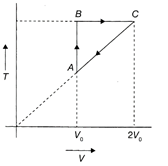
An ideal monatomic gas undergoes a cyclic process ABCA as shown in the figure. The ratio of heat absorbed during AB to the work done on the gas during BC is
A)
\[\frac{5}{2\,\text{in}\,2}\]
done
clear
B)
\[\frac{5}{3}\]
done
clear
C)
\[\frac{5}{3\,\text{in}\,2}\]
done
clear
D)
\[\frac{5}{6}\]
done
clear
View Answer play_arrow
Two identical rigid containers joined by a small pipe initially contain the same gas at pressure \[{{P}_{o}}\]and absolute temperature \[{{T}_{o}}\]One container is now maintained at the same temperature while the other is heated to \[2{{T}_{o}}.\]The common pressure of the gases will be
A)
\[\frac{3}{2}{{P}_{0}}\]
done
clear
B)
\[\frac{4}{3}{{P}_{0}}\]
done
clear
C)
\[\frac{5}{3}{{P}_{0}}\]
done
clear
D)
\[2{{P}_{0}}\]
done
clear
View Answer play_arrow
An ideal gas undergoes cyclic process ABC as shown in the temperature-volume graph. For the process, the ratio \[{{P}_{B}}/{{P}_{A}}\]equals (\[{{P}_{B}},{{P}_{A}}\]being pressure at B and A, respectively)
A)
1
done
clear
B)
\[\frac{1}{2}\]
done
clear
C)
\[\frac{1}{4}\]
done
clear
D)
2
done
clear
View Answer play_arrow
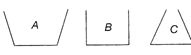
The three vessels shown in the figure, have same base area. Equal volumes of a liquid are poured in the three vessels. The force on the base will be
A)
Maximum in Vessel A
done
clear
B)
Maximum in Vessel B
done
clear
C)
Maximum in Vessel C
done
clear
D)
Equal in all the vessels
done
clear
View Answer play_arrow
A body having volume V and density p is attached to the bottom of a container as shown in the figure. Density of the liquid is d \[d(>\rho ).\] The container has a constant upward acceleration a. Tension in the string is
A)
\[V[dg-\rho (g+a)]\]
done
clear
B)
\[V(g+a)(d-\rho )\]
done
clear
C)
\[V(d-\rho )g\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
A point object is placed on the optic axis of a convex lens of focal length\[f\]at a distance of \[2f\]to the left of it. The diameter of the lens is d. An eye is placed at a distance of \[3f\] to the right of the lens and a distance h below the optic axis. The maximum value of h to see the image is
A)
\[\frac{d}{2}\]
done
clear
B)
\[d\]
done
clear
C)
\[\frac{d}{4}\]
done
clear
D)
\[\frac{d}{3}\]
done
clear
View Answer play_arrow
Ultraviolet light of wavelength 280 nm is used in an experiment on photoelectric effect with lithium of work function 2.5 eV The maximum and minimum ICE of the photoelectrons, if anode voltage is 3 V, is: \[(hc=1240\,eV\,nm)\]
A)
2.5 eV, 0
done
clear
B)
4.9 eV, 3eV
done
clear
C)
5.9 eV, 0.5 eV
done
clear
D)
5.5 eV, 3eV
done
clear
View Answer play_arrow
The displacement (in meters) of a body varies with time t (in seconds as\[y=(2/3){{t}^{2}}+16+2.\] How long does the body take to come to rest?
A)
8 seconds
done
clear
B)
10 seconds
done
clear
C)
12 seconds
done
clear
D)
16 seconds
done
clear
View Answer play_arrow
A solid sphere is rotating in free space. If the radius of the sphere is increased keeping mass same, which one of the following will not be affected?
A)
Moment of inertia
done
clear
B)
Angular momentum
done
clear
C)
Angular velocity
done
clear
D)
Rotational kinetic energy
done
clear
View Answer play_arrow
A ring starts from rest and acquires an angular speed of 10 rad/s in 2 s. The mass of the ring is 500 g and its radius is 20 cm. The torque on the ring is
A)
0.02 Nm
done
clear
B)
0.20 Nm
done
clear
C)
0.10 Nm
done
clear
D)
0.01 Nm
done
clear
View Answer play_arrow
If the gravitational force of attraction between any two bodies were to vary as instead of \[1/{{r}^{3}},\]instead of \[1/{{r}^{2}}.\] The period of revolution of a planet round the sun would vary as:
A)
\[\sqrt{r}\]
done
clear
B)
\[r\]
done
clear
C)
\[{{r}^{\frac{3}{2}}}\]
done
clear
D)
\[{{r}^{2}}\]
done
clear
View Answer play_arrow
The displacement\[x\](in centimeters) of an oscillating particle varies with time t (in seconds) as\[x=2\cos (0.5\pi t+\pi /3).\]The magnitude of the maximum acceleration of the particle in \[c{{m}^{-2}}\]is
A)
\[\frac{\pi }{2}\]
done
clear
B)
\[\frac{\pi }{4}\]
done
clear
C)
\[\frac{{{\pi }^{2}}}{2}\]
done
clear
D)
\[\frac{{{\pi }^{2}}}{4}\]
done
clear
View Answer play_arrow
An ambulance blowing a siren of frequency 700 Hz is travelling slowly towards a vertical reflecting wall with a speed of \[2\text{ }m{{s}^{-1}}.\] What is the reflected frequency as heard by the driver and the number of beats produced per second?(Velocity of sound\[=350\,m{{s}^{-1}}\])
A)
708 Hz, 8
done
clear
B)
692 Hz, 8
done
clear
C)
700 Hz, 0
done
clear
D)
696 Hz, 4
done
clear
View Answer play_arrow
The electric potential V (in volt) varies with x (in meter) according the relation\[V=5+4{{x}^{2}}.\] The force experienced by a negative charge of \[2\times {{10}^{-6}}C\]located at \[x=0.5\,m\]is:
A)
\[2\times {{10}^{-6}}N\]
done
clear
B)
\[4\times {{10}^{-6}}N\]
done
clear
C)
\[6\times {{10}^{-6}}N\]
done
clear
D)
\[8\times {{10}^{-6}}N\]
done
clear
View Answer play_arrow
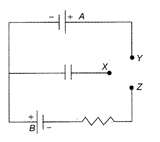
In the circuit shown the cells are ideal and of equal emfs, the capacitance of the capacitor is C and the resistance of the resistor is R. X is first joined to Y and then to Z. After a long time, the total heat produced in the resistor will be
A)
equal to the energy finally stored in the capacitor.
done
clear
B)
half of the energy finally stored in the capacitor.
done
clear
C)
twice the energy finally stored in the capacitor.
done
clear
D)
4 times the energy finally stored in the capacitor.
done
clear
View Answer play_arrow
In an ac generator, the number of turns in the coil is 2000 and the area of the coil is \[0.1\text{ }{{m}^{2}}.\] The coil is rotated at an angular frequency of \[200\,\text{rad}\,{{\text{s}}^{-1}}\] in a magnetic field of 0.2 T. What is the peak value of the induced emf?
A)
1000 V
done
clear
B)
2000V
done
clear
C)
4000V
done
clear
D)
8000V
done
clear
View Answer play_arrow
In an ac circuit the potential difference V and current\[I\]are given respectively by \[V=100\sin (100\,t)\]volt and\[I=100\sin (100t+\pi /3)\,mA.\]The power dissipated in the circuit will be
A)
\[~{{10}^{4}}W\]
done
clear
B)
10 W
done
clear
C)
2.5 W
done
clear
D)
5 W
done
clear
View Answer play_arrow
The angle of a prism is A and one of its refracting surfaces is silvered. Light rays falling at an angle of incidence 2A on the first surface return back through the same path after suffering reflection at the second (silvered) surface. The refractive of the material of the prism is
A)
2 sin A
done
clear
B)
\[2\cos A\]
done
clear
C)
\[\left( \frac{1}{2} \right)\cos A\]
done
clear
D)
\[\tan A\]
done
clear
View Answer play_arrow
A particle is projected from the ground at an angle of \[\theta \] with the horizontal with an initial speed of u. Time after which velocity vector of the projectile is perpendicular to the initial velocity is
A)
\[\frac{u}{(g\sin \theta )}\]
done
clear
B)
\[\frac{u}{(gcos\theta )}\]
done
clear
C)
\[\frac{2u}{(g\sin \theta )}\]
done
clear
D)
\[2u\,\tan \theta \]
done
clear
View Answer play_arrow
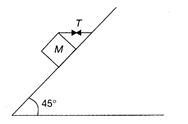
A block of mass 15 kg is resting on a rough inclined plane as shown in the figure. The block is tied by a horizontal sting, which has a tension of 50 N, The coefficient of friction between the surfaces of contact is \[(g=10\,m/{{s}^{2}})\]
A)
\[\frac{1}{2}\]
done
clear
B)
\[\frac{3}{4}\]
done
clear
C)
\[\frac{2}{3}\]
done
clear
D)
\[\frac{1}{4}\]
done
clear
View Answer play_arrow
A particle is acted upon by a force of constant magnitude, which is always perpendicular to the velocity of the particle, the motion of the particle takes plane in a plane. It follows that
A)
its velocity is constant.
done
clear
B)
its acceleration is constant.
done
clear
C)
its kinetic energy is constant.
done
clear
D)
it moves in a straight line.
done
clear
View Answer play_arrow
Consider elastic collision of a particle of mass m moving with a velocity u with another particle of the same mass at rest. After the collision the projectile and the struck particle move in directions making angles \[{{\theta }_{1}}\]and \[{{0}_{2}},\]respectively with the initial direction of motion. The sum of the angles \[{{\theta }_{1}}+{{\theta }_{2}},\] is
A)
\[45{}^\circ \]
done
clear
B)
\[90{}^\circ \]
done
clear
C)
\[135{}^\circ \]
done
clear
D)
\[180{}^\circ \]
done
clear
View Answer play_arrow
A solid cylinder of mass M and radius R rolls down an inclined plane without slipping. The speed of its center of mass when it reaches the bottom is (h is the height of inclined plane):
A)
\[\sqrt{2gh}\]
done
clear
B)
\[\sqrt{\frac{4}{3}gh}\]
done
clear
C)
\[\sqrt{\frac{3}{4}gh}\]
done
clear
D)
\[\sqrt{\frac{4g}{h}}\]
done
clear
View Answer play_arrow
A vertical meter stick of mass 400 gm is pivoted at the top and is then displaced through an angle \[60{}^\circ \]. The increase in its potential energy is
A)
2 J
done
clear
B)
\[3\,J\]
done
clear
C)
\[0\,J\]
done
clear
D)
\[1\,J\]
done
clear
View Answer play_arrow
A homogeneous cylinder of mass M and radius R is pulled on a horizontal plane by a horizontal force p acting through its mass center. Assuming rolling without slipping the angular acceleration of the cylinder is
A)
\[\frac{3F}{2MR}\]
done
clear
B)
\[\frac{2F}{3MR}\]
done
clear
C)
\[\frac{F}{2MR}\]
done
clear
D)
\[\frac{3F}{4MR}\]
done
clear
View Answer play_arrow
A radioactive substance is being produced at a constant rate 200 nuclei/s. The decay constant of the substance is \[1{{s}^{-1}}.\]After what time, the number of radioactive nuclei will become 100? Initially there are no nuclei present.
A)
\[1\,s\]
done
clear
B)
\[\frac{1}{\ell n\,2}s\]
done
clear
C)
\[(\ell n\,2)s\]
done
clear
D)
2 s
done
clear
View Answer play_arrow
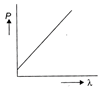
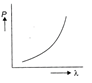
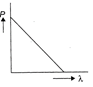
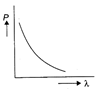
Which of the following figure represents the variation of particle momentum and associated de-Broglie wavelength?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
The velocity of the most energetic electron emitted from a metallic surface is doubled when the frequency v of incident radiation is doubled. The work function of this metal is
A)
\[\frac{2}{3}hv\]
done
clear
B)
\[\frac{hv}{2}\]
done
clear
C)
\[\frac{hv}{3}\]
done
clear
D)
0
done
clear
View Answer play_arrow
When a capillary tube of radius r is immersed in a liquid of density \[\rho ,\]the liquid rises to a height h in it. If m is the mass of the liquid in the capillary tube, the potential energy of this mass of the liquid in the tube is
A)
\[\frac{mgh}{4}\]
done
clear
B)
\[\frac{mgh}{2}\]
done
clear
C)
\[mgh\]
done
clear
D)
\[2mgh\]
done
clear
View Answer play_arrow
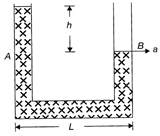
A liquid stands at the same level I arms A and B of a U-tube. If the U-tube is given a constant acceleration \[a(<g)\]towards the right as shown in the figure, the level of liquid in limb A rises to a height h above the level in limb B. If the length of the horizontal part of the U-tube is L, the value of h is given by
A)
\[\frac{La}{g}\]
done
clear
B)
\[\frac{Lg}{a}\]
done
clear
C)
\[L\left( 1+\frac{a}{g} \right)\]
done
clear
D)
\[L\left( 1-\frac{a}{g} \right)\]
done
clear
View Answer play_arrow
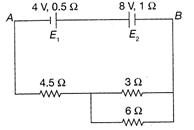
In the circuit shown in the figure the cells \[{{E}_{1}}\]and \[{{E}_{2}}\]have emfs. of 4 V and 8 V and internal resistance\[0.5\Omega \]and\[1\Omega ,\]respectively, Then the pd. Across \[{{E}_{1}}\]and\[{{E}_{2}}\] will be:
A)
\[4.25\,V;\,4.25\,V\]
done
clear
B)
\[3.75\,V;\,3.75\,V\]
done
clear
C)
\[4.25\,V;\,7.5\,V\]
done
clear
D)
\[3.75\,V;\,7.5\,V\]
done
clear
View Answer play_arrow
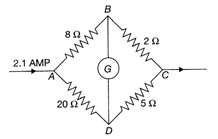
In the given figure when galvanomete shows on deflection. Current flowing through 50, resistance will be:
A)
0.5 A
done
clear
B)
0.6 A
done
clear
C)
1.5 A
done
clear
D)
2.0 A
done
clear
View Answer play_arrow
Three waves A, B, and C of frequencies 1600 kHz, 5 MHz, and 60 MHz, respectively are to be transmitted from one place to another. Which of the following is the most appropriate mode of communication:
A)
A is transmitted via space wave while B and C are transmitted via sky wave.
done
clear
B)
A is transmitted via ground wave, B via sky wave and C via space wave.
done
clear
C)
B and C are transmitted via ground wave while A is transmitted via sky wave.
done
clear
D)
B is transmitted via ground wave while A and C are transmitted via space wave.
done
clear
View Answer play_arrow
Which of the following is an insert gas?
A)
\[{{N}_{2}}\]
done
clear
B)
\[{{O}_{2}}\]
done
clear
C)
\[{{H}_{2}}\]
done
clear
D)
Argon
done
clear
View Answer play_arrow
If 0.1 M of a weak acid is taken and its percentage of degree of ionization is 1.34%, then its ionization constant will be
A)
\[0.182\times {{10}^{-5}}\]
done
clear
B)
\[1.79\times {{10}^{-5}}\]
done
clear
C)
\[0.8\times {{10}^{-5}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
If a substance with half-half 3 days is taken at other place in 12 days, then what amount of substance is left now?
A)
1/8
done
clear
B)
1/4
done
clear
C)
1/16
done
clear
D)
1/2
done
clear
View Answer play_arrow
To prepare a solution with concentration of 0.03 g/mL of \[AgN{{O}_{3}},\]what amount of \[AgN{{O}_{3}}\]should be added in 60 mL of solution?
A)
1.8 g
done
clear
B)
0.18 g
done
clear
C)
0.8 g
done
clear
D)
0.09 g
done
clear
View Answer play_arrow
How will you separate a solution (miscible) of benzene\[+\text{ }CHC{{l}_{3}}?\]
A)
Filtration
done
clear
B)
Sublimation
done
clear
C)
Distillation
done
clear
D)
Crystallization
done
clear
View Answer play_arrow
When alcohol reacts with concentrated \[{{H}_{2}}S{{O}_{4}},\]the intermediate compound formed is
A)
Carbonium ion
done
clear
B)
Alkyl hydrogen sulphate
done
clear
C)
Alkoxy ion
done
clear
D)
None of these
done
clear
View Answer play_arrow
According to the law of mass action rate of a chemical reaction is proportional to
A)
Concentration of reactants
done
clear
B)
Molar concentration of reactants
done
clear
C)
Molar concentration of products
done
clear
D)
Concentration of products
done
clear
View Answer play_arrow
In Hall's process the main reagent is mixed with
A)
\[~A1{{F}_{3}}\]
done
clear
B)
\[N{{a}_{3}}Al{{F}_{6}}\]
done
clear
C)
\[NaF\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
In the electrolysis of dilute \[{{H}_{2}}S{{O}_{4}}\]what is liberated at anode?
A)
\[S{{O}_{2}}\]
done
clear
B)
\[{{H}_{2}}\]
done
clear
C)
\[S{{O}_{4}}^{2-}\]
done
clear
D)
\[{{O}_{2}}\]
done
clear
View Answer play_arrow
A gas can be liquefied
A)
At its critical temperature
done
clear
B)
Above its critical temperature
done
clear
C)
Below its critical temperature
done
clear
D)
At any temperature
done
clear
View Answer play_arrow
Which of the following is hypnotic?
A)
Metaldehyde
done
clear
B)
Paraldehyde
done
clear
C)
Acetaldehyde
done
clear
D)
None of these
done
clear
View Answer play_arrow
By which of the following process permanent hardness of water can be removed?
A)
Washing soda
done
clear
B)
Soda lime
done
clear
C)
Sodium chloride
done
clear
D)
Sodium bicarbonate
done
clear
View Answer play_arrow
Vinegar obtained from sugarcane has
A)
\[C{{H}_{3}}COOH\]
done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}COOH\]
done
clear
C)
\[HCOOH\]
done
clear
D)
\[{{C}_{6}}{{H}_{5}}COOH\]
done
clear
View Answer play_arrow
What is the packet of energy called?
A)
Proton
done
clear
B)
Photon
done
clear
C)
Positron
done
clear
D)
Electron
done
clear
View Answer play_arrow
When an acid cell is charged, then
A)
Voltage of cell increases
done
clear
B)
Electrolyte of cell dilutes
done
clear
C)
Resistance of cell increases
done
clear
D)
None of these
done
clear
View Answer play_arrow
\[NaOH\] is prepared by the method
A)
Solvay process
done
clear
B)
Castner cell
done
clear
C)
Down cell
done
clear
D)
Castner-Kellner cell
done
clear
View Answer play_arrow
When toluene is treated with \[KMn{{O}_{4}}\]what is produced?
A)
Benzene
done
clear
B)
Benzaldehyde
done
clear
C)
Chlorobenzene
done
clear
D)
Benzoic acid
done
clear
View Answer play_arrow
Solder is an alloy of
A)
90% copper, 10% tin
done
clear
B)
30% lead, 70% tin
done
clear
C)
70% lead, 30% tin
done
clear
D)
80% lead, 20% tin
done
clear
View Answer play_arrow
Carbolic acid is
A)
\[{{C}_{6}}{{H}_{6}}\]
done
clear
B)
\[{{C}_{6}}{{H}_{5}}CHO\]
done
clear
C)
\[{{C}_{6}}{{H}_{5}}COOH\]
done
clear
D)
\[{{C}_{6}}{{H}_{5}}OH\]
done
clear
View Answer play_arrow
Alcohols are isomeric with
A)
Esters
done
clear
B)
Ethers
done
clear
C)
Acids
done
clear
D)
Aldehydes
done
clear
View Answer play_arrow
The group linkage present in fats is
A)
Peptide linkage
done
clear
B)
Ester linkage
done
clear
C)
Glycosidic linkage
done
clear
D)
None of these
done
clear
View Answer play_arrow
The group present in waxes are
A)
Acid group
done
clear
B)
Ester group
done
clear
C)
Alcoholic group
done
clear
D)
Ether group
done
clear
View Answer play_arrow
Which of the following is liquid at room temperature?
A)
\[C{{H}_{3}}I-66{{\,}^{o}}C,42{{\,}^{o}}C\]
done
clear
B)
\[C{{H}_{3}}F-115{{\,}^{o}}C,78{{\,}^{o}}C\]
done
clear
C)
\[{{C}_{2}}{{H}_{5}}Cl-94{{\,}^{o}}C,3{{\,}^{o}}C\]
done
clear
D)
\[{{C}_{2}}{{H}_{5}}Cl-139{{\,}^{o}}C,12{{\,}^{o}}C\]
done
clear
View Answer play_arrow
Which gas is liberated when\[A{{l}_{4}}C{{L}_{3}}\]is hydrolyzed?
A)
\[C{{H}_{4}}\]
done
clear
B)
\[C{{O}_{2}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
View Answer play_arrow
The only alcohol that can be prepared by the indirect hydration of alkene is
A)
Ethyl alcohol
done
clear
B)
Propyl alcohol
done
clear
C)
Isobutyl alcohol
done
clear
D)
Methyl alcohol
done
clear
View Answer play_arrow
Baking powder is
A)
\[NaHC{{O}_{3}}\]
done
clear
B)
\[N{{a}_{2}}C{{O}_{3}}\]
done
clear
C)
\[NaHC{{O}_{3}}.6{{H}_{2}}O\]
done
clear
D)
\[N{{a}_{2}}C{{O}_{3}}.10{{H}_{2}}O\]
done
clear
View Answer play_arrow
When washing soda is heated
A)
\[C{{O}_{2}}\] is released
done
clear
B)
Water vapor is released
done
clear
C)
\[C{{O}_{2}}\] is released
done
clear
D)
\[CO+C{{O}_{2}}\]is released
done
clear
View Answer play_arrow
Which of the following attacks glass
A)
\[HCl\]
done
clear
B)
\[HBr\]
done
clear
C)
\[HI\]
done
clear
D)
\[HF\]
done
clear
View Answer play_arrow
A colorless gas with the smell of rotten fish is
A)
\[S{{O}_{2}}\]
done
clear
B)
\[P{{H}_{3}}\]
done
clear
C)
\[{{H}_{2}}S\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
Salicylic acid is prepared form phenol by
A)
Reimer-Tiemann reaction
done
clear
B)
Kolbe electrolysis reaction
done
clear
C)
Kolbe's reaction
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Lucas test is done for
A)
Acids
done
clear
B)
Alcohols
done
clear
C)
Alkyl halides
done
clear
D)
Aldehydes
done
clear
View Answer play_arrow
Hydrogen can be fused to form helium at
A)
High temperature and high pressure
done
clear
B)
High temperature and low pressure
done
clear
C)
Low temperature and high pressure
done
clear
D)
Low temperature and low pressure
done
clear
View Answer play_arrow
When\[C{{O}_{2}}\]is bubbled through a solution of barium peroxide in water
A)
Carbonic acid is formed
done
clear
B)
\[{{O}_{2}}\]is released
done
clear
C)
\[{{H}_{2}}{{O}_{2}}\] is formed
done
clear
D)
No reaction occurs
done
clear
View Answer play_arrow
The most important ore of tin is
A)
Cassiterite
done
clear
B)
Cerussite
done
clear
C)
Cryolite
done
clear
D)
None of these
done
clear
View Answer play_arrow
Heating of ore in presence of air to remove sulphur impurities is called
A)
Smelting
done
clear
B)
Roasting
done
clear
C)
Calcination
done
clear
D)
None of these
done
clear
View Answer play_arrow
\[CaC{{O}_{3}}CaO+C{{O}_{2}}\]reaction in a line goes to completion because
A)
Backward reaction is very low
done
clear
B)
\[CaO\] does not react to \[C{{O}_{2}}\]to give\[CaC{{O}_{3}}\]
done
clear
C)
\[C{{O}_{2}}\]formed escapes out
done
clear
D)
None of the above
done
clear
View Answer play_arrow
If 30 mL of \[{{H}_{2}}\]and 20 mL of\[{{O}_{2}}\]reacts to from water, then what is left at the end of the reaction?
A)
10 mL of\[{{O}_{2}}\]
done
clear
B)
10 mL of\[{{H}_{2}}\]
done
clear
C)
5 mL of\[{{H}_{2}}\]
done
clear
D)
5 mL of \[{{O}_{2}}\]
done
clear
View Answer play_arrow
Which of the following is a highly corrosive salt?
A)
\[FeC{{l}_{2}}\]
done
clear
B)
\[HgC{{l}_{2}}\]
done
clear
C)
\[PbC{{l}_{2}}\]
done
clear
D)
\[H{{g}_{2}}C{{l}_{2}}\]
done
clear
View Answer play_arrow
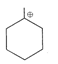
Which is the most stable Carbocation here
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Formula for tear gas is
A)
\[{{N}_{2}}O\]
done
clear
B)
\[CC{{l}_{3}}N{{O}_{2}}\]
done
clear
C)
\[COC{{l}_{2}}\]
done
clear
D)
None of these
done
clear
View Answer play_arrow
Which of the following is potassium ferricyanide?
A)
\[{{K}_{4}}[Fe{{(CN)}_{6}}]\]
done
clear
B)
\[{{K}_{3}}[Fe{{(CN)}_{6}}]\]
done
clear
C)
\[{{K}_{3}}[Fe{{(CN)}_{4}}]\]
done
clear
D)
\[{{K}_{3}}[Fe{{(CN)}_{3}}]\]
done
clear
View Answer play_arrow
Sodium nitroprusside when added to an alkaline solution of sulphide ions produce a
A)
Red colouration
done
clear
B)
Blue colouration
done
clear
C)
Purple colouration
done
clear
D)
Brown colouration
done
clear
View Answer play_arrow
The product obtained on reaction of \[{{C}_{2}}{{H}_{5}}Cl\]with hydrogen over palladium is
A)
\[{{C}_{2}}{{H}_{4}}\]
done
clear
B)
\[{{C}_{3}}{{H}_{8}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
D)
\[{{C}_{4}}{{H}_{10}}\]
done
clear
View Answer play_arrow
A solution has pH = 5, it is diluted 100 times, then it will become
A)
Neutral
done
clear
B)
More acidic
done
clear
C)
Basic
done
clear
D)
Unaffected
done
clear
View Answer play_arrow
Ketones react with Mg-Hg over water and gives
A)
Alcohols
done
clear
B)
Pinacols
done
clear
C)
Pinacolone
done
clear
D)
None of these
done
clear
View Answer play_arrow
\[x\]is heated with soda lime and gives ethane. What is\[x?\]
A)
Methanoic acid
done
clear
B)
Ethanoic acid
done
clear
C)
Propanoic acid
done
clear
D)
Either (a) or (c)
done
clear
View Answer play_arrow
Which is used to produce smoke screens?
A)
Calcium phosphide
done
clear
B)
Sodium carbonate
done
clear
C)
Zinc phosphide
done
clear
D)
Zinc sulphide
done
clear
View Answer play_arrow
The conversion of maltose to glucose is possible by the enzyme
A)
Zymase
done
clear
B)
Lactase
done
clear
C)
Maltase
done
clear
D)
Diastase.
done
clear
View Answer play_arrow
The product obtained on fusion of \[BaS{{O}_{4}}\]and \[N{{a}_{2}}C{{O}_{3}}\]is
A)
\[BaO\]
done
clear
B)
\[~BaC{{O}_{3}}\]
done
clear
C)
\[~BaHS{{O}_{4}}\]
done
clear
D)
\[Ba{{(OH)}_{2}}\]
done
clear
View Answer play_arrow
0.5 M of \[{{H}_{2}}S{{O}_{4}}\]is diluted from 1 litre to 10 litre, normality of the resulting solution is
A)
1 N
done
clear
B)
10 N
done
clear
C)
11 N
done
clear
D)
0.1 N
done
clear
View Answer play_arrow
Which of the following is incorrect about keys?
A)
Key is a taxonomical aid used for identification of plants and animals based on the similarities and dissimilarities.
done
clear
B)
Separate taxonomic keys are required for each taxonomic category such as family, genus and species for identification purposes.
done
clear
C)
Keys are generally analytical in nature.
done
clear
D)
Key is an another taxonomical aid used for nomenclature of plants and animals based on the similarities and dissimilarities.
done
clear
View Answer play_arrow
The molecular weight of RNA of viroid is
A)
High
done
clear
B)
Low
done
clear
C)
Very high
done
clear
D)
Any of the above
done
clear
View Answer play_arrow
Find the number of following fungus that belongs to the class Ascomycetes (count total number).
Rhizopus, Penicillium, Yeast, Mucor, Agaricus, Puccinia, Albugo, Claviceps, Neurospora, Alter- naria, Trichoderma, Aspergillus, Ustilago, Morels, Buffles, Colletotrichum, Toadstool.
A)
5
done
clear
B)
7
done
clear
C)
9
done
clear
D)
10
done
clear
View Answer play_arrow
Lichens are
A)
Pollution indicators
done
clear
B)
Symbiotic association between algae and fungus
done
clear
C)
Pioneer species in primary succession on rocks
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Which kingdom of Whittaker is not well-defined?
A)
Monera
done
clear
B)
Protista
done
clear
C)
Fungi
done
clear
D)
Animalia
done
clear
View Answer play_arrow
Find out the total number of false statements.
(A) Cyanpbacteria have chlorophyll 'a' like green plants. (B) Bacteria which oxidize various inorganic substances such as nitrates and ammonia and use the released energy for ATP production are chemosynthesis autotrophic bacteria. (C) Heterotrophic bacteria are less in abundance in nature. (D) Majority of the heterotrophic bacteria are decomposers. (E) Chemosynthetic autotrophic bacteria play an important role in recycling of nutrients like nitrogen, phosphorus, iron and sulphur.
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
Which is not a correct matching?
A)
Rust \[\to \] Puccinia
done
clear
B)
Yeast \[\to \] Bread and Beer
done
clear
C)
Chrysophyte\[\to \] Diatomaceous earth
done
clear
D)
Penicillium\[\to \]Red tide
done
clear
View Answer play_arrow
Which alga shows diplontic life cycle?
A)
Chara
done
clear
B)
Fucus
done
clear
C)
Ectocarpus
done
clear
D)
Polysiphonia
done
clear
View Answer play_arrow
In gymnosperms, the mycorrhizal association is found in
A)
Cycas
done
clear
B)
Pinus
done
clear
C)
Cedrus
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Bilaterally symmetrical, triploblastic, segmented, coelomate and covered by chitinous exoskeleton are features of
A)
Annelida
done
clear
B)
Vertebrata
done
clear
C)
Amphibia
done
clear
D)
Arthropoda
done
clear
View Answer play_arrow
Select the difference which is wrongly written.
A)
(Cartilaginous fish)
(Bony fishes)
Operculum absent
Operculum present
done
clear
B)
(Cartilaginous fish)
(Bony fishes)
Internal fertilization
External fertilization
done
clear
C)
(Cartilaginous fish)
(Bony fishes)
Possess 5 to 7 pair
of gills Posses 4 pair of gills
done
clear
D)
(Cartilaginous fish)
(Bony fishes)
Mostly oviparous
Mostly viviparous
done
clear
View Answer play_arrow
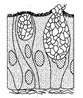
The excretory organ present in organism given in the figure is
A)
Rennet cells
done
clear
B)
Protonephridia or flame cells
done
clear
C)
Malpighian tubule
done
clear
D)
Organ of Bojanus
done
clear
View Answer play_arrow
Diadelphous condition is seen in all except
A)
Soya bean
done
clear
B)
Lupin
done
clear
C)
Brassica rapa
done
clear
D)
China rose
done
clear
View Answer play_arrow
Floral formula of Brassicaceae is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Select the incorrect matching:
A)
Indigofera - Sepals five (gamosepalous), corolla (vexillary aestivation)
done
clear
B)
Brinjal - Sepals five united (persistent, valvate aestivation), Petals five united (valvate aestivation)
done
clear
C)
Asparagus - Sepals three often united into tube (valvate aestivation)
done
clear
D)
Colchicine - Perianth present tepals six (valvate aestivation)
done
clear
View Answer play_arrow
Pinnately compound leaf is found in
A)
Lemon
done
clear
B)
Bombax
done
clear
C)
Desmodium
done
clear
D)
Neem
done
clear
View Answer play_arrow
A placentation in which ovules are born on central axis and the septa are absent is found in
A)
Argemone and mustard
done
clear
B)
Dianthus and primrose
done
clear
C)
Rose and tomato
done
clear
D)
Pea
done
clear
View Answer play_arrow
External protective tissues of plants (of dicot stem are)
A)
Cork and pericycle
done
clear
B)
Cortex and epidermis
done
clear
C)
Pericycle and cortex
done
clear
D)
Epidermis and cork
done
clear
View Answer play_arrow
External genitalia in cockroach is represented by
A)
Male gonapophysis
done
clear
B)
Phallomere
done
clear
C)
Ejaculatory duct
done
clear
D)
Both (a) and (B)
done
clear
View Answer play_arrow
The four sketches (A, B, C and D) given below, represent four different types of animal tissues. Which one of these is correctly identified in the options given, along with its correct location and function?
A)
Tissue Location Function Collagen fibres Cartilage Attach skeletal muscles to bones
done
clear
B)
Tissue Location Function Smooth muscle tissue Heart Heart contraction
done
clear
C)
Tissue Location Function Columnar epithelium Nephron Secretion and absorption
done
clear
D)
Tissue Location Function Glandular epithelium Intestine Secretion
done
clear
View Answer play_arrow
How is new mitochondria formed by the pre-existing ones?
A)
Mitosis
done
clear
B)
Fission
done
clear
C)
Conjugation
done
clear
D)
Budding
done
clear
View Answer play_arrow
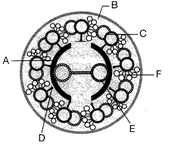
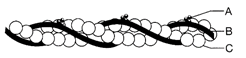
Identify A, B, C, D, E and F given in the figure.
A)
A: interdoublet Bridge, B: Peripheral microtubules (doublets), C: Central microtubule, D: Plasma membrane, E: Central bridge, F: Radial spoke.
done
clear
B)
A: Central sheath, B: Radial spoke, C: Interdoublet bridge, D: Central microtubule, E: Plasma membrane, F: Peripheral microtubules (doublets).
done
clear
C)
A: Central sheath, B: Plasma membrane, C: Peripheral microtubules (doublets), D: Radial spoke, E: Central microtubule, F: Interdoublet bridge.
done
clear
D)
A: Plasma membrane, B: Radial spoke, C: Peripheral microtubules (doublets), D: Central sheath, E: Central microtubule, F: Interdoublet bridge.
done
clear
View Answer play_arrow
A elaborate network of filamentous proteinaceous structures present in the cytoplasm is collectively known as
A)
Cilia
done
clear
B)
Flagella
done
clear
C)
Cytoskeleton
done
clear
D)
ER
done
clear
View Answer play_arrow
The site of active ribosomal RNA synthesis
A)
Nucleolus
done
clear
B)
Mitochondria
done
clear
C)
Cytoplasm
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Water soluble pigments found in plant cell vacuoles are
A)
Xanthophylls
done
clear
B)
Chlorophylls
done
clear
C)
Carotenoids
done
clear
D)
Anthocyanins
done
clear
View Answer play_arrow
The most abundant protein in the whole biosphere is
A)
RuBisCO
done
clear
B)
Collagen
done
clear
C)
Elastin
done
clear
D)
Albumin
done
clear
View Answer play_arrow
Which of the following is a ketogenic amino acid?
A)
Alanine
done
clear
B)
Glycine
done
clear
C)
Methionine
done
clear
D)
Leucine
done
clear
View Answer play_arrow
Most of the cell organelle duplicate during
A)
\[{{G}_{1}}-\]phase
done
clear
B)
S-phase
done
clear
C)
\[{{G}_{2}}-\] phase
done
clear
D)
M-phase
done
clear
View Answer play_arrow
Number of chromosomes present in pollen grains six. What shall be their number in leaf cells?
A)
12
done
clear
B)
24
done
clear
C)
6
done
clear
D)
3
done
clear
View Answer play_arrow
Which of the following is the longest phase of meiosis?
A)
Prophase I
done
clear
B)
Anaphase I
done
clear
C)
Prophase II
done
clear
D)
Metaphase II
done
clear
View Answer play_arrow
Choose the total number of correct statements from the following:
(A) Some channels in the membrane are always open (B) Porins allow the passing of molecule of size up to small protein. (C) Water channel made up of eight different type of aquaporins. (D) Facilitated diffusion is very specific.
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
Select the true and false statement:
(A) Morphological changes which are indicative of certain element deficiencies are known a deficiency symptoms. (B) For activity, the mobilized element deficiency symptoms first appear in older tissues. (C) When an element is relatively immobile, the deficiency symptom first appears in younge tissue. (D) The mineral nutrition study of plant has great significance to agriculture and horticulture.
A)
T T F F
done
clear
B)
T T T T
done
clear
C)
T F F T
done
clear
D)
F F F T
done
clear
View Answer play_arrow
Find out the similarity between \[{{C}_{3}}\] and \[{{C}_{4}}\] pathway
A)
Both are equally efficient
done
clear
B)
Organic acid is formed as the first product \[C{{O}_{2}}\]fixation
done
clear
C)
Both require one type of cell to occur
done
clear
D)
Both takes place in all the plants
done
clear
View Answer play_arrow
\[{{C}_{4}}\]plant is characterized by
A)
High tolerance to temperature
done
clear
B)
Lacks process of photorespiration
done
clear
C)
Greater productivity of biomass
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Light saturation occurs at __ per cent of the fall
A)
2
done
clear
B)
5
done
clear
C)
10
done
clear
D)
20
done
clear
View Answer play_arrow
How many of the following PGR helps to overcome apical dominance? Auxins, Gibberellins, Cytokinins, Ethylene
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
Bile is secreted by
A)
Glisson's Capsule
done
clear
B)
Gall bladder
done
clear
C)
Hepatic Cells
done
clear
D)
Kupfier cells
done
clear
View Answer play_arrow
Protection of mucosal epithelium from highly concentrated \[HCl\] in stomach is performed by
(A) \[HCO_{3}^{-}\]ion (B) Mucus (C) Pepsin (D) Intrinsic factor
A)
A and C only
done
clear
B)
B only
done
clear
C)
A and B only
done
clear
D)
A, B and C only
done
clear
View Answer play_arrow
Arrange the following in the order of increasing volume.
(1) Tidal volume (2) Residual volume (3) Expiratory reserve volume (4) Inspiratory reserve volume
A)
1 < 2 < 3 < 4
done
clear
B)
1 < 4 < 3 < 2
done
clear
C)
1 < 3 < 2 < 4
done
clear
D)
1 < 4 < 2 < 3
done
clear
View Answer play_arrow
What is correct statement about human respiration?
A)
About 90 per cent of \[C{{O}_{2}}\]is carried by haemoglobin as carbaminohaemoglobin.
done
clear
B)
Neural signals from pneumotaxic centre of pons can increase the duration of inspiration.
done
clear
C)
Workers in grinding and stone breaking industries may suffer from lung fibrosis.
done
clear
D)
Cigarette smoking leads to the inflammation of bronchi.
done
clear
View Answer play_arrow
When ventricular systole occurs
A)
Atrial diastole coincides
done
clear
B)
Tricuspid and bicuspid valves close
done
clear
C)
Semilunar valves guarding the pulmonary artery and aorta are forced to open.
done
clear
D)
All of the above
done
clear
View Answer play_arrow
In mammals, which blood vessel would normally carry the largest amount of urea?
A)
Renal Vein
done
clear
B)
Dorsal Aorta
done
clear
C)
Hepatic Vein
done
clear
D)
Hepatic Portal Vein
done
clear
View Answer play_arrow
Following is the figure of actin (thin) filaments. Identify A, B and C.
A)
A: Tropomyosin, B: Troponin, C: F actin
done
clear
B)
A: Tropomyosin, B: Myosin, C: F Tropomyosin
done
clear
C)
A: Troponin, B: Tropomyosin, C: Myosin
done
clear
D)
A: Troponin, B: Tropomyosin, C: F actin
done
clear
View Answer play_arrow
Which of the following occurs due to reflex action?
A)
Vomiting
done
clear
B)
Sneezing
done
clear
C)
Coughing
done
clear
D)
All of the above
done
clear
View Answer play_arrow
The features of cretinism includes
A)
Stunted growth
done
clear
B)
Mental retardation and low IQ
done
clear
C)
Abnormal skin and deaf mutism
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Select the correct statements from the following.
(A) Melatonin influences the menstrual cycle and our defence capability. (B) In adult women, hypothyroidism may cause menstrual cycle to become irregular. (C) Protein hormone secreted by thyroid, TCT (Thyrocalcitonin) regulates the blood calcium level. (D) Maintenance of water and the electrolytic balance is also influenced by thyroid hormone. (E) Oxytocin causes ejection of milk from the mammary gland.
A)
A, B, C only
done
clear
B)
A, B, C, E only
done
clear
C)
All except D
done
clear
D)
All statements are correct
done
clear
View Answer play_arrow
Which of the following is the effect of Cortisol?
A)
Anti-inflammatory
done
clear
B)
Immunosuppressant
done
clear
C)
Increased RBC production
done
clear
D)
All of the above
done
clear
View Answer play_arrow
The microsporangium cells which possess denser cytoplasm and have more than one nucleus is the characteristic of
A)
Middle layers
done
clear
B)
Tapetum
done
clear
C)
Endothecium
done
clear
D)
Epidermis
done
clear
View Answer play_arrow
Pollen grains
(A) Represent gametophytic phase of plant (B) Can cause severe allergies like asthma bronchitis (C) Are rich in nutrient (D) Are used as food supplements (E) Are available in form of tables and syrups in the market of western countries
A)
Only A is correct
done
clear
B)
All are correct
done
clear
C)
All are wrong
done
clear
D)
Only A, B and C are correct
done
clear
View Answer play_arrow
Which one of the following is incorrect?
(A) Parthenium or carrot gases causes pollen allergy. (B) Vegetative cell of pollen has abundant food reserve. (C) All pollen's cause severe allergies and bronchial afflictions. (D) Sporopollenin is the most resistant organic matter known.
A)
All of the above
done
clear
B)
A
done
clear
C)
B
done
clear
D)
C
done
clear
View Answer play_arrow
After three meiotic divisions in the functional mega- spore, the gametophyte (embryo sac) has how many cells?
A)
7 cells
done
clear
B)
4 cells
done
clear
C)
5 cells
done
clear
D)
8 cells
done
clear
View Answer play_arrow
In water pollinated species
(A) Pollen grains are long and ribbon-like (B) All pollen grains are protected from getting wet (C) Flowers are not very colourful (D) They do not produce nectar
A)
Only A and B are correct
done
clear
B)
Only A and B are wrong
done
clear
C)
All are correct
done
clear
D)
All are wrong
done
clear
View Answer play_arrow
The first menstruation which begins at puberty is called
A)
Menstrual cycle
done
clear
B)
Menarche
done
clear
C)
Oogenesis
done
clear
D)
Ovulation
done
clear
View Answer play_arrow
The tertiary follicle in ovary is characterized by the presence of
A)
Fundus
done
clear
B)
Antrum
done
clear
C)
Vacuole
done
clear
D)
Cavity
done
clear
View Answer play_arrow
The technique called Gamete Intra Fallopian Transfer (GIFT) is recommended for those females
A)
Who cannot produce an ovum.
done
clear
B)
Who cannot retain the foetus inside uterus.
done
clear
C)
Whose cervical canal is too narrow to allow passage for the sperms.
done
clear
D)
Who cannot provide suitable environment for fertilization.
done
clear
View Answer play_arrow
The following are the indicative measures, which should be considered for the better improvement of reproductive health of society. Choose the correct combination.
(i) Better awareness about sex related problems (ii) Better detection and cure of STD's (iii) Better post-natal care (iv) Increased number of couple with large families
A)
i and iv only
done
clear
B)
ii, iii and iv only
done
clear
C)
i and ii only
done
clear
D)
i, ii and iii only
done
clear
View Answer play_arrow
Match the following
Column - I Column - II (A) Haemophilia (1) Board palm with characteristic palm creased (B) Down's syndrome (2) Delayed clotting of blood (C) Klinefelter's syndrome (3) Male feminine development (D) Turner's Syndrome (4) Rudimentary ovaries
A)
A: 1, B: 3, C: 2, D: 4
done
clear
B)
A: 2, B: 1, C: 3, D: 4
done
clear
C)
A: 4, B: 2, C: 1, D: 3
done
clear
D)
A: 1, B: 2, C: 3, D: 4
done
clear
View Answer play_arrow
In the\[{{F}_{2}}\] generation of a Mendelian dihybrid cross, the number of phenotypes and genotypes are
A)
Phenotypes - 4; genotypes - 16
done
clear
B)
Phenotypes - 9; genotypes - 4
done
clear
C)
Phenotypes - 4; genotypes - 8
done
clear
D)
Phenotypes - 4; genotypes - 9
done
clear
View Answer play_arrow
If yellow body, white eyed drosophila is crossed with wild brown body red eyes drosophila. Then what would be the frequency of recombinants in \[{{F}_{1}}\]generation?
A)
100 %
done
clear
B)
1.3 %
done
clear
C)
98.7 %
done
clear
D)
0 %
done
clear
View Answer play_arrow
Monosomy and trisomy can be represented as
A)
2n + 1, 2n + 3
done
clear
B)
2n - 1, 2n - 2
done
clear
C)
2n, 2n + 1
done
clear
D)
2n - 1, 2n + 1
done
clear
View Answer play_arrow
Deletion or insertion of base pairs of DNA causes
A)
Point mutation
done
clear
B)
Frame-shift mutation
done
clear
C)
Transversion
done
clear
D)
All of the above
done
clear
View Answer play_arrow
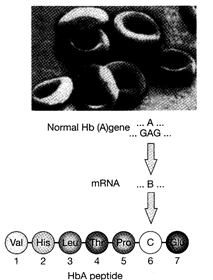
Identify A, B and C in the following figure?
A)
A: CTC, B: GAG, C: Glu
done
clear
B)
A: GAG, B: CTC, C: Pro
done
clear
C)
A: CTC, B: GAG, C: Thr
done
clear
D)
A: CTC, B: GAG, C: Leu
done
clear
View Answer play_arrow
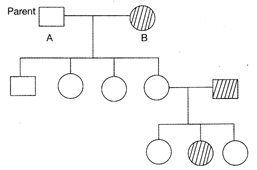
Give pedigree shows the inheritance of autosomal recessive gene. What is the genotype of given parents?
A)
AA, aa
done
clear
B)
Aa, AA
done
clear
C)
aa, aa
done
clear
D)
Aa, Aa
done
clear
View Answer play_arrow
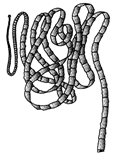
'Beads-on-string' structure can be viewed under
A)
Light microscope
done
clear
B)
Compound microscope
done
clear
C)
Confocal Laser microscope
done
clear
D)
Electron microscope
done
clear
View Answer play_arrow
Typically a nucleosome consists of bow many pairs?
A)
190
done
clear
B)
200
done
clear
C)
300
done
clear
D)
310
done
clear
View Answer play_arrow
In eukaryotes, RNA polymerase III is responsible for the synthesis of
A)
28 sRNA, 18 sRNA and 5.8 sRNA
done
clear
B)
tRNA, hnRNA, rRNA
done
clear
C)
tRNA, 5 sRNA, snRNA
done
clear
D)
hnRNA, tRNA, rRNA
done
clear
View Answer play_arrow
Dryopithecus and Ramapithecus existed.???? milion years ago.
A)
5
done
clear
B)
10
done
clear
C)
15
done
clear
D)
25
done
clear
View Answer play_arrow
Match the column:
(1) Pre-historic cave art 10,000 years back (2) Agriculture 18,000 years back (3) Neanderthal man 1.5 million years ago (4) Homo erectus 40,000 to 1,00,000 years back
A)
1:A, 2:C, 3:B, 4:D
done
clear
B)
1:D, 2:B, 3:C, 4:A
done
clear
C)
1:B, 2:A, 3:D, 4:C
done
clear
D)
1:C, 2:A, 3:D, 4:B
done
clear
View Answer play_arrow
This diagram shows
A)
Adaptive radiation
done
clear
B)
Genetic drift
done
clear
C)
Founder effect
done
clear
D)
Mutation
done
clear
View Answer play_arrow
Pneumonia is spread by
A)
Inhaling droplets/aerosols
done
clear
B)
Mosquito
done
clear
C)
Reusing syringes
done
clear
D)
Genetic disease
done
clear
View Answer play_arrow
Ringworm is caused by
A)
Round worm
done
clear
B)
Bacteria
done
clear
C)
Fungi
done
clear
D)
virus
done
clear
View Answer play_arrow
An antibody is represented as
A)
\[{{H}_{1}}{{L}_{2}}\]
done
clear
B)
\[{{H}_{2}}{{L}_{2}}\]
done
clear
C)
\[{{H}_{4}}{{L}_{1}}\]
done
clear
D)
\[{{H}_{4}}{{L}_{4}}\]
done
clear
View Answer play_arrow
When a quick immune response is required.
A)
Directly inject weakened pathogen at time of emergency
done
clear
B)
Directly inject preformed antigens
done
clear
C)
Directly inject preformed antibodies
done
clear
D)
Directly inject immunodepressants
done
clear
View Answer play_arrow
Select the correct matching
A)
Plant breeding: Purposeful manipulation of plant species in order to create desired plant types that are better suited for cultivation gives better yield and are disease resistant.
done
clear
B)
Gene bank: The entire collection of plant/seeds having all the diverse alleles for all genes in a given crop.
done
clear
C)
Fishery: Industry devoted to the catching, processing or selling of fish only.
done
clear
D)
MOET: It is used for herd improvement. In this method, a cow is administered hormones, with LH-like activity, which leads to super-ovulation (i.e., produce 6 to 8 eggs per cycle).
done
clear
View Answer play_arrow
Baculovirus belongs to the genes
A)
Retro Virus
done
clear
B)
Nucleohedero Virus
done
clear
C)
Nucleopolyhedro Virus
done
clear
D)
Polyhedron Virus
done
clear
View Answer play_arrow
The way to introduce alien DNA into host cell includes
A)
Disarmed pathogens
done
clear
B)
Biolistics or gene gun
done
clear
C)
Micro-injection
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Select the incorrect matching:
A)
pBR322 E. coli cloning vector
done
clear
B)
EcoRl, ClaI, - Hindlll Restriction enzyme
done
clear
C)
ROP Protein involved in the replication of the plasmid
done
clear
D)
PCR Technique in which multiple copies of the gene (or DNA) of interest in synthesized in vitro
done
clear
View Answer play_arrow
Restriction enzymes of E. coli are
A)
HindIII
done
clear
B)
Barn
done
clear
C)
EcoRI, EcoRll
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Select the correct statement from the following:
A)
Most Bt-toxins are insect group non-specific.
done
clear
B)
Bt-toxin gene is coded by the name 'CRY?.
done
clear
C)
Genes Cry 1 Ac and Cry2Ab forms protein that control cotton bollworms.
done
clear
D)
CrylAa gene forms protein that control corn borer.
done
clear
View Answer play_arrow
Golden rice is
A)
A variety of rice grown along the yellow river in China.
done
clear
B)
Long stored rice having yellow colour tint.
done
clear
C)
A transgenic rice having gene for (3-Carotene.
done
clear
D)
Wild variety of rice with yellow coloured grains.
done
clear
View Answer play_arrow
Carrying capacity K means _____
A)
Organism's capability of maximum reproduction.
done
clear
B)
Nature's limit for supporting maximum growth of a species.
done
clear
C)
Nature's limit for supporting maximum number of species.
done
clear
D)
Organism's capability to withstand environmental odds.
done
clear
View Answer play_arrow
Who showed that five closely related species of warblers living on the same tree were able to avoid competition and coexist due to behavioral differences in their forging activities?
A)
Connell
done
clear
B)
Me Arthur
done
clear
C)
Gause
done
clear
D)
Darwin
done
clear
View Answer play_arrow
Find out the incorrect statement from the following.
A)
In brood parasitism (of Koel and Crow), the eggs of parasitic bird have evolved to resemble the host's egg in size and colour to reduce the chance of the host bird detecting the foreign eggs and ejecting them from the host.
done
clear
B)
Majority of parasite harming the host.
done
clear
C)
Many parasites have evolved to be host specific in such a way that both host and the parasite tend to co-evolve.
done
clear
D)
Monarch butterfly require chemical by feeding on a poisonous weed in its adult stage, that chemical makes it highly distasteful to the birds.
done
clear
View Answer play_arrow
Which of the following represent simple GFC (Grazing Food Chain)?
A)
Grass \[\to \]Goat \[\to \]Man
done
clear
B)
Goat\[\to \]Grass \[\to \]Man
done
clear
C)
Detritus\[\to \]Fungi\[\to \]Man
done
clear
D)
Fungi\[\to \] Detritus \[\to \] Grass
done
clear
View Answer play_arrow
Nutrient cycling is known as
A)
Biogeochemical cycle
done
clear
B)
Calvin cycle
done
clear
C)
Hatch-Slack pathway
done
clear
D)
Krebs cycle
done
clear
View Answer play_arrow
Match the Column-I (Recently extinct animals) with Column-II (Places from where they are extinct).
Column-I Column-II (A) Dodo (1) Russia (B) Quagga (2) Australia (C) Thylacine (3) Africa (D) Steller's sea cow (4) Mauritius
A)
A: 1, B: 2, C: 3, D: 4
done
clear
B)
A: 4, B: 3, C: 2, D: 1
done
clear
C)
A: 4, B: 2, C: 3, D: 1
done
clear
D)
A: 4, B: 1, C: 2, D: 3
done
clear
View Answer play_arrow
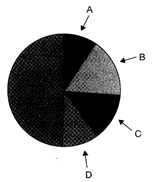
Following chart representing the proportionate number of species of major taxa of vertebrates, Identify A, B, C and D in following pie chart.
A)
A: Mammals, B: Birds, C: Reptile D: Amphibians
done
clear
B)
A: Amphibians, B: Birds, C: Mammal D: Reptiles
done
clear
C)
A: Reptiles, B: Amphibians, C: Bird D: mammals
done
clear
D)
A: Mammals, B: Reptiles, C: Birds, D: Amphibians
done
clear
View Answer play_arrow
What is the contribution of methane gas to the total global warming?
A)
60%
done
clear
B)
20%
done
clear
C)
14%
done
clear
D)
6%
done
clear
View Answer play_arrow
Match the column
Column-I Column-II (A) DV (1) Dobson unit (B) CFCs (2) Chlorofluorocarbons (C) BOD (3) Biological Oxygen Demand (D) PIL (4) Public Interest Litigation (E) CNG (5) Compressed Natural Gas
A)
A: 1, B: 3, C: 2, D: 4, E: 5
done
clear
B)
A: 4, B: 3, C: 2, D: 1, E: 5
done
clear
C)
A: 1, B: 2, C: 3, D: 4, E: 5
done
clear
D)
A: 1, B: 2, C: 4, D: 3, E: 5
done
clear
View Answer play_arrow
Rachel Carson's famous book 'Silent Spring? is related to
A)
Pesticide pollution
done
clear
B)
Noise Pollution
done
clear
C)
Population explosion
done
clear
D)
Ecosystem management
done
clear
View Answer play_arrow