If the error in the measurement of radius of a sphere is 2%, then the error in the determination of volume of the sphere will be
A)
4%
done
clear
B)
6%
done
clear
C)
8%
done
clear
D)
2%
done
clear
View Answer play_arrow
A body falls from a height h = 200 m (at New Delhi). The ratio of distance travelled in each \[2\sec \] during\[t=0\] to\[t=6\] seconds of the journey is
A)
1 : 4 : 9
done
clear
B)
1 : 2 : 4
done
clear
C)
1 : 3 : 5
done
clear
D)
1 : 2 : 3
done
clear
View Answer play_arrow
If \[\left| \vec{A}\times \vec{B} \right|=\sqrt{3}\vec{A}.\vec{B},\]then the value of \[\left| \vec{A}+\vec{B} \right|\]is:
A)
\[{{({{A}^{2}}+{{B}^{2}}+AB)}^{1/2}}\]
done
clear
B)
\[{{\left( {{A}^{2}}+{{B}^{2}}+\frac{AB}{\sqrt{3}} \right)}^{1/2}}\]
done
clear
C)
\[A+B\]
done
clear
D)
\[{{\left( {{A}^{2}}+{{B}^{2}}+\sqrt{3}AB \right)}^{1/2}}\]
done
clear
View Answer play_arrow
A man weighs 80 kg. He stands on a weighing scale in a lift which is moving upwards with a uniform acceleration of\[5m/{{s}^{2}}\]. What would be the reading on the scale?\[(g=10\,m/{{s}^{2}})\]
A)
400 N
done
clear
B)
800 N
done
clear
C)
1200 N
done
clear
D)
Zero
done
clear
View Answer play_arrow
A stationary particle explodes into two particles of a masses\[{{m}_{1}}\]and\[{{m}_{2}}\]which move in opposite directions with velocities \[{{v}_{1}}\]and \[{{v}_{2}}.\]The ratio of their kinetic energies\[{{E}_{1}}/{{E}_{2}}\] is
A)
\[{{m}_{1}}/{{m}_{2}}\]
done
clear
B)
1
done
clear
C)
\[{{m}_{1}}{{v}_{2}}/{{m}_{2}}{{v}_{1}}\]
done
clear
D)
\[{{m}_{2}}/{{m}_{1}}\]
done
clear
View Answer play_arrow
A solid cylinder of mass M and radius R rolls without slipping down an inclined plane of length \[L\] and height h. What is the speed of its centre of mass when the cylinder reaches its bottom
A)
\[\sqrt{\frac{3}{4}gh}\]
done
clear
B)
\[\sqrt{\frac{4}{3}gh}\]
done
clear
C)
\[\sqrt{4\,gh}\]
done
clear
D)
\[\sqrt{2\,gh}\]
done
clear
View Answer play_arrow
A round disc of moment of inertia\[{{I}_{2}}\]about its axis perpendicular to its plane and passing through its centre is placed over another disc of moment of inertia \[{{I}_{1}}\] rotating with an angular velocity \[\omega \]about the same axis. The final angular velocity of the combination of discs is
A)
\[\frac{{{I}_{2}}\omega }{{{I}_{1}}+{{I}_{2}}}\]
done
clear
B)
\[\omega \]
done
clear
C)
\[\frac{{{I}_{1}}\omega }{{{I}_{1}}+{{I}_{2}}}\]
done
clear
D)
\[\frac{({{I}_{1}}+{{I}_{2}})\omega }{{{I}_{1}}}\]
done
clear
View Answer play_arrow
If the density of a small planet is the same as that of earth, while the radius of the planet is 0.2 times that of the earth, the gravitational acceleration on the surface of that planet is:
A)
0.2 g
done
clear
B)
0.4 g
done
clear
C)
2 g
done
clear
D)
4 g
done
clear
View Answer play_arrow
The Earth is assumed to be a sphere of radius R. A platform is arranged at a height R from the surface of the earth. The escape velocity of a body from this platform is \[f{{v}_{e}},\]where \[{{v}_{e}}\]is escape velocity from the surface of the earth is. The value of\[f,\]is
A)
\[\sqrt{2}\]
done
clear
B)
\[\frac{1}{\sqrt{2}}\]
done
clear
C)
\[\frac{1}{3}\]
done
clear
D)
\[\frac{1}{2}\]
done
clear
View Answer play_arrow
A wire is stretched by 0.01 m by a certain force F. Another wire of the same material whose diameter and length are double to the original wire is stretched by the same force. Then its elongation will be
A)
0.005 m
done
clear
B)
0.01 m
done
clear
C)
0.02 m
done
clear
D)
0.002 m
done
clear
View Answer play_arrow
An iceberg of density\[900\text{ }kg/{{m}^{3}}\]is floating in water of density\[1000\text{ }kg/{{m}^{3}}.\] The percentage of volume of ice cube outside the water is
A)
20%
done
clear
B)
35%
done
clear
C)
10%
done
clear
D)
25%
done
clear
View Answer play_arrow
A cylindrical rod having temperature\[{{T}_{1}}\]and \[{{T}_{2}}\] at its ends. The rate of flow of heat is \[{{Q}_{1}}\] cal/sec. If all the linear dimensions are doubled keeping temperature constant, then rate of flow of heat \[{{Q}_{2}}\] will be
A)
\[4\,{{Q}_{1}}\]
done
clear
B)
\[2\,{{Q}_{1}}\]
done
clear
C)
\[\frac{{{Q}_{1}}}{4}\]
done
clear
D)
\[\frac{{{Q}_{1}}}{2}\]
done
clear
View Answer play_arrow
A black body radiates 20 W at temperature\[{{227}^{o}}C\]. If temperature of the black body is changed to\[{{727}^{o}}C\], then its radiating power will be:
A)
120 W
done
clear
B)
240 W
done
clear
C)
320 W
done
clear
D)
360 W
done
clear
View Answer play_arrow
A scientist says that the efficiency of his heat engine which operates at source temperature \[{{127}^{o}}C\] and sink temperature \[{{27}^{o}}C\] is 26%, then
A)
It is impossible
done
clear
B)
It is possible but less probable
done
clear
C)
It is quite probable
done
clear
D)
Data are incomplete
done
clear
View Answer play_arrow
A perfect gas at \[{{27}^{o}}C\] is heated at constant pressure to\[{{327}^{o}}C\]. If original volume of gas at \[{{27}^{o}}C\] is V, then volume at \[{{327}^{o}}C\] is
A)
V
done
clear
B)
3V
done
clear
C)
2V
done
clear
D)
VII
done
clear
View Answer play_arrow
Displacement between maximum potential energy position and maximum kinetic energy position for a particle executing S.H.M. is
A)
\[-a\]
done
clear
B)
\[+a\]
done
clear
C)
\[\pm \,a\]
done
clear
D)
\[\pm \,\frac{a}{4}\]
done
clear
View Answer play_arrow
In case of a forced vibration, the resonance wave becomes very sharp when the
A)
restoring force is small
done
clear
B)
applied periodic force is small
done
clear
C)
quality factor is small
done
clear
D)
damping force is small
done
clear
View Answer play_arrow
A string of 7m length has a mass of 0.035 kg. If tension in the string is 60.5 N, then speed of a wave on the string is
A)
77 m/s
done
clear
B)
102 m/s
done
clear
C)
110 m/s
done
clear
D)
165 m/s
done
clear
View Answer play_arrow
A whistle revolves in a circle with an angular speed of 20 rad/sec using a string of length 50 cm. If the frequency of sound from the whistle is 385 Hz, then what is the minimum frequency heard by an observer, which is far away from the centre in the same plane? (v = 340 m/s)
A)
333 Hz
done
clear
B)
374 Hz
done
clear
C)
385 Hz
done
clear
D)
394 Hz
done
clear
View Answer play_arrow
Cathode rays travelling from east to west enter into region of electric field directed towards north to south in the plane of paper. The deflection of cathode rays is towards
A)
East
done
clear
B)
South
done
clear
C)
West
done
clear
D)
North
done
clear
View Answer play_arrow
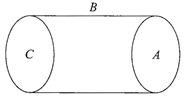
A hollow cylinder has a charge q coulomb within it. If\[\phi \]is the electric flux in unit of volt- meter associated with the curved surface B, the flux linked with the plane surface A in unit of voltmeter will be:
A)
\[\frac{1}{2}\left( \frac{q}{{{\varepsilon }_{0}}}-\phi \right)\]
done
clear
B)
\[\frac{q}{2{{\varepsilon }_{0}}}\]
done
clear
C)
\[\frac{\phi }{3}\]
done
clear
D)
\[\frac{q}{{{\varepsilon }_{0}}}-\phi \]
done
clear
View Answer play_arrow
The capacity of a parallel plate condenser is \[10\mu F,\]when the distance between its plates is 8 cm. If the distance between the plates is reduced to 4 cm, then the capacity of this parallel plate condenser will be
A)
\[5\,\mu F\]
done
clear
B)
\[10\,\mu F\]
done
clear
C)
\[20\,\mu F\]
done
clear
D)
\[40\,\mu F\]
done
clear
View Answer play_arrow
An electric dipole has the magnitude of its charge as q and its dipole moment is p. It is placed in a uniform electric field E. If its dipole moment is along the direction of the field, the force on it and its potential energy are respectively
A)
2qE and minimum
done
clear
B)
qE and pE
done
clear
C)
zero and minimum
done
clear
D)
qE and maximum
done
clear
View Answer play_arrow
A voltmeter has a range\[0-V\]with a series resistance R. With a series resistance 2R, the range is\[0-V'.\]The correct relation between V and V? is
A)
\[V'=2V\]
done
clear
B)
\[V'>2V\]
done
clear
C)
\[V'>>2V\]
done
clear
D)
\[V'<2V\]
done
clear
View Answer play_arrow
Masses of 3 wires of same metal are in the ratio 1:2:3 and their lengths are in the ratio 3 : 2 : 1. The electrical resistances are in the ratio
A)
1 : 4 : 9
done
clear
B)
9 : 4 : 1
done
clear
C)
1 : 2 : 3
done
clear
D)
27 : 6 : 1
done
clear
View Answer play_arrow
Electric bulb 50 W-100 V glowing at full power is to be used in parallel with battery 120 V, \[10\Omega .\]Maximum number of bulbs that can be connected so that they glow in full power is
A)
2
done
clear
B)
8
done
clear
C)
4
done
clear
D)
6
done
clear
View Answer play_arrow
In India electricity is supplied for domestic use at 220 V. It is supplied at 110 V in USA. If the resistance of a 60 W bulb for use in India is R, the resistance of a 60 W bulb for use in USA will be
A)
R
done
clear
B)
2R
done
clear
C)
\[\frac{R}{4}\]
done
clear
D)
\[\frac{R}{2}\]
done
clear
View Answer play_arrow
Lorentz force can be calculated by using the formula
A)
\[\vec{F}=q(\vec{E}+\vec{v}\times \vec{B})\]
done
clear
B)
\[\vec{F}=q(\vec{E}-\vec{v}\times \vec{B})\]
done
clear
C)
\[\vec{F}=q(\vec{E}+\vec{v}.\vec{B})\]
done
clear
D)
\[\vec{F}=q(\vec{E}\times \vec{B}+\vec{v})\]
done
clear
View Answer play_arrow
A galvanometer acting as a voltmeter will have
A)
a high resistance in parallel with its coil
done
clear
B)
a high resistance in series with its coil
done
clear
C)
a low resistance in parallel with its coil
done
clear
D)
a low resistance in series with its coil
done
clear
View Answer play_arrow
The direction of the null points is on the equatorial line of a bar magnet, when the north pole of the magnet is pointing
A)
North
done
clear
B)
South
done
clear
C)
East
done
clear
D)
West
done
clear
View Answer play_arrow
If the magnetic dipole moment of an atom of diamagnetic material, paramagnetic material and ferromagnetic material are denoted by\[{{\mu }_{d}},\,{{\mu }_{p}}\]and \[{{\mu }_{f}}\]respectively, then
A)
\[{{\mu }_{d}}\ne 0\]and\[{{\mu }_{f}}\ne 0\]
done
clear
B)
\[{{\mu }_{p}}\ne 0\] and \[{{\mu }_{f}}\ne 0\]
done
clear
C)
\[{{\mu }_{d}}\ne 0\]and \[{{\mu }_{p}}\ne 0\]
done
clear
D)
\[{{\mu }_{d}}\ne 0\]and\[{{\mu }_{p}}\ne 0\]
done
clear
View Answer play_arrow
A coil of resistance\[10\Omega \] and an inductance 5 H is connected to a 100 volt battery. Then energy stored in the coil is
A)
125 erg
done
clear
B)
125 J
done
clear
C)
250 erg
done
clear
D)
250 J
done
clear
View Answer play_arrow
Two coils of self-inductances 2 mH and 8 mH are placed so close together that the effective flux in one coil is completely linked with the other. The mutual inductance between these coils is
A)
10 mH
done
clear
B)
6 mH
done
clear
C)
4 mH
done
clear
D)
16 mH
done
clear
View Answer play_arrow
If the value of potential in an AC, circuit is 10 V, then the peak value of potential is
A)
\[\frac{10}{\sqrt{2}}\]
done
clear
B)
\[10\sqrt{2}\]
done
clear
C)
\[20\sqrt{2}\]
done
clear
D)
\[\frac{20}{\sqrt{2}}\]
done
clear
View Answer play_arrow
The core of a transformer is laminated be- Cause
A)
energy losses due to Eddy currents may be minimised
done
clear
B)
the weight of the transformer may be reduced
done
clear
C)
rusting of the core may be prevented
done
clear
D)
ratio of voltage in primary and secondary may be increased
done
clear
View Answer play_arrow
Which of the following shows greenhouse effect?
A)
Ultraviolet rays
done
clear
B)
Infrared rays
done
clear
C)
X-rays
done
clear
D)
None of these
done
clear
View Answer play_arrow
The velocity of electromagnetic radiation in a medium of permittivity \[{{\varepsilon }_{0}}\]and permeability\[{{\mu }_{0}}\]is given by
A)
\[\sqrt{\frac{{{\varepsilon }_{0}}}{{{\mu }_{0}}}}\]
done
clear
B)
\[\sqrt{{{\mu }_{0}}{{\varepsilon }_{0}}}\]
done
clear
C)
\[\sqrt{\frac{1}{\sqrt{{{\mu }_{0}}{{\varepsilon }_{0}}}}}\]
done
clear
D)
\[\sqrt{\frac{{{\mu }_{0}}}{{{\varepsilon }_{0}}}}\]
done
clear
View Answer play_arrow
Four lenses of focal length +15 cm, +20 cm, +150 cm and +250 cm are available for making an astronomical telescope. To produce the largest magnification, the focal length of the eye-piece should be
A)
+15 cm
done
clear
B)
+20 cm
done
clear
C)
+150 cm
done
clear
D)
+250 cm
done
clear
View Answer play_arrow
An object is at a distance of 0.5 m in front of a plane mirror. Distance between the object and image is
A)
0.5 m
done
clear
B)
1 m
done
clear
C)
0.25 m
done
clear
D)
1.5 m
done
clear
View Answer play_arrow
If two light waves having the same frequency have intensity ratio 4 : 1 and they interfere, the ratio of maximum to minimum intensity in the pattern will be
A)
9 : 1
done
clear
B)
3 : 1
done
clear
C)
25 : 9
done
clear
D)
16 : 25
done
clear
View Answer play_arrow
In Young's double-slit experiment, the slits are 2 mm apart and are illuminated by photons of two wavelengths\[{{\lambda }_{1}}=12000\,\overset{\text{o}}{\mathop{\text{A}}}\,\]and\[{{\lambda }_{2}}=10000\overset{\text{o}}{\mathop{\text{A}}}\,\]. At what minimum distance from the common central bright fringe on the screen 2 m from the slit will a bright fringe from one interference pattern coincide with a bright fringe from the other?
A)
8 mm
done
clear
B)
6 mm
done
clear
C)
4 mm
done
clear
D)
3 mm
done
clear
View Answer play_arrow
Two identical metal plates show photoelectric effect by a light of wavelength\[{{\lambda }_{A}}\]falls on plate A and\[{{\lambda }_{B}}\]on plate\[B({{\lambda }_{A}}=2{{\lambda }_{B}}).\]The maximum kinetic energy is
A)
\[2{{K}_{A}}={{K}_{B}}\]
done
clear
B)
\[{{K}_{A}}<{{K}_{B}}/2\]
done
clear
C)
\[{{K}_{A}}=2{{K}_{B}}\]
done
clear
D)
\[{{K}_{A}}={{K}_{B}}/2\]
done
clear
View Answer play_arrow
The work function of a metal is 4.2 eV, its threshold wavelength will be
A)
\[4000\,\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
B)
\[~3500\text{ }\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
C)
\[~2955\text{ }\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
D)
\[~2500\text{ }\overset{\text{o}}{\mathop{\text{A}}}\,\]
done
clear
View Answer play_arrow
In Bohr's model, the atomic radius of the first orbit is \[{{r}_{0}},\]then the radius of the third orbit is
A)
\[\frac{{{r}_{0}}}{9}\]
done
clear
B)
\[{{r}_{0}}\]
done
clear
C)
\[9{{r}_{0}}\]
done
clear
D)
\[3{{r}_{0}}\]
done
clear
View Answer play_arrow
The frequency of 1st line of Balmer series in \[{{H}_{2}}\]atom is \[{{v}_{0}}.\]The frequency of line emitted by singly ionised He atom is
A)
\[2{{v}_{0}}\]
done
clear
B)
\[4{{v}_{0}}\]
done
clear
C)
\[{{v}_{0}}/2\]
done
clear
D)
\[{{v}_{0}}/4\]
done
clear
View Answer play_arrow
Molarity of liquid HCl with density equal to g/mL is:
A)
36.5
done
clear
B)
18.25
done
clear
C)
32.05
done
clear
D)
4.65
done
clear
View Answer play_arrow
Which of the following is isoelectronic?
A)
\[C{{O}_{2}},N{{O}_{2}}\]
done
clear
B)
\[NO_{2}^{-},C{{O}_{2}}\]
done
clear
C)
\[C{{N}^{-}},CO\]
done
clear
D)
\[S{{O}_{2}},C{{O}_{2}}\]
done
clear
View Answer play_arrow
Among K, Ca, Fe and Zn, the element which can form more than one binary compound with chlorine is:
A)
Fe
done
clear
B)
Zn
done
clear
C)
K
done
clear
D)
Ca
done
clear
View Answer play_arrow
Which one of the following molecules will form a linear polymeric structure due to H-bonding?
A)
HCl
done
clear
B)
HF
done
clear
C)
\[{{H}_{2}}O\]
done
clear
D)
\[~N{{H}_{3}}\]
done
clear
View Answer play_arrow
The surface tension of which of the following liquid is maximum?
A)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
B)
\[C{{H}_{3}}OH\]
done
clear
C)
\[{{H}_{2}}O\]
done
clear
D)
\[{{C}_{6}}{{H}_{6}}\]
done
clear
View Answer play_arrow
\[Pb{{O}_{2}}\xrightarrow{{}}pbO,\Delta {{G}_{298}}<0\]\[Sn{{O}_{2}}\xrightarrow{{}}SnO,\Delta {{G}_{298}}>0\]. Most probable oxidation states of Pb and Sn will be
A)
\[P{{b}^{4+}}+S{{n}^{4+}}\]
done
clear
B)
\[P{{b}^{4+}}+S{{n}^{2+}}\]
done
clear
C)
\[P{{b}^{2+}},S{{n}^{2+}}\]
done
clear
D)
\[P{{b}^{2+}},S{{n}^{4+}}\]
done
clear
View Answer play_arrow
Heat of combustion\[\Delta {{H}^{o}}\]for\[C(s),{{H}_{2}}(g)\]and \[C{{H}_{4}}(g)\]are -94, -68 and -213 kcal/mol. Then \[\Delta {{H }^{o}}\] for \[C(s)+2{{H}_{2}}(g)\Delta C{{H}_{4}}(g)\]is
A)
-17 kcal
done
clear
B)
-111 kcal
done
clear
C)
-170 kcal
done
clear
D)
-85 kcal
done
clear
View Answer play_arrow
Reaction\[Ba{{O}_{2}}(s)\rightleftharpoons BaO(s)+{{O}_{2}}(g);\Delta H =+ve\]. In equilibrium condition, pressure of 02 depends on
A)
increased mass of \[Ba{{O}_{2}}\]
done
clear
B)
increased mass of BaO
done
clear
C)
increased temperature of equilibrium
done
clear
D)
increased mass of\[Ba{{O}_{2}}\]and BaO both
done
clear
View Answer play_arrow
If the concentration of\[O{{H}^{-}}\]ions in the reaction\[Fe{{(OH)}_{3}}(s)\rightleftharpoons F{{e}^{3+}}(aq)+3\bar{O}H\]is decreased to \[\frac{1}{4}\] times, then equilibrium concentration of \[F{{e}^{3+}}\] will be increased to:
A)
16 times
done
clear
B)
64 times
done
clear
C)
4 times
done
clear
D)
8 times
done
clear
View Answer play_arrow
Solubility of\[{{M}_{2}}S\]type salt is \[3.5\times {{10}^{-6}},\] then find out its solubility product
A)
\[1.7\times {{10}^{-6}}\]
done
clear
B)
\[1.7\times {{10}^{-16}}\]
done
clear
C)
\[1.7\times {{10}^{-18}}\]
done
clear
D)
\[1.7\times {{10}^{-12}}\]
done
clear
View Answer play_arrow
The solubility product of a sparingly soluble salt \[A{{X}_{2}}\]is \[3.2\times {{10}^{-11}}.\] Its solubility (in mol/L) is
A)
\[5.6\times {{10}^{-6}}\]
done
clear
B)
\[3.1\times {{10}^{-4}}\]
done
clear
C)
\[2\times {{10}^{-4}}\]
done
clear
D)
\[4\times {{10}^{-4}}\]
done
clear
View Answer play_arrow
In which of the following compounds, nitrogen exhibits the highest oxidation state?
A)
\[{{N}_{3}}H\]
done
clear
B)
\[~N{{H}_{2}}OH\]
done
clear
C)
\[~{{N}_{2}}{{H}_{4}}\]
done
clear
D)
\[~N{{H}_{3}}\]
done
clear
View Answer play_arrow
The pair of compounds that can exist together is
A)
\[FeC{{l}_{3}},SnC{{l}_{2}}\]
done
clear
B)
\[HgC{{l}_{2}},SnC{{l}_{2}}\]
done
clear
C)
\[FeC{{l}_{2}},SnC{{l}_{2}}\]
done
clear
D)
\[FeC{{l}_{3}},KI\]
done
clear
View Answer play_arrow
At its melting point ice is lighter than water because
A)
\[{{H}_{2}}O\]molecules are more closely packed in solid state
done
clear
B)
Ice crystals have hollow hexagonal arrangement of \[{{H}_{2}}O\]molecules
done
clear
C)
On melting of ice the \[{{H}_{2}}O\]molecule shrinks in size
done
clear
D)
Ice forms mostly heavy water on first melting
done
clear
View Answer play_arrow
The volume strength of \[1.5\,N\,{{H}_{2}}{{O}_{2}}\] solution is
A)
4.8 L
done
clear
B)
5.2 L
done
clear
C)
8.4 L
done
clear
D)
8.8 L
done
clear
View Answer play_arrow
Which of the statements is not true?
A)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] solution in acidic medium is orange
done
clear
B)
\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] solution becomes yellow on increasing the pH beyond 7
done
clear
C)
On passing H2S through acidified \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]solution, a milky color is observed
done
clear
D)
\[N{{a}_{2}}C{{r}_{2}}{{O}_{7}}\]is preferred over\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]in volumetric analysis
done
clear
View Answer play_arrow
A solid compound 'X' on heating gives \[C{{O}_{2}}\]gas and a residue. The residue mixed with water forms 'Y' on passing an excess of \[C{{O}_{2}}\]through 'Y' in water, a clear solution 'Z' is obtained. On boiling 'Z' compound, 'X' is reformed. The compound 'X' is
A)
\[Ca{{(HC{{O}_{3}})}_{2}}\]
done
clear
B)
\[CaC{{O}_{3}}\]
done
clear
C)
\[N{{a}_{2}}C{{O}_{3}}\]
done
clear
D)
\[{{K}_{2}}C{{O}_{3}}\]
done
clear
View Answer play_arrow
Which of the following structure is similar to graphite?
A)
B
done
clear
B)
\[{{B}_{4}}C\]
done
clear
C)
\[{{B}_{2}}{{H}_{6}}\]
done
clear
D)
BN
done
clear
View Answer play_arrow
Which property is common in diamond and graphite?
A)
Electrical conductivity
done
clear
B)
Crystal structure
done
clear
C)
Relative atomic mass
done
clear
D)
Density
done
clear
View Answer play_arrow
The most suitable method of separation of 1:1 mixture of ortho and para nitrophenols is:
A)
distillation
done
clear
B)
crystallization
done
clear
C)
sublimation
done
clear
D)
chromatography
done
clear
View Answer play_arrow
An organic compound contains carbon, hydro- gen and oxygen. Its elemental analysis gave C, 38.71% and H, 9.67%. The empirical formula of the compound would be:
A)
\[C{{H}_{2}}O\]
done
clear
B)
CHO
done
clear
C)
\[C{{H}_{4}}O\]
done
clear
D)
\[C{{H}_{3}}O\]
done
clear
View Answer play_arrow
The most stable carbocation is
A)
\[\overset{+}{\mathop{C}}\,{{H}_{3}}\]
done
clear
B)
\[C{{H}_{3}}\overset{+}{\mathop{C}}\,{{H}_{2}}\]
done
clear
C)
\[{{(C{{H}_{3}})}_{2}}\overset{+}{\mathop{C}}\,H\]
done
clear
D)
\[{{(C{{H}_{3}})}_{3}}\overset{+}{\mathop{C}}\,\]
done
clear
View Answer play_arrow
A)
[c] > (2) > (1)
done
clear
B)
[a] > (3) > (2)
done
clear
C)
[a] > (2) > (3)
done
clear
D)
[b] > (1) > (3)
done
clear
View Answer play_arrow
Which one of the following pairs represent; stereoisomerism?
A)
Structural and geometrical isomerism
done
clear
B)
Linkage and geometrical isomerism
done
clear
C)
Chain and rotational isomerism
done
clear
D)
Optical and geometrical isomerism
done
clear
View Answer play_arrow
How many stereoisomers does this molecule has? \[C{{H}_{3}}CH=CHC{{H}_{2}}CHBrC{{H}_{3}}\]
A)
6
done
clear
B)
8
done
clear
C)
4
done
clear
D)
2
done
clear
View Answer play_arrow
In preparation of alkene from alcohol using \[A{{l}_{2}}{{O}_{3}}\]which is effective factor?
A)
Temperature
done
clear
B)
Concentration
done
clear
C)
Surface area of\[A{{l}_{2}}{{O}_{3}}\]
done
clear
D)
Porosity of \[A{{l}_{2}}{{O}_{3}}\]
done
clear
View Answer play_arrow
Reaction of HBr with propene in the presence of peroxides gives
A)
isopropyi bromide
done
clear
B)
3-bromopropane
done
clear
C)
allyl bromide
done
clear
D)
n-propyl bromide
done
clear
View Answer play_arrow
Which of the following is/are the hazardous pollutant(s) present in automobile exhaust gases?
(i)\[{{N}_{2}}\] (ii) CO (iii)\[~C{{H}_{4}}\] (iv) Oxides of nitrogen
A)
(ii) and (iii)
done
clear
B)
(i) and (ii)
done
clear
C)
(ii) and (iv)
done
clear
D)
(i) and (iii)
done
clear
View Answer play_arrow
Roasting of sulphides gives the gas X as a byproduct. This is a colorless gas with choking smell of burnt sulphur and causes great damage to respiratory organs as it results in acid rain. Its aqueous solution is acidic and acts as a reducing agent, and its acid has never been isolated. The gas X is
A)
\[S{{O}_{2}}\]
done
clear
B)
\[C{{O}_{2}}\]
done
clear
C)
\[S{{O}_{3}}\]
done
clear
D)
\[{{H}_{2}}S\]
done
clear
View Answer play_arrow
In the nuclear change \[{{\,}_{92}}{{U}^{235}}+{{\,}_{0}}{{n}^{1}}\xrightarrow{{}}\] Fission products + neutron + \[3.20\times {{10}^{-11}}\,J,\] the energy released when 1 g of\[{{\,}_{92}}{{U}^{235}}\]finally undergoes fission is
A)
\[12.75\times {{10}^{8}}\,kJ\]
done
clear
B)
\[18.60\,\times {{10}^{9}}kJ\]
done
clear
C)
\[8.21\times {{10}^{7}}\,kJ\]
done
clear
D)
\[6.55\times {{10}^{6}}\,kJ\]
done
clear
View Answer play_arrow
The radioisotope \[\,_{27}^{60}Co\]used in the treatment of cancer can be made by \[(n,p)\] reaction. For this reaction, the target nucleus is
A)
\[_{28}^{59}Ni\]
done
clear
B)
\[_{27}^{59}Ni\]
done
clear
C)
\[_{28}^{60}Ni\]
done
clear
D)
\[_{27}^{60}Ni\]
done
clear
View Answer play_arrow
When molten zinc is cooled to solid state it assumes hcp structure. Then the number of nearest neighbours of zinc atom will be
A)
4
done
clear
B)
6
done
clear
C)
8
done
clear
D)
12
done
clear
View Answer play_arrow
Which of the following statements is not correct?
A)
Molecular solids are generally volatile
done
clear
B)
The number of carbon atoms in an unit cell of diamond is 4
done
clear
C)
The number o Bravais lattices in which a crystal can be categorized is 14
done
clear
D)
The faction of the total volume occupied by the atoms in a primitive cell is 0.48
done
clear
View Answer play_arrow
A solution has a 1 : 4 mole ratio of pentane to hexane. The vapour pressures of pure hydrocarbons at \[20{}^\circ C\] are 440 mm Hg for pentane and 120 mm Hg for hexane. The mole fraction of pentane in vapour phase would be:
A)
0.786
done
clear
B)
0.478
done
clear
C)
0.549
done
clear
D)
0.200
done
clear
View Answer play_arrow
A solution containing 10 g per dm3 of urea (molar mass = 60) is isotonic with a 5% (mass by vol.) solution of a non-volatile solute. The molar mass (in g\[mo{{l}^{-1}}\]) of non-volatile solute is
A)
350
done
clear
B)
300
done
clear
C)
250
done
clear
D)
200
done
clear
View Answer play_arrow
Electrode potential of \[Z{{n}^{2+}}/Zn\]is -0.76 V and that of \[C{{u}^{2+}}/Cu\]is +0.34 V. The EMF of the cell constructed between these two electrodes is
A)
1.10 V
done
clear
B)
0.42 V
done
clear
C)
-1.1 V
done
clear
D)
-0.42 V
done
clear
View Answer play_arrow
The standard electrode potential of the half cells is given below:
\[Z{{n}^{2+}}+2{{e}^{-}}\xrightarrow{{}}Zn;E=-7.62\,V,\] \[F{{e}^{2+}}+2{{e}^{-}}\xrightarrow{{}}Fe;E=-7.81\,V,\] The emf of the cell \[F{{e}^{2+}}+Zn\xrightarrow{{}}Z{{n}^{2+}}+Fe\]is
A)
1.54 V
done
clear
B)
-1.54 V
done
clear
C)
-0.19 V
done
clear
D)
+0.19 V
done
clear
View Answer play_arrow
The given reaction \[2NO+{{O}_{2}}\to 2N{{O}_{2}}\] is an example of
A)
first-order reaction
done
clear
B)
second-order reaction
done
clear
C)
third-order reaction
done
clear
D)
none of these
done
clear
View Answer play_arrow
If the rate of the reaction is equal to the rate constant, the order of the reaction is
A)
3
done
clear
B)
0
done
clear
C)
1
done
clear
D)
2
done
clear
View Answer play_arrow
If \[x\]is amount of adsorbate and m is amount of adsorbent, which of the following relations is related to adsorption process?
A)
\[x/m=P\times T\]
done
clear
B)
\[x/m=f(P)\]at constant T
done
clear
C)
\[x/m=f(T)\] at constant P
done
clear
D)
\[P=f(T)\] at constant \[(x/m)\]
done
clear
View Answer play_arrow
Catalyst
A)
lowers activation energy
done
clear
B)
increases activation energy
done
clear
C)
may increase or may decrease activation energy
done
clear
D)
brings out equilibrium
done
clear
View Answer play_arrow
Which one of the following ores is a chloride?
A)
Horn silver
done
clear
B)
Zincite
done
clear
C)
Bauxite
done
clear
D)
Felspar
done
clear
View Answer play_arrow
Which of the following is not an ore of magnesium?
A)
Magnesite
done
clear
B)
Dolomite
done
clear
C)
Gypsum
done
clear
D)
Carnalite
done
clear
View Answer play_arrow
Which one has the highest percentage of nitro-gen?
A)
Urea
done
clear
B)
Ammonium sulphate
done
clear
C)
Ammonium nitrate
done
clear
D)
Calcium nitrate
done
clear
View Answer play_arrow
\[{{H}_{3}}P{{O}_{2}}\]has the name and basicity respectively:
A)
phosphorus acid and two
done
clear
B)
hypophosphorus acid and two
done
clear
C)
hypophosphorus acid and one
done
clear
D)
hypophosphoric acid and two
done
clear
View Answer play_arrow
In five kingdom system, the main basis of classification is:
A)
Structure of nucleus
done
clear
B)
Nutrition
done
clear
C)
Structure of cell wall
done
clear
D)
Asexual reproduction
done
clear
View Answer play_arrow
Plants reproducing by spores such as mosses and ferns are grouped under the general term:
A)
Cryptogams
done
clear
B)
Bryophytes
done
clear
C)
Sporophytes
done
clear
D)
Thallophytes
done
clear
View Answer play_arrow
ICBN stands for:
A)
Indian Code of Botanical Nomenclature
done
clear
B)
Indian Congress of Biological Names
done
clear
C)
International code of Botanical Nomenclature
done
clear
D)
International Congress of Biological Names
done
clear
View Answer play_arrow
Cauliflower mosaic virus contains:
A)
ss RNA
done
clear
B)
ds RNA
done
clear
C)
ds DNA
done
clear
D)
ss DNA
done
clear
View Answer play_arrow
Extranuclear inheritance occurs in:
A)
Killer paramaecium
done
clear
B)
Killer Amoeba
done
clear
C)
Euglena
done
clear
D)
Hydra
done
clear
View Answer play_arrow
In bacteria, plasmid is:
A)
Extra chromosomal material
done
clear
B)
Main DNA
done
clear
C)
Nonfunctional DNA
done
clear
D)
Repetitive gene
done
clear
View Answer play_arrow
In a moss the sporophyte
A)
is partially parasitic on the gametophyte
done
clear
B)
produces gametes that given rise to the Gametophyte
done
clear
C)
Arises from a spore produced from the gametophyte
done
clear
D)
Manufactures food for itself, as well as for the gametophyte
done
clear
View Answer play_arrow
If you are asked to classify the various algae into distinct groups, which of the following characters you should choose?
A)
Chemical composition of the cell wall
done
clear
B)
Types of pigments present in the cell
done
clear
C)
Nature of stored food materials in the cell
done
clear
D)
Structural organization of thallus
done
clear
View Answer play_arrow
In which one of the following, male and female gametophytes do not have free-living independent existence?
A)
Polytrichum
done
clear
B)
Cedrus
done
clear
C)
Pteris
done
clear
D)
Funaria
done
clear
View Answer play_arrow
In which of the following notochord is present in embryonic stage:
A)
All chordates
done
clear
B)
Some chordates
done
clear
C)
Vertebrates
done
clear
D)
Non-chordates
done
clear
View Answer play_arrow
Ommatidia serve the purpose of photoreception in:
A)
Cockroach
done
clear
B)
Frog
done
clear
C)
Humans
done
clear
D)
Sunflower
done
clear
View Answer play_arrow
The animals with bilateral symmetry in young stage and radial pentamerous symmetry in the adult stage, belong to the phylum:
A)
Mollusca
done
clear
B)
Cnidaria
done
clear
C)
Echinodermata
done
clear
D)
Annelida
done
clear
View Answer play_arrow
Which is correct pair for edible part?
A)
Tomato - Thalamus
done
clear
B)
Maize - Cotyledons
done
clear
C)
Guava - Mesocarp
done
clear
D)
Date palm - Pericarp
done
clear
View Answer play_arrow
The aleurone layer in maize grain is specially
A)
Proteins
done
clear
B)
Starch
done
clear
C)
Lipids
done
clear
D)
Auxins
done
clear
View Answer play_arrow
The filamentous threads protruding at the end young cob of maize are
A)
Anthers
done
clear
B)
Styles
done
clear
C)
Ovaries
done
clear
D)
Hairs
done
clear
View Answer play_arrow
Axillary bud and terminal bud derived from the activity of:
A)
Lateral meristem
done
clear
B)
Intercalary meristem
done
clear
C)
Apical meristem
done
clear
D)
Parenchyma
done
clear
View Answer play_arrow
The apical meristem of the root is present:
A)
Only in radicals
done
clear
B)
Only in tap roots
done
clear
C)
Only in adventitious roots
done
clear
D)
In all the rook
done
clear
View Answer play_arrow
In a woody dicotyledonous tree, which of the following parts will mainly consist of primary tissues:
A)
Stem and root
done
clear
B)
All parts
done
clear
C)
Shoot tips and root tips
done
clear
D)
Flowers, fruit and leaves
done
clear
View Answer play_arrow
Which cells do not form layer and remains structurally separate?
A)
Epithelial cells
done
clear
B)
Muscle cells
done
clear
C)
Nerve cells
done
clear
D)
Gland cells
done
clear
View Answer play_arrow
Bowman's glands are found in
A)
Olfactory epithelium
done
clear
B)
External auditory canal
done
clear
C)
Cortical nephrons only
done
clear
D)
Juxtamedullary nephrons
done
clear
View Answer play_arrow
The kind of tissue that forms the supportive structure in our pinna (external ears) is also found in:
A)
Tip of the nose
done
clear
B)
Vertebrae
done
clear
C)
Nails
done
clear
D)
Ear ossicles
done
clear
View Answer play_arrow
Microtubules are absent in
A)
Mitochondria
done
clear
B)
Flagella
done
clear
C)
Spindle fibres
done
clear
D)
Centriole
done
clear
View Answer play_arrow
In which one of the following, nitrogen is not a constituent?
A)
Idioblast
done
clear
B)
Bacteriochlorophyll
done
clear
C)
Invertase
done
clear
D)
Pepsin
done
clear
View Answer play_arrow
According to widely accept "fluid mosaic model" cell membranes are semi-fluid, where lipids and integral proteins can diffuse randomly. In recent years, this model has been modified in several respects. In this regard, which of the following statements are incorrect:
A)
Proteins can also undergo flip-flop movements in the lipid bilayer
done
clear
B)
Many proteins remain completely embedded within the lipid bilayer
done
clear
C)
Proteins in cell membranes can travel within the lipid bilayer
done
clear
D)
Proteins can remain confined within certain domains of the membranes
done
clear
View Answer play_arrow
Most abundant organic compound on earth is:
A)
Protein
done
clear
B)
Cellulose
done
clear
C)
Lipids
done
clear
D)
Steroids
done
clear
View Answer play_arrow
Lipids are insoluble in water because lipids molecules are:
A)
Hydrophilic
done
clear
B)
Hydrophobic
done
clear
C)
Neutral
done
clear
D)
Zwitter ions
done
clear
View Answer play_arrow
Which of the following is the simplest amino acid?
A)
Alanine
done
clear
B)
Asparagine
done
clear
C)
Glycine
done
clear
D)
Tyrosine
done
clear
View Answer play_arrow
If a diploid cell is treated with colchicine, then it becomes:
A)
Triploid
done
clear
B)
Tetraploid
done
clear
C)
Diploid
done
clear
D)
Monoploid
done
clear
View Answer play_arrow
If you are provided with root-tips of onion in your class and are asked to count the chromosomes which of the following stages can you most conveniently look into:
A)
Telophase
done
clear
B)
Anaphase
done
clear
C)
Prophase
done
clear
D)
Metaphase
done
clear
View Answer play_arrow
Synapsis occurs between:
A)
two homolog chromosomes
done
clear
B)
a male and a female gamete
done
clear
C)
mRNA and ribosomes
done
clear
D)
spindle fibres and centromere
done
clear
View Answer play_arrow
When water moves through a semipermeable membrane, then which of the following pressure develops?
A)
OP
done
clear
B)
SP
done
clear
C)
TP
done
clear
D)
WP
done
clear
View Answer play_arrow
Potometer works on the principle of:
A)
Potential difference between the tip of the tube and that of the plant
done
clear
B)
Amount of water absorbed equals the amount transpired
done
clear
C)
Osmotic pressure
done
clear
D)
Root pressure
done
clear
View Answer play_arrow
The rupture and fractionation do not usually occur in the water column in vessel/tracheids during the ascent of sap because of:
A)
weak gravitational pull
done
clear
B)
transpiration pull
done
clear
C)
lignified thick walls
done
clear
D)
cohesion and adhesion
done
clear
View Answer play_arrow
In plants inulin and pectin are:
A)
Reserved material
done
clear
B)
Wastes
done
clear
C)
Excretory material
done
clear
D)
Insect attracting material
done
clear
View Answer play_arrow
Boron in green plants assists in:
A)
Activation of enzymes
done
clear
B)
Acting of enzyme cofactor
done
clear
C)
Photosynthesis
done
clear
D)
Sugar transport
done
clear
View Answer play_arrow
The major role of minor elements inside living organisms is to act as:
A)
co-factors of enzymes
done
clear
B)
Building blocks of important amino acids
done
clear
C)
Constituent of hormones
done
clear
D)
Binder of cell structure
done
clear
View Answer play_arrow
Which pair is wrong?
A)
\[{{C}_{3}}-\]Maize
done
clear
B)
\[{{C}_{4}}-\]Kranz anatomy
done
clear
C)
Calvin cycle - PGA
done
clear
D)
Hatch and Slake cycle -O.A.A.
done
clear
View Answer play_arrow
Which element is located at the centre of the porphyrin ring in chlorophyll?
A)
Calcium
done
clear
B)
Magnesium
done
clear
C)
Potassium
done
clear
D)
Manganese
done
clear
View Answer play_arrow
Photosynthesis in\[{{C}_{4}}\]plants is relatively less limited by atmospheric\[C{{O}_{2}}\]levels because:
A)
The primary fixation of\[C{{O}_{2}}\]is mediated via PEP carboxylase
done
clear
B)
Effective pumping of \[C{{O}_{2}}\]into bundle sheath cells
done
clear
C)
Four carbon acids are the primary initial\[C{{O}_{2}}\] fixation products
done
clear
D)
Rubisco in\[{{C}_{4}}\]plants has higher Inanity for \[C{{O}_{2}}\]
done
clear
View Answer play_arrow
In which one of the following do the two names refer to one and the same thing:
A)
Krebs cycle and Calvin cycle
done
clear
B)
Tricarboxylic acid cycle and citric acid Cycle
done
clear
C)
Citric acid cycle and Calvin cycle
done
clear
D)
Tricarboxylic acid cycle and urea cycle
done
clear
View Answer play_arrow
In glycolysis, during oxidation electrons are removed by:
A)
Glyceraldehyde-3 -phosphate
done
clear
B)
\[~NA{{D}^{+}}\]
done
clear
C)
Molecular oxygen
done
clear
D)
ATP
done
clear
View Answer play_arrow
The overall goal of glycolysis, Krebs cycle and the electron transport system is the formation of:
A)
Nucleic acids
done
clear
B)
ATP in small stepwise units
done
clear
C)
ATP in one large oxidation reaction
done
clear
D)
Sugars
done
clear
View Answer play_arrow
Proteinaceous pigment which control the activities concerned with light:
A)
Phytochrome
done
clear
B)
Chlorophyll
done
clear
C)
Anthocyanin
done
clear
D)
Carotenoids
done
clear
View Answer play_arrow
Seed dormancy is due to the:
A)
Ethylene
done
clear
B)
Abscisic acid
done
clear
C)
IAA
done
clear
D)
Starch
done
clear
View Answer play_arrow
One set of a plant was grown at 12 hours day and 12 hours night period cycles and it flowered while in the other set night phase was interrupted by flash of light and it did not produce flower. Under which one of the following categories will you place this plant?
A)
Darkness neutral
done
clear
B)
Day neutral
done
clear
C)
Short day
done
clear
D)
Long day
done
clear
View Answer play_arrow
Hydrolytic enzymes which act on low pH are called:
A)
Protease
done
clear
B)
a-Amylase
done
clear
C)
Hydrolases
done
clear
D)
Peroxidase
done
clear
View Answer play_arrow
Duodenum has characteristic Brunner's glands which secrete two hormones called
A)
Secretin, cholecystokinin
done
clear
B)
Prolactin, parathormone
done
clear
C)
Extradiol, progesterone
done
clear
D)
Kinase, estrogen
done
clear
View Answer play_arrow
Examination of blood of a person suspected of having anemia shows large, immature, nucleated erythrocytes without haemoglobin. Supplementing his diet with which of the following is likely to alleviate his symptoms?
A)
Thiamine
done
clear
B)
Folic acid and cobalamin
done
clear
C)
Riboflavin
done
clear
D)
Iron compounds
done
clear
View Answer play_arrow
Which one of the following statements is incorrect?
A)
The residual air in lungs slightly decreases the efficiency of respiration in mammals
done
clear
B)
The presence of non-respiratory air sacs, increases the efficiency of respiration in birds
done
clear
C)
In insects, circulating body fluids serve to distribute oxygen to tissues
done
clear
D)
The principle of countercurrent flow facilitates efficient respiration in gills of fishes
done
clear
View Answer play_arrow
A large proportion of oxygen is left unused in the human blood even after its uptakes by the body tissues. This \[{{O}_{2}}:\]
A)
helps in releasing more\[{{O}_{2}}\]to the epithelial tissues
done
clear
B)
acts as a reserve during muscular exercise
done
clear
C)
raises the\[pC{{O}_{2}}\]of blood to 75 mm of Hg
done
clear
D)
is enough to keep oxyhaemoglobin saturation at 96%
done
clear
View Answer play_arrow
People who have migrated from the planes to an area adjoining Rohtang Pass about six months back:
A)
Suffer from altitude sickness with symp- toms like nausea, fatigue, etc.
done
clear
B)
Have the usual RBC count but their hae- moglobin has very high binding affinity to\[{{O}_{2}}\]
done
clear
C)
Have more RBCs and their haemoglobin has a lower binding affinity to\[{{O}_{2}}\]
done
clear
D)
Are not physically fit to play games like football
done
clear
View Answer play_arrow
Sickle cell anaemia is due to:
A)
Change of amino acid in\[\alpha -\]chain of hae- moglobin
done
clear
B)
Change of amino acid in\[\beta -\]chain of haemoglobin
done
clear
C)
Change of amino acid in both\[\alpha \]and \[\beta \]chain of haemoglobin
done
clear
D)
Change of amino acid either\[\alpha \]or\[\beta \]chain of haemoglobin
done
clear
View Answer play_arrow
Bundle of His is a network of:
A)
Muscle fibres distributed throughout the heart walls
done
clear
B)
Muscle fibres found only in the ventricle wall
done
clear
C)
Nerve fibres distributed in ventricles
done
clear
D)
Nerve fibres found throughout the heart
done
clear
View Answer play_arrow
In humans, blood passes from the post caval to the diastolic right atrium of heart due to:
A)
stimulation of the sino auricular node
done
clear
B)
pressure difference between the post caval and atrium
done
clear
C)
pushing open of the venous valves
done
clear
D)
suction pull
done
clear
View Answer play_arrow
In Hydra, waste material of food digestion and nitrogenous waste material removed from:
A)
Mouth and mouth
done
clear
B)
Body wall and body wall
done
clear
C)
Mouth and body wall
done
clear
D)
Mouth and tentacles
done
clear
View Answer play_arrow
In omithine cycle which of the following wastes are removed from the blood:
A)
\[C{{O}_{2}}\]and ammonia
done
clear
B)
Ammonia and urea
done
clear
C)
\[C{{O}_{2}}\]and urea
done
clear
D)
Urea and urine
done
clear
View Answer play_arrow
What will happen if the stretch receptors of the urinary bladder wall are totally removed?
A)
There will be no micturition
done
clear
B)
Urine will continue to collect normally in bladder
done
clear
C)
Micturition will continue
done
clear
D)
Urine will not collect in the bladder
done
clear
View Answer play_arrow
What will happen if ligaments are cut or bro- ken?
A)
Bones will move freely at joints
done
clear
B)
No movement at joint
done
clear
C)
Bone will become unfix
done
clear
D)
Bone will become fixed
done
clear
View Answer play_arrow
Elbow joint is an example of:
A)
Ball and socket joint
done
clear
B)
Pivot joint
done
clear
C)
Hinge joint
done
clear
D)
Gliding joint
done
clear
View Answer play_arrow
Which of the following is not a function of the skeletal system?
A)
Locomotion
done
clear
B)
Production of erythrocytes
done
clear
C)
Storage of minerals
done
clear
D)
Production of body heat
done
clear
View Answer play_arrow
Characteristics character of human cornea is:
A)
Secreted by conjurctiva and glandular
done
clear
B)
It has lacrimal gland which secrete tears
done
clear
C)
Blood circulation is absent in cornea
done
clear
D)
In old age it become harden and white layer deposit on it which causes the cataract
done
clear
View Answer play_arrow
In the resting state of the neural membrane, diffusion due to concentration gradients, if allowed, would drive:
A)
\[{{K}^{+}}\]and\[N{{a}^{+}}\]out of the cell
done
clear
B)
\[N{{a}^{+}}\]into the cell
done
clear
C)
\[N{{a}^{+}}\]out of the cell
done
clear
D)
\[{{K}^{+}}\]into the cell
done
clear
View Answer play_arrow
Bowman's glands are found in:
A)
Olfactory epithelium
done
clear
B)
External auditory canal
done
clear
C)
Cortical nephrons only
done
clear
D)
Juxtamedullary nephrons
done
clear
View Answer play_arrow
Which steroid is used for transformation?
A)
Cortisol
done
clear
B)
Cholesterol
done
clear
C)
Testosterone
done
clear
D)
Progesterone
done
clear
View Answer play_arrow
Melanin protect from:
A)
UV rays
done
clear
B)
Visible rays
done
clear
C)
Infrared rays
done
clear
D)
X-rays
done
clear
View Answer play_arrow
Which one of the following does not act as a neurotransmitter?
A)
Acetylcholine
done
clear
B)
Epinephrine
done
clear
C)
Norepinephrine
done
clear
D)
Cortisone
done
clear
View Answer play_arrow
What is true for cleavage?
A)
Size of embryo increase
done
clear
B)
Size of cells decrease
done
clear
C)
Size of cells increase
done
clear
D)
Size of embryo decrease
done
clear
View Answer play_arrow
Which one of the following is correctly matched?
A)
Chlamydomonas - Conidia
done
clear
B)
Yeast - Zoospores
done
clear
C)
Onion-Bulb
done
clear
D)
Ginger - Sucker
done
clear
View Answer play_arrow
Monoecious plant of Chara shows occurrence of:
A)
Antheridiophore and archegoniophore on the same plant
done
clear
B)
Stamen and carpel on the same plant
done
clear
C)
Upper antheridium and lower oogonium on the same plant
done
clear
D)
Upper oogonium and lower antheridium on the same plant
done
clear
View Answer play_arrow
Anemophily type of pollination is found in:
A)
Salvia
done
clear
B)
Bottle brush
done
clear
C)
Vallisneria
done
clear
D)
Coconut
done
clear
View Answer play_arrow
What is the direction of micropyle in anatropous ovule?
A)
Upward
done
clear
B)
Downward
done
clear
C)
Right
done
clear
D)
Left
done
clear
View Answer play_arrow
In which pair both the plants can be vegetatively propagated by leaf pieces:
A)
Bryophyllum and Kalanchoe
done
clear
B)
Agave and Kalanchoe
done
clear
C)
Asparagus and Bryophyllum
done
clear
D)
Chrysanthemum and Agave
done
clear
View Answer play_arrow
Grey crescent is the area:
A)
At the point of entry of sperm into ovum
done
clear
B)
Just opposite to the site of entry of sperm into ovum
done
clear
C)
At the animal pole
done
clear
D)
At the vegetal pole
done
clear
View Answer play_arrow
Which extraembryonic membrane in humans prevents desiccation of the embryo inside the uterus?
A)
Yolk sac
done
clear
B)
Amnion
done
clear
C)
Chorion
done
clear
D)
Allantois
done
clear
View Answer play_arrow
Foetal ejection reflex in human female is induced by:
A)
Differentiation of mammary glands
done
clear
B)
Pressure exerted by amniotic fluid
done
clear
C)
Release of oxytocin from pituitary
done
clear
D)
Fully developed foetus and placenta
done
clear
View Answer play_arrow
Cu ions released from copper-releasing intra- uterine devices (lUDs):
A)
make uterus unsuitable for implantation
done
clear
B)
increase phagocytosis of sperms
done
clear
C)
suppress sperm motility
done
clear
D)
prevent ovulation
done
clear
View Answer play_arrow
The test-tube baby programme employs which one of the mowing techniques?
A)
Gamete intra fallopian transfer (GIFT)
done
clear
B)
Zygote intra fallopian transfer (ZIFT)
done
clear
C)
Intra cytoplasmic sperm injection (ICSI)
done
clear
D)
Intrauterine insemination (IUI)
done
clear
View Answer play_arrow
Assisted reproductive technology, IVF involves transfer of:
A)
Ovum into the fallopian tube
done
clear
B)
Zygote into the fallopian tube
done
clear
C)
Zygote into the uterus
done
clear
D)
Embryo with 16 blastomeres into the fal- lopian tube
done
clear
View Answer play_arrow
A and B genes are linked. What shall be genotype of progeny in a cross between AB/ab and ab/ab:
A)
AAbb and aabb
done
clear
B)
AaBb and aabb
done
clear
C)
AABB and aabb
done
clear
D)
None
done
clear
View Answer play_arrow
Ratio of complementary genes is:
A)
9 : 3 : 4
done
clear
B)
12 : 3 : 1
done
clear
C)
9 : 3 : 3 : 4
done
clear
D)
9 : 7
done
clear
View Answer play_arrow
There are three genes a, b and c. The percentage of crossing over between a and b is 20%, b and c is 28% and a and c is 8%. What is the sequence of genes on chromosome?
A)
b, a, c
done
clear
B)
a, b, c
done
clear
C)
a, c, b
done
clear
D)
None
done
clear
View Answer play_arrow
At the time of organogenesis genes regulate the process at different levels and at different time due to:
A)
Promoter
done
clear
B)
Regulator
done
clear
C)
Intron
done
clear
D)
Exon
done
clear
View Answer play_arrow
Exon part of m-RNAs have code for:
A)
Protein
done
clear
B)
Lipid
done
clear
C)
Carbohydrate
done
clear
D)
Phospholipid
done
clear
View Answer play_arrow
In E. coli, during lactose metabolism represser binds to:
A)
Regulator gene
done
clear
B)
Operator gene
done
clear
C)
Structural gene
done
clear
D)
Promoter gene
done
clear
View Answer play_arrow
Half-life period of\[{{C}^{14}}\]is:
A)
500 years
done
clear
B)
5000 years
done
clear
C)
50 years
done
clear
D)
\[5\times {{10}^{4}}\]years
done
clear
View Answer play_arrow
According to fossils which discovered up to present time, origin and evolution of man was started from which country
A)
France
done
clear
B)
Java
done
clear
C)
Africa
done
clear
D)
China
done
clear
View Answer play_arrow
Sequence of which of the following is used to know the phylogeny:
A)
m-RNA
done
clear
B)
r-RNA
done
clear
C)
t-RNA
done
clear
D)
DNA
done
clear
View Answer play_arrow
Which statement correct about centre of origin of plant?
A)
More diversity in improved variety
done
clear
B)
Frequency of dominant gene is more
done
clear
C)
Climatic condition more favourable
done
clear
D)
None
done
clear
View Answer play_arrow
Which one of the following is correct match?
A)
Reserpine - Tranquilliser
done
clear
B)
Cocaine - opiatic narcotic
done
clear
C)
Morphine - Hallucinogenic
done
clear
D)
Bhang-Analgesic
done
clear
View Answer play_arrow
Short-lived immunity acquired from mother to foetus across placenta or through mother's milk to the infant is categorised as:
A)
Active immunity
done
clear
B)
Passive immunity
done
clear
C)
Cellular immunity
done
clear
D)
Innate non-specific immunity
done
clear
View Answer play_arrow