-
question_answer1)
Which compound is formed when \[C{{H}_{3}}OH\] reacts with \[C{{H}_{3}}-Mg-X\] [CPMT 1977, 89]
A)
Acetone done
clear
B)
Alcohol done
clear
C)
Methane done
clear
D)
Ethane done
clear
View Solution play_arrow
-
question_answer2)
A compound X of formula \[{{C}_{3}}{{H}_{8}}O\]yields a compound \[{{C}_{3}}{{H}_{6}}O\], on oxidation. To which of the following classes of compounds could X being [Pb. PMT 2000]
A)
Secondary alcohol done
clear
B)
Alkene done
clear
C)
Aldehyde done
clear
D)
Tertiary alcohol done
clear
View Solution play_arrow
-
question_answer3)
The boiling point of alcohol are ?. than corresponding thiols [Pb. PMT 2000]
A)
More done
clear
B)
Same done
clear
C)
Either of these done
clear
D)
Less done
clear
View Solution play_arrow
-
question_answer4)
Methyl alcohol can be distinguished from ethyl alcohol using [KCET 1984; BHU 2000]
A)
Fehling solution done
clear
B)
Schiff's reagent done
clear
C)
Sodium hydroxide and iodine done
clear
D)
Phthalein fusion test done
clear
View Solution play_arrow
-
question_answer5)
A compound X with molecular formula \[{{C}_{3}}{{H}_{8}}O\] can be oxidised to a compound Y with the molecular formula \[{{C}_{3}}{{H}_{6}}{{O}_{2}}\] X is most likely to be [MP PMT 1991]
A)
Primary alcohol done
clear
B)
Secondary alcohol done
clear
C)
Aldehyde done
clear
D)
Ketone done
clear
View Solution play_arrow
-
question_answer6)
An alcohol on oxidation is found to give \[C{{H}_{3}}COOH\] and \[C{{H}_{3}}C{{H}_{2}}COOH.\] The structure of the alcohol is [BIT 1990]
A)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OH\] done
clear
B)
\[{{(C{{H}_{3}})}_{2}}C(OH)C{{H}_{2}}C{{H}_{3}}\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}CHOHC{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}CH(OH)C{{H}_{2}}C{{H}_{2}}C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer7)
An organic liquid A containing \[C,H\] and O has a pleasant odour with a boiling point of \[{{78}^{o}}C\] On boiling A with conc. \[{{H}_{2}}S{{O}_{4}}\] a colourless gas is produced which decolourises bromine water and alkaline \[KMn{{O}_{4}}.\] One mole of this gas also takes one mole of \[{{H}_{2}}.\] The organic liquid A is [KCET 1993]
A)
\[{{C}_{2}}{{H}_{5}}Cl\] done
clear
B)
\[{{C}_{2}}{{H}_{5}}CHO\] done
clear
C)
\[{{C}_{2}}{{H}_{6}}\] done
clear
D)
\[{{C}_{2}}{{H}_{5}}OH\] done
clear
View Solution play_arrow
-
question_answer8)
An aromatic amine A was treated with alcoholic potash and another compound Y when foul smelling gas was formed with formula \[{{C}_{6}}{{H}_{5}}NC.\] Y was formed by reacting a compound Z with \[C{{l}_{2}}\] in the presence of slaked lime. The compound Z is [CBSE PMT 1990]
A)
\[{{C}_{6}}{{H}_{5}}N{{H}_{2}}\] done
clear
B)
\[{{C}_{2}}{{H}_{5}}OH\] done
clear
C)
\[C{{H}_{3}}OC{{H}_{3}}\] done
clear
D)
\[CHC{{l}_{3}}\] done
clear
View Solution play_arrow
-
question_answer9)
Rectified spirit obtained by fermentation contains 4.5% of water. So in order to remove it, rectified spirit is mixed with suitable quantity of benzene and heated. Benzene helps because [KCET 1987]
A)
It is dehydrating agent and so removes water done
clear
B)
It forms the lower layer which retains all the water so that alcohol can be distilled off done
clear
C)
It forms an azeotropic mixture having high boiling point and thus allows the alcohol to distill over done
clear
D)
It forms low boiling azeotropic mixtures which distill over, leaving behind pure alcohol which can then be distilled done
clear
View Solution play_arrow
-
question_answer10)
\[{{C}_{6}}{{H}_{5}}OH+ClCOC{{H}_{3}}\xrightarrow{\text{aq}\text{. }NaOH}{{C}_{6}}{{H}_{5}}OCOC{{H}_{3}}\] is an example of [BHU 1984]
A)
Dow's reaction done
clear
B)
Reimer-Tiemann reaction done
clear
C)
Schotten-Baumann reaction done
clear
D)
Kolbe's reaction done
clear
View Solution play_arrow
-
question_answer11)
Ortho-nitrophenol is steam volatile whereas para-nitrophenol is not. This is due to [CBSE PMT 1989]
A)
Intramolecular hydrogen bonding present in ortho-nitrophenol done
clear
B)
Intermolecular hydrogen bonding done
clear
C)
Intramolecular hydrogen bonding present in para-nitrophenol done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer12)
Reaction of phenol with dil. \[HN{{O}_{3}}\] gives [KCET 1993; RPMT 1997]
A)
p and m-nitrophenols done
clear
B)
o- and p-nitrophenols done
clear
C)
Picric acid done
clear
D)
o- and m-nitrophenols done
clear
View Solution play_arrow
-
question_answer13)
Phenol is less acidic than [IIT-JEE 1986; UPSEAT 2003; Orissa JEE 2004]
A)
Acetic acid done
clear
B)
p-nitrophenol done
clear
C)
Both a and b done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer14)
The strongest acid among the following aromatic compounds is [NCERT 1978]
A)
ortho-nitrophenol done
clear
B)
para-chlorophenol done
clear
C)
para-nitrophenol done
clear
D)
meta-nitrophenol done
clear
View Solution play_arrow
-
question_answer15)
Diazo-coupling is useful to prepare some [CBSE PMT 1994]
A)
Pesticides done
clear
B)
Proteins done
clear
C)
Dyes done
clear
D)
Vitamins done
clear
View Solution play_arrow
-
question_answer16)
Glycerol reacts with \[{{P}_{4}}+{{I}_{2}}\] to form [CBSE PMT 1991]
A)
Aldehyde done
clear
B)
Allyl iodide done
clear
C)
Allyl alcohol done
clear
D)
Acetylene done
clear
View Solution play_arrow
-
question_answer17)
When glycerine is added to a litre of water which of the following behaviour is observed [NCERT 1977; BHU 1979]
A)
Water evaporates more easily done
clear
B)
The temperature of water is increased done
clear
C)
The freezing point of water is lowered done
clear
D)
The viscosity of water is lowered done
clear
View Solution play_arrow
-
question_answer18)
Final product formed on reduction of glycerol by hydroiodic acid is [CPMT 1987]
A)
Propane done
clear
B)
Propanoic acid done
clear
C)
Propene done
clear
D)
Propyne done
clear
View Solution play_arrow
-
question_answer19)
Glycerol was distilled with oxalic acid crystals and the products were led into Fehling solution and warmed. Cuprous oxide was precipitated. It is due to [KCET 1987]
A)
CO done
clear
B)
HCHO done
clear
C)
\[C{{H}_{3}}CHO\] done
clear
D)
HCOOH done
clear
View Solution play_arrow
-
question_answer20)
Kolbe-Schmidt reaction is used for [CBSE PMT 1991]
A)
Salicylic acid done
clear
B)
Salicylaldehyde done
clear
C)
Phenol done
clear
D)
Hydrocarbon done
clear
View Solution play_arrow
-
question_answer21)
Which of the following explains the viscous nature of glycerol [JIPMER 1997]
A)
Covalent bonds done
clear
B)
Hydrogen bonds done
clear
C)
Vander Wall's forces done
clear
D)
Ionic forces done
clear
View Solution play_arrow
-
question_answer22)
On heating glycerol with conc. \[{{H}_{2}}S{{O}_{4}},\] a compound is obtained which has a bad odour. The compound is [CPMT 1974; CBSE PMT 1994]
A)
Glycerol sulphate done
clear
B)
Acrolein done
clear
C)
Formic acid done
clear
D)
Allyl alcohol done
clear
View Solution play_arrow
-
question_answer23)
Isopropyl alcohol on oxidation forms [CPMT 1971, 81, 94; RPMT 2002]
A)
Acetone done
clear
B)
Ether done
clear
C)
Ethylene done
clear
D)
Acetaldehyde done
clear
View Solution play_arrow
-
question_answer24)
Benzenediazonium chloride on reaction with phenol in weakly basic medium gives [IIT-JEE 1998]
A)
Diphenyl ether done
clear
B)
p-hydroxyazobenzene done
clear
C)
Chlorobenzene done
clear
D)
Benzene done
clear
View Solution play_arrow
-
question_answer25)
The alcohol that produces turbidity immediately with \[ZnC{{l}_{2}}+\] conc. HCl at room temperature [EAMCET 1997; MP PMT 1989, 99; IIT JEE 1981, 86; CBSE PMT 1989; CPMT 1989;MP PET 1997; JIPMER 1999]
A)
1-hydroxybutane done
clear
B)
2-hydroxybutane done
clear
C)
2-hydroxy-2-methylpropane done
clear
D)
1-hydroxy-2-methylpropane done
clear
View Solution play_arrow
-
question_answer26)
The reagent which easily reacts with ethanol and propanol is [MP PET 1989]
A)
Fehling solution done
clear
B)
Grignard reagent done
clear
C)
Schiff's reagent done
clear
D)
Tollen's reagent done
clear
View Solution play_arrow
-
question_answer27)
Propene is the product obtained by dehydrogenation of [KCET (Engg.) 2001]
A)
2-propanol done
clear
B)
1-propanol done
clear
C)
Propanal done
clear
D)
n-propyl alcohol done
clear
View Solution play_arrow
-
question_answer28)
Which of the following statements is correct [BHU 1997]
A)
Phenol is less acidic than ethyl alcohol done
clear
B)
Phenol is more acidic than ethyl alcohol done
clear
C)
Phenol is more acidic than carboxylic acid done
clear
D)
Phenol is more acidic than carbonic acid done
clear
View Solution play_arrow
-
question_answer29)
Boiling point of alcohol is comparatively higher than that corresponding alkane due to [MH CET 2002]
A)
Intermolecular hydrogen bonding done
clear
B)
Intramolecular hydrogen bonding done
clear
C)
Volatile nature done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer30)
When Phenol is heated with phthalic anhydride in concentrated sulphuric acid and the hot reaction mixture is poured into a dilute solution of sodium hydroxide, the product formed is [MP PET 1997, 2003; RPMT 1999; KCET (Med.) 2000; CPMT 1981; CBSE PMT 1988]
A)
Alizarin done
clear
B)
Methyl orange done
clear
C)
Fluorescein done
clear
D)
Phenolphthalein done
clear
View Solution play_arrow
-
question_answer31)
\[C{{H}_{3}}-CH=CH-CH(OH)-C{{H}_{3}}\underset{\text{reagent}}{\mathop{\xrightarrow{\text{Jon }\!\!'\!\!\text{ s}}}}\,\]X, Product X is [RPET 2000]
A)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}CH(OH)C{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}CH=CHCOC{{H}_{3}}\] done
clear
C)
Both A and B are correct done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}COC{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer32)
Reaction : \[C{{H}_{3}}OH+{{O}_{2}}\underset{Ag}{\mathop{\xrightarrow{{{600}^{0}}C}}}\,\]product The product is [RPET 2000]
A)
\[C{{H}_{2}}=C=O\] done
clear
B)
\[{{H}_{2}}C=O\] done
clear
C)
\[{{C}_{2}}{{H}_{4}}\] done
clear
D)
\[{{C}_{2}}{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer33)
Ethylene glycol, on oxidation with per-iodic acid, gives [NCERT 1983; CPMT 1983]
A)
Oxalic acid done
clear
B)
Glycol done
clear
C)
Formaldehyde done
clear
D)
Glycollic acid done
clear
View Solution play_arrow
-
question_answer34)
An unknown compound ?D?, first oxidised to aldehyde and then acitic acid by a dilute solution of\[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\]and\[{{H}_{2}}S{{O}_{4}}\]. The unknown compound ?D? is [BHU 2000]
A)
\[C{{H}_{3}}CHO\] done
clear
B)
\[C{{H}_{2}}C{{H}_{3}}OH\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}OH\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer35)
The reaction of ethylene glycol with \[P{{I}_{3}}\]gives [MP PMT 2000]
A)
\[IC{{H}_{2}}C{{H}_{2}}I\] done
clear
B)
\[C{{H}_{2}}=C{{H}_{2}}\] done
clear
C)
\[C{{H}_{2}}=CHI\] done
clear
D)
\[ICH=CHI\] done
clear
View Solution play_arrow
-
question_answer36)
The compound ?A? when treated with ceric ammonium nitrate solution gives yellow ppt. The compound ?A? is [MP PET 2002]
A)
Alcohol done
clear
B)
Aldehyde done
clear
C)
Acid done
clear
D)
Alkane done
clear
View Solution play_arrow
-
question_answer37)
Which of the following product is formed, when ether is exposed to air [AIIMS 2000; RPMT 2002]
A)
Oxide done
clear
B)
Alkanes done
clear
C)
Alkenes done
clear
D)
Peroxide of diethyl ether done
clear
View Solution play_arrow
-
question_answer38)
During dehydration of alcohols to alkenes by heating with conc.\[{{H}_{2}}S{{O}_{4}}\] the initiation step is [AIEEE 2003]
A)
Protonation of alcohol molecule done
clear
B)
Formation of carbocation done
clear
C)
Elimination of water done
clear
D)
Formation of an ester done
clear
View Solution play_arrow
-
question_answer39)
Phenol is less acidic than [MNR 1995]
A)
Ethanol done
clear
B)
Methanol done
clear
C)
o-nitrophenol done
clear
D)
p-methylphenol done
clear
View Solution play_arrow
-
question_answer40)
The compound which gives the most stable carbonium on dehydration is [MNR 1995]
A)
\[\underset{\underset{C{{H}_{3}}\,\,\,\,\,}{\mathop{|\,\,\,\,\,\,\,\,\,}}\,}{\mathop{C{{H}_{3}}-CH-C{{H}_{2}}OH}}\,\] done
clear
B)
\[\begin{matrix} \,\,\,\,\,\,\,C{{H}_{3}} \\ | \\ C{{H}_{3}}-C-OH\,\, \\ | \\ \,\,\,\,\,\,C{{H}_{3}} \\ \end{matrix}\] done
clear
C)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}OH\] done
clear
D)
\[\underset{\underset{OH\,\,\,\,\,\,\,\,\,\,\,\,\,}{\mathop{|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,}}\,}{\mathop{C{{H}_{3}}-CH-C{{H}_{2}}-C{{H}_{3}}}}\,\] done
clear
View Solution play_arrow
-
question_answer41)
At higher temperature, iodoform reaction is given by [AIIMS 2003]
A)
\[C{{H}_{3}}C{{O}_{2}}C{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}C{{O}_{2}}{{C}_{2}}{{H}_{5}}\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}C{{O}_{2}}C{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}C{{O}_{2}}{{C}_{6}}{{H}_{5}}\] done
clear
View Solution play_arrow
-
question_answer42)
Cresol has [CPMT 2003]
A)
Alcoholic ? \[OH\] done
clear
B)
Phenolic ? \[OH\] done
clear
C)
? \[COOH\] done
clear
D)
? \[CHO\] done
clear
View Solution play_arrow
-
question_answer43)
In \[C{{H}_{3}}C{{H}_{2}}OH\underset{{{350}^{o}}C}{\mathop{\xrightarrow{X}}}\,C{{H}_{2}}=C{{H}_{2}}+{{H}_{2}}O;\] 'X' is
A)
NaCl done
clear
B)
\[CaC{{l}_{2}}\] done
clear
C)
\[{{P}_{2}}{{O}_{5}}\] done
clear
D)
\[A{{l}_{2}}{{O}_{3}}\] done
clear
View Solution play_arrow
-
question_answer44)
Sodium phenoxide reacts with \[C{{O}_{2}}\] at \[400K\] and 4-7 atm pressure to give [MP PET 1996]
A)
Sodium salicylate done
clear
B)
Salicylaldehyde done
clear
C)
Catechol done
clear
D)
Benzoic acid done
clear
View Solution play_arrow
-
question_answer45)
The reaction of \[{{C}_{2}}{{H}_{5}}OH\] with \[{{H}_{2}}S{{O}_{4}}\] does not give [MP PET 1996]
A)
Ethylene done
clear
B)
Diethyl ether done
clear
C)
Acetylene done
clear
D)
Ethyl hydrogen sulphate done
clear
View Solution play_arrow
-
question_answer46)
The order of stability of carbonium ions is [MP PET 1996]
A)
Methyl > ethyl > iso-propyl > tert-butyl done
clear
B)
Tert-butyl > iso-propyl > ethyl > methyl done
clear
C)
Iso-propyl > tert-butyl > ethyl > methyl done
clear
D)
Tert-butyl > ethyl > iso-propyl > methyl done
clear
View Solution play_arrow
-
question_answer47)
Which statement is not correct about alcohol [AFMC 1997]
A)
Alcohol is lighter than water done
clear
B)
Alcohol evaporates quickly done
clear
C)
Alcohol of less no. of carbon atoms is less soluble in water than alcohol of high no. of carbon atoms done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer48)
An organic compound A reacts with sodium metal and forms B. On heating with conc. \[{{H}_{2}}S{{O}_{4}},\] A gives diethyl ether. A and B are [AFMC 1998]
A)
\[{{C}_{2}}{{H}_{5}}OH\] and \[{{C}_{2}}{{H}_{5}}ONa\] done
clear
B)
\[{{C}_{3}}{{H}_{7}}OH\] and \[C{{H}_{3}}ONa\] done
clear
C)
\[C{{H}_{3}}OH\] and \[C{{H}_{3}}ONa\] done
clear
D)
\[{{C}_{4}}{{H}_{9}}OH\] and \[{{C}_{4}}{{H}_{9}}ONa\] done
clear
View Solution play_arrow
-
question_answer49)
In the Liebermann's nitroso reaction, sequential changes in the colour of phenol occurs as [AFMC 1998; BHU 1999]
A)
Brown or red \[\to \] green \[\to \] red \[\to \] deep blue done
clear
B)
Red \[\to \] deep blue \[\to \] green done
clear
C)
Red \[\to \] green \[\to \] white done
clear
D)
White \[\to \] red \[\to \] green done
clear
View Solution play_arrow
-
question_answer50)
Which one of the following reactions does not yield an alkyl halide [EAMCET 1998]
A)
Diethyl ether \[+C{{l}_{2}}\] done
clear
B)
Diethyl ether \[+HI\] done
clear
C)
Diethyl ether and \[PC{{l}_{5}}\] done
clear
D)
Diethyl ether \[\xrightarrow{\text{Reduction}}X\xrightarrow{S{{O}_{2}}C{{l}_{2}}}\] done
clear
View Solution play_arrow
-
question_answer51)
Compound A reacts with \[PC{{l}_{5}}\]to give B which on treatment with KCN followed by hydrolysis gave propionic acid. What is A and B respectively [EAMCET 1998]
A)
\[{{C}_{3}}{{H}_{8}}\] and \[{{C}_{3}}{{H}_{7}}Cl\] done
clear
B)
\[{{C}_{2}}{{H}_{6}}\] and \[{{C}_{2}}{{H}_{5}}Cl\] done
clear
C)
\[{{C}_{2}}{{H}_{5}}Cl\] and \[{{C}_{2}}{{H}_{5}}Cl\] done
clear
D)
\[{{C}_{2}}{{H}_{5}}OH\] and \[{{C}_{2}}{{H}_{5}}Cl\] done
clear
View Solution play_arrow
-
question_answer52)
The increasing order of acidity among phenol, p-methylphenol, m-nitrophenol and p-nitrophenol is [CBSE PMT 1995; RPMT 2002]
A)
m-nitrophenol, p-nitrophenol, phenol, p-methylphenol done
clear
B)
p-methylphenol, m-nitrophenol, phenol, p-nitrophenol done
clear
C)
p-methylphenol, phenol, m-nitrophenol, p-nitrophenol done
clear
D)
Phenol, p-methylphenol, p-nitrophenol, m-nitrophenol done
clear
View Solution play_arrow
-
question_answer53)
Which of the following is not characteristic of alcohols [AFMC 1992]
A)
Lower alcohols are stronger and have bitter taste done
clear
B)
Higher alcohols are stronger and have bitter taste done
clear
C)
The boiling points of alcohols increase with increasing molecular mass done
clear
D)
The lower alcohols are soluble in water done
clear
View Solution play_arrow
-
question_answer54)
In reaction of alcohols with alkali metal, acid etc. which of the following alcohol will react fastest [BHU 1984]
A)
Secondary done
clear
B)
Tertiary done
clear
C)
Primary done
clear
D)
All equal done
clear
View Solution play_arrow
-
question_answer55)
Order of reactivity of alcohols towards sodium metal is [Pb. CET 1985]
A)
Pri > Sec > Ter done
clear
B)
Pri > Sec < Ter done
clear
C)
Pri < Sec > Ter done
clear
D)
Pri < Sec < Ter done
clear
View Solution play_arrow
-
question_answer56)
23 g of Na will react with methyl alcohol to give [NCERT 1972]
A)
One mole of oxygen done
clear
B)
One mole of \[{{H}_{2}}\] done
clear
C)
\[\frac{1}{2}\text{ mole of }{{H}_{2}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer57)
Which reagent is useful in converting 1-butanol to 1-bromobutane [EAMCET 1989]
A)
\[CHB{{r}_{3}}\] done
clear
B)
\[B{{r}_{2}}\] done
clear
C)
\[C{{H}_{3}}Br\] done
clear
D)
\[PB{{r}_{3}}\] done
clear
View Solution play_arrow
-
question_answer58)
The \[-OH\] group of methyl alcohol cannot be replaced by chlorine by the action of [KCET 1989]
A)
Chlorine done
clear
B)
Hydrogen chloride done
clear
C)
Phosphorus trichloride done
clear
D)
Phosphorus pentachloride done
clear
View Solution play_arrow
-
question_answer59)
Which of the following gives ketone on oxidation [EAMCET 1987; BIT 1992]
A)
\[{{(C{{H}_{3}})}_{3}}COH\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OH\] done
clear
C)
\[{{(C{{H}_{3}})}_{2}}CHC{{H}_{2}}OH\] done
clear
D)
\[C{{H}_{3}}CHOHC{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer60)
Phenol is treated with bromine water and shaken well. The white precipitate formed during the process is [KCET (Med.) 2001; BIT 1992; AIIMS 1996; KCET 2001]
A)
m-bromophenol done
clear
B)
2, 4-dibromophenol done
clear
C)
2, 4, 6-tribromophenol done
clear
D)
A mixture of o- and p-bromophenols done
clear
View Solution play_arrow
-
question_answer61)
Which compound has the highest boiling point [MP PET 2003]
A)
Acetone done
clear
B)
Diethyl ether done
clear
C)
Methanol done
clear
D)
Ethanol done
clear
View Solution play_arrow
-
question_answer62)
When vapour of ethanol are passed over platinised asbestos in excess of air, the compound formed is [CPMT 1983]
A)
\[C{{H}_{3}}CHO\] done
clear
B)
\[C{{H}_{3}}COC{{H}_{3}}\] done
clear
C)
\[{{C}_{2}}{{H}_{2}}\] done
clear
D)
\[C{{H}_{3}}COOH\] done
clear
View Solution play_arrow
-
question_answer63)
Dehydration of ethanol gives [CPMT 1985; BHU 1989]
A)
Acetic acid done
clear
B)
Ethane done
clear
C)
Ethylene done
clear
D)
Acetylene done
clear
View Solution play_arrow
-
question_answer64)
Which of the following compound will give positive iodoform test [MP PMT 1986, 99; SCRA 1991; CPMT 1994]
A)
\[C{{H}_{3}}OH\] done
clear
B)
\[\underset{\underset{C{{H}_{3}}}{\mathop{|}}\,}{\mathop{\overset{\overset{H}{\mathop{|}}\,}{\mathop{C{{H}_{3}}-C-OH}}\,}}\,\] done
clear
C)
\[\underset{\underset{C{{H}_{3}}}{\mathop{|}}\,}{\mathop{\overset{\overset{C{{H}_{3}}}{\mathop{|}}\,}{\mathop{C{{H}_{3}}-C-OH}}\,}}\,\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OH\] done
clear
View Solution play_arrow
-
question_answer65)
Absolute ethanol cannot be obtained by simple fraction of a solution of ethanol and water because [KCET 1984; MP PMT 1987]
A)
Their B.P.'s are very nearer done
clear
B)
Ethanol remains dissolved in water done
clear
C)
They form a constant boiling mixture done
clear
D)
Ethanol molecules are solvated done
clear
View Solution play_arrow
-
question_answer66)
The alcohol which easily reacts with conc. HCl is [MP PMT 1985]
A)
\[C{{H}_{3}}-CHOH-C{{H}_{2}}-C{{H}_{3}}\] done
clear
B)
\[{{(C{{H}_{3}})}_{3}}-C-OH\] done
clear
C)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-OH\] done
clear
D)
\[{{(C{{H}_{3}})}_{3}}-CH-C{{H}_{2}}OH\] done
clear
View Solution play_arrow
-
question_answer67)
In the following series of chemical reactions, identify Z \[{{C}_{3}}{{H}_{7}}OH\underset{160-{{180}^{o}}C}{\mathop{\xrightarrow{Conc.{{H}_{2}}S{{O}_{4}}}}}\,X\underset{{}}{\mathop{\xrightarrow{B{{r}_{2}}}}}\,Y\underset{Alc.KOH}{\mathop{\xrightarrow{\text{Excess of}}}}\,Z\] [Manipal MEE 1995]
A)
\[\underset{\underset{\,\,\,\,\,\,\,\,\,\,\,\,N{{H}_{2}}\,\,\,\,N{{H}_{2}}}{\mathop{\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,|}}\,}{\mathop{C{{H}_{3}}-CH-C{{H}_{2}}}}\,\] done
clear
B)
\[\underset{\underset{\,\,\,\,\,\,\,\,\,\,OH\,\,\,\,\,OH}{\mathop{\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,|}}\,}{\mathop{C{{H}_{3}}-CH-C{{H}_{2}}}}\,\] done
clear
C)
\[\underset{\underset{OH}{\mathop{|}}\,}{\mathop{C{{H}_{3}}-C=C{{H}_{2}}}}\,\] done
clear
D)
\[C{{H}_{3}}C\equiv CH\] done
clear
View Solution play_arrow
-
question_answer68)
Alcohols of low molecular weight are [CPMT 1976, 89; Pb. PMT 2000]
A)
Soluble in water done
clear
B)
Soluble in all solvents done
clear
C)
Insoluble in all solvents done
clear
D)
Soluble in water on heating done
clear
View Solution play_arrow
-
question_answer69)
Which of the following compounds is oxidised to prepare methyl ethyl ketone [DCE 2001]
A)
2 - propanol done
clear
B)
1 - butanol done
clear
C)
2 - butanol done
clear
D)
Tert-butyl alcohol done
clear
View Solution play_arrow
-
question_answer70)
Which of the following is acidic [CBSE PMT 2001; MH CET 2001]
A)
\[C{{H}_{3}}OH\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}OH\] done
clear
C)
\[{{(C{{H}_{3}})}_{2}}CHOH\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}OH\] done
clear
View Solution play_arrow
-
question_answer71)
With excess bromine, phenol reacts of form [BHU 2001]
A)
B)
C)
D)
Mixture of A and B done
clear
View Solution play_arrow
-
question_answer72)
Which is obtained on treating phenol, with dilute \[HN{{O}_{3}}\] [BVP 2003]
A)
B)
C)
D)
None of these done
clear
View Solution play_arrow
-
question_answer73)
Primary alcohols on dehydration give [NCERT 1986]
A)
Alkenes done
clear
B)
Alkanes done
clear
C)
Both A and B done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer74)
Primary and secondary alcohols on action of reduced copper give [CPMT 1982; MP PMT 1985; EAMCET 1987, 93; MP PET 1995]
A)
Aldehydes and ketones respectively done
clear
B)
Ketones and aldehydes respectively done
clear
C)
Only aldehydes done
clear
D)
Only ketones done
clear
View Solution play_arrow
-
question_answer75)
Methyl alcohol on oxidation with acidified \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] gives [MNR 1987]
A)
\[C{{H}_{3}}COC{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}CHO\] done
clear
C)
\[HCOOH\] done
clear
D)
\[C{{H}_{3}}COOH\] done
clear
View Solution play_arrow
-
question_answer76)
Ethyl alcohol on oxidation with \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] gives [MNR 1987; Bihar CEE 1995; UPSEAT 2000]
A)
Acetic acid done
clear
B)
Acetaldehyde done
clear
C)
Formaldehyde done
clear
D)
Formic acid done
clear
View Solution play_arrow
-
question_answer77)
Lucas test is used for [CBSE PMT 1990; AIIMS 2002; AFMC 2005]
A)
Alcohols done
clear
B)
Amines done
clear
C)
Diethyl ether done
clear
D)
Glacial acetic acid done
clear
View Solution play_arrow
-
question_answer78)
When phenol reacts with ammonia in presence of \[ZnC{{l}_{2}}\]at 300°C, it gives [AFMC 2001]
A)
Primary amine done
clear
B)
Secondary amine done
clear
C)
Tertiary amine done
clear
D)
Both B and C done
clear
View Solution play_arrow
-
question_answer79)
Azo-dyes are prepared from [CPMT 2001]
A)
Aniline done
clear
B)
Benzaldehye done
clear
C)
Benzoic acid done
clear
D)
Phenol done
clear
View Solution play_arrow
-
question_answer80)
A compound that easily undergoes bromination is [KCET (Engg.) 2002]
A)
Phenol done
clear
B)
Toluene done
clear
C)
Benzene done
clear
D)
Benzoic acid done
clear
View Solution play_arrow
-
question_answer81)
Which of the following has lowest boiling point [MH CET 1999]
A)
p-nitrophenol done
clear
B)
m-nitrophenol done
clear
C)
o-nitrophenol done
clear
D)
phenol done
clear
View Solution play_arrow
-
question_answer82)
In esterification, the reactivity of alcohols is [DPMT 2000]
A)
\[1{}^\circ >2{}^\circ >3{}^\circ \] done
clear
B)
\[3{}^\circ >2{}^\circ >1{}^\circ \] done
clear
C)
Same in all cases done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer83)
The role of conc. \[{{H}_{2}}S{{O}_{4}}\]in the esterification process is [RPMT 1999]
A)
Catalyst done
clear
B)
Dehydrating agent done
clear
C)
Hydrolysing agent done
clear
D)
Dehydrating agent and catalyst done
clear
View Solution play_arrow
-
question_answer84)
Methanol and ethanol are distinguished by the [MP PET 1999]
A)
Action of HCl done
clear
B)
Iodoform test done
clear
C)
Solubility in water done
clear
D)
Sodium done
clear
View Solution play_arrow
-
question_answer85)
For phenol, which of the following statements is correct [MP PMT 1995]
A)
It is insoluble in water done
clear
B)
It has lower melting point compared to aromatic hydrocarbons of comparable molecular weight done
clear
C)
It has higher boiling point than toluene done
clear
D)
It does not show acidic property done
clear
View Solution play_arrow
-
question_answer86)
The reaction of Lucas reagent is fast with [MP PMT 2000]
A)
\[{{\left( C{{H}_{3}} \right)}_{3}}COH\] done
clear
B)
\[{{(C{{H}_{3}})}_{2}}CHOH\] done
clear
C)
\[C{{H}_{3}}{{(C{{H}_{2}})}_{2}}OH\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}OH\] done
clear
View Solution play_arrow
-
question_answer87)
Which of the following reagents convert the propene to 1-propanol [CBSE PMT 2000]
A)
\[{{H}_{2}}O,\ {{H}_{2}}S{{O}_{4}}\] done
clear
B)
Aqueous \[KOH\] done
clear
C)
\[MgS{{O}_{4}},\ NaB{{H}_{4}}/{{H}_{2}}O\] done
clear
D)
\[{{B}_{2}}{{H}_{6}},\ {{H}_{2}}{{O}_{2}},\ O{{H}^{-}}\] done
clear
View Solution play_arrow
-
question_answer88)
Compound ?A? reacts with \[PC{{l}_{5}}\] to give ?B? which on treatment with \[KCN\]followed by hydrolysis gave propanoic acid as the product. What is ?A? [CBSE PMT 2002]
A)
Ethane done
clear
B)
Propane done
clear
C)
Ethyl chloride done
clear
D)
Ethyl alcohol done
clear
View Solution play_arrow
-
question_answer89)
Which reagent can convert acetic acid into ethanol [BVP 2003]
A)
\[Na+\]alcohol done
clear
B)
\[LiAl{{H}_{4}}+\] ether done
clear
C)
\[{{H}_{2}}+Pt\] done
clear
D)
\[Sn+HCl\] done
clear
View Solution play_arrow
-
question_answer90)
Which of the following would undergo dehydration most readily [UPSEAT 2000]
A)
1-phenyl-1butanol done
clear
B)
2-phenyl-2-butanol done
clear
C)
1-phenyl-2-butanol done
clear
D)
2-phenyl-1-butanol done
clear
View Solution play_arrow
-
question_answer91)
Phenol and benzoic acid is distinguished by [BHU 2003]
A)
\[NaOH\] done
clear
B)
\[NaHC{{O}_{3}}\] done
clear
C)
\[N{{a}_{2}}C{{O}_{3}}\] done
clear
D)
\[{{H}_{2}}S{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer92)
Electrophilic substitution reaction in phenol take place at [RPMT 2002]
A)
p- position done
clear
B)
m- position done
clear
C)
o- position done
clear
D)
o- and p- position done
clear
View Solution play_arrow
-
question_answer93)
Liebermann's test is answered by [KCET 1998]
A)
Aniline done
clear
B)
Methylamine done
clear
C)
Ethyl benzoate done
clear
D)
Phenol done
clear
View Solution play_arrow
-
question_answer94)
In the sequence of the following reactions [MP PMT 2002] \[C{{H}_{3}}C{{H}_{2}}OH\]\[\underset{\text{ChromicAcid}}{\mathop{\xrightarrow{[\text{O }\!\!]\!\!\text{ }}}}\,X\underset{\text{ChromicAcid}}{\mathop{\xrightarrow{[O]}}}\,\]\[C{{H}_{3}}COOH\] X is
A)
\[C{{H}_{3}}COC{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}CHO\] done
clear
C)
\[C{{H}_{3}}OC{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}COOH\] done
clear
View Solution play_arrow
-
question_answer95)
The boiling point of glycerol is more than propanol because of [CPMT 1997, 2002]
A)
Hydrogen bonding done
clear
B)
Hybridisation done
clear
C)
Resonance done
clear
D)
All the above done
clear
View Solution play_arrow
-
question_answer96)
Which of the following produces violet colour with \[FeC{{l}_{3}}\] solution
A)
Enols done
clear
B)
Ethanol done
clear
C)
Ethanal done
clear
D)
Alkyl halides done
clear
View Solution play_arrow
-
question_answer97)
When heated with \[N{{H}_{3}}\] under pressure alone or in presence of zinc chloride phenols are converted into [RPMT 1997]
A)
Aminophenols done
clear
B)
Aniline done
clear
C)
Nitrobenzene done
clear
D)
Phenyl hydroxylamine done
clear
View Solution play_arrow
-
question_answer98)
Because of resonance the oxygen atom of \[-OH\] group of phenol
A)
Acquires positive charge done
clear
B)
Acquires negative charge done
clear
C)
Remains uneffected done
clear
D)
Liberates done
clear
View Solution play_arrow
-
question_answer99)
When glycerol is heated with \[KHS{{O}_{4}}\] it gives [CPMT 1974, 85; MP PMT 1988, 90, 91, 92, 94; MP PET 1988, 92]
A)
\[C{{H}_{2}}=CH-C{{H}_{3}}\] done
clear
B)
\[C{{H}_{2}}=CH-C{{H}_{2}}OH\] done
clear
C)
\[C{{H}_{2}}=CH-CHO\] done
clear
D)
\[C{{H}_{2}}=C=C{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer100)
An organic compound X on treatment with acidified \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\] gives a compound Y which reacts with \[{{I}_{2}}\] and sodium carbonate to form tri-odomethane. The compound X is [KCET 1996]
A)
\[C{{H}_{3}}OH\] done
clear
B)
\[C{{H}_{3}}-CO-C{{H}_{3}}\] done
clear
C)
\[C{{H}_{3}}CHO\] done
clear
D)
\[C{{H}_{3}}CH(OH)C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer101)
The reaction of conc. \[HN{{O}_{3}}\] and phenol forms [MP PMT/PET 1988; BHU 1988; MP PMT 1999; Pb. PMT 2000]
A)
Benzoic acid done
clear
B)
Salicylic acid done
clear
C)
o-and p-nitrophenol done
clear
D)
Picric acid done
clear
View Solution play_arrow
-
question_answer102)
Phenol is [MP PMT 1990; UPSEAT 1999]
A)
A weaker base than \[N{{H}_{3}}\] done
clear
B)
Stronger than carbonic acid done
clear
C)
Weaker than carbonic acid done
clear
D)
A neutral compound done
clear
View Solution play_arrow
-
question_answer103)
Phenol at \[{{25}^{o}}C\] is
A)
A white crystalline solid done
clear
B)
A transparent liquid done
clear
C)
A gas done
clear
D)
Yellow solution done
clear
View Solution play_arrow
-
question_answer104)
At low temperature phenol reacts with \[B{{r}_{2}}\] in \[C{{S}_{2}}\] to form [MP PET 1991; CPMT 1981; MP PMT 1990; IIT 1982; RPMT 2000]
A)
m-bromophenol done
clear
B)
o-and p-bromophenol done
clear
C)
p-bromophenol done
clear
D)
2, 4, 6-tribromophenol done
clear
View Solution play_arrow
-
question_answer105)
Oxidation of ethanol by chromic acid forms [MP PET 1992]
A)
Ethanol done
clear
B)
Methanol done
clear
C)
2-propanone done
clear
D)
Ethanoic acid done
clear
View Solution play_arrow
-
question_answer106)
Which of the following not gives effervescence with \[NaHC{{O}_{3}}\] [MP PET 1992]
A)
Phenol done
clear
B)
Benzoic acid done
clear
C)
2, 4-dinitrophenol done
clear
D)
2, 4, 6-trinitrophenol done
clear
View Solution play_arrow
-
question_answer107)
Conc. \[{{H}_{2}}S{{O}_{4}}\] reacts with \[{{C}_{2}}{{H}_{5}}OH\] at \[{{170}^{o}}C\] to form [MP PMT 1991; MP PET 1991; IIT-JEE 1981; EAMCET 1979; KCET 2001]
A)
\[C{{H}_{3}}COC{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}COOH\] done
clear
C)
\[C{{H}_{3}}CHO\] done
clear
D)
\[{{C}_{2}}{{H}_{4}}\] done
clear
View Solution play_arrow
-
question_answer108)
Which compound has hydrogen bonding [MP PMT 1992; MP PET 1991]
A)
Toluene done
clear
B)
Phenol done
clear
C)
Chlorobenzene done
clear
D)
Nitrobenzene done
clear
View Solution play_arrow
-
question_answer109)
Which statement is true [MP PMT 1991]
A)
\[{{C}_{6}}{{H}_{5}}OH\] is more acidic than \[{{C}_{2}}{{H}_{5}}OH\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}OH\] is less acidic than \[{{C}_{2}}{{H}_{5}}OH\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}OH\] react with \[NaHC{{O}_{3}}\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}OH\] gives oxime with \[N{{H}_{2}}OH\] and \[HCl\] done
clear
View Solution play_arrow
-
question_answer110)
Read the following statements carefully : A secondary alcohol on oxidation gives a ketone Ethanol reacts with conc. \[{{H}_{2}}S{{O}_{4}}\] at \[{{180}^{o}}C\] to yield ethylene Methanol reacts with iodine and sodium hydroxide to give a yellow precipitate of iodoform Hydrogen gas is liberated when sodium is added to alcohol. Select the correct statements from the above set:
A)
A, B done
clear
B)
C, D done
clear
C)
A, B, D done
clear
D)
A, C, D done
clear
View Solution play_arrow
-
question_answer111)
The following reaction :
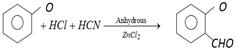
is known as [MP PET 1997]
A)
Perkin reaction done
clear
B)
Gattermann reaction done
clear
C)
Kolbe reaction done
clear
D)
Gattermann-Koch reaction done
clear
View Solution play_arrow
-
question_answer112)
Carbylamine test is done by heating alcoholic KOH with [IIT-JEE 1984; BIT 1992; CBSE PMT 1992]
A)
Chloroform and silver powder done
clear
B)
Trihalogen methane and primary amine done
clear
C)
Alkyl halide and primary amine done
clear
D)
Alkyl cyanide and primary amine done
clear
View Solution play_arrow
-
question_answer113)
Isopropyl alcohol heated at \[{{300}^{o}}C\] with copper catalyst to form [AFMC 1990; MP PMT 1986, 89, 92; JIPMER 2000]
A)
Acetone done
clear
B)
Dimethyl ether done
clear
C)
Acetaldehyde done
clear
D)
Ethane done
clear
View Solution play_arrow
-
question_answer114)
Dehydrogenation of \[\underset{\underset{OH}{\mathop{|}}\,}{\mathop{C{{H}_{3}}-CH-C{{H}_{3}}}}\,\] gives [MP PMT 2002]
A)
Acetone done
clear
B)
Acetaldehyde done
clear
C)
Acetic acid done
clear
D)
Acetylene done
clear
View Solution play_arrow
-
question_answer115)
In the sequence of the following reactions \[C{{H}_{3}}OH\xrightarrow{HI}C{{H}_{3}}I\xrightarrow{KCN}\]\[C{{H}_{3}}CN\xrightarrow{\text{reduction}}X\xrightarrow{HN{{O}_{3}}}Y\] X and Y are respectively [MP PMT 2002]
A)
\[C{{H}_{3}}C{{H}_{2}}N{{H}_{2}}\] and \[C{{H}_{3}}C{{H}_{2}}OH\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}N{{H}_{2}}\] and \[C{{H}_{3}}COOH\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}OH\] and \[C{{H}_{3}}CHO\] done
clear
D)
\[C{{H}_{3}}OC{{H}_{3}}\] and \[C{{H}_{3}}CHO\] done
clear
View Solution play_arrow
-
question_answer116)
Alcohols (i) \[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OH,\] (ii) \[C{{H}_{3}}-CHOH-C{{H}_{3}}\] and (iii) \[C{{H}_{3}}-C(C{{H}_{3}})(OH)-C{{H}_{3}}\] were treated with Lucas reagent (Conc. \[HCl+ZnC{{l}_{2}}).\] What results do you expect at room temperature
A)
(ii) and (iii) react immediately and (i) in about 5 minutes done
clear
B)
(iii) reacts immediately, (ii) reacts in about 5 minutes and (i) not at all done
clear
C)
(i) reacts immediately, (ii) reacts in about 5 minutes and (iii) not at all done
clear
D)
(i) reacts in about 5 minutes, (ii) reacts in about 15 minutes and (iii) not at all done
clear
View Solution play_arrow
-
question_answer117)
Ethylene may be obtained by dehydration of which of the following with concentrated \[{{H}_{2}}S{{O}_{4}}\]at 160 ? 170°C [DPMT 2000; MP PET 2001]
A)
\[{{C}_{2}}{{H}_{5}}OH\] done
clear
B)
\[C{{H}_{3}}OH\] done
clear
C)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OH\] done
clear
D)
\[{{(C{{H}_{3}})}_{2}}CHC{{H}_{2}}OH\] done
clear
View Solution play_arrow
-
question_answer118)
The final product of the oxidation of ethyl alcohol is [KCET (Med.) 1999 ]
A)
Ethane done
clear
B)
Acetone done
clear
C)
Acetaldehyde done
clear
D)
Acetic acid done
clear
View Solution play_arrow
-
question_answer119)
The compound obtained by heating salicylic acid with phenol in the presence of phosphorus oxychloride is [KCET (Med.) 1999]
A)
Salol done
clear
B)
Aspirin done
clear
C)
Oil of wintergreen done
clear
D)
o-chlorobenzoyl chloride done
clear
View Solution play_arrow
-
question_answer120)
When phenol is allowed to react with \[B{{r}_{2}}\] in (i) \[C{{S}_{2}}\] solution and (ii) in aqueous solution, the resulting compounds are
A)
(i) 2, 4, 6-tribromophenol and (ii) o-and p-bromophenol done
clear
B)
(i) m-bromophenol and (ii) 2, 3, 4-tribromophenol done
clear
C)
s (i) o-and p-bromophenol and (ii) 2, 4, 6-tribromophenol done
clear
D)
(i) o- and m-bromophenol and (ii) 2, 3, 4-tribromophenol done
clear
View Solution play_arrow
-
question_answer121)
Which of the following is not true in case of reaction with heated copper at \[{{300}^{o}}C\] [CPMT 1999]
A)
Phenol \[\to \] Benzyl alcohol done
clear
B)
Primary alcohol \[\to \] Aldehyde done
clear
C)
Secondary alcohol \[\to \] Ketone done
clear
D)
Tertiary alcohol \[\to \] Olefin done
clear
View Solution play_arrow
-
question_answer122)
Which of the following is the most suitable method for removing the traces of water from ethanol [CPMT 1999]
A)
Heating with Na metal done
clear
B)
Passing dry HCl through it done
clear
C)
Distilling it done
clear
D)
Reacting with Mg done
clear
View Solution play_arrow
-
question_answer123)
With oxalic acid, glycerol at \[{{260}^{o}}C\] gives [BHU 1996]
A)
Allyl alcohol done
clear
B)
Glyceryl mono-oxalate done
clear
C)
Formic acid done
clear
D)
Glyceraldehyde done
clear
View Solution play_arrow
-
question_answer124)
Absolute alcohol cannot be prepared by fractional distillation of rectified spirit since
A)
It forms azeotropic mixture done
clear
B)
It is used as power alcohol done
clear
C)
It is used in wines done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer125)
The reagent used for the dehydration of an alcohol is [MP PET/PMT 1998]
A)
Phosphorus pentachloride done
clear
B)
Calcium chloride done
clear
C)
Aluminium oxide done
clear
D)
Sodium chloride done
clear
View Solution play_arrow
-
question_answer126)
Which one of the following compounds gives a positive iodoform test [MP PMT 1997]
A)
Pentanal done
clear
B)
1-phenyl ethanol done
clear
C)
2-phenyl ethanol done
clear
D)
3-pentanol done
clear
View Solution play_arrow
-
question_answer127)
What amount of bromine will be required to convert 2 g of phenol into 2, 4, 6-tribromophenol [MP PET/PMT 1998]
A)
4.00 done
clear
B)
6.00 done
clear
C)
10.22 done
clear
D)
20.44 done
clear
View Solution play_arrow
-
question_answer128)
Ethyl alcohol exhibits acidic character on reacting with [MP PMT 1995]
A)
Acetic acid done
clear
B)
Sodium metal done
clear
C)
Hydrogen iodide done
clear
D)
Acidic potassium dichromate done
clear
View Solution play_arrow
-
question_answer129)
The mixture of ethanol and water cannot be separated by distillation because [KCET 1984]
A)
They form a constant boiling mixture done
clear
B)
Alcohol molecules are solvated done
clear
C)
Their boiling points are very near done
clear
D)
Alcohol remains dissolved in water done
clear
View Solution play_arrow
-
question_answer130)
The reaction between an alcohol and an acid with the elimination of water molecule is called [MH CET 1999]
A)
Esterification done
clear
B)
Saponification done
clear
C)
Etherification done
clear
D)
Elimination done
clear
View Solution play_arrow
-
question_answer131)
The compound with the highest boiling point is [MNR 1985]
A)
\[C{{H}_{4}}\] done
clear
B)
\[C{{H}_{3}}OH\] done
clear
C)
\[C{{H}_{3}}Cl\] done
clear
D)
\[C{{H}_{3}}Br\] done
clear
View Solution play_arrow
-
question_answer132)
The boiling point of ethyl alcohol should be less than that of [Pb. CET 1985]
A)
Propane done
clear
B)
Formic acid done
clear
C)
Dimethyl ether done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer133)
Which of the following is not characteristic of alcohols [AIIMS 1980]
A)
They are lighter than water done
clear
B)
Their boiling points rise fairly uniformly with increasing molecular weight done
clear
C)
Lower members are insoluble in water and organic solvents but solubility regularly increases with molecular weight done
clear
D)
Lower members have pleasant smell and burning taste, while higher members are odorless and tasteless done
clear
View Solution play_arrow
-
question_answer134)
At room temperature the alcohol that do not reacts with Lucas reagent is
A)
Primary alcohol done
clear
B)
Secondary alcohol done
clear
C)
Tertiary alcohol done
clear
D)
All these three done
clear
View Solution play_arrow
-
question_answer135)
By means of calcium chloride which of following can be dried
A)
Methanol done
clear
B)
Ethanol done
clear
C)
Both A and B done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer136)
Lucas test is used to distinguish between [MP PET 1994]
A)
\[{{1}^{o}},\,{{2}^{o}}\] and \[{{3}^{o}}\] alcohols done
clear
B)
\[{{1}^{o}},\,\,{{2}^{o}}\] and \[{{3}^{o}}\] amines done
clear
C)
Aldehydes and ketones done
clear
D)
Alkenes and alkynes done
clear
View Solution play_arrow
-
question_answer137)
Among the following, the compound that undergoes nitration readily is [NCERT 1984]
A)
Benzoic acid done
clear
B)
Toluene done
clear
C)
Phenol done
clear
D)
Nitrobenzene done
clear
View Solution play_arrow
-
question_answer138)
\[\text{Phenol }\underset{\text{Distillation}}{\mathop{\xrightarrow{Zn}}}\,A\underset{\text{Conc}\text{. }HN{{O}_{3}}}{\mathop{\xrightarrow{\text{Conc}\text{. }{{H}_{2}}S{{O}_{4}}}}}\,B\underset{NaOH}{\mathop{\xrightarrow{Zn}}}\,C\]In the above reaction A, B and C are the following compounds [MP PMT/PET 1988]
A)
\[{{C}_{6}}{{H}_{6}},{{C}_{6}}{{H}_{5}}N{{O}_{2}}\] and aniline done
clear
B)
\[{{C}_{6}}{{H}_{6}},\] dinitrobenzene and metanitroaniline done
clear
C)
Toluene, metanitrobenzene and metatoluedine done
clear
D)
\[{{C}_{6}}{{H}_{6}},\,{{C}_{6}}{{H}_{5}}N{{O}_{2}}\] and hydrazobenzene done
clear
View Solution play_arrow
-
question_answer139)
\[C{{H}_{3}}-O-{{C}_{3}}{{H}_{7}}\] and \[{{C}_{2}}{{H}_{5}}-O-{{C}_{2}}{{H}_{5}}\] exhibit which type of isomerism [MP PMT 1989]
A)
Metamerism done
clear
B)
Position done
clear
C)
Chain done
clear
D)
Functional done
clear
View Solution play_arrow
-
question_answer140)
Phenol reacts with \[CC{{l}_{4}}\] in presence of aqueous alkali and forms a product which on hydrolysis gives [MP PMT 1990]
A)
Salicylaldehyde done
clear
B)
Salicylic acid done
clear
C)
Benzaldehyde done
clear
D)
Benzoic acid done
clear
View Solution play_arrow
-
question_answer141)
In fermentation by zymase, alcohol and \[C{{O}_{2}}\] are obtained from the following sugar [MP PMT/PET 1988]
A)
Glucose done
clear
B)
Invert sugar done
clear
C)
Fructose done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer142)
The order of melting point of ortho, para, meta-nitrophenol is [Orissa JEE 2003]
A)
o > m > p done
clear
B)
p > m > o done
clear
C)
m > p > o done
clear
D)
p > o > m done
clear
View Solution play_arrow
-
question_answer143)
The alcohol which does not give a stable compound on dehydration is [MP PET 1997]
A)
Ethyl alcohol done
clear
B)
Methyl alcohol done
clear
C)
n-propyl alcohol done
clear
D)
n-butyl alcohol done
clear
View Solution play_arrow
-
question_answer144)
When ethyl alcohol \[({{C}_{2}}{{H}_{5}}OH)\] is mixed with ammonia and passed over heated alumina, the compound formed is [DPMT 1981; CBSE PMT 1989]
A)
\[{{C}_{2}}{{H}_{5}}N{{H}_{2}}\] done
clear
B)
\[{{C}_{2}}{{H}_{4}}\] done
clear
C)
\[{{C}_{2}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}\] done
clear
D)
\[C{{H}_{3}}OC{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer145)
A mixture of methanol vapours and air is passed over heated copper. The products are [KCET 1988]
A)
Carbon monoxide and hydrogen done
clear
B)
Formaldehyde and water vapour done
clear
C)
Formic acid and water vapour done
clear
D)
Carbon monoxide and water vapour done
clear
View Solution play_arrow
-
question_answer146)
In the esterification reaction of alcohols [Bihar CEE 1995]
A)
\[O{{H}^{-}}\] is replaced by \[C{{H}_{3}}COO\] group done
clear
B)
\[O{{H}^{-}}\] is replaced by chlorine done
clear
C)
\[{{H}^{-}}\] is replaced by sodium metal done
clear
D)
\[O{{H}^{-}}\]is replaced by \[{{C}_{2}}{{H}_{5}}OH\] done
clear
View Solution play_arrow
-
question_answer147)
A compound A on oxidation gave acetaldehyde, then again on oxidation gave acid. After first oxidation it was reacted with ammoniacal \[AgN{{O}_{3}}\] then silver mirror was produced. A is likely to be [DPMT 1996]
A)
Primary alcohol done
clear
B)
Tertiary alcohol done
clear
C)
Acetaldehyde done
clear
D)
Acetone done
clear
View Solution play_arrow
-
question_answer148)
Phenol \[\underset{{{\text{H}}^{+}}}{\mathop{\xrightarrow{\text{CHCl}{{ & }_{\text{3}}}\text{/NaOH}}}}\,\]Salicyldehyde The above reaction is known as [Pb. PMT 2002]
A)
Riemer Tiemann reaction done
clear
B)
Bucherer reaction done
clear
C)
Gattermann synthesis done
clear
D)
Perkin reaction done
clear
View Solution play_arrow
-
question_answer149)
Alcohol which gives red colour with Victor Meyer test is [RPMT 2003]
A)
\[{{C}_{2}}{{H}_{5}}OH\] done
clear
B)
\[C{{H}_{3}}-\underset{OH\,\,\,\,\,\,\,\,\,\,\,\,\,}{\mathop{\underset{|\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,}{\mathop{CH-C{{H}_{3}}}}\,}}\,\] done
clear
C)
\[C{{(C{{H}_{3}})}_{3}}OH\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer150)
Conc. \[{{H}_{2}}S{{O}_{4}}\] heated with excess of \[{{C}_{2}}{{H}_{5}}OH\] at \[{{140}^{o}}C\] to form [MP PMT 1990; RPMT 2000; AFMC 2002]
A)
\[C{{H}_{3}}C{{H}_{2}}-O-C{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}-O-C{{H}_{2}}C{{H}_{3}}\] done
clear
C)
\[C{{H}_{3}}-O-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{3}}\] done
clear
D)
\[C{{H}_{2}}=C{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer151)
Rate of substitution reaction in phenol is [MP PMT 1989]
A)
Slower than the rate of benzene done
clear
B)
Faster than the rate of benzene done
clear
C)
Equal to the rate of benzene done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer152)
Phenol reacts with dilute \[HN{{O}_{3}}\] at normal temperature to form [MP PMT 1989]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer153)
One mole of phenol reacts with bromine to form tribromophenol. How much bromine is used [MP PMT 1989]
A)
1. 5 mol done
clear
B)
3 mol done
clear
C)
4.5 mol done
clear
D)
6 mol done
clear
View Solution play_arrow
-
question_answer154)
In presence of NaOH, phenol react with \[CHC{{l}_{3}}\] to form o-hydroxy benzaldehyde. This reaction is called [BIT 1992; MP PMT 1990, 2002; AIIMS 1992; MP PET 1994; JIPMER 1999]
A)
Riemer-Tiemann's reaction done
clear
B)
Sandmeyer's reaction done
clear
C)
Hoffmann's degradation reaction done
clear
D)
Gattermann's aldehyde synthesis done
clear
View Solution play_arrow
-
question_answer155)
Which of the following vapours passed over heated copper to form acetone [BIT 1992]
A)
\[{{H}_{3}}C-C{{H}_{2}}-C{{H}_{2}}OH\] done
clear
B)
\[\underset{\underset{OH}{\mathop{|}}\,}{\mathop{C{{H}_{3}}-CH-C{{H}_{3}}}}\,\] done
clear
C)
\[\begin{matrix} C{{H}_{3}} & - & {} & {} & {} \\ C{{H}_{3}} & > & C & - & OH \\ C{{H}_{3}} & {} & {} & {} & {} \\ \end{matrix}\] done
clear
D)
\[C{{H}_{2}}=CH-C{{H}_{2}}OH\] done
clear
View Solution play_arrow
-
question_answer156)
Methyl alcohol (methanol), ethyl alcohol (ethanol) and acetone (propanone) were treated with iodine and sodium hydroxide solutions. Which substances will give iodoform test
A)
Only ethyl alcohol done
clear
B)
Only methyl alcohol and ethyl alcohol done
clear
C)
Only ethyl alcohol and acetone done
clear
D)
Only acetone done
clear
View Solution play_arrow
-
question_answer157)
TNT has the structure [UPSEAT 2000]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer158)
The vapour pressure of aqueous solution of methanal is [UPSEAT 2000]
A)
Equal to water done
clear
B)
Equal to methanal done
clear
C)
More than water done
clear
D)
Less than water done
clear
View Solution play_arrow
-
question_answer159)
Glycerol reacts with conc. \[HN{{O}_{3}}\] and conc. \[{{H}_{2}}S{{O}_{4}}\] to form [CPMT 1983; MP PMT/PET 1988]
A)
Glycerol mononitrate done
clear
B)
Glycerol dinitrate done
clear
C)
Glycerol trinitrate done
clear
D)
Acrolein done
clear
View Solution play_arrow
-
question_answer160)
Glycerol heated with oxalic acid at \[{{110}^{o}}C\] to form [CPMT 1986, 90, 91, 97; JIPMER 1997]
A)
Formic acid done
clear
B)
Oxalic acid done
clear
C)
Allyl alcohol done
clear
D)
Glycerol trioxalate done
clear
View Solution play_arrow
-
question_answer161)
Dimethyl ether and ethyl alcohol are [CPMT 1986; Manipal MEE 1995]
A)
Branched isomer done
clear
B)
Position isomer done
clear
C)
Functional isomer done
clear
D)
Tautomer done
clear
View Solution play_arrow
-
question_answer162)
The process of manufacture of absolute alcohol from rectified spirit is [CPMT 1986, 87; Kurukshetra CEE 2002]
A)
Fractional distillation done
clear
B)
Steam distillation done
clear
C)
Azeotropic distillation done
clear
D)
Vacuum distillation done
clear
View Solution play_arrow
-
question_answer163)
When ethyl alcohol reacts with acetic acid, the products formed are [CPMT 1989]
A)
Sodium ethoxide + hydrogen done
clear
B)
Ethyl acetate + water done
clear
C)
Ethyl acetate + soap done
clear
D)
Ethyl alcohol + water done
clear
View Solution play_arrow
-
question_answer164)
Picric acid is (at \[{{25}^{o}}C\])
A)
A white solid done
clear
B)
A colourless liquid done
clear
C)
A gas done
clear
D)
A bright yellow solid done
clear
View Solution play_arrow
-
question_answer165)
Phenol on distillation with zinc dust gives [MP PET 1991; CPMT 1997; MP PMT 1999, 2001; Pb. PMT 2000]
A)
\[{{C}_{6}}{{H}_{6}}\] done
clear
B)
\[{{C}_{6}}{{H}_{12}}\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}O{{C}_{6}}{{H}_{5}}\] done
clear
D)
\[{{C}_{6}}{{H}_{5}}-{{C}_{6}}{{H}_{5}}\] done
clear
View Solution play_arrow
-
question_answer166)
Methanol and ethanol are miscible in water due to [MP PET/PMT 1988; CPMT 1989; CBSE PMT 1991]
A)
Covalent character done
clear
B)
Hydrogen bonding character done
clear
C)
Oxygen bonding character done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer167)
By distilling glycol with fuming sulphuric acid, which of following is obtained
A)
Glycerol done
clear
B)
Pinacol done
clear
C)
Dioxan done
clear
D)
Ethylene oxide done
clear
View Solution play_arrow
-
question_answer168)
The compound which gives the most stable carbonium ion on dehydration is [DCE 2000]
A)
\[C{{H}_{3}}-\underset{\underset{C{{H}_{3}}}{\mathop{|\,\,\,\,\,\,\,}}\,}{\mathop{CH}}\,-C{{H}_{2}}OH\] done
clear
B)
\[C{{H}_{3}}-\underset{\underset{C{{H}_{3}}}{\mathop{|}}\,}{\mathop{\overset{\overset{C{{H}_{3}}}{\mathop{|}}\,}{\mathop{C}}\,}}\,-OH\] done
clear
C)
\[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}OH\] done
clear
D)
\[C{{H}_{3}}-\overset{\overset{C{{H}_{3}}}{\mathop{|\,\,\,\,\,\,}}\,}{\mathop{CH-}}\,C{{H}_{2}}-C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer169)
In \[C{{H}_{3}}C{{H}_{2}}OH\] which bond dissociates heterolytically [IIT-JEE 1988; CPMT 1996]
A)
C ? C done
clear
B)
C ? O done
clear
C)
C ? H done
clear
D)
O ? H done
clear
View Solution play_arrow
-
question_answer170)
Which compound is soluble in water [IIT-JEE 1980; CPMT 1993; RPET 1999]
A)
\[C{{S}_{2}}\] done
clear
B)
\[{{C}_{2}}{{H}_{5}}OH\] done
clear
C)
\[CC{{l}_{4}}\] done
clear
D)
\[CHC{{l}_{3}}\] done
clear
View Solution play_arrow
-
question_answer171)
Which of the following is most soluble in water [MP PMT 1995]
A)
Normal butyl alcohol done
clear
B)
Isobutyl alcohol done
clear
C)
Tertiary butyl alcohol done
clear
D)
Secondary butyl alcohol done
clear
View Solution play_arrow
-
question_answer172)
Which of the following gives negative iodoform test
A)
\[C{{H}_{3}}C{{H}_{2}}OH\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OH\] done
clear
C)
\[\underset{\underset{OH}{\mathop{|}}\,}{\mathop{{{C}_{6}}{{H}_{5}}-CH-C{{H}_{3}}}}\,\] done
clear
D)
\[\underset{\underset{OH}{\mathop{|}}\,}{\mathop{C{{H}_{3}}-CH-C{{H}_{3}}}}\,\] done
clear
View Solution play_arrow
-
question_answer173)
If ethanol dissolves in water, then which of the following would be done [MP PET 1989]
A)
Absorption of heat and contraction in volume done
clear
B)
Emission of heat and contraction in volume done
clear
C)
Absorption of heat and increase in volume done
clear
D)
Emission of heat and increase in volume done
clear
View Solution play_arrow
-
question_answer174)
A migration of hydrogen with a pair of electrons is called
A)
Alkyl shift done
clear
B)
Hydride shift done
clear
C)
Hydrogen ion formation done
clear
D)
Dehydrogenation done
clear
View Solution play_arrow
-
question_answer175)
When rectified spirit and benzene are distilled together, the first fraction obtained is
A)
A ternary azeotrope done
clear
B)
Absolute alcohol done
clear
C)
A binary azeotrope done
clear
D)
Denatured spirit done
clear
View Solution play_arrow
-
question_answer176)
Alcohols react with Grignard reagent to form [DPMT 1986]
A)
Alkanes done
clear
B)
Alkenes done
clear
C)
Alkynes done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer177)
Action of diazomethane on phenol liberates
A)
\[{{O}_{2}}\] done
clear
B)
\[{{H}_{2}}\] done
clear
C)
\[{{N}_{2}}\] done
clear
D)
\[C{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer178)
The ring deuteration of phenol
A)
Lowers the acidity done
clear
B)
Increases the acidity done
clear
C)
Imparts no effect done
clear
D)
Causes amphoteric nature done
clear
View Solution play_arrow
-
question_answer179)
In esterification of an acid, the other reagent is [CPMT 1988]
A)
Aldehyde done
clear
B)
Alcohol done
clear
C)
Amine done
clear
D)
Water done
clear
View Solution play_arrow
-
question_answer180)
Maximum solubility of alcohol in water is due to [MP PMT/ PET 1988; MP PMT 1989]
A)
Covalent bond done
clear
B)
Ionic bond done
clear
C)
H-bond with \[{{H}_{2}}O\] done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer181)
Alcohols can be distinguished from alkenes by
A)
Dissolving in cold concentrated \[{{H}_{2}}S{{O}_{4}}\] done
clear
B)
Decolourizing with bromine in \[CC{{l}_{4}}\] done
clear
C)
Oxidizing with neutral permanganate solution done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer182)
At \[{{25}^{o}}C\] Ethylene glycol is a
A)
Solid compound done
clear
B)
Liquid done
clear
C)
Gas done
clear
D)
Brown solid done
clear
View Solution play_arrow
-
question_answer183)
When primary alcohol is oxidised with chlorine, it produces [AFMC 1999]
A)
\[HCHO\] done
clear
B)
\[C{{H}_{3}}CHO\] done
clear
C)
\[CC{{l}_{3}}CHO\] done
clear
D)
\[{{C}_{3}}{{H}_{7}}CHO\] done
clear
View Solution play_arrow
-
question_answer184)
Alcohols combine with acetylene in the presence of mercury compounds as catalyst to form
A)
Acetals done
clear
B)
Xanthates done
clear
C)
Vinyl ethers done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer185)
The compound which will give negative iodoform test is [CPMT 1993, 99]
A)
\[C{{H}_{3}}CHO\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}OH\] done
clear
C)
Isopropyl alcohol done
clear
D)
Benzyl alcohol done
clear
View Solution play_arrow
-
question_answer186)
Which of the following is most acidic [CPMT 1999]
A)
Phenol done
clear
B)
Benzyl alcohol done
clear
C)
m-chlorophenol done
clear
D)
Cyclohexanol done
clear
View Solution play_arrow
-
question_answer187)
Number of metamers represented by molecular formula \[{{C}_{4}}{{H}_{10}}O\] is [Tamil Nadu CET 2001]
A)
4 done
clear
B)
3 done
clear
C)
2 done
clear
D)
1 done
clear
View Solution play_arrow
-
question_answer188)
When ether is exposed in air for sometime an explosive substance produced is [RPMT 2002]
A)
Peroxide done
clear
B)
TNT done
clear
C)
Oxide done
clear
D)
Superoxide done
clear
View Solution play_arrow
-
question_answer189)
Ether which is liquid at room temperature is [BVP 2002]
A)
\[{{C}_{2}}{{H}_{5}}OC{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}OC{{H}_{3}}\] done
clear
C)
\[{{C}_{2}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}\] done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer190)
In the following reaction \[{{C}_{2}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}+4[H]\xrightarrow{\text{Red P}+\text{HI}}2X+{{H}_{2}}O,\] X is [MP PMT 2002]
A)
Ethane done
clear
B)
Ethylene done
clear
C)
Butane done
clear
D)
Propane done
clear
View Solution play_arrow
-
question_answer191)
Diethyl ether absorbs oxygen to form [DPMT 1984]
A)
Red coloured sweet smelling compound done
clear
B)
Acetic acid done
clear
C)
Ether suboxide done
clear
D)
Ether peroxide done
clear
View Solution play_arrow
-
question_answer192)
Diethyl ether can be decomposed by heating with [CPMT 1980, 81, 89]
A)
HI done
clear
B)
NaOH done
clear
C)
Water done
clear
D)
\[KMn{{O}_{4}}\] done
clear
View Solution play_arrow
-
question_answer193)
On boiling with concentrated hydrobromic acid, phenyl ethyl ether will yield [AIIMS 1992]
A)
Phenol and ethyl bromide done
clear
B)
Phenol and ethane done
clear
C)
Bromobenzene and ethanol done
clear
D)
Bromobenzene and ethane done
clear
View Solution play_arrow
-
question_answer194)
Ether is formed when ethyl alcohol is heated with conc. \[{{H}_{2}}S{{O}_{4}}.\] The conditions are [KCET 1984]
A)
Excess of \[{{H}_{2}}S{{O}_{4}}\] and \[{{170}^{o}}C\] done
clear
B)
Excess of \[{{C}_{2}}{{H}_{5}}OH\] and \[{{140}^{o}}C\] done
clear
C)
Excess of \[{{C}_{2}}{{H}_{5}}OH\] and \[{{180}^{o}}C\] done
clear
D)
Excess of conc. \[{{H}_{2}}S{{O}_{4}}\] and \[{{100}^{o}}C\] done
clear
View Solution play_arrow
-
question_answer195)
The ether
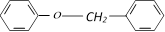
When treated with HI produces [IIT-JEE 1999]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer196)
Addition of alcohols to aldehydes in presence of anhydrous acids yield [CET Pune 1998]
A)
Carboxylic acids done
clear
B)
Ethers done
clear
C)
Cyclic ethers done
clear
D)
Acetals done
clear
View Solution play_arrow
-
question_answer197)
In which of the following reaction, phenol or sodium phenoxide is not formed [CPMT 1996]
A)
\[{{C}_{6}}{{H}_{5}}{{N}_{2}}Cl+alco.\,KOH\to \] done
clear
B)
\[{{C}_{6}}{{H}_{5}}OCl+NaOH\to \] done
clear
C)
\[{{C}_{6}}{{H}_{5}}{{N}_{2}}Cl+aq.\,NaOH\to \] done
clear
D)
\[{{C}_{6}}{{H}_{5}}NNCl\underset{\Delta }{\mathop{\xrightarrow{{{H}_{2}}O}}}\,\] done
clear
View Solution play_arrow
-
question_answer198)
Dimethyl ether when heated with excess HI gives [CPMT 1996]
A)
\[C{{H}_{3}}I\] and \[C{{H}_{3}}OH\] done
clear
B)
\[C{{H}_{3}}I\] and \[{{H}_{2}}O\] done
clear
C)
\[{{C}_{2}}{{H}_{6}}+C{{H}_{3}}I\] and \[C{{H}_{3}}OH\] done
clear
D)
\[C{{H}_{3}}I\] and HCHO done
clear
View Solution play_arrow
-
question_answer199)
The ether that undergoes electrophilic substitution reactions is [JIPMER 2001]
A)
\[C{{H}_{3}}O{{C}_{2}}{{H}_{5}}\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}OC{{H}_{3}}\] done
clear
C)
\[C{{H}_{3}}OC{{H}_{3}}\] done
clear
D)
\[{{C}_{2}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}\] done
clear
View Solution play_arrow
-
question_answer200)
Acetyl chloride does not react with [MNR 1995]
A)
Diethyl ether done
clear
B)
Aniline done
clear
C)
Phenol done
clear
D)
Ethanol done
clear
View Solution play_arrow
-
question_answer201)
The products formed in the following reaction \[{{C}_{6}}{{H}_{5}}-O-C{{H}_{3}}+HI\xrightarrow{\text{heat}}\] are [IIT 1995]
A)
\[{{C}_{6}}{{H}_{5}}-I\] and \[C{{H}_{3}}-OH\] done
clear
B)
\[{{C}_{6}}{{H}_{5}}-OH\] and \[C{{H}_{3}}-I\] done
clear
C)
\[{{C}_{6}}{{H}_{5}}-C{{H}_{3}}\] and HOI done
clear
D)
\[{{C}_{6}}{{H}_{6}}\] and \[C{{H}_{3}}OI\] done
clear
View Solution play_arrow
-
question_answer202)
Etherates are
A)
Ethers done
clear
B)
Solution in ether done
clear
C)
Complexes of ethers with Lewis acid done
clear
D)
Complexes of ethers with Lewis base done
clear
View Solution play_arrow
-
question_answer203)
An ether is more volatile than an alcohol having the same molecular formula. This is due to [AIEEE 2003]
A)
Dipolar character of ethers done
clear
B)
Alcohols having resonance structures done
clear
C)
Inter-molecular hydrogen bonding in ethers done
clear
D)
Inter-molecular hydrogen bonding in alcohols done
clear
View Solution play_arrow
-
question_answer204)
When ether is reacted with \[{{O}_{2}},\] it undergoes explosion due to [CPMT 1996]
A)
Peroxide done
clear
B)
Acid done
clear
C)
Ketone done
clear
D)
TNT done
clear
View Solution play_arrow
-
question_answer205)
The compound which does not react with sodium is [CBSE PMT 1994]
A)
\[{{C}_{2}}{{H}_{5}}OH\] done
clear
B)
\[C{{H}_{3}}-O-C{{H}_{3}}\] done
clear
C)
\[C{{H}_{3}}COOH\] done
clear
D)
\[C{{H}_{3}}-CHOH-C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer206)
Methyl-terbutyl ether on heating with HI of one molar concentration gives [MP PET 1997]
A)
\[C{{H}_{3}}I+{{(C{{H}_{3}})}_{3}}COH\] done
clear
B)
\[C{{H}_{3}}OH+{{(C{{H}_{3}})}_{3}}Cl\] done
clear
C)
\[C{{H}_{3}}I+{{(C{{H}_{3}})}_{3}}Cl\] done
clear
D)
None of the above done
clear
View Solution play_arrow
-
question_answer207)
A substance \[{{C}_{4}}{{H}_{10}}O\] yields on oxidation a compound \[{{C}_{4}}{{H}_{8}}O\] which gives an oxime and a positive iodoform test. The original substance on treatment with conc. \[{{H}_{2}}S{{O}_{4}}\] gives \[{{C}_{4}}{{H}_{8}}\]. The structure of the compound is [SCRA 2000]
A)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}OH\] done
clear
B)
\[C{{H}_{3}}CH(OH)C{{H}_{2}}C{{H}_{3}}\] done
clear
C)
\[{{(C{{H}_{3}})}_{3}}COH\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}-O-C{{H}_{2}}C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer208)
Ethylene glycol reacts with excess of \[PC{{l}_{5}}\] to give [Kerala PMT 2004]
A)
1, 1-dichloroethane done
clear
B)
1, 2-dicholoroethane done
clear
C)
1, 1, 1-trichloroethane done
clear
D)
1, 1, 2, 2-tetrachloroethane done
clear
E)
2, 2-dichloroethane done
clear
View Solution play_arrow
-
question_answer209)
Which of the following will not react with NaOH [CPMT 2004]
A)
B)
\[C{{H}_{3}}CON{{H}_{2}}\] done
clear
C)
\[CH{{(CN)}_{3}}\] done
clear
View Solution play_arrow
-
question_answer210)
The boiling point of methanol is greater than that of methyl thiol because [Kerala PMT 2004]
A)
There is intramolecular hydrogen bonding in methanol and intermolecular hydrogen bonding in methyl thiol done
clear
B)
There is intermolecular hydrogen bonding in methanol and no hydrogen bonding in methyl thiol done
clear
C)
There is no hydrogen bonding in methanol and intermolecular hydrogen bonding in methyl thiol done
clear
D)
There is intramolecular hydrogen bonding in methanol and no hydrogen bonding in methyl thiol done
clear
E)
There is no hydrogen bonding in methanol and intramolecular hydrogen bonding in methyl thiol done
clear
View Solution play_arrow
-
question_answer211)
In the reaction \[\underset{C{{H}_{2}}OH}{\overset{C{{H}_{2}}OH}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,HOH\,}}}\,+\underset{COOH}{\overset{COOH}{\mathop{|\ \ \ \ \ \ \ \ }}}\,\xrightarrow{110{}^\circ C}(A)\] product A will be [Pb . CET 2001]
A)
Glycerol monoformate done
clear
B)
Allyl alcohol done
clear
C)
Formaldehyde done
clear
D)
Acetic acid done
clear
View Solution play_arrow
-
question_answer212)
Which of the following will not form a yellow precipitate on heating with an alkaline solution of iodine [CBSE PMT 2004]
A)
\[C{{H}_{3}}OH\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}OH\] done
clear
C)
\[C{{H}_{3}}CH(OH)C{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}CH(OH)C{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer213)
In Friedal-Crafts acylation, besides \[AlC{{l}_{3}}\], the other reactants are [DPMT 2004]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer214)
Which of the following reagents will produce salicyldehyde on reaction with phenol [DPMT 2004]
A)
\[CHC{{l}_{3}}/NaOH\] done
clear
B)
\[CC{{l}_{4}}/NaOH\] done
clear
C)
\[C{{H}_{2}}C{{l}_{2}}/NaOH\] done
clear
D)
\[C{{H}_{3}}Cl/NaOH\] done
clear
View Solution play_arrow
-
question_answer215)
At 530 K, glycerol reacts with oxalic acid to produce [Pb. CET 2002]
A)
Allyl alcohol done
clear
B)
Formic acid done
clear
C)
Glyceraldehyde done
clear
D)
Glycerol monooxalate done
clear
View Solution play_arrow
-
question_answer216)
With anhydrous zinc chloride, ethylene glycol gives [MP PMT 2004]
A)
Formaldehyde done
clear
B)
Acetylene done
clear
C)
Acetaldehyde done
clear
D)
Acetone done
clear
View Solution play_arrow
-
question_answer217)
Which of the following compound give yellow precipitate with \[{{I}_{2}}\] and NaOH [Pb. CET 2003]
A)
\[C{{H}_{3}}OH\] done
clear
B)
\[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}OH\] done
clear
C)
\[{{C}_{2}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}OH\] done
clear
View Solution play_arrow
-
question_answer218)
Amongst the following, HBr reacts fastest with [IIT-JEE 1986; JIPMER 2000; DCE 2003]
A)
Propane-1-ol done
clear
B)
Propane-2-ol done
clear
C)
2-methyl propane-1-ol done
clear
D)
2-methyl propane-2-ol done
clear
View Solution play_arrow
-
question_answer219)
Which of the following react with benzoic acid to form ethyl benzoate [Pb. CET 2001]
A)
Ethyl alcohol done
clear
B)
Cinnamic acid done
clear
C)
Sodium ethoxide done
clear
D)
Ethyl chloride done
clear
View Solution play_arrow
-
question_answer220)
When phenyl magnesium bromide reacts with t-butanol, the product would be
A)
Benzene done
clear
B)
Phenol done
clear
C)
t-butyl benzene done
clear
D)
t-butyl ether done
clear
View Solution play_arrow
-
question_answer221)
Which of the following is used as catalyst for preparing Grignard reagent [Pb. CET 2002]
A)
Iron powder done
clear
B)
Dry ether done
clear
C)
Activated charcoal done
clear
D)
\[Mn{{O}_{2}}\] done
clear
View Solution play_arrow
-
question_answer222)
Ethyl alcohol is heated with conc. \[{{H}_{2}}S{{O}_{4}}\]. The product formed is [DCE 2004]
A)
\[C{{H}_{3}}-\overset{O}{\mathop{\overset{||}{\mathop{C}}\,}}\,-O{{C}_{2}}{{H}_{5}}\] done
clear
B)
\[{{C}_{2}}{{H}_{6}}\] done
clear
C)
\[{{C}_{2}}{{H}_{4}}\] done
clear
D)
\[{{C}_{2}}{{H}_{2}}\] done
clear
View Solution play_arrow
-
question_answer223)
Dehydration of 2-butanol yield [Pb. CET 2004]
A)
1-butene done
clear
B)
2-butene done
clear
C)
2-butyne done
clear
D)
Both A and B done
clear
View Solution play_arrow
-
question_answer224)
Fats, on alkaline hydrolysis, gives [MH CET 2003]
A)
Oils done
clear
B)
Soaps done
clear
C)
Detergents done
clear
D)
Glycol + acid done
clear
View Solution play_arrow
-
question_answer225)
When vapours of an alcohol are passed over hot reduced copper, alcohol is converted into alkene quickly, the alcohol is [CPMT 1985]
A)
Primary done
clear
B)
Secondary done
clear
C)
Tertiary done
clear
D)
None of these done
clear
View Solution play_arrow
-
question_answer226)
The adduct of the compound 'A' obtained by the reaction with excess of isopropyl magnesium iodide, upon hydrolysis gives a tertiary alcohol. The compound 'A' is [MP PET 1985]
A)
An ester done
clear
B)
A secondary alcohol done
clear
C)
A primary alcohol done
clear
D)
An aldehyde done
clear
View Solution play_arrow
-
question_answer227)
If there be a compound of the formula \[C{{H}_{3}}C{{(OH)}_{3}}\] which one of the following compounds would be obtained from it without reaction with any reagent [CPMT 1983]
A)
\[C{{H}_{3}}OH\] done
clear
B)
\[{{C}_{2}}{{H}_{5}}OH\] done
clear
C)
\[C{{H}_{3}}COOH\] done
clear
D)
HCHO done
clear
View Solution play_arrow
-
question_answer228)
Which of the following can work as a dehydrating agent for alcohols [BHU 1980]
A)
\[{{H}_{2}}S{{O}_{4}}\] done
clear
B)
\[A{{l}_{2}}{{O}_{3}}\] done
clear
C)
\[{{H}_{3}}P{{O}_{4}}\] done
clear
D)
All of these done
clear
View Solution play_arrow
-
question_answer229)
What is formed when glycerol reacts with HI [DCE 2002]
A)
\[\underset{C{{H}_{2}}OH}{\overset{C{{H}_{2}}OH}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,HI\ \ \ \ \,}}}\,\] done
clear
B)
\[\underset{C{{H}_{2}}I}{\overset{C{{H}_{2}}\ }{\mathop{\underset{|}{\overset{||}{\mathop{C}}}\,H\ \ }}}\,\] done
clear
C)
\[\underset{C{{H}_{3}}}{\overset{C{{H}_{3}}}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,{{H}_{2}}}}}\,\] done
clear
D)
\[\underset{C{{H}_{3}}\ \ \ \ }{\overset{C{{H}_{2}}OH}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,=O\ \ }}}\,\] done
clear
View Solution play_arrow
-
question_answer230)
The dehydration of 2-methyl butanol with conc. \[{{H}_{2}}S{{O}_{4}}\] gives [UPSEAT 2004]
A)
2-methyl butene as major product done
clear
B)
Pentene done
clear
C)
2-methyl but-2-ene as major product done
clear
D)
2-methyl pent-2-ene done
clear
View Solution play_arrow
-
question_answer231)
Which alcohol reacts with fatty acids to form fats [MP PMT/PET 1988; MP PET 1991]
A)
Ethanol done
clear
B)
Glycerol done
clear
C)
Methanol done
clear
D)
Isopropanol done
clear
View Solution play_arrow
-
question_answer232)
Which will dehydrate easily [Roorkee 1995]
A)
3-methyl-2-butanol done
clear
B)
Ethyl alcohol done
clear
C)
2-methyl propane-2-ol done
clear
D)
2-methyl butanol-2 done
clear
View Solution play_arrow
-
question_answer233)
\[A\underset{\Delta }{\mathop{\xleftarrow{Cu}}}\,C{{H}_{3}}C{{H}_{2}}OH\underset{\Delta }{\mathop{\xrightarrow{A{{l}_{2}}{{O}_{3}}}}}\,B.\] A and B respectively are \[\] [RPMT/PET 2000]
A)
Alkene, alkanal done
clear
B)
Alkyne, alkanal done
clear
C)
Alkanal, alkene done
clear
D)
Alkene, alkyne done
clear
View Solution play_arrow
-
question_answer234)
Which one of the following reactions would produce secondary alcohol [MP PET 1994]
A)
\[\overset{\overset{\,\,O}{\mathop{\,\,\,||}}\,}{\mathop{{{C}_{6}}{{H}_{5}}CC{{H}_{3}}}}\,\underset{2.{{H}^{+}}}{\mathop{\xrightarrow{1.C{{H}_{3}}MgBr}}}\,\] done
clear
B)
\[\overset{\overset{\,\,O}{\mathop{\,\,\,||}}\,}{\mathop{{{C}_{6}}{{H}_{5}}CC{{H}_{3}}}}\,\underset{2.{{H}^{+}}}{\mathop{\xrightarrow{1.LiAl{{H}_{4}}}}}\,\] done
clear
C)
\[C{{H}_{3}}CHO\underset{2.{{H}^{+}}}{\mathop{\xrightarrow{1.LiAl{{H}_{4}}}}}\,\] done
clear
D)
\[\overset{\overset{O}{\mathop{||}}\,}{\mathop{C{{H}_{3}}CC{{H}_{3}}}}\,\underset{2.B{{r}_{2}}}{\mathop{\xrightarrow{1.O{{H}^{-}}}}}\,\] done
clear
View Solution play_arrow
-
question_answer235)
On reaction with hot conc. \[{{H}_{2}}S{{O}_{4}},\] which one of the following compounds loses a molecule of water [CPMT 1989]
A)
\[C{{H}_{3}}COC{{H}_{3}}\] done
clear
B)
\[C{{H}_{3}}COOH\] done
clear
C)
\[C{{H}_{3}}OC{{H}_{3}}\] done
clear
D)
\[C{{H}_{3}}C{{H}_{2}}OH\] done
clear
View Solution play_arrow
-
question_answer236)
The best method to prepare cyclohexene from cyclohexanol is by using [IIT 2005]
A)
Conc. HCl + ZnCl2 done
clear
B)
Conc. H3PO4 done
clear
C)
HBr done
clear
D)
Conc. HCl done
clear
View Solution play_arrow
-
question_answer237)
Which of the following compound is most acidic [BCECE 2005]
A)
\[C{{H}_{4}}\] done
clear
B)
\[{{C}_{2}}{{H}_{6}}\] done
clear
C)
\[CH\equiv CH\] done
clear
D)
\[{{C}_{2}}{{H}_{5}}OH\] done
clear
View Solution play_arrow
-
question_answer238)
\[{{C}_{2}}{{H}_{5}}OH\] can be differentiated from \[C{{H}_{3}}OH\] by [MP PMT 1994]
A)
Reaction with HCl done
clear
B)
Reaction with \[N{{H}_{3}}\] done
clear
C)
By iodoform test done
clear
D)
By solubility in water done
clear
View Solution play_arrow
-
question_answer239)
A compound does not react with 2.4 di-nitrophenyl hydrazine and Na , compound is [UPSEAT 2003]
A)
Acetone done
clear
B)
Acetaldehyde done
clear
C)
\[C{{H}_{3}}OH\] done
clear
D)
\[C{{H}_{2}}=CHOC{{H}_{3}}\] done
clear
View Solution play_arrow
-
question_answer240)
Which of the following reaction is correctly represented [Orissa JEE 2005]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer241)
Tertiary butyl alcohol gives tertiary butyl chloride on treatment with [Orissa JEE 2005]
A)
Conc. \[HCl\]/anhydrous \[ZnC{{l}_{2}}\] done
clear
B)
\[KCN\] done
clear
C)
\[NaOCl\] done
clear
D)
\[C{{l}_{2}}\] done
clear
View Solution play_arrow
-
question_answer242)
[DPMT 2005]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer243)
In which of the following reactions carbon carbon bond formation takes place [DPMT 2005]
A)
Cannizzaro done
clear
B)
Reimer-Tiemann done
clear
C)
HVZ reaction done
clear
D)
Schmidt reaction done
clear
View Solution play_arrow
-
question_answer244)
Reaction of phenol with chloroform/sodium hydroxide to give \[o\]-hydroxy benzaldehyde involves the formation of [J & K 2005]
A)
Dichloro carbene done
clear
B)
Trichloro carbene done
clear
C)
Chlorine atoms done
clear
D)
Chlorine molecules done
clear
View Solution play_arrow
-
question_answer245)
Which is not correct [J & K 2005]
A)
Phenol is more acidic than acetic acid done
clear
B)
Ethanol is less acidic than phenol done
clear
C)
Ethanol has lower boiling point than ethane done
clear
D)
Ethyne is a non-linear molecule done
clear
View Solution play_arrow
 done
clear
done
clear
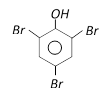 done
clear
done
clear
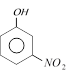 done
clear
done
clear
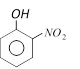 done
clear
done
clear
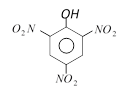 done
clear
done
clear
 is known as [MP PET 1997]
is known as [MP PET 1997] 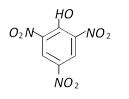 done
clear
done
clear
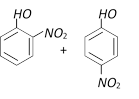 done
clear
done
clear
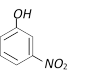 done
clear
done
clear
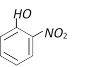 done
clear
done
clear
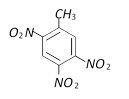 done
clear
done
clear
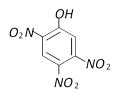 done
clear
done
clear
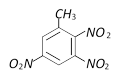 done
clear
done
clear
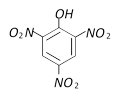 done
clear
done
clear
![]() When treated with HI produces [IIT-JEE 1999]
When treated with HI produces [IIT-JEE 1999] 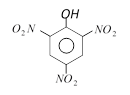 \[{{C}_{2}}{{H}_{5}}OH\] done
clear
\[{{C}_{2}}{{H}_{5}}OH\] done
clear
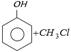 done
clear
done
clear
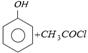 done
clear
done
clear
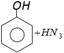 done
clear
done
clear
![]() [DPMT 2005]
[DPMT 2005] 