The intermolecular attractive forces between the liquid molecules can be decreased by:
A)
Cooling the liquid
done
clear
B)
Heating the liquid
done
clear
C)
Turning the liquid into solid
done
clear
D)
All of these
done
clear
E)
None of these
done
clear
View Answer play_arrow
The minerals required by a plant in very small quantities are called:
A)
Macronutrients
done
clear
B)
micronutrients
done
clear
C)
Fertilizers
done
clear
D)
manures
done
clear
E)
None of these
done
clear
View Answer play_arrow
In transverse waves, the particles of the medium move _______ to the direction of motion of the wave.
A)
Parallel
done
clear
B)
Perpendicular
done
clear
C)
Curved
done
clear
D)
Both parallel and perpendicular
done
clear
E)
None of these
done
clear
View Answer play_arrow
Evaporation of a liquid occurs at:
A)
Its boiling point
done
clear
B)
Room temperature
done
clear
C)
All temperatures
done
clear
D)
A temperature higher than its boiling point
done
clear
E)
None of these
done
clear
View Answer play_arrow
36 g of sodium chloride is dissolved in 100 g of water at 293 K. Find its concentration at this temperature.
A)
25%
done
clear
B)
26.47%
done
clear
C)
23.5%
done
clear
D)
27.23%
done
clear
E)
None of these
done
clear
View Answer play_arrow
Which one of the following is not a property of suspension?
A)
A suspension is not transparent to light
done
clear
B)
Suspended particles are visible through naked eye
done
clear
C)
Filter paper can be used to separate components of a suspension
done
clear
D)
A suspension is homogeneous in nature
done
clear
E)
None of these
done
clear
View Answer play_arrow
Carbon is stored in our planet as:
A)
Organic molecules, in the living and dead organisms found in the biosphere
done
clear
B)
Carbon dioxide in atmosphere
done
clear
C)
Organic matters in soil
done
clear
D)
All of these
done
clear
E)
None of these
done
clear
View Answer play_arrow
Murrah is a high yielding breed of:
A)
Cow
done
clear
B)
Hen
done
clear
C)
Buffalo
done
clear
D)
Sheep
done
clear
E)
None of these
done
clear
View Answer play_arrow
The movement of colloidal particles under the influence of electric field is known as:
A)
Brownian movement
done
clear
B)
Electrophoresis
done
clear
C)
Tyndall effect
done
clear
D)
Coagulation
done
clear
E)
None of these
done
clear
View Answer play_arrow
Which one of the following is a mixture?
A)
Sugar
done
clear
B)
Water
done
clear
C)
Blood
done
clear
D)
Glucose
done
clear
E)
None of these
done
clear
View Answer play_arrow
Which of the following is a degenerative disease?
A)
Osteoporosis
done
clear
B)
Kidney failure
done
clear
C)
Myopia
done
clear
D)
All of these
done
clear
E)
None of these
done
clear
View Answer play_arrow
The electrons present in outermost shell of an atom are called:
A)
Valence electrons
done
clear
B)
Core electrons
done
clear
C)
Charged electrons
done
clear
D)
Free electrons
done
clear
E)
None of these
done
clear
View Answer play_arrow
Which one of the following is a communicable disease?
A)
Diabetes
done
clear
B)
Arthritis
done
clear
C)
Cancer
done
clear
D)
Marasmus
done
clear
E)
None of these
done
clear
View Answer play_arrow
The life-supporting zone of the earth where the atmosphere, the hydrosphere and the lithosphere interact and make life possible, is known as:
A)
Asthenosphere
done
clear
B)
Biosphere
done
clear
C)
Ionosphere
done
clear
D)
Exosphere
done
clear
E)
None of these
done
clear
View Answer play_arrow
Give the name of elements present in the compound of baking soda.
A)
Sodium, hydrogen, carbon, oxygen
done
clear
B)
Sodium, hydrogen, oxygen
done
clear
C)
Bromine, calcium, oxygen, carbon
done
clear
D)
Potassium, sulphur, oxygen
done
clear
E)
None of these
done
clear
View Answer play_arrow
What is the atomicity of the molecule \[Si{{H}_{4}}\]?
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
E)
None of these
done
clear
View Answer play_arrow
Calculate the mass percentage of each element present in water.
A)
H = 22.22% and O = 77.78%
done
clear
B)
H = 77.78% and O = 22.22%
done
clear
C)
H = 11.11% and O = 88.89%
done
clear
D)
H = 88.89% and O = 11.11%
done
clear
E)
None of these
done
clear
View Answer play_arrow
Which one of the following is not a characteristic of isotopes?
A)
The isotopes of an element have the same number of protons and electrons but different numbers of neutrons
done
clear
B)
The isotopes of an element have same mass numbers
done
clear
C)
The isotopes of an element exhibit different physical properties such as density, melting point, boiling point etc.
done
clear
D)
The isotopes of an element possess the same electronic configuration and the same number of valence electrons and exhibit same chemical properties
done
clear
E)
None of these
done
clear
View Answer play_arrow
Which one of the following is a scalar quantity?
A)
Force
done
clear
B)
Length
done
clear
C)
Displacement
done
clear
D)
Velocity
done
clear
E)
None of these
done
clear
View Answer play_arrow
Jellyfish belongs to which one of the following phyla?
A)
Cnidaria
done
clear
B)
Porifera
done
clear
C)
Arthropoda
done
clear
D)
Platyhelminthes
done
clear
E)
None of these
done
clear
View Answer play_arrow
Find the odd one out from the following.
A)
Mosses
done
clear
B)
Liverworts
done
clear
C)
Ferns
done
clear
D)
Hornworts
done
clear
E)
None of these
done
clear
View Answer play_arrow
When an object undergoes acceleration:
A)
Its speed always remains constant
done
clear
B)
Its speed always decreases
done
clear
C)
It always falls towards earth
done
clear
D)
A force always acts upon it
done
clear
E)
None of these
done
clear
View Answer play_arrow
An observer standing between two cliffs hears two successive echoes at 2 s and 3 s. Calculate the distance between cliffs (speed of sound in air =\[340\text{ }m/s\]).
A)
340 m
done
clear
B)
850 m
done
clear
C)
510 m
done
clear
D)
790 m
done
clear
E)
None of these
done
clear
View Answer play_arrow
Which one of the following is not a part of animal cell?
A)
Plasma membrane
done
clear
B)
Nucleus
done
clear
C)
Mitochondria
done
clear
D)
Ribosome
done
clear
E)
None of these
done
clear
View Answer play_arrow
Which one of the following statements is incorrect about plasma membrane?
A)
It gives definite shape to the cell
done
clear
B)
It provides protection to the internal contents of the cell
done
clear
C)
It helps in the storage of proteins, starch and lipids
done
clear
D)
All of these
done
clear
E)
None of these
done
clear
View Answer play_arrow
Genes are located on the:
A)
Nuclear membrane
done
clear
B)
Chromosomes
done
clear
C)
Lysosomes
done
clear
D)
Ribosomes
done
clear
E)
None of these
done
clear
View Answer play_arrow
Find the odd one out from the following.
A)
Tracheids
done
clear
B)
Xylem parenchyma
done
clear
C)
Xylem fibres
done
clear
D)
Companion cells
done
clear
E)
None of these
done
clear
View Answer play_arrow
Which one of the following is a type of connective tissue?
A)
Blood
done
clear
B)
Bone
done
clear
C)
Cartilage
done
clear
D)
All of these
done
clear
E)
None of these
done
clear
View Answer play_arrow
The given animal belongs to which one of the following classes?
A)
Pisces
done
clear
B)
Amphibia
done
clear
C)
Reptilia
done
clear
D)
Aves
done
clear
E)
None of these
done
clear
View Answer play_arrow
A car acquires a velocity of 48 km/h in 8 seconds just after the start. Calculate the acceleration of the car.
A)
\[3.15\text{ }m/{{s}^{2}}\]
done
clear
B)
\[4.16\text{ }m/{{s}^{2}}\]
done
clear
C)
\[1.67\text{ }m/{{s}^{2}}\]
done
clear
D)
\[6.18\text{ }m/{{s}^{2}}\]
done
clear
E)
None of these
done
clear
View Answer play_arrow
The velocity-time graph for a car is shown in the figure below.
Using this graph, calculate the average speed of the car.
A)
\[40\,km/h\]
done
clear
B)
\[30\text{ }km/h\]
done
clear
C)
\[64\text{ }km/h\]
done
clear
D)
\[80\text{ }km/h\]
done
clear
E)
None of these
done
clear
View Answer play_arrow
32. Calculate the number of molecules of chloroform \[(CHC{{I}_{3}})\]weighing \[0.0239\text{ }g\text{ }(H=1,C=12,\,Cl=35.5)\].
A)
\[1.2046\times {{10}^{22}}\]
done
clear
B)
\[2.2046\times {{10}^{19}}\]
done
clear
C)
\[1.4046\times {{10}^{17}}\]
done
clear
D)
\[1.2046\times {{10}^{20}}\]
done
clear
E)
None of these
done
clear
View Answer play_arrow
A car weighing 1600 kg moving with a velocity of \[30\text{ }m/s\]retards uniformly coming to rest in 20 s. Calculate the magnitude of the force applied.
A)
\[-600\,\,N\]
done
clear
B)
\[-1200\text{ }N\]
done
clear
C)
\[-2000\text{ }N\]
done
clear
D)
\[-2400\text{ }N\]
done
clear
E)
None of these
done
clear
View Answer play_arrow
Calculate the gravitational force between two bodies A and B having mass 80 kg each and situated at a distance of 100 cm from each other.
A)
\[4.3\times {{10}^{-15}}N\]
done
clear
B)
\[2.3\times {{10}^{-7}}N\]
done
clear
C)
\[4.3\times {{10}^{-7}}N\]
done
clear
D)
\[8.3\times {{10}^{-5}}N\]
done
clear
E)
None of these
done
clear
View Answer play_arrow
How much work is done, when a force of 200 N displaces a body by 30 m in the direction of applied force?
A)
6000 J
done
clear
B)
8000 J
done
clear
C)
4000 J
done
clear
D)
2000 J
done
clear
E)
None of these
done
clear
View Answer play_arrow
What is the work done by the force of gravity on a satellite moving around the earth?
A)
0
done
clear
B)
2400 J
done
clear
C)
36000 J
done
clear
D)
1000 J
done
clear
E)
None of these
done
clear
View Answer play_arrow
A driver accelerates his car first at the rate of \[1.8\text{ }m/{{s}^{2}}\]and then at the rate of\[1.2\text{ }m/{{s}^{2}}\]. The ratio of the two forces exerted by the engine in two cases will be:
A)
1 : 2
done
clear
B)
2 : 1
done
clear
C)
4 : 3
done
clear
D)
3 : 2
done
clear
E)
None of these
done
clear
View Answer play_arrow
What is the work to be done to increase the velocity of a car from 36 km/h to 72 km/h if the mass of the car is 1000 kg?
A)
50000 J
done
clear
B)
200000 J
done
clear
C)
150000 J
done
clear
D)
250000 J
done
clear
E)
None of these
done
clear
View Answer play_arrow
A submarine emits a sonar pulse, which returns from an underwater cliff in 1.02s. If the speed of sound in salt water is\[1531\text{ }m/s\], how far away is the cliff?
A)
648 m
done
clear
B)
780.8 m
done
clear
C)
1025 m
done
clear
D)
2125 m
done
clear
E)
None of these
done
clear
View Answer play_arrow
The average atomic mass of a sample of an element X is 16.2 u. What are the percentages of isotopes \[{}_{8}^{16}X\,\,and\,\,{}_{8}^{18}X\] in the sample?
A)
Percentage of \[{}_{8}^{16}X=80\] and percentage of \[{}_{8}^{18}X=20\]
done
clear
B)
Percentage of \[{}_{8}^{16}X=70\]and percentage of \[{}_{8}^{18}X=30\]
done
clear
C)
Percentage of \[{}_{8}^{16}X=90\]and percentage of \[{}_{8}^{18}X=10\]
done
clear
D)
Percentage of \[{}_{8}^{16}X=95\]and percentage of \[{}_{8}^{18}X=5\]
done
clear
E)
None of these
done
clear
View Answer play_arrow

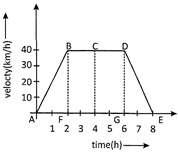 Using this graph, calculate the average speed of the car.
Using this graph, calculate the average speed of the car. 