-
question_answer1)
Given\[l\text{/}a=0.5\,c{{m}^{-1}}\], R = 50 ohm, N = 1.0. The equivalent conductance of the electrolytic cell is
A)
\[100\,oh{{m}^{-1}}c{{m}^{2}}g\,E{{q}^{-1}}\] done
clear
B)
\[20\,oh{{m}^{-1}}c{{m}^{2}}g\,E{{q}^{-1}}\] done
clear
C)
\[300\,oh{{m}^{-1}}c{{m}^{2}}g\,E{{q}^{-1}}\] done
clear
D)
\[10\,oh{{m}^{-1}}c{{m}^{2}}g\,E{{q}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer2)
The resistivity of aluminum is \[2.834\times {{10}^{-8}}\Omega m.\] Thus, conductance across a piece of aluminum wire, that is 4.0 mm in diameter and 2.00 m long is (assume current=1.25 A)
A)
111.0 S done
clear
B)
1.11 S done
clear
C)
222.05 S done
clear
D)
1111 S done
clear
View Solution play_arrow
-
question_answer3)
Molar conductance's of \[BaC{{l}_{2,}}\]\[{{H}_{2}}S{{O}_{4}}\], and HCl at infinite dilutions are \[{{x}_{1}}\],\[{{x}_{2}}\], and \[{{x}_{3}}\], respectively. Equivalent conductance of \[BaS{{O}_{4}}\] at infinite dilution will be:
A)
\[\frac{[{{x}_{1}}+{{x}_{2}}-{{x}_{3}}]}{2}\] done
clear
B)
\[\frac{[{{x}_{1}}-{{x}_{2}}-{{x}_{3}}]}{2}\] done
clear
C)
\[2({{x}_{1}}+{{x}_{2}}-{{2x}_{3}})\] done
clear
D)
\[\frac{[{{x}_{1}}+{{x}_{2}}-2{{x}_{3}}]}{2}\] done
clear
View Solution play_arrow
-
question_answer4)
In acidic medium, \[MnO_{4}^{-}\] is converted to \[M{{n}^{+2}}\] when acts as an oxidizing agent. The quantity of electricity required to reduce 0.05 mol of \[MnO_{4}^{-}\] would be
A)
0.01 F done
clear
B)
0.05 F done
clear
C)
0.25 F done
clear
D)
0.15 F done
clear
View Solution play_arrow
-
question_answer5)
| What product are formed during the electrolysis of a concentrated aqueous solution of sodium chloride using an electrolytic cell in which electrodes are separated by a porous pot? |
| I. \[NaOH(aq)\] |
| II. \[C{{l}_{2}}(g)\] |
| III. \[NaCl{{O}_{3}}(aq)\] |
| IV. \[{{H}_{2}}(g)\] |
| V. \[NaClO(aq)\] |
| Select the correct choice. |
A)
I, II and V done
clear
B)
I, II and IV done
clear
C)
I and II done
clear
D)
I, III and V done
clear
View Solution play_arrow
-
question_answer6)
When a lead storage battery is discharged, then
A)
\[S{{O}_{2}}\] is evolved done
clear
B)
lead is formed done
clear
C)
lead sulphate is consumed done
clear
D)
sulphuric acid is consumed done
clear
View Solution play_arrow
-
question_answer7)
| The standard potential at 298 K for the following half reactions are given against each: |
| \[Z{{n}^{2+}}(aq)+2{{e}^{-}}\rightleftharpoons Zn(s)\] \[-\,0.762\text{ }V\] |
| \[2{{H}^{+}}(aq)+2{{e}^{-}}\rightleftharpoons {{H}_{2}}(g)\] 0.000 V |
| \[C{{r}^{3+}}(aq)+3{{e}^{-}}\rightleftharpoons Cr(s)\] \[-\,0.740\text{ }V\] |
| \[F{{e}^{3+}}(aq)+2{{e}^{-}}\rightleftharpoons F{{e}^{2+}}(aq)\] 0.770 V |
| Which is the strongest reducing agent? |
A)
Zn(s) done
clear
B)
Cr(s) done
clear
C)
\[{{H}_{2}}\left( g \right)\] done
clear
D)
\[F{{e}^{2+}}\left( aq \right)\] done
clear
View Solution play_arrow
-
question_answer8)
The rusting of iron takes place as follows \[2{{H}^{+}}+2{{e}^{-}}+1/2{{O}_{2}}\to {{H}_{2}}O(l);\] \[{{E}^{o}}=+1.23\,V\] \[F{{e}^{2+}}+2{{e}^{-}}\to Fe(s);\,\,{{E}^{o}}=-\,0.44\,V\] Calculate \[\Delta G{}^\circ \] for the net process.
A)
\[-\,152\] kJ \[mo{{l}^{-1}}\] done
clear
B)
\[-\,161\]kJ \[mo{{l}^{-1}}\] done
clear
C)
\[-\,322\]kJ \[mo{{l}^{-1}}\] done
clear
D)
\[-\,76\]kJ \[mo{{l}^{-1}}\] done
clear
View Solution play_arrow
-
question_answer9)
Given, \[{{E}^{o}}_{C{{r}_{2}}O_{4}^{2-}/C{{r}^{3+}}}=1.34\,V,\,\,\,\,{{E}^{o}}_{Cl/C{{l}^{-}}}=\,\,1.37\,V\] \[{{E}^{o}}_{C{{u}^{3+}}/C{{u}^{+}}}=-\,0.74\,V,\,\,\,{{E}^{o}}_{MnO_{4}^{-}/M{{n}^{2+}}}=\,1.51\,\,V,\] Based on the data given above the strongest oxidizing agent will be
A)
\[MnO_{4}^{-}\] done
clear
B)
\[M{{n}^{2+}}\] done
clear
C)
\[C{{l}_{2}}\] done
clear
D)
\[C{{r}^{3+}}\] done
clear
View Solution play_arrow
-
question_answer10)
Electrode potential data are given below. \[F{{e}^{3+}}~\left( aq \right)\text{+}{{\text{e}}^{-}}\to F{{e}^{2+}}\left( aq \right);\] \[E{}^\circ =+\,0.77\] \[A{{l}^{3+}}\left( aq \right)\text{+3}{{\text{e}}^{-}}\to \text{Al(s);}\] \[E{}^\circ \text{ }=-\,1.66\text{ }V\] \[B{{r}_{2}}(aq)+2{{e}^{-}}\to 2B{{r}^{-}}(aq);\] \[E{}^\circ =+\,1.08V\] Based on the data given above, reducing power of \[F{{e}^{2+}}\], Al and \[B{{r}^{-}}\] will increase in the order
A)
\[B{{r}^{-}}<F{{e}^{2+}}<Al\] done
clear
B)
\[F{{e}^{2+}}<Al<B{{r}^{-}}\] done
clear
C)
\[Al<B{{r}^{-}}<F{{e}^{2+}}\] done
clear
D)
\[Al<F{{e}^{2+}}<B{{r}^{-}}\] done
clear
View Solution play_arrow
-
question_answer11)
The standard reduction potentials for \[Z{{n}^{2+}}\]/Zn, \[N{{i}^{2+}}\]/Ni and \[F{{e}^{2+}}\]/Fe are \[-\text{ }0.74,\] \[-\text{ }0.22\text{ }V\]and \[-\text{ }0.44\text{ }V\]respectively. The reaction \[X+{{Y}^{2+}}\to {{X}^{2+}}+Y\] Will be spontaneous when
A)
X = Ni, Y = Fe done
clear
B)
X = Ni, Y = Zn done
clear
C)
X = Fe, Y = Zn done
clear
D)
X = Zn, Y = Ni done
clear
View Solution play_arrow
-
question_answer12)
For\[Ag\to A{{g}^{+}}+{{e}^{-}},\] \[E{}^\circ =-0.798V\] \[{{V}^{2+}}+V{{O}^{2+}}+2{{H}^{+}}\to 2{{V}^{3+}}+{{H}_{2}}O,\] \[E\text{ }\!\!{}^\circ\!\!\text{ }=-0.614\text{ }V\] \[{{V}^{3+}}+A{{g}^{+}}+{{H}_{2}}O\to V{{O}^{2+}}+2{{H}^{+}}+Ag,\] \[E{}^\circ =-\,0.438\text{ }V\] Then \[E{}^\circ \]for the reaction \[{{V}^{3+}}+{{e}^{-}}\to {{V}^{2+}}\] is
A)
+ 0.255V done
clear
B)
\[-\,0.255\,V\] done
clear
C)
\[-\,0.254\,V\] done
clear
D)
\[-\,1.055\,V\] done
clear
View Solution play_arrow
-
question_answer13)
The solubility product of silver iodide is \[8.3\times {{10}^{-17}}\] and the standard reduction potential of Ag/\[A{{g}^{+}}\] electrode is + 0.8 volts at \[25{}^\circ \text{ }C.\] The standard reduction potential of Ag, \[AgI\text{/}{{I}^{-}}\]electrode from these data is
A)
\[-\,0.30\,V\] done
clear
B)
+ 0.16V done
clear
C)
+0.10 V done
clear
D)
\[-\,0.16\,V\] done
clear
View Solution play_arrow
-
question_answer14)
Given standard electrode potentials: \[F{{e}^{3+}}+3{{e}^{-}}\to Fe;\] \[{{E}^{o}}=-\,0.036\text{ }volt\] \[F{{e}^{2+}}+2{{e}^{-}}\to Fe;\] \[{{E}^{o}}~=-\,0.440\text{ }volt\] The standard electrode potential \[E{}^\circ \]for \[F{{e}^{3+}}+{{e}^{-}}\to F{{e}^{2+}}\]
A)
\[-\,0.476\]volt done
clear
B)
\[-\,0.404\]volt done
clear
C)
0.440 volt done
clear
D)
0.772 volt done
clear
View Solution play_arrow
-
question_answer15)
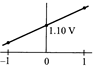
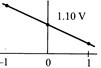
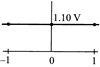
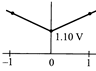
The overall cell reaction of Daniell cell is: \[Zn(s)+C{{u}^{2+}}\rightleftharpoons Z{{n}^{2+}}+Cu(s);\] \[E{}^\circ =+\,1.10\text{ }V\] \[Q=\frac{[Z{{n}^{2+}}]}{[C{{u}^{2+}}]}\] When \[{{\log }_{10}}\]Q is plotted on X-axis and \[{{E}_{cell}}\] on Y-axis, then which of the following will correctly represent the variation?
A)
B)
C)
D)
View Solution play_arrow
-
question_answer16)
Standard reduction electrode potentials of three metals A, B and C are +0.5 V, \[-\,3.0\]V, and \[-\,1.2\] V respectively. The reducing power of these metals are
A)
\[B>C>A\] done
clear
B)
\[A>B>C\] done
clear
C)
\[C>B>A\] done
clear
D)
\[A>C<B\] done
clear
View Solution play_arrow
-
question_answer17)
When during electrolysis of a solution of \[AgN{{O}_{3}},\] 9650 coulombs of charge pass through the electroplating bath the mass of silver deposited on the cathode will be
A)
1.08 g done
clear
B)
10.8 g done
clear
C)
21.6 g done
clear
D)
108 g done
clear
View Solution play_arrow
-
question_answer18)
The standard emf of a cell having one electron change found to be 0.591 V at \[25{}^\circ C\]. The equilibrium constant the reaction is
A)
\[1.0\times {{10}^{30}}\] done
clear
B)
\[1.0\times {{10}^{5}}\] done
clear
C)
\[1.0\times {{10}^{10}}\] done
clear
D)
\[1.0\times {{10}^{1}}\] done
clear
View Solution play_arrow
-
question_answer19)
In a cell that utilizes the reaction; \[Zn(s)+2{{H}^{+}}(aq.)\to Z{{n}^{2+}}(aq)+{{H}_{2}}(g),\]addition of \[{{H}_{2}}S{{O}_{4}}\] to cathode compartment will
A)
increase the E and shift equilibrium to the left done
clear
B)
lower the E and shift equilibrium to the right done
clear
C)
increase the E and shift equilibrium to the right done
clear
D)
lower the E and shift equilibrium to the left done
clear
View Solution play_arrow
-
question_answer20)
The equivalent conductances of two strong electrolytes a infinite dilution in \[{{H}_{2}}O\] (where ions move freely througl a solution) at \[25{}^\circ C\]are given below \[{{\Lambda }^{o}}_{C{{H}_{3}}COONa}=91.0\,\,S\,\,c{{m}^{2}}\text{/}\,Eq\] \[{{\Lambda }^{o}}_{HCl}=426.2\,\,S\,\,c{{m}^{2}}\text{/}\,Eq\] What additional information/ quantity one needs to calculate \[\Lambda {}^\circ \]of an aqueous solution of aceti acid?
A)
\[\Lambda {}^\circ \]of \[NaCl\] done
clear
B)
\[\Lambda {}^\circ \]of\[C{{H}_{3}}\]COOK done
clear
C)
The limiting equivalent conductance of \[{{H}^{+}}(\lambda _{{{H}^{+}}}^{\circ })\] done
clear
D)
\[\Lambda {}^\circ \]of chloroaceti cacid (Cl\[C{{H}_{2}}\]COOH) done
clear
View Solution play_arrow
-
question_answer21)
The molar conductivity of acetic acid at infnite dilution is 390.7 and for 0.01 M acetic acid is 3.907 S \[c{{m}^{2}}\] \[mo{{l}^{-1}}\]. What is the pH of solution?
View Solution play_arrow
-
question_answer22)
0.01 M aqueous solution of a dibasic acid is diluted to 0.004N such that equivalent conductance is x times. What is the value of x?
View Solution play_arrow
-
question_answer23)
pH of the solution in the anode compartment of the ( following cell at 298 K is x when \[{{E}_{cell}}\]\[-\]\[E_{cell}^{o}\]= 0.0591 V Pt.(\[{{H}_{2}}\])|pH = x||\[N{{i}^{2+}}\](1M)|Ni. Find the value of x.
View Solution play_arrow
-
question_answer24)
B In a fuel cell, following reactions takes place and electricity is produced. Anodic: \[{{H}_{2}}\]+\[2O{{H}^{-}}\]\[\to \]\[2{{H}_{2}}O\]+\[2{{e}^{-}}\] Cathodic: \[{{O}_{2}}\] + \[2{{H}_{2}}O\] + \[4{{e}^{-}}\]\[\to \] \[4O{{H}^{-}}\] If 100.8 L of \[{{H}_{2}}\]at STP reacts in 96500 s, what is the average current produced (in ampere)?
View Solution play_arrow
-
question_answer25)
An expensive but lighter alternate to the lead storage battery is the silver-zinc battery. \[A{{g}_{2}}O(s)+Zn(s)+{{H}_{2}}O(l)\to Zn(O{{H}_{2}})(s)+2Ag(s)\]\[{{E}^{0}}\]=1.5544V Numerical value of\[\Delta G{}^\circ \]in scientific notation is \[x\times {{10}^{y}}\]J. What is the value of x+y?
View Solution play_arrow
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
