question_answer 1)
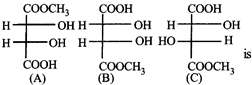
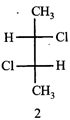
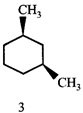
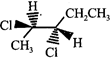
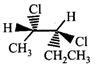
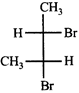
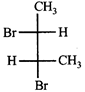
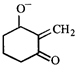
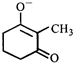
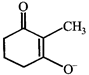
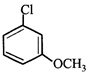
The correct statement about the compounds A, B, and C
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 2)
\[{{C}_{6}}{{H}_{5}}CHO+HCN\xrightarrow{{}}{{C}_{6}}{{H}_{5}}-\underset{H}{\overset{H}{\mathop{\underset{|}{\overset{|}{\mathop{C}}}\,}}}\,-CN\] The product would be
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 3)
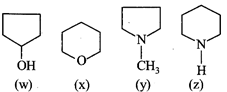
Arrange the following (w, x, y, z) in decreasing order of their boiling points:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
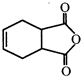
question_answer 4)
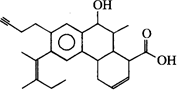
How many degrees of unsaturation are there in the following compound?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 5)
Arrange in the order of increasing acidity.
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 6)
Which of the following is optically active?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 7)
Keto-enol tautomerism is observed in:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 8)
The change in the state of hybridization of the asterisked carbon in the following reaction. \[C{{H}_{3}}\overset{*}{\mathop{C}}\,N\xrightarrow{{{H}_{2}}O}C{{H}_{3}}\overset{*}{\mathop{C}}\,ON{{H}_{2}}\] is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 9)
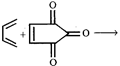
In the reaction shown below the six membered ring is generated by shifting which bond
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 10)
Which among the following is substitution reaction?
A)
B)
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 11)
In allene \[({{C}_{3}}{{H}_{4}})\], the type(s) of hybridization of the carbon atoms is (are):
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
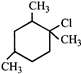
question_answer 12)
The correct IUPAC name of the following compound
is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 13)
Which molecule will be most reactive for \[{{S}_{N}}1\] reaction?
A)
B)
C)
D)
View Solution play_arrow
question_answer 14)
Arrange the following in decreasing order of solubility in water
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 15)
The correct priorities for the substituents shown below, according to the E-Z sequence rule is
I. \[-CN\] II. \[-CBr{{\left( C{{H}_{3}} \right)}_{2}}\] III. \[-COOH\] IV. \[-C{{H}_{2}}-\overset{O}{\mathop{\overset{\parallel }{\mathop{C}}\,}}\,-OC{{H}_{3}}\] V. \[-\overset{O}{\mathop{\overset{\parallel }{\mathop{C}}\,}}\,-H\]
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 16)
The alcohol product(s) of the reduction of 2-methyl-3-pentanone with \[LiAl{{H}_{4}}\] is (are)
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 17)
Identity which of the strucutre below are meso structures?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 18)
Select the most stable carbocation amongst the following
A)
B)
C)
D)
View Solution play_arrow
question_answer 19)
Which of the following reactions is elimination reaction?
A)
B)
done
clear
C)
D)
done
clear
View Solution play_arrow
question_answer 20)
In the following groups
\[\underset{(I)}{\mathop{-OAc}}\,\] \[\underset{(II)}{\mathop{-O}}\,Me\]
\[-\underset{(III)}{\mathop{OS{{O}_{2}}}}\,Me\] \[-\underset{(IV)}{\mathop{OS{{O}_{2}}}}\,C{{F}_{3}}\]
the order of leaving group ability is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 21)
A mixture containing the following four compounds is extracted with 1M\[HCl\]. The compound that goes to aqueous layer is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 22)
Most stable carbocation among the following is:
A)
B)
C)
D)
done
clear
View Solution play_arrow
question_answer 23)
Which of the following cannot be represented by resonance structures?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
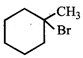
question_answer 24)
The IUPAC name of the compound
is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 25)
The correct relation between the bond lengths a and b is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 26)
Which of the following compounds are meso forms?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 27)
Allyl is ocyanide has
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 28)
Which one of the following is a free-radical substitution reaction?
A)
done
clear
B)
C)
D)
View Solution play_arrow
question_answer 29)
The most stable canonical structure among the given structure is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 30)
Which of the following is the IUPAC name of the compound
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
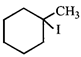
question_answer 31)
The IUPAC name of the following compound is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 32)
Dipole moment is shown by
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 33)
The optically inactive compound from the following is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 34)
Tautomerism is exhibited by -
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 35)
Which of the following represents the correct order of stability of the given carbocations?
A)
B)
C)
D)
View Solution play_arrow
question_answer 36)
Which of the following behaves both as a nucleophile and as an electrophile?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 37)
What is the decreasing order of strength of the bases \[O{{H}^{-}},NH_{2}^{-},HC\equiv {{C}^{-}}~\] and \[C{{H}_{3}}CH_{2}^{-}\]?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 38)
Pick up the correct statement regarding the following resonating structures of the anilinium ion
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 39)
Which of the following numberings is correct?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 40)
The two compounds given below are
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 41)
The number of structural isomers for \[{{C}_{6}}{{H}_{14}}\] is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 42)
Which of the following pairs of compounds are positional isomers?
A)
done
clear
B)
done
clear
C)
done
clear
D)
View Solution play_arrow
question_answer 43)
The correct nucleophilicity order is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 44)
The correct order of heats of combustion of above compounds is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 45)
Which of the following optically active compounds racemizes in dil. \[KOH/C{{H}_{3}}OH\]
A)
B)
C)
D)
View Solution play_arrow
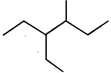
question_answer 46)
The correct number of chiral centres in
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 47)
An optically active compound (A) is treated with NaI/acetone (B) is obtained
What is true about (B)?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 48)
Which of the following is correct set of physical properties of the geometrical isomers?
A)
B)
C)
D)
View Solution play_arrow
question_answer 49)
Which of the following resonance structure is lowest in energy?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 50)
A solution of(+) - 2 - chloro - 2 - phenylethane in toluene racemises slowly in the presence of small amounts of \[SbC{{l}_{5}}\] due to the formation of
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 51)
In the following compounds
the order of basicity is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 52)
Which one of the following compounds has non zero dipole moment?
A)
B)
C)
D)
View Solution play_arrow
question_answer 53)
Dipole moment of which ketone is maximum?
A)
B)
C)
D)
View Solution play_arrow
question_answer 54)
Which of the following orders is correct for heat of hydrogenation of these compounds?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 55)
Which one of the following acids does not exhibit optical isomerism?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
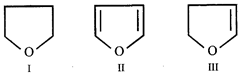
question_answer 56)
How many cyclic structures are possible for \[{{C}_{4}}{{H}_{6}}\]?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 57)
Maleic acid and fumaric acids are
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 58)
One among the following compounds will not give effervescence with sodium carbonate:
A)
done
clear
B)
C)
done
clear
D)
View Solution play_arrow
question_answer 59)
Which of the following alkenes is the most stable?
A)
B)
C)
D)
View Solution play_arrow
question_answer 60)
Which of the following statements is not correct?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 61)
The enol form of acetone after treatment with \[{{D}_{2}}O\] gives:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 62)
Indicate whether each of the following pairs are identical or not?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 63)
Which of the following compound has plane of symmetry? \[R=-\underset{C{{H}_{3}}}{\mathop{\underset{|}{\mathop{CH}}\,}}\,-Cl\] , \[S=-\underset{Br}{\mathop{\underset{|}{\mathop{CH}}\,}}\,-Cl\]
A)
B)
C)
D)
done
clear
View Solution play_arrow
question_answer 64)
Which of the following is not a resonance form of the enolate ion formed in the following acid base equilibrium
A)
B)
C)
D)
View Solution play_arrow
question_answer 65)
The order of stability of the following carbocations:
(I) \[C{{H}_{2}}=CH-\overset{+}{\mathop{C}}\,{{H}_{2}}\]
(II) \[C{{H}_{3}}-C{{H}_{2}}-\overset{+}{\mathop{C}}\,{{H}_{2}}\]
(III)
(IV) \[\overset{+}{\mathop{C}}\,{{H}_{3}}\] is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 66)
1.4 kg of an organic compound was digested according to Kjeldah method and the ammonia evolved was absorbed in 60 mL of M/ 10 \[{{H}_{2}}S{{O}_{4}}\]solution. The excess sulphuric acid required 20 mL of M/10 \[NaOH\] solution for neutralization. The percentage of nitrogen in the compound is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 67)
Which of the following would react most readily with nucleophiles?
A)
B)
C)
D)
View Solution play_arrow
question_answer 68)
The shape of transition state is -
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
question_answer 69)
Which one of the following is most stable?
A)
B)
C)
D)
View Solution play_arrow
question_answer 70)
In nucleophilic substitution reaction, order of halogens as incoming (attacking) nucleophile is:
\[{{I}^{-}}>B{{r}^{-}}>C{{l}^{-}}\] The order of halogens as departing nucleophile should be:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Solution play_arrow
![]()
 done
clear
done
clear
 is:
is: 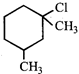 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
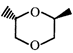
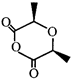
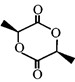
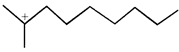 done
clear
done
clear
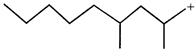 done
clear
done
clear
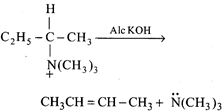 done
clear
done
clear
![]() is
is 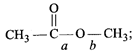 The correct relation between the bond lengths a and b is:
The correct relation between the bond lengths a and b is: 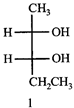
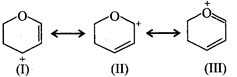 The most stable canonical structure among the given structure is:
The most stable canonical structure among the given structure is: 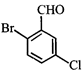
![]()
![]()
![]()
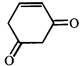
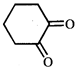
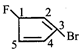
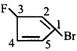
![]()
![]()
![]()
![]()
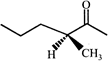
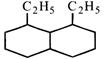
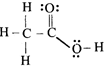
 The correct order of heats of combustion of above compounds is:
The correct order of heats of combustion of above compounds is: 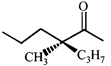 done
clear
done
clear
 done
clear
done
clear
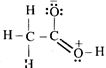
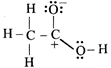
![]()
![]()
![]()
![]()
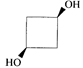 done
clear
done
clear
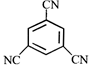 done
clear
done
clear
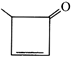 done
clear
done
clear
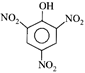 done
clear
done
clear
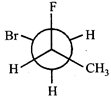
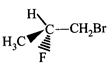
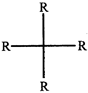 done
clear
done
clear
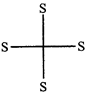 done
clear
done
clear
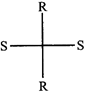 done
clear
done
clear
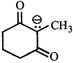 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
 done
clear
done
clear
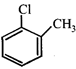 done
clear
done
clear
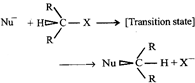
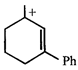 done
clear
done
clear
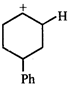 done
clear
done
clear
