-
question_answer1)
The coordination number and oxidation state of \[Cr\] in \[{{K}_{3}}\left[ Cr{{\left( {{C}_{2}}{{O}_{4}} \right)}_{3}} \right]\] are, respectively [CBSE PMT 1995]
A)
4 and +2 done
clear
B)
6 and +3 done
clear
C)
3 and +3 done
clear
D)
3 and 0 done
clear
View Solution play_arrow
-
question_answer2)
The complex compounds which result from the coordination of carbon monoxide are known as [BHU 1999]
A)
Electronic done
clear
B)
Carbonyls done
clear
C)
Carbonates done
clear
D)
Carbon permono done
clear
View Solution play_arrow
-
question_answer3)
Mixture \[X=0.02\,mol\] of \[[Co{{(N{{H}_{3}})}_{5}}S{{O}_{4}}]\,Br\] and 0.02 mol of \[[Co{{(N{{H}_{3}})}_{5}}Br]S{{O}_{4}}\] was prepared in 2 litre of solution 1 litre of mixture X + excess \[AgN{{O}_{3}}\to Y\]. 1 litre of mixture X + excess \[BaC{{l}_{2}}\to Z\] Number of moles of Y and Z are [IIT JEE 2003]
A)
0.01, 0.01 done
clear
B)
0.02, 0.01 done
clear
C)
0.01, 0.02 done
clear
D)
0.02, 0.02 done
clear
View Solution play_arrow
-
question_answer4)
Which of the following organometallic compound is \[\sigma \] and \[\pi \] bonded [MH CET 2001; PCET 2002]
A)
\[Fe{{(C{{H}_{3}})}_{3}}\] done
clear
B)
\[[Fe{{({{\eta }^{5}}-{{C}_{5}}{{H}_{5}})}_{2}}]\] done
clear
C)
\[{{[Co{{(CO)}_{5}}N{{H}_{3}}]}^{2+}}\] done
clear
D)
\[K[PtC{{l}_{3}}({{n}^{2}}-{{C}_{2}}{{H}_{4}})]\] done
clear
View Solution play_arrow
-
question_answer5)
In \[\left[ Ni{{\left( N{{H}_{3}} \right)}_{4}} \right]S{{O}_{4}},\] the valency and coordination number of \[Ni\] will be respectively
A)
3 and 6 done
clear
B)
2 and 4 done
clear
C)
4 and 2 done
clear
D)
4 and 4 done
clear
View Solution play_arrow
-
question_answer6)
Co-ordination number of \[Fe\] in the complexes \[{{\left[ Fe{{\left( CN \right)}_{6}} \right]}^{4-}},\] \[{{\left[ Fe{{\left( CN \right)}_{6}} \right]}^{3-}}\] and \[{{\left[ FeC{{l}_{4}} \right]}^{-}}\] would be respectively [MP PET 2003]
A)
2, 3, 3 done
clear
B)
6, 6, 4 done
clear
C)
6, 3,3 done
clear
D)
6, 4, 6 done
clear
View Solution play_arrow
-
question_answer7)
On hydrolysis \[{{(Me)}_{2}}SiC{{l}_{2}}\] will produce [IIT-JEE 2003]
A)
\[{{(Me)}_{2}}Si{{(OH)}_{2}}\] done
clear
B)
\[{{(Me)}_{2}}Si=O\] done
clear
C)
\[-{{[-O-{{(Me)}_{2}}Si-O-]}_{n}}-\] done
clear
D)
\[M{{e}_{2}}SiCl(OH)\] done
clear
View Solution play_arrow
-
question_answer8)
Which of the following represents a chelating ligand [JIPMER 2002]
A)
\[{{H}_{2}}O\] done
clear
B)
\[O{{H}^{-}}\] done
clear
C)
\[DMG\] done
clear
D)
\[C{{l}^{-}}\] done
clear
View Solution play_arrow
-
question_answer9)
\[CuS{{O}_{4}}\] reacts with \[KCN\] solution and forms [MP PMT 1992; IIT 1996; UPSEAT 2001, 02]
A)
\[Cu{{(CN)}_{2}}\] done
clear
B)
\[Cu(CN)\] done
clear
C)
\[{{K}_{2}}\left[ Cu{{(CN)}_{4}} \right]\] done
clear
D)
\[{{K}_{3}}\left[ Cu{{\left( CN \right)}_{4}} \right]\] done
clear
View Solution play_arrow
-
question_answer10)
In the process of extraction of gold, Roasted gold ore\[+C{{N}^{-}}+{{H}_{2}}O\xrightarrow{{{O}_{2}}}[X]+O{{H}^{-}}\] \[[X]+Zn\to [Y]+Au\] Identify the complexes [X] and [Y] [IIT-JEE 2003]
A)
\[X={{[Au{{(CN)}_{2}}]}^{-}},\,Y={{[Zn{{(CN)}_{4}}]}^{2-}}\] done
clear
B)
\[X={{[Au{{(CN)}_{4}}]}^{3-}},\,Y={{[Zn{{(CN)}_{4}}]}^{2-}}\] done
clear
C)
\[X={{[Au{{(CN)}_{2}}]}^{-}},\,Y=Zn{{(CN)}_{6}}{{]}^{4-}}\] done
clear
D)
\[X={{[Au{{(CN)}_{4}}]}^{-}},\,Y={{[Zn{{(CN)}_{4}}]}^{2-}}\] done
clear
View Solution play_arrow
-
question_answer11)
A solution of potassium ferrocyanide would contains ...... ions [KCET 1990]
A)
2 done
clear
B)
3 done
clear
C)
4 done
clear
D)
5 done
clear
View Solution play_arrow
-
question_answer12)
Which one of the following complexes is an outer orbital complex [AIEEE 2004]
A)
\[{{[Co{{(N{{H}_{3}})}_{6}}]}^{3+}}\] done
clear
B)
\[{{[Mn{{(CN)}_{6}}]}^{4-}}\] done
clear
C)
\[{{[Fe{{(CN)}_{6}}]}^{4-}}\] done
clear
D)
\[{{[Ni{{(N{{H}_{3}})}_{6}}]}^{2+}}\] Atomic nos: \[Mn=25,\,Fe=26,\,Co=27,\,Ni=28\] done
clear
View Solution play_arrow
-
question_answer13)
Which one of the following has largest number of isomers? [AIEEE 2004]
A)
\[{{[Ir{{(P{{P}_{3}})}_{2}}H(CO)]}^{2+}}\] done
clear
B)
\[{{[Co{{(N{{H}_{3}})}_{5}}Cl]}^{2+}}\] done
clear
C)
\[{{[Ru{{(N{{H}_{3}})}_{4}}C{{l}_{2}}]}^{+}}\] done
clear
D)
\[{{[Co{{(en)}_{2}}C{{l}_{2}}]}^{+}}\] (R = alkyl group; en = ethylenediamine) done
clear
View Solution play_arrow
-
question_answer14)
Which kind of isomerism is exhibited by octahedral \[Co{{(N{{H}_{3}})}_{4}}B{{r}_{2}}Cl\] ? [IIT-JEE Screening 2005]
A)
Geometrical and Ionization done
clear
B)
Geometrical and Optical done
clear
C)
Optical and Ionization done
clear
D)
Geometrical only done
clear
View Solution play_arrow
-
question_answer15)
Which one of the following is expected to exhibit optical isomerism (en = ethylenediamine)
A)
cis - \[[Pt{{(N{{H}_{3}})}_{2}}C{{l}_{2}}]\] done
clear
B)
trans - \[[Co{{(en)}_{2}}C{{l}_{2}}]\] done
clear
C)
trans - \[[Pt{{(N{{H}_{3}})}_{2}}C{{l}_{2}}]\] done
clear
D)
cis - \[[Co{{(en)}_{2}}C{{l}_{2}}]\] done
clear
View Solution play_arrow
-
question_answer16)
\[{{[\text{EDTA}]}^{4-}}\] is a: [UPSEAT 2004]
A)
Monodentate ligand done
clear
B)
Bidentate ligand done
clear
C)
Quadridentate ligand done
clear
D)
Hexadentate ligand done
clear
View Solution play_arrow
-
question_answer17)
Which of the following statements is incorrect? [KCET 2004]
A)
In \[{{K}_{3}}[Fe{{(CN)}_{6}}],\] the ligand has satisfied only the secondary valency of ferric ion. done
clear
B)
In \[{{K}_{3}}[Fe{{(CN)}_{6}}],\] the ligand has satisfied both primary and secondary valencies of ferric ion. done
clear
C)
In \[{{K}_{4}}[Fe{{(CN)}_{6}}],\] the ligand has satisfied both primary and secondary valencies of ferrous ion. done
clear
D)
In \[[Cu{{(N{{H}_{3}})}_{4}}]S{{O}_{4}},\] the ligand has satisfied only the secondary valency of copper. done
clear
View Solution play_arrow
-
question_answer18)
Which of the following is considered as an anticancer species. [CBSE PMT 2004]
A)
B)
C)
D)
View Solution play_arrow
-
question_answer19)
An aqueous solution of \[CoC{{l}_{2}}\] on addition of excess of concentrated HCl turns blue due to formation of [AIIMS 2005]
A)
\[[Co{{({{H}_{2}}O)}_{4}}C{{l}_{2}}]\] done
clear
B)
\[{{[Co({{H}_{2}}O)2C{{l}_{4}}]}^{2-}}\] done
clear
C)
\[{{[CoC{{l}_{4}}]}^{2-}}\] done
clear
D)
\[[Co({{H}_{2}}O)2C{{l}_{2}}]\] done
clear
View Solution play_arrow
-
question_answer20)
The correct order for the wavelength of absorption in the visible region is [AIIMS 2005]
A)
\[{{[Ni{{(N{{O}_{2}})}_{6}}]}^{4-}}<{{[Ni{{(N{{H}_{3}})}_{6}}]}^{2+}}<{{[Ni{{({{H}_{2}}O)}_{6}}]}^{2+}}\] done
clear
B)
\[{{[Ni{{(N{{O}_{2}})}_{6}}]}^{4-}}<{{[Ni{{({{H}_{2}}O)}_{6}}]}^{2+}}<{{[Ni{{(N{{H}_{3}})}_{6}}]}^{2+}}\] done
clear
C)
\[{{[Ni{{({{H}_{2}}O)}_{6}}]}^{2+}}<{{[Ni{{(N{{H}_{3}})}_{6}}]}^{2+}}<{{[Ni{{(N{{O}_{2}})}_{6}}]}^{4-}}\] done
clear
D)
\[{{[Ni{{(N{{H}_{3}})}_{6}}]}^{2+}}<{{[Ni{{({{H}_{2}}O)}_{6}}]}^{2+}}<{{[Ni{{(N{{O}_{2}})}_{6}}]}^{4-}}\] done
clear
View Solution play_arrow
-
question_answer21)
In which of the following pairs both the complexes show optical isomerism [AIIMS 2005]
A)
cis-\[{{[Cr{{({{C}_{2}}{{O}_{4}})}_{2}}C{{l}_{2}}]}^{3-}}\], cis-\[[Co{{(N{{H}_{3}})}_{4}}C{{l}_{2}}]\] done
clear
B)
\[[Co{{(en)}_{3}}]C{{l}_{3}}\], cis-\[[Co{{(en)}_{2}}C{{l}_{2}}]Cl\] done
clear
C)
\[[PtCl(dien)]Cl\], \[{{[NiC{{l}_{2}}B{{r}_{2}}]}^{2-}}\] done
clear
D)
\[[Co{{(N{{O}_{3}})}_{3}}{{(N{{H}_{3}})}_{3}}]\], cis-\[[Pt{{(en)}_{2}}C{{l}_{2}}]\] done
clear
View Solution play_arrow
-
question_answer22)
Which of the following compounds shows optical isomerism [AIEEE 2005; CBSE PMT 2005]
A)
\[{{[Cu{{(N{{H}_{3}})}_{4}}]}^{2+}}\] done
clear
B)
\[{{[ZnC{{l}_{4}}]}^{2-}}\] done
clear
C)
\[{{[Cr{{({{C}_{2}}{{O}_{4}})}_{3}}]}^{3-}}\] done
clear
D)
\[{{[Co{{(CN)}_{6}}]}^{3-}}\] done
clear
View Solution play_arrow
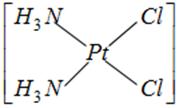 done
clear
done
clear
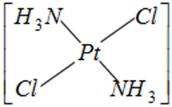 done
clear
done
clear
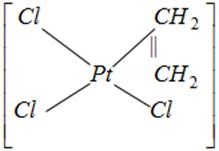 done
clear
done
clear
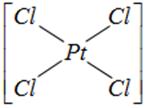 done
clear
done
clear
